Abstract
The reasons why some individuals, when exposed chronically to neuroleptic drugs, develop tardive dyskinesia (TD) are still incompletely understood [1]. It has been hypothesized that the occurrence of TD may reflect a vulnerability to the development of extrapyramidal side-effects (EPSE) in general, evidence for which could come from the presence of greater early neurolepticinduced EPSE in those who later develop TD [2]. Direct investigations of this hypothesis have been few, with some [3–5] but not all [6] studies supporting it. Kane and colleagues [3–5] showed in two prospective studies that the occurrence of EPSE was associated with a threefold increase in the risk of TD. Jus et al. [6] obtained retrospective histories on 300 patients and found no difference of acute EPSE in those with and without TD.
A suggested variation of this hypothesis, which has not been directly examined, is that patients with hyperkinetic (i.e. akathisia) rather than hypokinetic (i.e. bradykinesia and rigidity) EPSE are more likely to develop TD [7]. DeVeaugh-Geiss [8] suggested that akathisia may in fact be a stage in the progression from parkinsonism to TD. There have been some case reports of acute akathisia proceeding to the development of TD [9], [10], but no prospective study has examined this. Moreover, there may be a dissociation between oro-facial and limb-truncal dyskinesia, with the latter being better predicted by early EPSE [1]. This report is of a prospective study of patients, first investigated when they were initiated on conventional neuroleptics, to examine the above hypotheses, with an assessment of both early parkinsonism and akathisia.
Method
Subjects
One hundred patients, admitted consecutively to a psychiatric unit with a primary psychotic disorder and free of neuroleptic medication at baseline, were studied intensively for 2 weeks for the development of akathisia and EPSE as reported previously [11]. They were followed up longitudinally and 78 subjects completed the study, with a mean (SD) follow-up of 41.2 (5.9) months (range 28–52 months). All subjects provided written informed consent and the study was approved by the Ethics Committee of the South-Eastern Sydney Area Health Service.
Baseline assessment
Baseline assessment has been described previously in detail [11]. The preneuroleptic assessment comprised a full psychiatric evaluation, the Abnormal Involuntary Movement Scale (AIMS) [12], the Simpson-Angus Scale for Extrapyramidal Side-effects (EPSE) [13], the Prince Henry Hospital Akathisia Rating Scale (PHH-ARS) [14] and the Brief Psychiatric Rating Scale (BPRS) [15]. The EPSE and PHH-ARS ratings were repeated at 1 and 2 weeks after the initiation of neuroleptic treatment. Akathisia was diagnosed if the score on PHH-ARS was > 4.
Follow-up assessments
The subjects were routinely followed at the community health centres affiliated with the hospitals. They were assessed by the research team whenever a hospital admission occurred (mean number of repeat admissions 2.6, range 0–13, with 23 subjects 29.5% having no further admissions) with the rating scales being repeated. They were formally assessed 2–4 years after initial assessment for the development of TD using the AIMS examination. Subjects meeting diagnostic criteria for TD [16] were re-examined within a month to confirm the diagnosis.
Analysis
A diagnosis of TD was made if the subject met Schooler-Kane research diagnostic criteria [16] on two occasions and the informant confirmed the presence of dyskinetic movements. The TD and non-TD groups were contrasted on relevant variables using independent sample t-tests, after testing for equality of variance, for parametric and χ2 tests for categorical data (Table 1), using 2-tailed tests with α = 0.05. A logistic regression model was examined for presence or absence of TD entering four variables simultaneously (median neuroleptic dose, duration of neuroleptic use, severity of EPSE and akathisia severity at day 14). Because of the significant correlation between EPSE and akathisia scores (r = 0.3, p < 0.01), and our interest in akathisia, a second model to test the importance of akathisia was examined with the variables akathisia (presence/absence) in the first 2 weeks, akathisia severity on days 7 and 14, and CPZE load.
Socio-demographic and clinical data for subjects with (TD) and without (NTD) tardive dyskinesia at follow-up
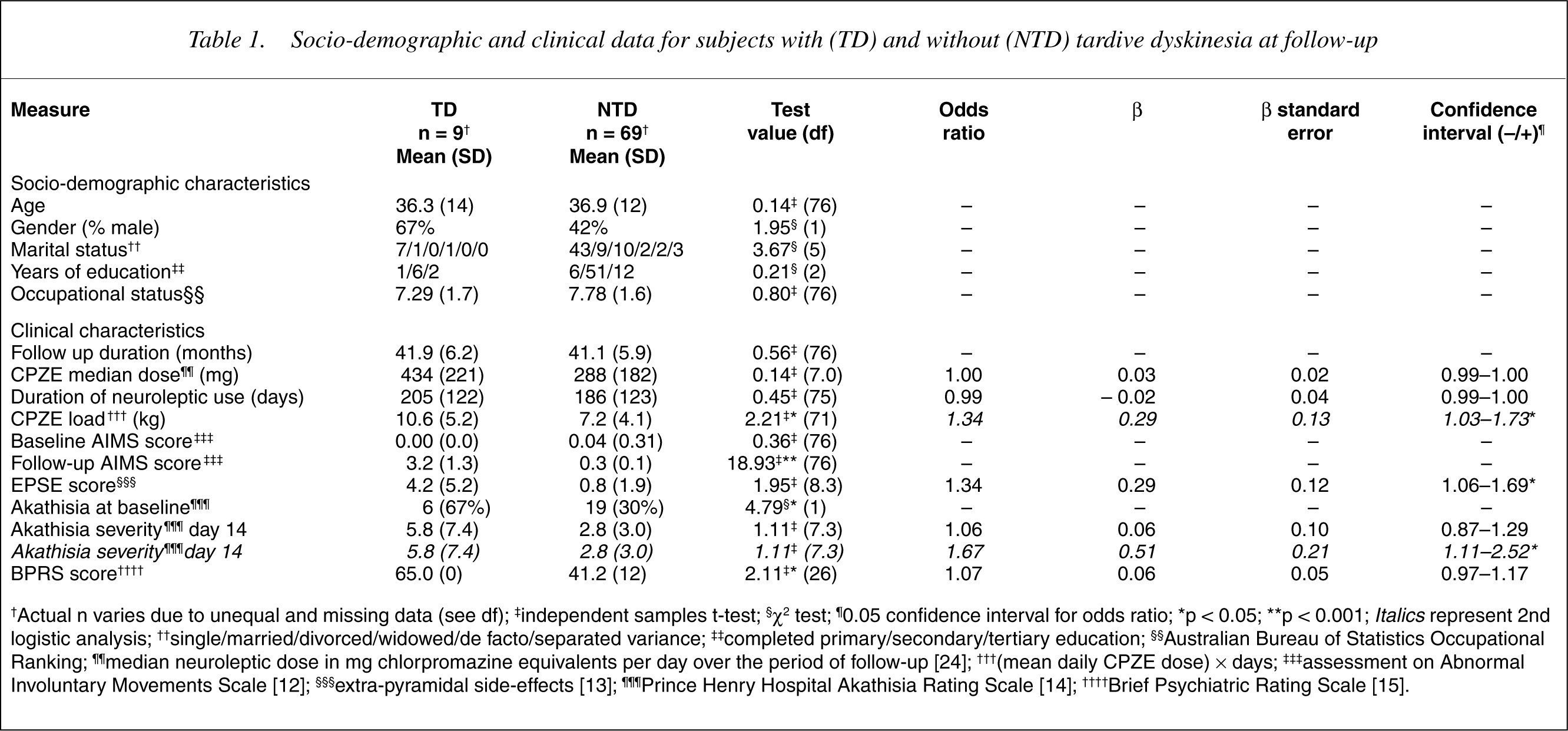
Results
The 78 subjects with follow-up data were compared to the 22 without. The two groups did not differ significantly on age (t = 0.11, df = 98, p = 0.91), sex ratio (χ2 = 0.03, df = 1, p = 0.85), marital status (χ2 = 1.66, df = 2, p = 0.44) and baseline BPRS scores (t = 0.53, df = 98, p = 0.59). The followed-up sample had a mean age 36.8 years (SD = 11.9), with 35 men (44.9%), 14 (17.9%) with tertiary education, and 27 (34.6%) currently employed. The DSM-IV primary diagnoses were: schizophrenic disorder, 60 (77%); other psychoses, 12 (15%); and bipolar disorder 6 (8%). The subjects were followed up for a mean 41.9 months (SD = 5.9) with the TD and non-TD groups not being different in the duration of follow-up (t = 0.56, df = 76, p = 0.72).
All subjects were maintained on conventional neuroleptics, except six subjects who were switched to clozapine at a mean 26.2 months after initial assessment. The neuroleptic drugs used, in order of frequency, were: haloperidol (n = 40); fluphenazine decanoate (n = 15); flupenthixol deconoate (n = 12); trifluoperazine (n = 16); chlorpromazine (n = 14); thioridazine (n = 10); clozapine (n = 6); and pimozide (n = 3). The neuroleptic use was continuous in 28 (36%) and intermittent in the rest. The total neuroleptic load was estimated based on the total duration of actual use and the median dose for the subjects. Anticholinergic drugs were used by 40 (51.3%) subjects at some time during follow-up, and 22 (28.3%) were on an anticholinergic drug at the time of final assessment. The reasons for switching to clozapine were the development of TD in two subjects and resistance to conventional drugs in the other four, two of whom also had TD.
Nine (11.5%) subjects were diagnosed to have TD, with mild severity in six (7.7%) and moderate in three (3.8%). Eight had oro-facial dyskinesia, with two additionally affected with limb-truncal TD. One subject had limb-truncal TD only. Three of these subjects had acute akathisia at baseline. All of these went on to have subsequent episodes of akathisia, with akathisia being chronic in one subject. The total neuroleptic load was higher for the TD group (t = 2.21, df = 71, p = 0.03). The correlations between EPSE and total akathisia scores at 1 week and 2 weeks of neuroleptic treatment were 0.30 and 0.30, respectively.
A logistic regression analysis, entering four variables simultaneously, indicated that EPSE scores at baseline were significant predictors of TD group membership, with the odds of being in the TD group increasing by 34% for a single unit increase in EPSE scores. The model correctly classified 100% of non-TD but only 33% (3 out of 9) TD subjects, corresponding to an overall accuracy of 92.3% (χ2 = 12.48, df = 1, p < 0.05). In the second logistic regression model without EPSE scores, akathisia severity on day 14 and CPZE load were significant predictors of non-TD (presented in italics in Table 1). The model correctly classified 60/61 (98%) non-TD and 4/8 (50%) of TD subjects, an overall accuracy of 93% (χ2 = 20.87, df = 1, p = 0.004).
Discussion
The study provides empirical evidence for the hypothesis that acute EPSE is a predictor of the later development of TD. This was true for both parkinsonism and akathisia, although only the former was significant in a regression model that contained both variables. The TD seen in these subjects was predominantly oro-facial, but limb-truncal TD was also represented, the number of the latter being too small to perform a separate analysis. The increase in risk is relatively small, however, with an OR of 1.34, and is lower than suggested in some earlier studies [3–5].
The strength of the study is that subjects were neuroleptic-free at baseline, even though the majority (83%) had previously been exposed to neuroleptic drugs [11]. They were free of dyskinesia and EPSE before the initiation of the drugs and were followed up intensively for 2 weeks after initiation. A limitation is that the subsequent follow-up was irregular, but it was frequent, and the proportion who developed TD in the mean 41.2 months of follow-up was consistent with the incident rates of TD previously published [1], [3], [5]. The subjects lost to follow-up did not differ significantly on clinical characteristics from those included in the study, thereby reducing the likelihood of bias. Another limitation is that the study subjects were treated with typical neuroleptics and the findings therefore cannot be extended to patients being treated with atypical drugs.
We had hypothesized that akathisia and parkinsonism would be independent predictors of TD. The pathophysiology of akathisia is arguably different from that of parkinsonism [11]. While the latter is clearly related to the antagonism of dopamine D2 receptors in the striatum, the basis of the former is incompletely understood, with frontal cortical dopamine antagonism being one hypothesis [17]. Parkinsonism and akathisia had a significant correlation in our study, and we performed a second regression analysis to examine akathisia separately. Moreover, the rates of acute akathisia in the two groups were significantly different (67% vs. 30%), which supported our decision for a second analysis.
Our study does not suggest that drug-induced parkinsonism and akathisia differentially predict TD. The theoretical implication of this is that these acute and delayed side-effects may reflect a general vulnerability to extrapyramidal reactions to neuroleptic drugs. The study does not suggest a definitive mechanism by which this may happen, but many possibilities can be raised. These include individual differences in drug-handling [18], concomitant brain pathology [19], genetic polymorphisms underlying neuroleptic-sensitivity [20] and so on. Acute and tardive side-effects have been considered to reflect different, even opposing, pathomechanisms such as dopamine receptor blockade and hypersensitivity, respectively, but their frequent cooccurrence in patients suggests that this may be too simplistic an explanation [21]. Recent evidence that TD subjects have abnormal caudate volume [22], and that an increase in caudate volume is in fact an acute effect of neuroleptic medication [23], is one lead to understanding pathomechanisms common to acute and tardive sideeffects. At the synaptic level, sensitivity to dopamine blockade may also relate to increased dopamine turnover, increased supersensitivity, or other mechanisms that have been implicated in the pathophysiology of TD [1].
Our study may have some clinical implications. While it is impossible from the presence of acute EPSE to predict which individual will definitely go on to develop TD, the presence of severe EPSE early in the treatment should alert the clinician to an increased risk. Such patients should arguably be managed with lower doses of antipsychotic drugs, preferably atypical ones.
Footnotes
Acknowledgements
I thank Celia Loneragan, Jane Kruk, Stuart Cathcart and Angie Russell. The study was supported by grants from the National Health and Medical Research Council of Australia and the Rebecca Cooper Foundation.
