Abstract
Acute tryptophan depletion (ATD) is an experimental procedure that has been widely used over the last decade to investigate the role of serotonin (5-HT) in a variety of disorders and in the effects of treatment. The following two articles provide an update of this technique; the first part examines the pharmacology of 5-HT synthesis and outlines the reasoning behind the development of the ATD method; the second part reviews the clinical application of ATD to the field of psychiatry.
Tryptophan and serotonin synthesis
The technique of ATD examines the behavioural and cognitive consequences of acute reductions in plasma tryptophan (TRP) levels. It is based on the premise that depletion of TRP, the precursor of 5-HT, leads to depletion of brain 5-HT.
Tryptophan is an essential amino acid that mammals acquire from dietary sources [1]. Plasma TRP levels are determined by the balance between the dietary intake of TRPand its removal from the plasma by protein synthesis. Most of the TRP in plasma is bound to albumin, with 5% being left free and available for transport into the central nervous system (CNS) [2] – thus central brain TRP levels are more accurately predicted by free TRP than total (free+protein bound) TRPplasma levels [3]. Tryptophan is transported across the blood-brain barrier (BBB) by a specific carrier for which TRP and all other large neutral amino acids (LNAAs) (valine, leucine, isoleucine, methionine, phenylalanine and tyrosine) also compete [4]. The ratio of plasma TRP/plasma LNAAs has been noted to be a more accurate predictor of brain TRP levels than the absolute plasma TRP concentration [5]. The relationship between plasma TRP levels and those of the other LNAAs also explains why foods which elevate brain TRP are thosewith high carbohydrate content rather than those that are particularly high in protein and TRP. This is because carbohydrate-rich meals promote insulin secretion which preferentially stimulates uptake of LNAAs other than TRP into tissue leading to their depletion in plasma and consequently an increase in the plasma TRP/LNAA ratio and therefore reduced competition for the uptake of TRP into the brain [5],[6]. Once in the brain, TRP is converted into 5-hydroxytryptophan (5-HTP) by the enzyme TRP hydroxylase; 5-HTP is then decarboxylated by the enzyme, aromatic acid decarboxylase to 5-HT. Under normal conditions the enzyme TRP hydroxylase is only about 50% saturated which means that the reaction this enzyme catalyses is the rate-limiting step. Serotonin synthesis is therefore dependent on the availability of TRP; increasing or decreasing its availability correlates highly with the amount of 5-HT produced [7].
Methods of reducing serotonin synthesis
From this discussion and Fig. 1, it is clear that three factors are important in determining the rate of brain 5- HT synthesis: (i) the concentration of free TRP in the plasma; (ii) how much of that free TRP crosses the BBB; and (iii) the activity of the TRP hydroxylase enzyme. Serotonin synthesis can be influenced by interfering with any or all of these factors.
Factors that determine the rate of serotonin (5-HT) synthesis. Points at which 5-HT synthesis may be controlled:1, TRP availability: dietary restriction;2, active transport: competition for the LNAA protein shuttle;3, synthesis: inhibition of tryptophan hydroxylase. Best acute tryptophan depletion (ATD) results are achieved using a combination of control at points 1 and 2.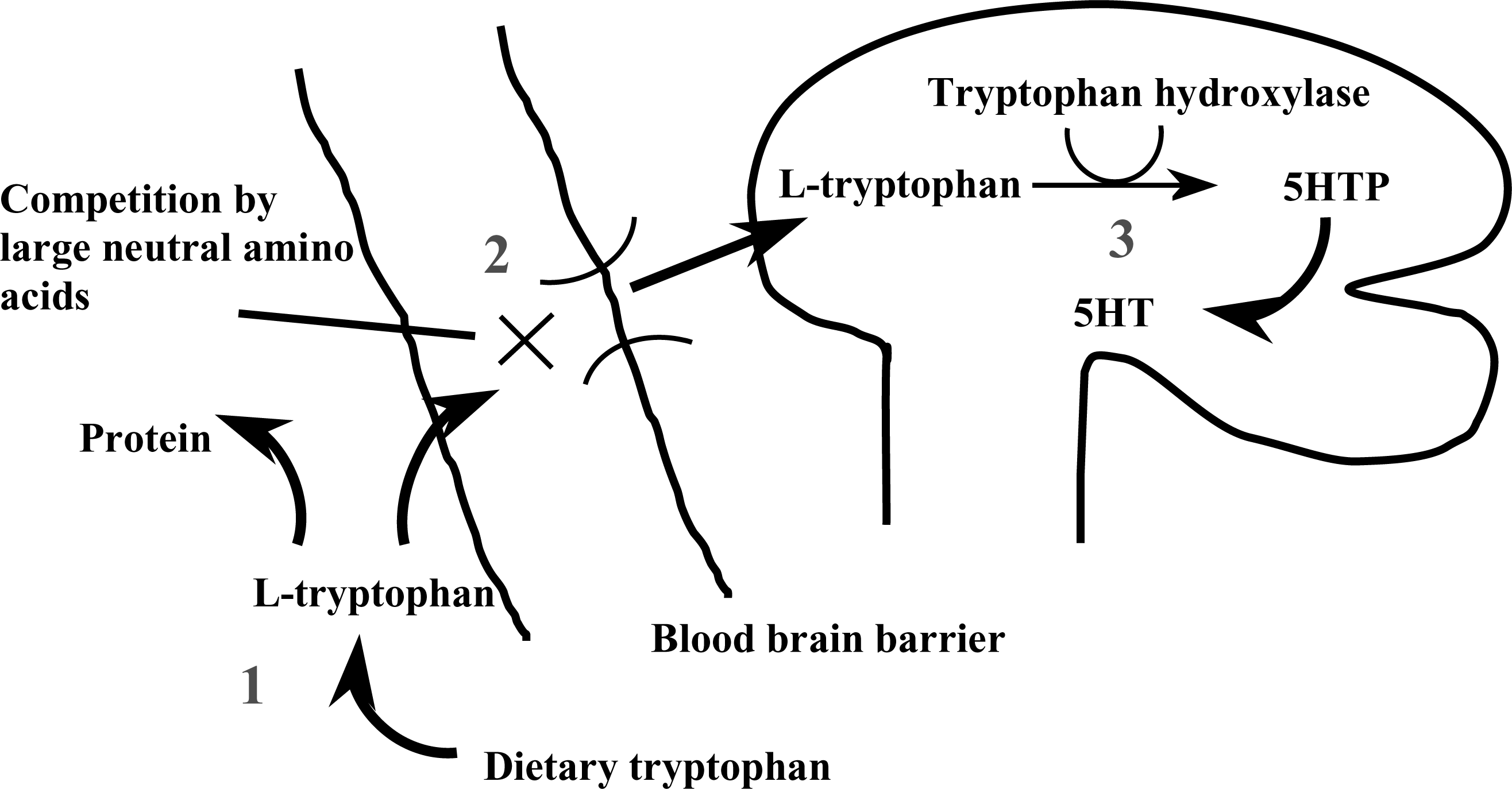
Early studies took the third approach, using para-chlorophenylalanine (PCPA), a drug that inhibits the enzyme TRP hydroxylase and therefore the hydroxylation of TRP to 5-HTP. This methodwas successful in reducing 5-HIAA (5-hydroxyindole acetic acid), the metabolite of 5-HT in urine, by 72–88% [8]. In two small studies of depressed patients who had responded to treatment with imipramine [9] or tranylcypromine [10], PCPA produced a transient return of depressive symptoms which remitted on discontinuation of PCPA. These preliminary studies suggested that both the tricyclics and monoamine oxidase inhibitors depended on 5-HT to exert their effect. Further studies were unfortunately not pursued because side effects such as fatigue, dizziness and constipation were marked and difficult to distinguish from those occurring during a depressive relapse. Para-chlorophenylalanine has also been reported to have a high incidence of allergic eosinophilia [11]. Subsequent interest has therefore focused on techniques influencing the first two factors.
Reducing TRP slowly by administration of long-term, low TRP or low calorie (1000–1200 kcal a day) and low carbohydrate diets has been shown to reduce plasma TRP levels by only 15–20% and to have few behavioural consequences in rats [12],[13] or human healthy volunteers [14]. In the human studies, women (but not men) were, however, reported to have an increased prolactin response to intravenous TRP infusion as a result of the dieting [15], possibly suggesting that women have an increased vulnerability to these effects. Longer periods of dieting were deemed both unethical and impractical. It was therefore concluded that a diet alone was not a strong enough technique to alter central 5-HT transmission acutely.
A much bigger fall in plasma TRP can be achieved by giving subjects an amino acid load which does not contain TRP, in the form of a drink. This amino acid load has two effects: (i) it stimulates protein synthesis in the liver, which uses up plasma TRP; and (ii) the amino acids that are given include the other LNAAs which compete with TRP for transport across the BBB and thus reduce the entry of TRP into the brain. The effect of giving an LNAA load alone has been demonstrated by studies in animals and humans using the LNAA valine with no other intervention. The animal studies reported reduced brain entry of TRP and decreased 5-HT release [16],[17]. The human study reported that plasma TRP/LNAA ratio was reduced, but the procedure resulted in only mild behavioural effects [17].
Acute tryptophan depletion
Acute tryptophan depletion is a technique that uses a combination of a low TRP diet and a TRP-deficient protein load containing large amounts of the other LNAAs to produce maximal brain TRP depletion [reviewed in 18].
The diet is typically 2500 calories, 48 g protein, 160 mg TRP/day and is consumed over the 24 hours before the test days [19]. After 10.00 pm on the night before the testing the subjects are allowed only to drink water. This overnight fasting extends through the test day until the experiment is over. This diet has been shown to reduce cerebrospinal fluid (CSF) TRP levels by 15–25% and is used to standardize dietary intake before and during the test days.
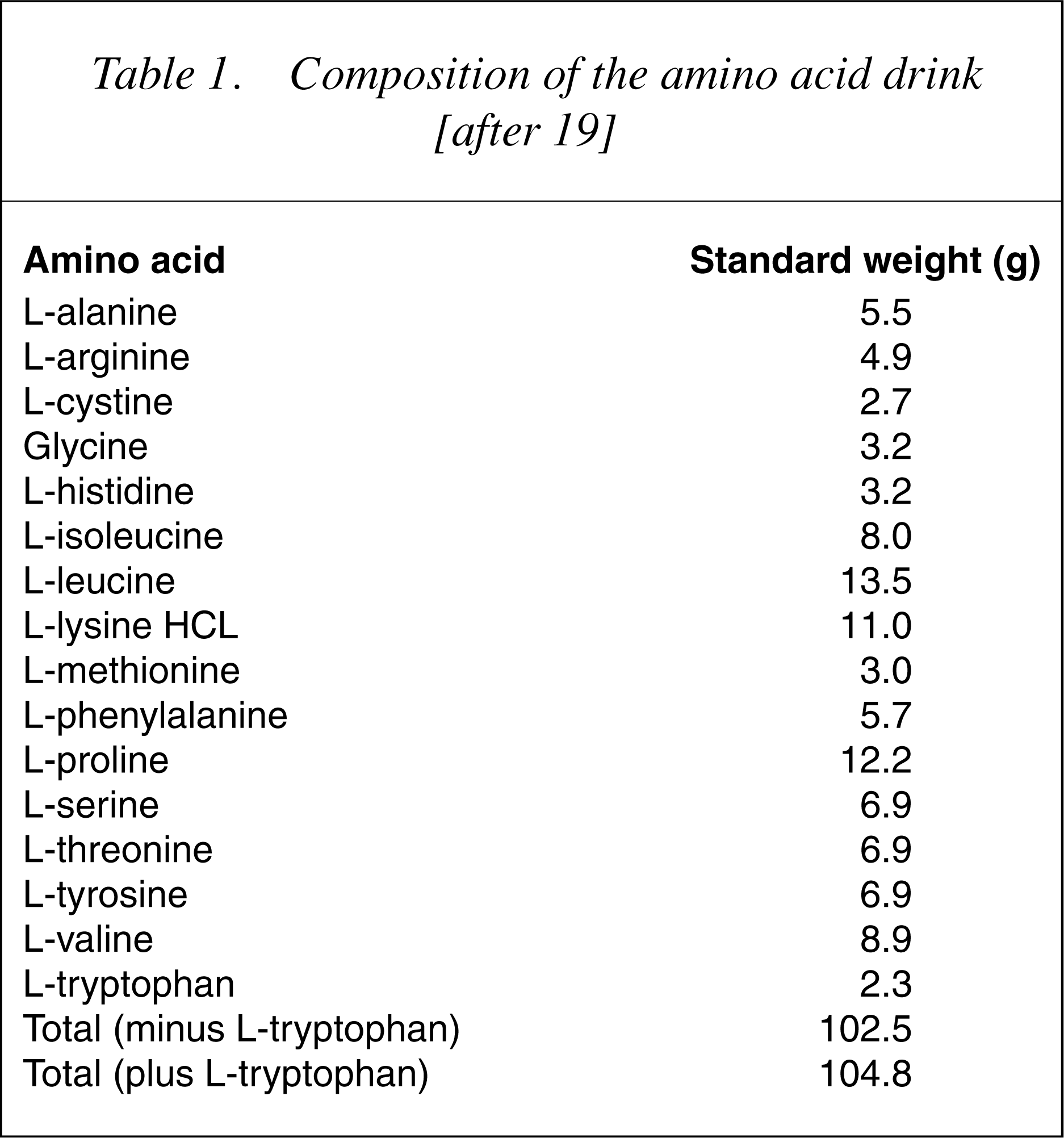
The amino acid drink contains 15 amino acids in the same proportion as human milk, except that TRP, aspartic acid and glutamic acid are omitted (the latter two because of concerns about toxicity) [20] (20]. In other cases, the amino acids that contain sulphur and have an unpleasant taste (methionine, cystine and arginine) are not included in the drink and are given separately in an encapsulated form. Some researchers take into account the lower average body weight of women and they reduce the amount of the total drink given to them to 80%.
Composition of the amino acid drink [after 19]
Recently, a gelatin-based mixture has become available for preclinical and clinical TRP depletion studies (Blokland, A., pers. comm. 2004). By using a gelatin hydrolysate in combination with carbohydrate, the nutritional mixture no longer consists of individual amino acids. Nevertheless, except for the deficiency of TRP, this gelatin-based protein comprises the entire range of amino acids in the form of peptides, as seen in normal food. Robust TRP depletion effects have been demonstrated in preclinical [21],[22] and clinical studies (Blokland, A., pers. comm. 2004). Such a mixture has the potential advantages of ease of preparation, high quality control and improved tolerance for subjects.
A common procedure for ATD involves two test days about 1 week apart in a double-blind, cross-over design. Subjects follow the dietary schedule described earlier. On one test day, subjects consume the amino acid drink (or the gelatin-based mixture) containing a 100 g load of 15 amino acids that does not contain TRP, and on the other test day they have a ‘control’ drink. The sequence of the TRP depletion and control tests are randomly assigned. Free and total plasma TRP levels and behavioural measures are recorded throughout the test days. On completion of the test days the subjects return to a normal dietary intake.
Following ingestion of the TRP depleting drink/gelatinbased mixture peak effects are usually seen at 5–7 hours which is the point of maximal depletion of plasma TRP. Tryptophan levels (both free and total) are reduced by 70–90% when a 100 g LNAA load or the gelatin-based mixture are used [23],[24]. Although not routinely reported, studies that measure the TRP/LNAA ratio report that this is reduced by a similar percentage.
In most studies the control drink contains 2.3 g TRP. This produces a marked increase in plasma TRP (100–300%). The plasma TRP/LNAA ratio is, however, decreased by 40–50% [25]. The increase in plasma TRP (total and free) produced by the control drink containing 2.3 g of TRP has generated some debate around whether this drink is indeed neutral or whether it might have a protective effect, perhaps even inducing 5-HT synthesis. However, as mentioned earlier, the TRP/LNAA ratio is reduced with both control drinks suggesting that increased TRP entry to the brain and stimulation of 5-HT synthesis would therefore be highly unlikely with either drink.
In the mid-1990s a quarter strength, that is, containing identical proportions of the amino acids as the original TRP depletion drink but only 25% by weight of each amino acid, was introduced [26]. This was because the Food and Drug Administration (FDA) in the US banned the use of TRP following an outbreak of eosinophilia myalgia syndrome associated with its use. This quarter strength drink causes smaller reductions in plasma TRP (40–50%) and similar reductions in the plasma, that is, 40–50% TRP/LNAA ratio [27],[28].
26] and has been used as such in several studies [27–29]. In these studies it has produced TRP depletions of 40–50% and has had little effect on mood. Very recent studies have used this lack of effect on mood to examine the changes to emotional and cognitive processing that occur with these smaller changes to TRP. These studies have shown that even small changes to TRP levels produce cognitive changes without causing depression [30].
Summary of effects of different drinks on plasma tryptophan (TRP) and TRP/large neutral amino acid (LNAA) ratio
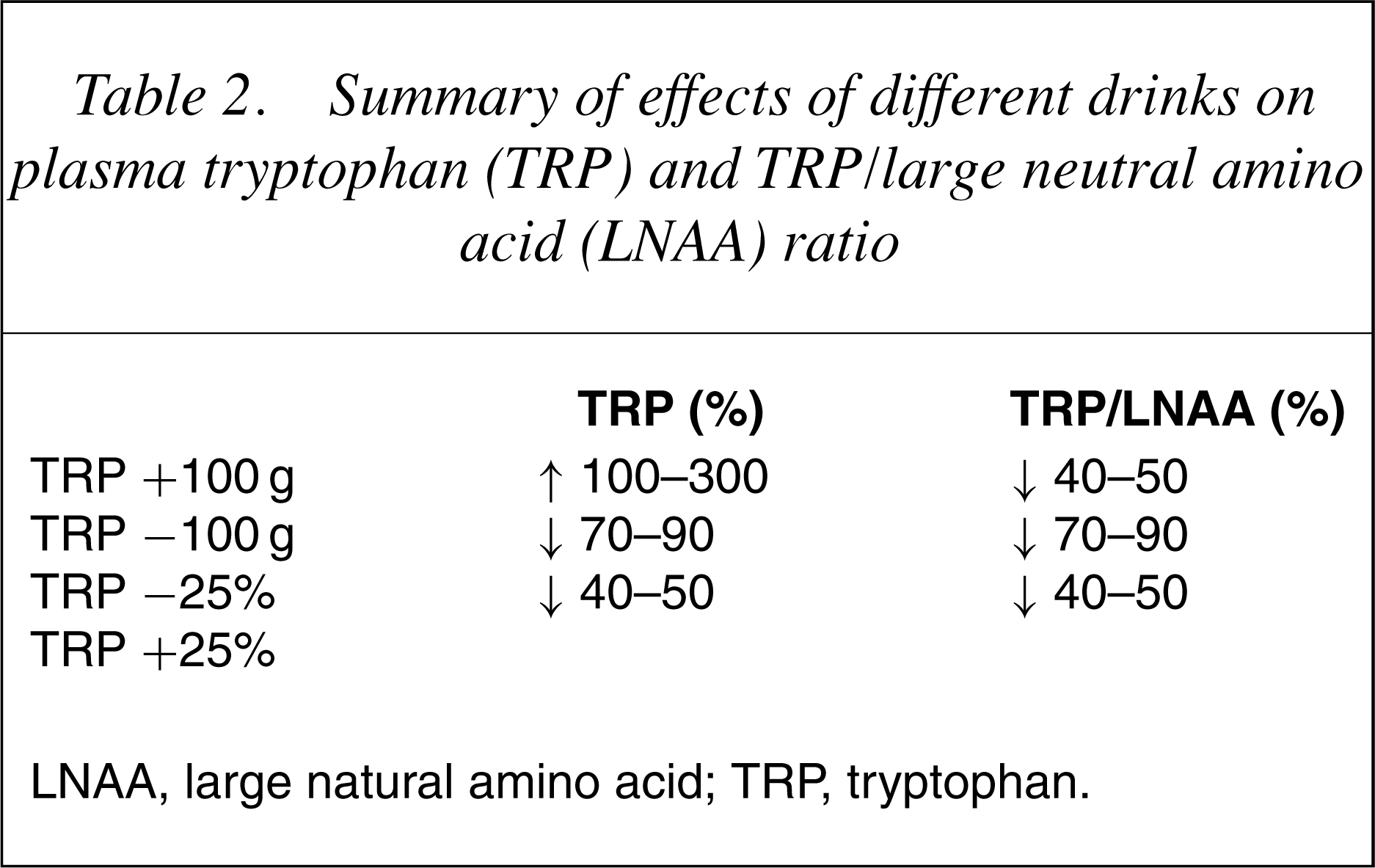
LNAA, large natural amino acid; TRP, tryptophan.
There has been debate about the relationship between the degree of plasma TRP depletion and the effects on mood. As described earlier, it does seem that greater reductions in plasma TRP have increased effects on mood. The effect does not, however, seem to be linear [31]. It has been argued that there may be a threshold effect, that is, mood effects are not produced unless about a 60% reduction in plasma TRP is achieved [29],[32].
Although not consistent, there is some suggestion that ATD produces greater reductions to plasma TRP in women compared with men [28],[31]. These trends have been reported in both healthy volunteers [33],[34] and patient groups [31].
It can be understood that there is a delay from the time plasma TRP reaches its minimum level (about 5–6 h after the ingestion of the drink) to the time of maximum central serotonergic effect. The maximum effect in the CSF TRP levels is seen from 7 to 10 hours, while CSF 5-HIAA continued to decrease after 12–14 hours following the onset of the experiment [35],[36]. This delay is an important consideration when designing an ATD study, therefore, the challenges and the measurements of their effects are usually performed at least 5 hours after the ingestion of the drink.
Effect of acute tryptophan depletion on serotonin release
The ATD paradigm is based on the hypothesis that reducing plasma TRP produces a consequent reduction in brain 5-HT synthesis and release. This has been confirmed by a large body of preclinical data showing that depletion of TRP specifically reduces brain 5-HT content and function. Ingestion of TRP-free amino acid mixtures in laboratory animals and monkeys [37] has been shown to produce marked reductions in plasma TRP and brain 5-HT content [38–40]. Acute tryptophan depletion has also been shown to alter behavioural indices of 5-HT function (increasing pain sensitivity, acoustic startle, motor activity and aggression), effects which are reversed once brain TRP is restored. In rats, in vivo microdialysis studies also showed that the peripheral reduction of TRP during ATD leads to a lowering of hippocampal 5-HT release, while increased TRP ingestion has the opposite effect [16],[41],[42]. Changes may be most marked when 5-HT levels are enhanced, for example, in presence of a selective serotonin re-uptake inhibitor [43].
The evidence that ATD produces a decline in central 5-HT release from human studies has until recently been more circumstantial than the animal literature. Recent reports involving continuous CSF sampling in healthy volunteers have shown that ATD reduces central levels of TRP by 80–90% [35],[36]. 5-HIAA levels (reflecting 5-HT turnover) aremoremoderately reduced by the technique, that is, by 24–40%. The time courses of the central responses were also slightly delayed compared with the plasma findings, with the nadir for CSF TRP being 7–10 hours post-ATD and even longer (12–14 h) for 5- HIAA. There has been one study examining the rate of 5-HT synthesis in vivo using positron emission tomography (PET) that has reported more dramatic effects of ATD than those described earlier with reductions in the rate of synthesis of a factor of about 9.5 in men and about 40 inwomen [33]. The discrepancies between these studies may be explained by the different techniques employed, that is, the CSF studies measure 5-HT catabolism and the PET study 5-HT synthesis.
Effects of acute tryptophan depletion: findings from imaging studies
Four studies have examined the effects of ATD on brain function using PET. Two were in medicated patients remitted from an episode of depression and reported similar findings with ATD resulting in decreased metabolism in the orbitofrontal, cingulate and parietal cortex and the caudate nucleus [44],[45]. One study in remitted patients on no medication for at least 3 months reported increased metabolism in similar (although not identical) brain regions [46]. The discrepancies in the direction of change and the circuitry involved may be explained by the effects of medication in the earlier studies. It is interesting, however, that the areas identified are those traditionally associated with depression [47]. It is not clear at present whether these changes occurred as a result of ATD or whether they are specific to (recovered) depressed patients. Another recent study in healthy women reported that 5-HT2 receptor binding showed a significant decrease following ATD [48]. The study reported no mood effects and the authors suggested that this downregulation was adaptive and protected against depressive symptoms.
Effects of tryptophan depletion: findings from neuroendocrine studies
In healthy volunteers, ATD has been shown to attenuate the prolactin response to a D-fenfluramine challenge indicating that ATD reduces 5-HT levels in the releasable pool of 5-HT in the presynaptic neurone [49]. There is a suggestion thatATDmay have greater behavioural effects in subjects with impaired 5-HT1A autoreceptor function [50]. This may be because the presynaptic 5-HT1A autoreceptor works homeostatically to normalize synaptic concentrations; reduced function may result in TRP manipulations producing more prolonged changes to synaptic 5-HT levels.
Two neuroendocrine studies in depressed patients have examined the effect of ATD on prolactin, cortisol and growth hormone responses to challenges with IV-mchlorophenylpiperazine (m-CPP) [51] and IV-TRP [52]. These reported some enhancement of hormonal responses suggesting that ATD results in postsynaptic receptor supersensitivity involving 5-HT2 and possibly 5-HT1A receptors.
The effects of dietary restriction on central 5-HT function have also been investigated using neuroendocrine challenges with IV-TRP [14],[15],[53], m-CPP [54] and fenfluramine [55]. These reported reduced plasma TRP levels and increased endocrine responses following dieting in healthy volunteers, particularly women, suggesting adaptive upregulation of postsynaptic mechanisms in response to impaired presynaptic 5-HT release. One of these studies which included a group of women with a past history of depression reported that, although dieting produced a similar reduction in plasma TRP levels in women with and without a history of depression, there was no increase in endocrine responses in the group who had previously been depressed, suggesting that women with a vulnerability to depression do not respond to decreased precursor availability by upregulating 5-HT pathways [53]. The precise mechanism underlying the increased endocrine responses following dieting have not been established. These studies suggest the involvement of postsynaptic 5-HT2 (and possibly 5-HT1A) receptors. Animal studies report that a low TRP diet causes an upregulation of 5-HT2 receptor function [56].
These results suggest that ATD (or a low TRP diet) challenges the 5-HT system that results in a compensatory change to 5-HT receptors in healthy controls. In contrast, vulnerable individuals, for example, those with depression (or a past history of depression) may not be able to mount this compensatory response, reflecting a biological vulnerability of the 5-HT system.
Are there non-serotonergic explanations for the effects of acute tryptophan depletion?
There are some caveats to the assumption that the effects of ATD are explained by effects to central 5-HT, only some of which have been addressed.
The effects of ATD do seem to be related to changes in 5-HT metabolism rather than changes to other neurotransmitters (e.g. catecholamines). Although the LNAA load produces an increase in plasma and CSF tryosine, changes in other LNAAs result in the tyrosine/LNAA ratio remaining stable. Studies have reported that measures of metabolites of dopamine, that is, homovanellic acid (HVA), are not changed [35] therefore a direct effect on dopamine transmission is very unlikely [57],[58]. A modulatory effect on other transmitters resulting from the alterations in 5-HT function, however, cannot be excluded.
In addition to being the precursor of 5-HT, TRP is also required for the synthesis of many proteins (including various hormones and neuropeptides) and nicotinamide via the kynurenine pathway. It is possible that reducing TRP has effects on the synthesis of these products, which might also be partly responsible for the effects of ATD.
Another question has been whether the effects of ATD could be explained by changes to brain protein synthesis consequent on the amino acid load. One study has addressed this by examining the effects of an amino acid mixture deficient in lysine rather than TRP, which also results in increased protein synthesis and reported no effects on mood or memory compared with ATD [59]. One can therefore conclude that ATD is a technique specific in manipulating central 5-HT function.
Conclusions
Despite recent advances in neuroimaging and limitations of validation in human populations, ATD remains a useful psychiatric research tool for exploring 5-HT systems. Acute tryptophan depletion is an active topic of discussion at major international symposia almost two decades after its introduction [60],[61]. It remains a relatively inexpensive, non-invasive method of serotonergic manipulation that is acceptable to human populations [27].
This article has explained the psychopharmacological rationale of this technique and described current methodology. The second part of this series will review the application of this procedure in clinical populations.
