Abstract
In the last decade several studies demonstrated that comorbid cannabis abuse worsens the course of schizophrenia [1]. In patients with an established psychotic disorder cannabis abuse is associated with a higher risk of psychotic relapses [2] and with poorer social outcome [3]. Many patients continue to consume cannabis products in spite of their clinicians' efforts to educate them about the consequences. Systematic surveys suggest that patients purposefully seek the psychotropic effects of cannabis. They experience cannabis as relaxing, mood-elevating, and distancing from life's problems just as do non-schizophrenic cannabis abusers [4].
At least in chronic samples, high comorbidity rates between cannabis misuse and schizophrenia were found [5]. In different settings within the United States, the rates of cannabis use or abuse in patients with schizophrenia varied between 31% [6], 32% [7] and 41.8% [8], depending on the inclusion criterion of abuse versus use. Much lower rates were reported from Germany: in a sample of 630 consecutive admissions of patients with schizophrenia, Soyka et al. [9] found prevalence rates of cannabis abuse or dependence of about 5% (lifetime) and 3.5% (3-month prevalence). These data illustrate that transnational differences in comorbidity rates are relevant in substance abuse disorders. Secular trends have to be considered, too. From 1990 to 1992 the rates for cannabis and stimulant abuse were found to decrease in USA samples, while the rate of cocaine abuse increased [10,11].
Large population surveys in the United States have been published which report on abuse or dependence of illicit drugs in general. In the Epidemiologic Catchment Area Study 27.5% of the individuals with a lifetime diagnosis of schizophrenia had a comorbid drug abuse disorder [12]. Marijuana abuse and/or dependence was associated with a significantly increased odds ratio of 4.8 for schizophrenia. The National Comorbidity Survey [13] reported a lifetime comorbidity rate of 44.8% of non-affective psychosis and drug abuse and/or dependence. Comorbidity rates with cannabis abuse alone can be estimated to be between 15% and 25% (based on the reported clinical samples).
In summary, cannabis abuse has been reported in 5% to 40% of all schizophrenic patients, and sampling and other methodological differences should be considered. Epidemiological research strongly depends on the validity of the employed diagnostic categories. This is a particular problem for substance abuse. DSM-IV as well as ICD-10 give rather broad, unspecific, and hardly operationalised categories for this condition [14]. Diagnostic validity is further challenged by the fact that substance abuse can be particularly difficult to detect in patients with schizophrenia [15,16].
The clinical observation of relapsing, cannabisabusing schizophrenic patients and of self-medicating schizophrenic patients mirrors the controversial discussion about the reasons for the high comorbidity rates. Several hypotheses have been proposed (e.g. [17–19]). They belong to two logical groups postulating different directions of causality: (i) causation models state that substance abuse precipitates or causes schizophrenia, at least in vulnerable individuals (e.g. [10,11,20]) and (ii) self-medication models postulate the opposite direction of causality: patients use drugs to counter distressing symptoms of the illness or side-effects of the therapy (e.g. [5,6,21,22]). Hambrecht & Häfner [23] reviewed the methodology of these studies: some lacked adequate control groups, and some investigated small, selected and often hetergeneous samples with regard to the stage of the disorder (i.e. first admissions vs chronic cases).
A few studies investigated the temporal sequence of the onset of schizophrenia and cannabis abuse in order to test the direction of this relationship. A Swedish case register study [17] investigated 112 patients with a diagnosis of schizophrenia and with a diagnosis of cannabis dependence. In 69% of these cases, heavy cannabis abuse had occurred at least 1 year before the onset of psychotic symptoms. The reverse order was seen in only 11% of the sample. A Dutch study [2] found that in 23 out of 24 cannabisabusing, recent onset patients with schizophrenia the abuse preceded the onset of the first psychotic symptoms by at least 1 year.
These sophisticated and important studies, however, have a shortcoming: defining the onset of schizophrenia as the onset of psychotic symptoms oversimplifies the onset of schizophrenia. The large majority of schizophrenic disorders begin insidiously. Stage models ([24] for review) conceptualise psychotic symptoms as the last step in the development of the disorder. The onset of schizophrenia, therefore, should be seen as a gradual process rather than as a single point in time.
The aim of the present study was to examine the prevalence, clinical characteristics and temporal sequence of schizophrenia and comorbid cannabis abuse at the very onset of psychosis in order to contribute to the understanding of a possible causality between the two disorders.
Methods
The operationalisation of the different stages in early psychosis was one major focus of the Age, Beginning and Course (ABC) of Schizophrenia Study [25,26]. The ABC Study investigated a representative sample of 232 first admissions with first episode of schizophrenia or paranoid disorder (i.e. ICD-9 definitions: 295, 297, 298.3/298.4). Between 1987 and 1989, 392 persons (age 12–59 years, German citizenship, no organic brain syndrome or severe mental retardation) had been admitted for the first time with one of these diagnoses to the psychiatric hospitals serving the defined catchment area (population 1.5 million). Due to refusals or too short hospitalisations, 125 patients (32%) could not be completely assessed, but participants and non-participants did not differ significantly with regard to sociodemographic variables and clinical diagnosis. Among the participating 267 patients, 232 (87%) had just experienced the first psychotic episode and were included in the present analyses. Thirty-five patients (13%) were excluded because of an earlier psychotic episode.
The presence of a first psychotic episode was identified by means of a structured clinical assessment. The previous history of any sign or symptom of mental disturbance was carefully investigated with special emphasis on the exact timing of the first appearance of the symptoms. This was accomplished with the Interview for the Retrospective Assessment of the Onset of Schizophrenia (IRAOS) [27]. This semistructured interview was developed on the basis of the Present State Examination [28], the Scale for the Assessment of Negative Symptoms [29], the Disability Assessment Schedule [30] and other instruments. It retrospectively assesses the presence, onset and course of 66 symptoms during the early course of schizophrenia. The IRAOS determines the points in time, when the first symptom of schizophrenia, the first positive and the first negative symptom had emerged. The definition of the ‘first symptom of schizophrenia’ takes the differing specificity of symptoms for schizophrenia into account: Non-specific symptoms were only considered as ‘first symptoms’ if they persisted continuously into the psychotic episode, negative symptoms were considered if they continued into or recurred in the psychotic episode, positive symptoms were always considered as ‘first symptoms’ if they appeared earlier than other symptoms.
Patients' and relatives' reports on these ‘milestones’ during the early course of schizophrenia correlated significantly (e.g. Pearson correlation of 0.77 for the first symptom, 0.93 for the first positive symptom, 0.73 for the first negative symptom) demonstrating the validity of the data [26]. A history of drug abuse was also reported quite congruently by patients and relatives as demonstrated by a kappa of 0.52 and a Pearson correlation for first onset of drug abuse of 0.69 [31].
Within the IRAOS a special section addresses substance misuse, differentiating substances and the frequency of their consumption during various intervals. Drug abuse was defined as consumption of illegal drugs more than once a week over at least 1 month. The onset of drug abuse was defined as the first month in which the criteria for abuse were fulfilled. If the patient quit substance abuse for at least 1 month and then started again, two separate episodes of substance abuse were rated.
For an estimate of the base rate of substance abuse in this region, a control group was drawn from the general population, matched to those 57 first-episode patients who lived in the city of Mannheim, the centre of the catchment area. In 1991, for every patient four controls of the same age and sex were randomly selected from the files of the municipal registration office and contacted by telephone in random order until the first agreed to participate in the interviews. (About one-third of the contacted persons refused to participate.) The 57 controls had no history of schizophrenia or paranoid disorder and were carefully interviewed with the IRAOS. A history of cannabis abuse was found in four controls (7.0%). A history of alcohol abuse was found in 12% of the controls, but no other substance abuse.
Results
A lifetime history of drug abuse was found in 33 patients with a first episode of schizophrenia (14.2%). This was twice the rate of drug abuse in matched controls. The difference missed statistical significance due to the small size of the control sample. Cannabis was prominent among abused drugs (Table 1). Almost 90% of illicit drug abusers had abused cannabis at some time, 37% had taken solely cannabis in their substance-abuse history. Other relevant drugs were hallucinogens, cocaine and stimulants. Multiple substance abuse was frequent, particularly concomitant alcohol abuse by drug-abusing patients.
Substances abused by first-episode schizophrenic patients
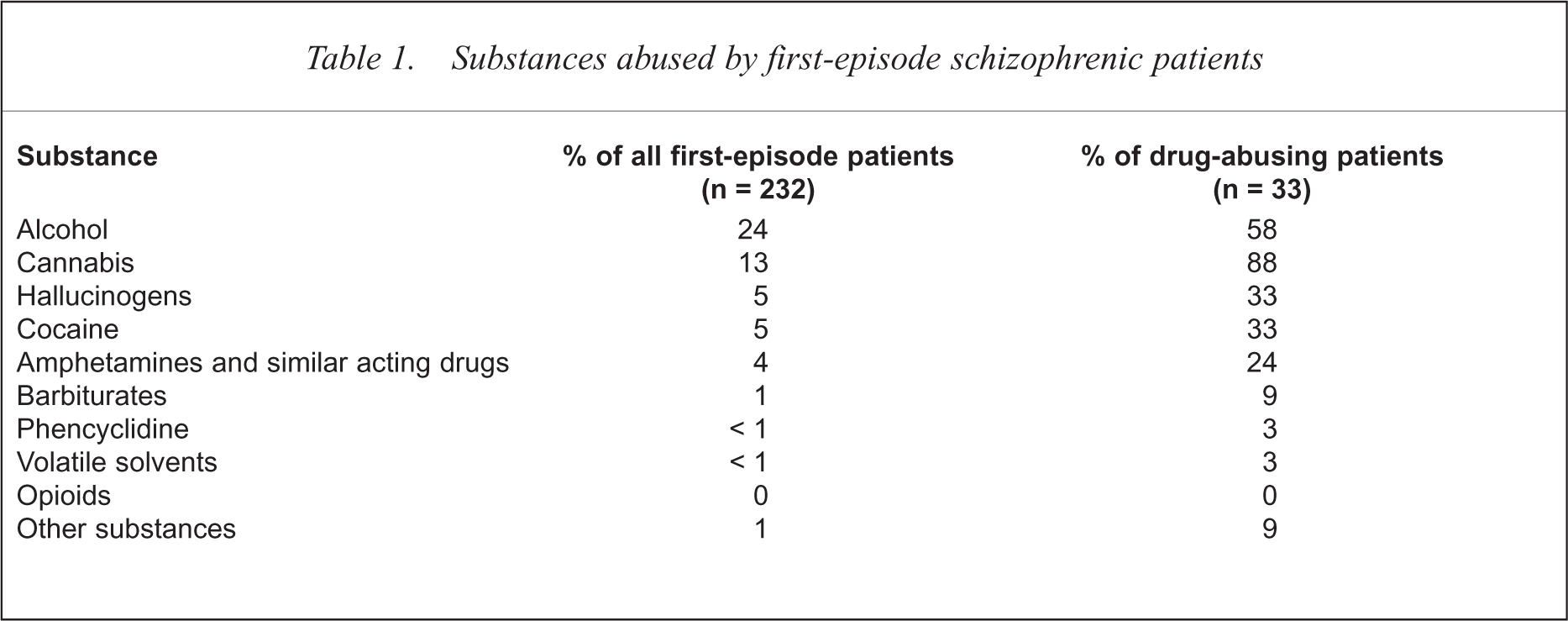
About 60% of the patients with a history of drug abuse reported one or two episodes of drug abuse, while 40% had had three or four episodes. A typical pattern of consumed substances was found during these episodes. Again, cannabis was most used. Within a first (or single) episode of abuse, 80% of the drug-abusing patients took cannabis. Of those drug-abusing patients who reported a second episode of abuse, 39% consumed hallucinogens. Among those who changed the substance from the first to the second episode, 53% changed to hallucinogens. Cocaine dominated the abuse pattern when it came to a third episode of abuse, with 62% then consuming cocaine.
Male sex, younger age of onset, unemployment, comorbid alcohol abuse and the catatonic subtype of schizophrenia were significantly associated with drug abuse in first-episode schizophrenic patients (Table 2).
Sociodemographic and diagnostic characteristics over-represented in schizophrenic patients with comorbid drug abuse

Four cases with catatonic subtype of schizophrenia in the total first-episode sample (two cases with comorbid cannabis abuse and two cases without a history of substance abuse).
First-episode schizophrenic patients with a history of drug abuse were compared with randomly selected, age and sex matched, first-episode patients without such a history (Table 3). The two groups differed in a number of symptoms prior to first admission as assessed by the IRAOS. As expected from the literature, depression was more often seen in drug-abusers than in non-abusers. Negative symptoms did not differ between non-abusers and abusers, but social behaviours in many ways did. Substance abuse is associated with social conflicts and antisocial behaviours. Drug-abusing schizophrenic patients more often report about occultism and similar sub-culturally influenced magical ideas. Clinical experience indicates that this pattern of asociality and affiliation with sub-cultural groups fosters a worse outcome in substance-abusing schizophrenic patients, partly mediated by continuous substance abuse, partly mediated by non-compliance with antipsychotic medication.
Differences in symptomatology between schizophrenic patients with (n = 33) and matched patients without (n = 33) drug-abuse history

p < 0.05,
p < 0.01 (χ2). IRAOS, Retrospective Assessment of the Onset of Schizophrenia.
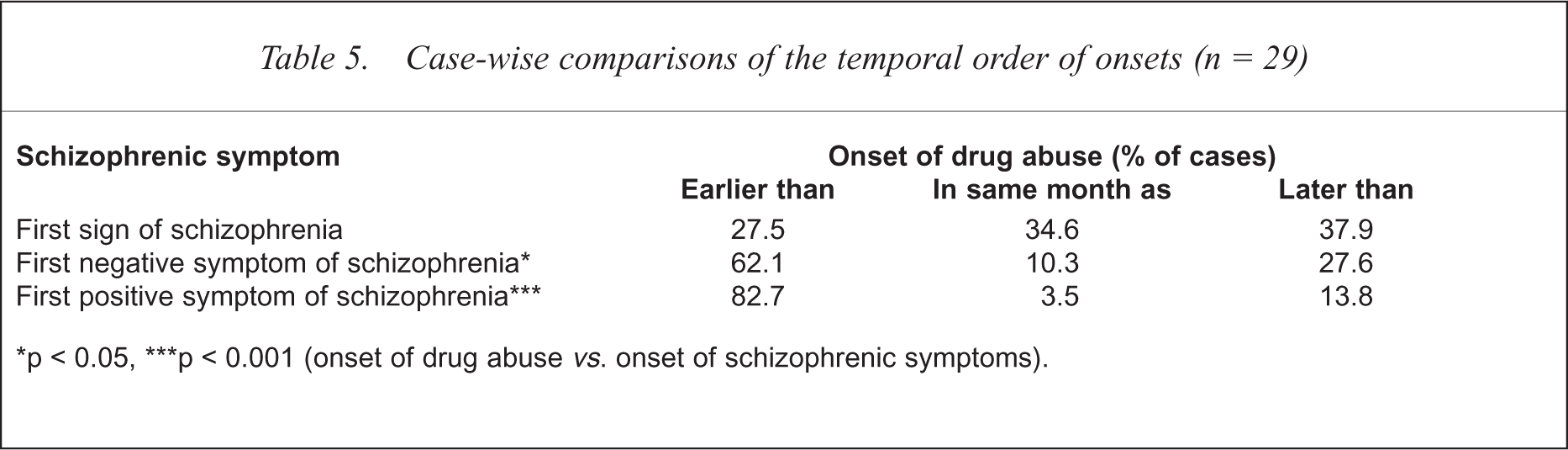
A precise assessment of the temporal order of onsets requires a differentiation of the relevant points in time during the early course of schizophrenia, namely first sign, first negative and first positive symptom. The temporal sequence of the first occurrence of these events, and of the onset of drug abuse, was compared group-wise for all comorbid cases (t-test for paired samples) and also individually in each patient (Wilcoxon signed rank test). The results are shown in Tables 4 and 5. The mean age of onset of drug abuse did not differ from the mean age at the first symptom of schizophrenia, but occurred significantly earlier than the first negative symptom and earlier than the first positive symptom.
Comparison of mean ages at onset of drug abuse and schizophrenic symptoms in substance abusers (n = 29)

p < 0.05,
p < 0.001 (onset of drug abuse vs onset of schizophrenic symptoms).
Case-wise comparisons of the temporal order of onsets (n = 29)
p < 0.05,
p < 0.001 (onset of drug abuse vs. onset of schizophrenic symptoms).
The comparison of group means shows the general trends, but may blur the temporal order in individual cases. We therefore looked at the numbers of patients with onsets of substance abuse before, within the same month and after the described ‘milestones’ during the early course of schizophrenia. The analysis of the sequence of the onset of drug (i.e. cannabis) abuse and of the first symptom of schizophrenia was of special interest, since the group means for these two events did not differ significantly. The analysis of the sequence in the individual cases (see Table 3) revealed that drug-abusing, first-episode schizophrenic patients can be subdivided in three groups of almost equal size: 27.5% had a drug problem more than 1 year (most often more than 5 years) before the first sign of schizophrenia. In 34.6% the first signs of schizophrenia and drug abuse started at almost the same time. In 37.9% drug abuse began after the first symptoms of schizophrenia.
Discussion
The vulnerability-stress-coping model of schizophrenia, introduced by Zubin [32] and expanded by Nuechterlein [33], is generally accepted as the concept for the understanding of schizophrenia. The model is useful in providing a framework for the communication between schizophrenia researchers and clinicians from diverse backgrounds with neurobiological and psychosocial approaches to schizophrenia. This model suggests three possible interpretations of the ABC Study's findings on subgroups of comorbid patients.
Group 1, with an onset of cannabis abuse years before the onset of schizophrenic symptoms, could be named the ‘vulnerability group’. These patients might suffer from a chronic deterioration of mental health caused by cannabis that reduces the vulnerability threshold and/or their coping resources. Neurobiological studies (see [34] for review) have demonstrated the effects of cannabis on the brain. Regular heavy ingestion of cannabis causes a sub-acute encephalopathy. Without psychotic features these patients present with an ‘amotivational syndrome’, impaired coping behaviours and cognitive deficits [35], all quite similar to the negative syndrome of schizophrenia. Some patients develop additional positive symptoms, some do not.
Group 2, with an onset of both disorders at about the same time, can be named the ‘stress group’. This group consists of individuals who had already been vulnerable to schizophrenia due to genetic, pre- or perinatal influences. Cannabis consumption then acts as the dopaminergic stress factor and precipitates the onset of psychosis [36].
Group 3, with an onset of cannabis abuse after the first symptoms of schizophrenia, can be called the ‘coping group’. These patients consume cannabis predominantly for self-medication against (or for coping with) symptoms of schizophrenia, particularly negative and depressive symptoms [5,6,21,22]. The patients learn to counterbalance an unpleasant hypodopaminergic prefrontal state by the dopaminergic effects of cannabis.
Further research is needed to validate this preliminary proposal for a differentiation of comorbid cases. For the following reasons, the conclusions, particularly regarding group 1, have to be tentative at this stage.
A third risk factor, such as neurodevelopmental and morphological brain anomalies, frequently preceding the onset of both schizophrenia and substance abuse cannot be ruled out. Prospective studies that could clarify this issue have yet to be done.
The existence of confounding variables, such as subtle cognitive, perceptual, affective and social deficiencies, preceding schizophrenia that might also contribute to an increased risk of preschizophrenic substance abuse is difficult to assess in retrospect, and can not be ruled out either.
Due to the small size of the comorbid sample, confounding effects of other drugs abused by some patients in addition to cannabis could not be excluded. Since 80% of the patients consumed cannabis as their first drug, this issue is less relevant in groups 2 and 3.
The randomly selected case–control group from the general population was added to the original ABC Schizophrenia Study to give an estimate of the base rate of substance abuse. The existence of a base rate implies that comorbidity of schizophrenia and substance abuse can be a coincidental association in some patients. The control group, however, was small and representative only for the central region of the catchment area. Due to administrative reasons, the controls could not be drawn from the population registers of all catchment area communities and were only obtained from the largest city. This geographical bias, however, appears to be small because of short distances, demographical homogeneity and high recreational mobility within this region [26]. Since cannabis consumption increased in Germany during the 1980s [37], the delayed recruitment of the controls (on average 3 years after the patients, but with the same instruments by the same research group) may rather result in an overestimation, rather than an underestimation, of the base rate of cannabis abuse in the general population, and therefore diminish the real difference between patients and controls. Future, more sophisticated, epidemiological investigations are necessary to consolidate the results of this pilot study.
Neurobiological variables will contribute to further characterise the three groups whose actual size has to be determined in larger samples. It is most likely that a patient can belong to more than one group, longitudinally as well as cross-sectionally. During the course of the illness, a patient may shift from one group to another (e.g. from group 2 to group 3) and even, at a given point in time, all three relations of cannabis to psychosis may converge to produce comorbidity. Prospective studies and sophisticated case studies need to further explore this issue.
Conclusions
Already at first hospitalisation, the prevalence of drug abuse, and particularly cannabis abuse, is high in patients with schizophrenia. Compared with a matched non-psychiatric control sample, the prevalence of drug abuse appears to be about twice as large in the first-episode patients. The results support earlier studies that discerned male sex and young age as risk factors for substance abuse in schizophrenic patients. Multiple substance abuse is frequent, particularly concomitant alcohol abuse. In accordance with other findings (e.g. [9,10]), cannabis was found to be the most frequently abused illegal drug in schizophrenic patients. Already during the early course of schizophrenia, social adaptation and integration in many aspects is considerably worse in comorbid patients.
Several fields of psychiatric research, involving biological, psychopathological or epidemiological studies, contribute to a better understanding of the role of psychotropic substances in the aetiology of psychoses. Biological studies in animals and man have been able to illuminate the pathogenetic action of psychotropic drugs. Epidemiological studies in first-episode patients such as the ABC Schizophrenia Study can test whether these experimentally derived models are applicable to the natural course of schizophrenia, particularly the order of onset of substance abuse and schizophrenic symptoms. The differentiation of important steps during the early course of schizophrenia (namely the appearance of the first sign, the first negative and the first positive symptom) is necessary for a sophisticated analysis of the temporality of onsets. Of course, the determination of the temporal sequence of events may not prove, but falsify causal hypotheses. While Rosenthal [1] rightly summarised in a recent review that ‘no compelling evidence to date has been presented for causality’, the ABC Study suggests that causal links between schizophrenia and cannabis may go both ways, and that the vulnerability-stress-coping model of schizophrenia is a helpful framework for the interpretation of these findings.
