Abstract
Some three hundred mainly steel shipwrecks from both World Wars lie buried at shallow depths along the Belgian North Sea coast. They were examined recently to estimate corrosion rates over periods in excess of 100 years. The rate was estimated by comparing the measured in-situ steel plate thicknesses with archived ship information. The estimates show distinctly lower long-term corrosion rate compared to that predicted by the Melchers (Modeling of marine immersion corrosion for mild and low alloy steels – part 1: phenomenological model. Corrosion. 2003;59(4):319-334) corrosion model, when parameterised for local North Sea conditions. Concretion after 50 years has a multi-layer structure for which SEM-EDS and XRD measurements show the innermost layer, close to the metal surface, consisting of akaganeite, and the outer layer mostly of calcium carbonates, silicates, and some siderite. In between there is a considerable layer of hard magnetite. The latter is proposed as the reason for the low long-term corrosion rate (0.016 mm y–1) in an environment with calcium carbonate available in abundance.
Keywords
Introduction
Some three hundred mainly steel shipwrecks from both World Wars lie buried at shallow depths along the Belgian North Sea coast. Many contain the remains of their crews and therefore are protected by Belgian law. Besides their cultural and historical value, these wrecks have a biological, environmental, and economic importance. Moreover, given the detailed information available on the history of these wrecks, they are a source of information for corrosion rates over periods in excess of 100 years in a very specific environment. This is of interest since there are very few long-term data available for corrosion in actual sea conditions [1].
Marine corrosion is a major issue for infrastructure such as bridges, industrial facilities, coastal and harbour structures, pipelines especially when positioned in or near the sea. It also is an issue for offshore structures and ships. A good understanding of the corrosion processes and rates allows better estimates of the expected lifetime and reliability of such structures. It also can provide optimisation of maintenance planning, construction design (corrosion-resistant materials, acceptable corrosion rate, protective coatings, sacrificial anodes, impressed current systems, etc.) and the use of monitoring systems and tools [2]. Corrosion, particularly marine corrosion, is often considered a complicated process governed by many parameters such as temperature, water velocity, pH, dissolved oxygen, nitrate concentration, water depth, hydrostatic pressure, bacterial activity, pollutants, erosion, and surface roughness [2,3]. Most corrosion studies focus on the initial corrosion processes, leading to assessments of corrosion rates which cannot be extrapolated to longer time periods, as required for the use of marine and maritime structures, which typically have design life times of 20-30 or more years. Only a limited number of models have been proposed to describe corrosion for long-term exposures. Initial modelling efforts assumed a constant corrosion rate and showed limited consistency with reality [4]. Models already applied in the 1930s and 1940s include the well-known ‘Power Model’, originally proposed only empirically but later derived formally, with some mathematical approximations, by Tammann [5], originally for the atmospheric corrosion of copper roofs. Later models [6 8] proposed non-linear relations leading asymptotically to a zero-corrosion rate. Some included also the retarding effect of a protective coating, as reviewed by Melchers [4].
The present study applies the bi-modal model originally proposed by Melchers [9], largely because it is based on physical and electrochemical principles. The model is robust, is related to probabilistic principles, and can take into account uniform as well as pitting corrosion, aerobic and anaerobic conditions, as well as bacterial, environmental, and anthropogenic influences. The model consists of five phases (Figure 1). Phases 0, 1 and 2 are predominantly oxygen controlled while phases 3 and 4 are largely under anaerobic conditions. In the idealised case, the aerobic corrosion process starts with an initial instantaneous corrosion rate r0. Owing to a progressive reduction of oxygen available at the corrosion interface the corrosion rate diminishes and becomes nearly zero, at ta. However, the corrosion rate then increases under the influence of anaerobic and possibly bacterial corrosion processes with an initial corrosion rate of ra to reach a constant rate rs at time point B.
Multi-phase model for long-term corrosion in marine environments with indication of the parameters defined in the text and in Table 1. Long-term anaerobic corrosion rate becomes linear at t > B, due to a possibly increasing importance of microbiological influences over the involvement of oxygen. Figure based on Melchers [10].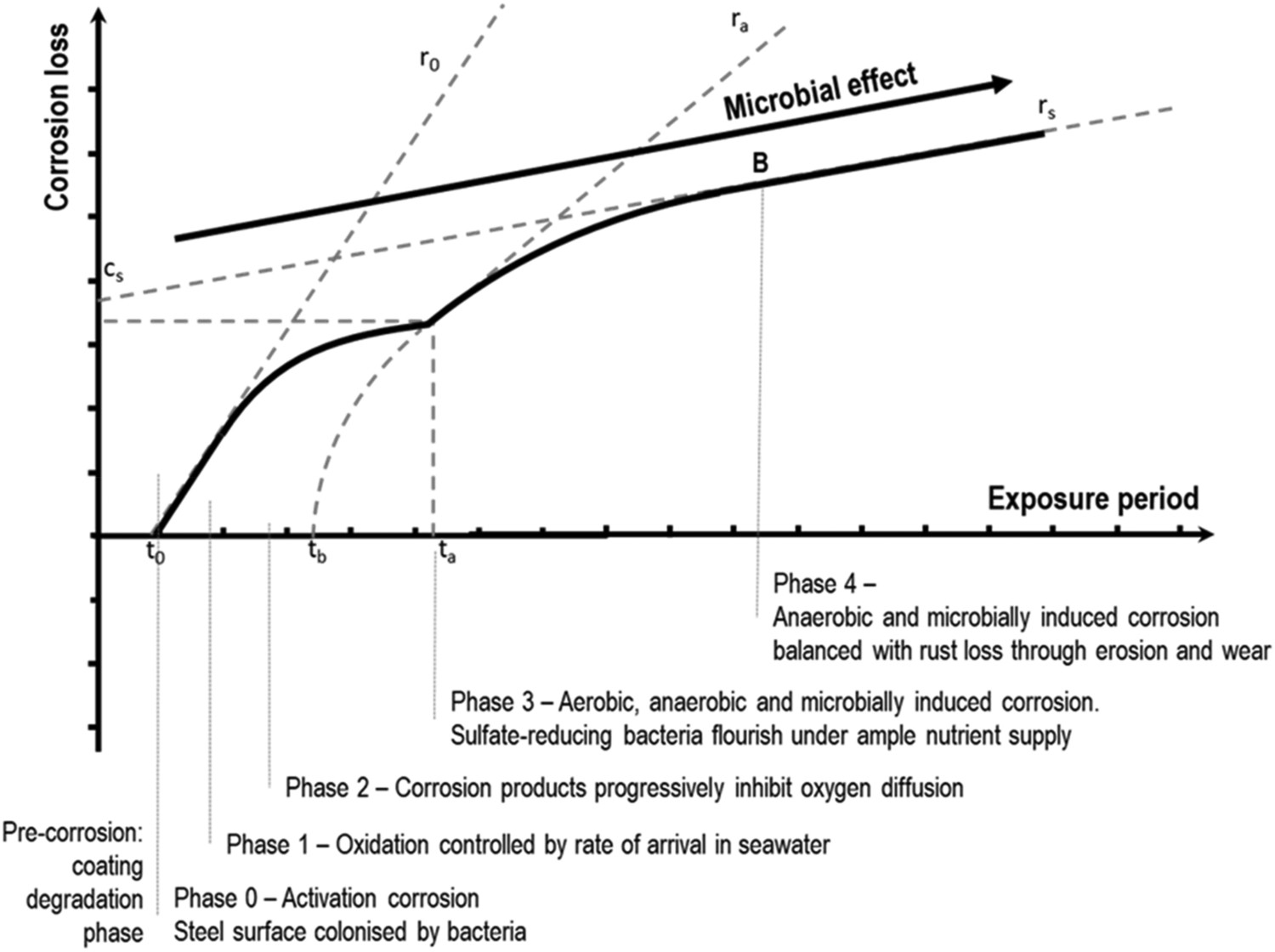
The model is based on a large variety of field and relevant laboratory exposure tests, and it has been calibrated for a considerable number of environmental variables. As reviewed earlier [2], it differs considerably from all other models proposed in the literature since these are all, essentially, entirely empirical when applied to the longer-term exposure of physical infrastructure. Many of these have been applied to cases with much longer exposure periods than for which the power law was originally intended, largely because it was not incorporated that the underlying corrosion processes change from initially being aerobic at the corrosion interface to being governed eventually by the hydrogen evolution cathodic reaction [2]. Additionally, this transition does not occur for the very special case of pure copper, for which the power law was developed, but it can be seen in a variety of other metals and alloys including steels, stainless steels, aluminium, and copper alloys. Thus, simplistic curvilinear models are not appropriate, especially not when studying longer-term effects or complex materials.
At the present time it appears that the Melchers model is the best suited for long-term corrosion modelling. In addition, the model allows for longer-term exposure of interest here (i.e. well into phase 4). For the present study, this model has been parameterised using data from the Belgian North Sea shipwrecks and compared with the earlier calibration of that model. The next section describes the field investigations conducted during 2015-2019. This is followed by detailed observations and then by interpretations of the observations and proposals for further investigations.
Material and methods
Parametrisation of the Melchers model
For long-term corrosion processes, the Melchers model described in the introduction can be approximated by a linear curve with slope rs and intercept cs (Figure 1):
Position of the wrecks used in this study, in the Belgian Part of the North Sea and the French coast. Model parameters for general corrosion and pitting under immersion conditions as a function of the average sea water temperature T, as well as applied for the North Sea (T = 12.5°C). Equations taken from Melchers (2005)[36].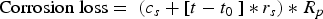
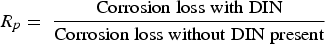

Corrosion measurements on shipwrecks
Overview of the shipwrecks used in this study.
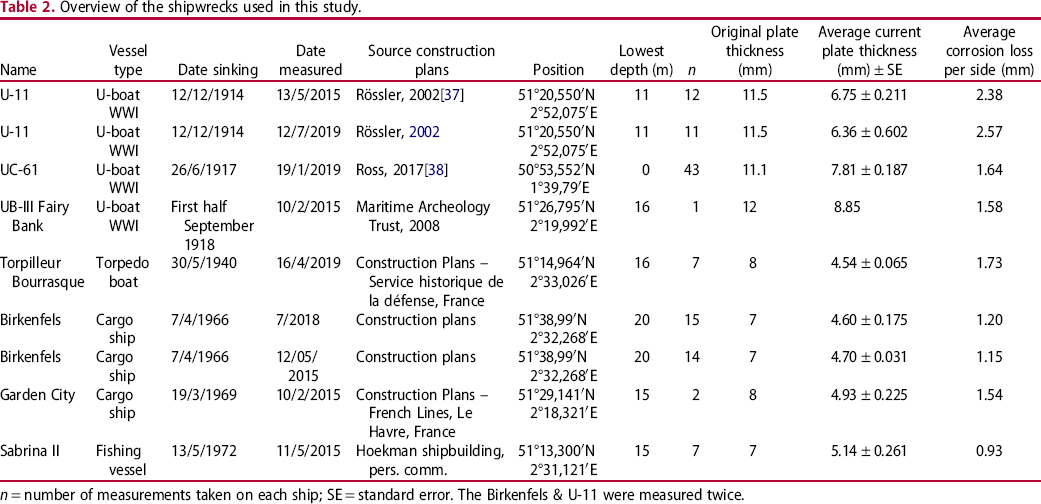
n = number of measurements taken on each ship; SE = standard error. The Birkenfels & U-11 were measured twice.
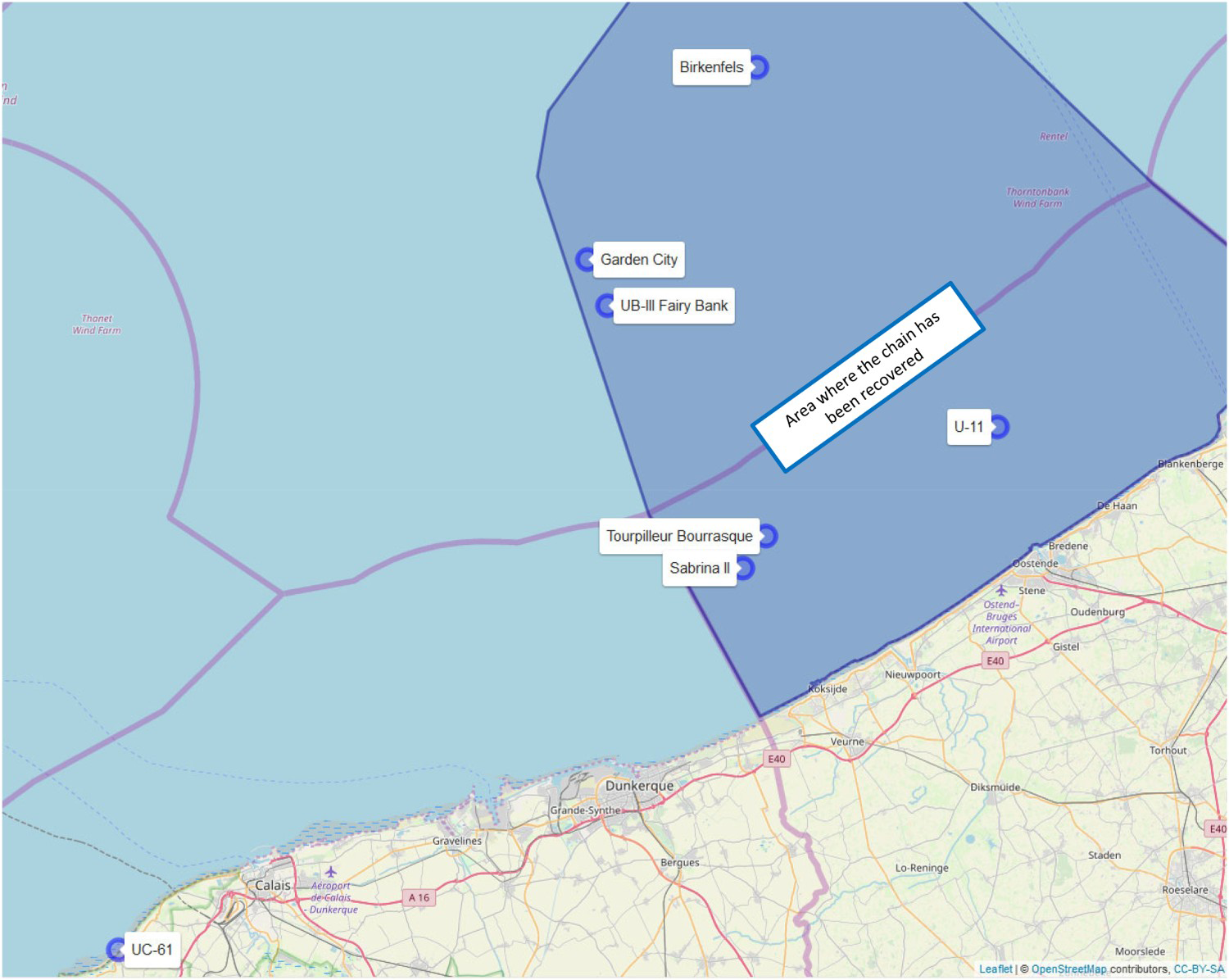
The oldest wrecks in this study are three German submarines dating from the First World War. The U-11 is a German long-distance submarine that sank on 12/12/1914 after colliding with a mine. The wreck is considered to be a war grave. The UC-61 is a German minelaying submarine of the UC-II type. She was built in 1916 and beached near Boulogne on 26 July 1917 before the crew surrendered to French authorities. The identity of the remains of the third German UB-III class submarine, situated on the Fairy Bank, is not certain. The wreck may be a submarine of the series UB-54 till UB-59 or UB-103 till UB-117. Most probably, the submarine sank after colliding with a mine in the Dover Mine Field in the first half of September 1918. The four other wrecks are shipwrecks, dating between 1940 and 1972. The Torpilleur Bourrasque is one of a series of 12 French destroyers. She was built by Ateliers et Chantiers de France at Dunkirk; she collided with a mine and was lost off Nieuwpoort on 30 May 1940 during Operation Dynamo. The Birkenfels is a German merchant vessel built in 1951. On 7 April 1966, while travelling from Bremen via Antwerp to the Persian Gulf with a cargo of 8.750 tons steel and general cargo, she collided with the German MV Marie Luise Bolton in dense fog, and sank, at about one mile south of Noordhinder Lightship. The Garden City was built at ‘Le Moyne d'Iberville’ in France in 1949. She sank on 19 March 1969 after a collision with a Polish vessel MV Zaglebie Dabrowskie. Finally, the Sabrina II is a Belgian trawler built in 1963, which capsized on 12 May 1972 during fishing.
Diving excursions took place between February 2015 and April 2019 and were performed by recognised scientific divers/historic wreck divers (co-author Sven Van Haelst). For the wrecks close to the shore a rigid inflatable craft (RIB) was used while for those further away the research vessel Simon Stevin of the Flanders Maritime Institute (VLIZ) was engaged. The underwater, in situ, plate thickness measurements were performed with a Cygnus M2-Dive equipped with the S2C single ultrasonic (UTS) Crystal Probe (Cygnus Instruments, USA) with a gauge, requiring three equally spaced echoes per spot in order to calculate a thickness measurement value.
For each of the wrecks, an easily recognisable place was selected for the measurements and for which the original plate thickness was known. Also, for all measurements in Table 2, both sides of the plate were exposed to open sea water (not buried) allowing an identical corrosion rate on front and back. A few cm2 of the fouling and the concretion were removed with a chipping hammer and scraper used to remove loose material and to provide the UTS probe good contact, preferably direct with the metal surface, to allow measurements to be taken. In all cases these were consistent with accepted industry standards for UTS testing
Further ultrasonic measurements were taken in a similar manner at distances of about 20 cm around the first reading, resulting in n measurements per wreck (as specified in Table 2). Comparison with the original plate thickness allowed for a calculation of the corrosion rate at each individual spot.
More detailed information regarding these UTS measurement available via mail to kris.de.baere@hzs.be or sven.vanhaelst@vliz.be.
SEM-EDS and XRD analysis
On 5 August 2013, Belgium ratified the UNESCO Convention [12] on the protection of the underwater cultural heritage. This new law covers not only wrecks and wreckages but also all other forms of underwater cultural heritage both within the territorial sea and on the continental shelf. Collecting pieces of an actual wreck became morally and legally impossible. Therefore, the present study was obliged to use a work-around and instead used a piece of anchor chain not belonging to one of the shipwrecks. That piece of chain was salvaged during UXO (Unexploded Ordnance) surveys in March 2018, recovered in an area between Oostdyck and Akkaertbank at a depth of approximately 30 m and was buried approximately 50 cm in mainly sands and shells. Based on the thickness of the concretion layer (Figure 3) and the characteristics (diameter and length/width ratio) of the link [13] the chain was dated as being from the first half of the twentieth century. This corresponds closely with the age of the shipwrecks used in this study (Table 2). In turn this suggests that the steel was similar in composition and in corrosion characteristics, consistent with the literature [14,15]. Also, the chain would initially have lain on the seafloor and only gradually have become buried in the local sands, most likely well after the early corrosion products had been formed (i.e. in phases 1-3 of Figure 1). Thereafter corrosion would have been largely anaerobic, in an environment under well-developed rusts and thus largely independent of the external environment. Importantly, sea water and by implication dissolved oxygen and calcareous material are likely to have been present almost as much as if the links had been on the surface instead of shallow burial. From this it was concluded that examination of the chain links was an adequate surrogate for examination of the rusts on the wrecks.
Piece of historic chain used for SEM-EDX and XRD analysis of the concretion layers.
In a first series of measurements, X-ray diffraction (XRD) was used to determine a preliminary composition of the concretion around the metal chain shackle a D8 Discover XRD (Bruker), set-up for phase identification with a Co tube (Kα1 = 1.78897 Å) with a generator at 35 kV and 40 mA, a 1 mm slit behind the Polycap, long soller slits 0.35° in front of the detector, the lynx eye detector in 0 D mode with an opening of 13 mm, and a q/q configuration (continuous locked couple scan). The crystalline phases present in the analysed sample were identified by comparing the positions of diffraction peaks in the experimental pattern with those present in (experimental and simulated) patterns of known samples available in a database (Powder Diffraction File, International Centre for Diffraction Data, Swarthmore/USA).
Next, the chain was cut into pieces of approximately 5 cm. One of the pieces was forwarded to the University of Newcastle in Australia for detailed analysis. Upon reception, the sample was stored in a dry and secure location under ambient conditions for about 6 weeks prior to testing. The chain pieces were dried in an oven at 110°C for 24 h prior to SEM/EDS and XRD testing. Target areas of interest were marked and aligned in the PANalytical X'Pert Pro MRD XRD for analysis, which was used with a continuous scan type, step time of 135 s, at 25°C using a Cu anode with beam generator set to 40 mA, 40 kV.
To prepare the sample for Zeiss SIGMA VP SEM/EDS analysis, the chain was ground flat using a 400 grit silica carbide belt sander and then broken open to obtain suitable samples for Scanning Electron Spectroscopy (SEM) with Electron Dispersive Spectroscopy (EDS) analysis. The Back Scatter Electron Detector was used for observation when necessary, while the analysis was primary completed using the Secondary Electron Detector at a 20 keV beam intensity. When performing EDS scans, all zones had the same magnification and the scan was performed for the same duration; sufficient time was allowed (approx. 10 min) until no further changes to peak count were detectable.
Results and discussion
Model parametrisation
The Melchers model was parametrised for the North Sea, with parameter values as estimated and presented in Figure 1 based on the equations in Table 1. As most of the wrecks of interest have been immersed for a very long time (>15 years), the linear approximation of the Melchers model (Equation (1)) is appropriate. The value for Rp in this equation can be determined with an empirical relation between Rp and the concentration of DIN (dissolved inorganic nitrogen) for long term exposures [10]:
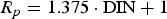
Melchers [10] obtained a strong relationship between the availability of DIN for microbiological activity and the severity of the resulting MIC, noting that the DIN in the water is only a proxy for MIC. This is the case also for the waters surrounding the wrecks. Since sulfate reducing bacteria (SRB) usually are considered the most common corrosion-accelerating bacteria, attention was given to estimating their presence. They are known to produce sulphides, mainly in the form of iron sulphides, in their main energy metabolism, and this can be used as a marker for their presence. In this context, it is noted that earlier XRD analysis in 2019 of a piece of an iron chain conserved under the same conditions as the shipwrecks failed to demonstrate the presence of iron sulphides [19]. That sample did contain SiO2, CaCO3, K2CaMg(SO4)3, FeOOH, K3FeO2, Fe0.94O, CaFeO3 and Fe8O8(OH)8Cl. For the present study DNA sampling was attempted on a sample taken from within the concretion layer around the iron chain shackle. The extraction yielded very low DNA concentrations. The subsequent PCR of 16S rRNA genes did not result in any amplification of the genetic material. This is probably due to very low amounts of bacteria, and presumably nutrients, within the concretion layer. How this relates to the elevated DIN conditions outside the concretion layer, that is, within the local sea water, remains an interesting question. In this context it is noted there could be the presence of inhibitory compounds in the North Sea sea water [16]. Overall, these results suggest that MIC may be insignificant for the corrosion of the shipwrecks considered here. The factor Rp can therefore be kept at a value of 1. Hence, according to the Melchers model (Table 1) the long-term corrosion rate, that is after a period of approximately 15 years on the sea bottom at 12.5°C, is predicted to be rs, = 0.053 mm y−1.
Estimation of shipwreck corrosion rate
To make an estimate of the long-term corrosion rate rs specifically for the Belgian North Sea wrecks, the results of the average measured corrosion losses on the shipwrecks were plotted against the period of immersion. The empirically obtained trend (Figure 4) was compared with the multi-phase model of Melchers at 12.5°C in the absence of MIC [2].
Corrosion loss per side (mm) versus time in the water in years; the blue line corresponds with the linear North Sea model with a green 95% confidence and prediction band; the orange line corresponds with the Melchers model as parametrised for the North Sea; the red dotted line corresponds with the North Sea power model.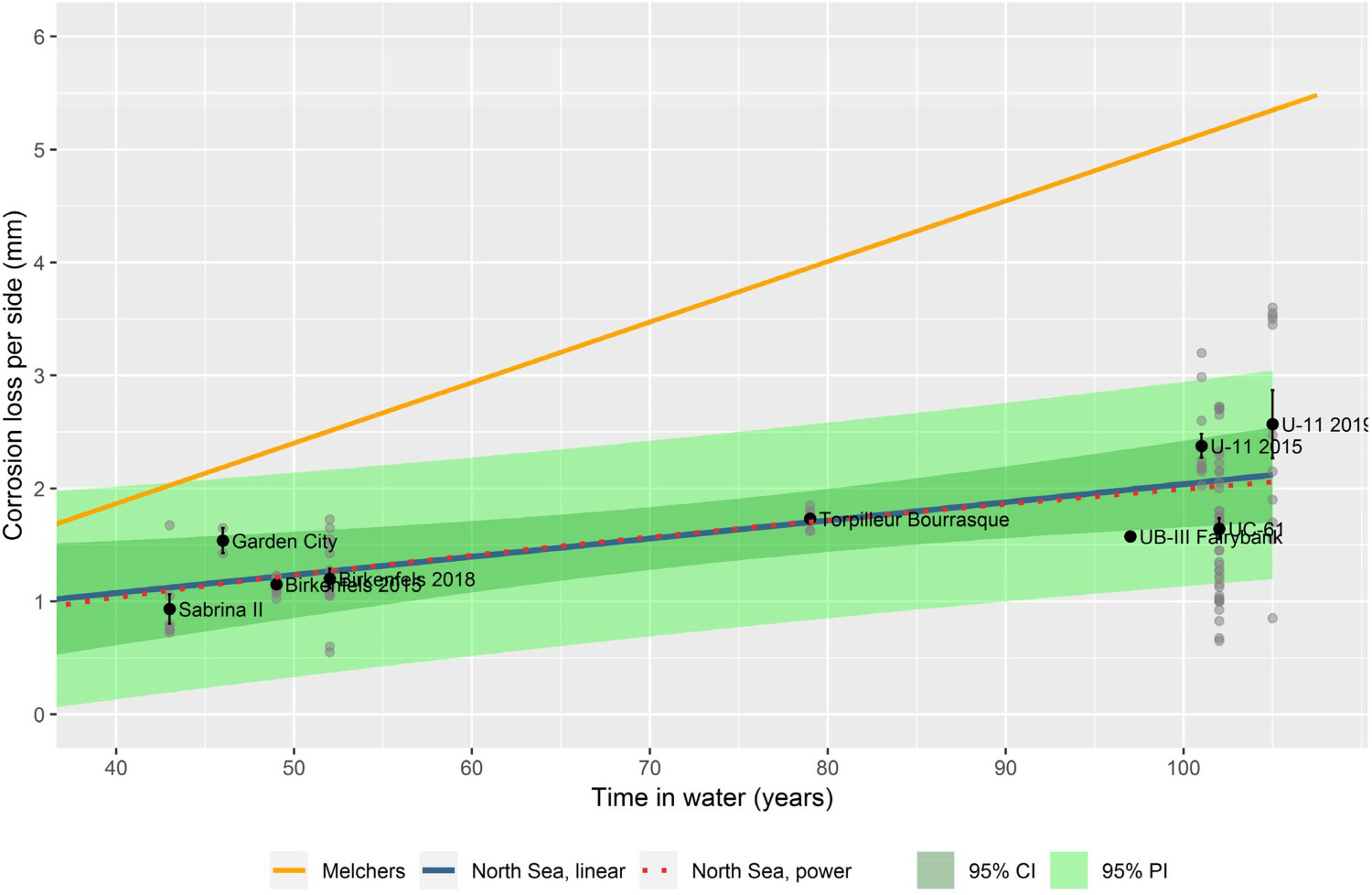
For the predominantly anaerobic part of the model (phases 3 and 4), there is a steep increase in corrosion loss, starting for phase 3, at 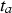 , followed eventually by a linear function (phase 4). The exact value of
, followed eventually by a linear function (phase 4). The exact value of 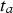 depends mainly on water temperature but cannot be estimated from the data at hand, as all considered shipwrecks have been immersed for many years; the minimum being 43 years (Sabrina II). In all cases, it is likely that the vessels were coated, and the likely life of such coatings needs to be considered. This has never been studied in any detail for shipwrecks. Even estimating the durability of modern coatings poses significant problems. A survey by Verstraelen et al. [20] suggests that, on average, ballast tank coatings remain sufficiently intact for a period of 10 years. Another survey suggests coating breakdown occurs much earlier and that 10 years is consistent with complete coating breakdown for plates but with a range of ±3 years around the mean, and larger scatter for edges and welds [21]. Calculations based on the equations given in Table 1 give a time interval between
depends mainly on water temperature but cannot be estimated from the data at hand, as all considered shipwrecks have been immersed for many years; the minimum being 43 years (Sabrina II). In all cases, it is likely that the vessels were coated, and the likely life of such coatings needs to be considered. This has never been studied in any detail for shipwrecks. Even estimating the durability of modern coatings poses significant problems. A survey by Verstraelen et al. [20] suggests that, on average, ballast tank coatings remain sufficiently intact for a period of 10 years. Another survey suggests coating breakdown occurs much earlier and that 10 years is consistent with complete coating breakdown for plates but with a range of ±3 years around the mean, and larger scatter for edges and welds [21]. Calculations based on the equations given in Table 1 give a time interval between 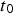 and
and 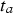 of approximately 2.2 years, so that
of approximately 2.2 years, so that 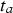 = 12.3 years (taking into account a coating life of 10.1 years) is considered reasonable. This was allowed in fitting the trend to the data. It could be approximated by the power law as follows:
= 12.3 years (taking into account a coating life of 10.1 years) is considered reasonable. This was allowed in fitting the trend to the data. It could be approximated by the power law as follows:
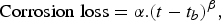
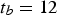 years,
years, 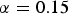 (95% confidence interval [
(95% confidence interval [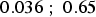 ]) and
]) and 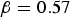 (95% confidence interval [
(95% confidence interval [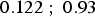 ]). This model is statistically significant (p = 0.006 < 0.05). For this model,
]). This model is statistically significant (p = 0.006 < 0.05). For this model, 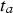 is reached at 12.3 years after sinking, when the tangent to the model has a slope of
is reached at 12.3 years after sinking, when the tangent to the model has a slope of 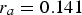 , based on the equations in Table 1.
, based on the equations in Table 1.
A long-term linear model was also fitted on the data (Equation (5)). As can be seen in Figure 4, the long-term evolution of average corrosion loss differs little between the linear and the non-linear trends. The linear model was therefore adopted:
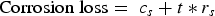
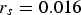 mm y−1 (95% confidence interval
mm y−1 (95% confidence interval 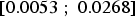 ), while the intercept is given by
), while the intercept is given by 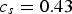 (95% confidence interval
(95% confidence interval 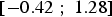 ). This model is statistically significant (p = 0.009 < 0.05).
). This model is statistically significant (p = 0.009 < 0.05).
A comparison of the long-term linear model (displayed along with its 95% confidence and prediction bands in Figure 4) with the linear stage 4 of Melchers model (as defined in Figure 1) shows that the linear part of the latter has a much higher long-term corrosion rate. The corrosion rate for the ship wrecks considered, all located in the Belgian part of the North Sea (BNS), is less than one-third of the corrosion rate predicted by Melchers’ multi-phase model (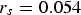 mm y−1) (Table 1).
mm y−1) (Table 1).
Although it is sometimes claimed that it is difficult to generalise corrosion rates of iron and steel in sea water [22], it should be remembered that the Melchers model was based on a very large data set, including other data in the North Sea (near Scotland) [9]. Because of the non-linearity displayed in the bi-model model for corrosion simple comparisons of corrosion rates without allowing for the early part of the model can give quite misleading interpretations. This can be seen by consideration of data reported by others. For example, field corrosion rate tests in shallow water (28 m) and in the deep sea (2511 m) in the Sea of Japan obtained corrosion rates in deep water of 0.055 mm y−1 after (only) 1.4 y, decreasing to 0.035 mm y−1 after 8.6 y [23]. In shallow water they obtained a rate of 0.229 mm y−1 after 124 days. Macleod [24] reported a corrosion rate of 0.095 mm y−1 on the wreck of the Yubae Maru, found in Chuuk lagoon at a depth of 28 m. These rates are all considerably larger than the long-term rate in the Belgian North Sea, but do not account for the variation with time as indicated by the model.
The present findings suggest that it is necessary to investigate more fully the characteristics of corrosion specifically in the Belgian part of the North Sea and perhaps other locations that have similar local conditions. To throw some light on what might be different about steel corrosion in the Belgian part of the North Sea, the next sections report detailed observations of the rusts on the wrecks and similar objects in this part of the North Sea.
Analysis of corrosion product layers
SEM/EDS analysis
One of the features seen on the corroded wrecks (and other steel objects) in the Belgian part of the North Sea is the heavy coverage with calcareous deposits (concretion layer). These have been observed in some other immersion corrosion, particularly off Western Australia [25] and some sites in the US [26] but overall are not common for marine corrosion. To obtain some understanding of the characteristics of the concretion layers, several distinct parts of steel chain link available for analysis (see above) were selected, based on their position relative to the metal surface and their difference in colour (Figure 5). This shows that there is a substantial amount of calcium ions in the outer, whitish-coloured, corrosion product. The layers closer to the original steel are high in Fe as expected.
SEM/EDS analysis of five locations ranging from the metal surface towards the outer layers of the concretion. The values in table are expressed in atomic weight %.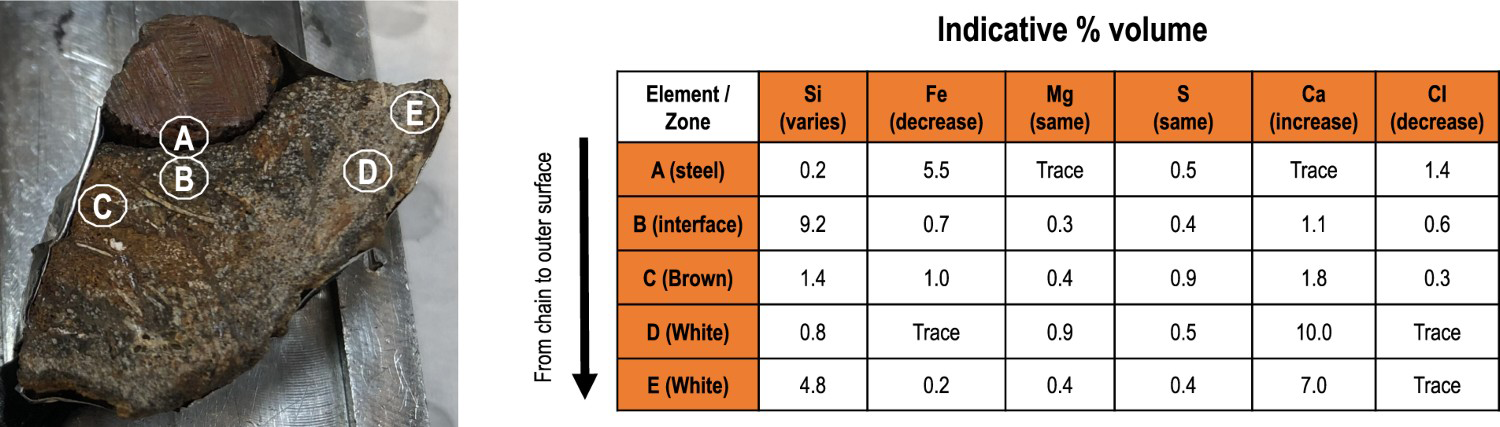
XRD analysis
For the XRD analyses, six distinct zones were considered of interest, starting at the metal structure, and then outward towards the outer surface of the concretion (see Figure 6). Two locations (A and B) were chosen directly near the steel chain surface and inside the concretion, assumed representative of the direct corrosion products. At both locations, the products consist almost entirely of akaganeite (β-FeOOH). On the exterior surface, at location F (taken at the outer layers of the accretion) and E (taken in between two parts of the chain link), deposits of calcium and magnesium minerals were noted, mixed with silica oxides and silicates with mineral structures of quartz (SiO2), tilleyite (Ca5Si2O7(CO3)2), dolomite (CaMg(CO3)2) and gypsum (CaSO4), next to large amounts of calcite and aragonite (forms of CaCO3) 17% and 57% at location 1, respectively, and for 10% and 36% at location 3. Both locations also contained 8% and 6% of siderite, respectively. At locations C and D, black and orange rusts were found. These rusts were akaganeite (46% and 32%), mixed with goethite (12% and 17%) and magnetite (19% and 11%). Some quartz and gypsum were also observed. Table 3 summarises the results. For comparison, some results are shown also for long-term rust developed on ferrous objects buried in coastal region soils some 100 km south-west of the Belgian North Sea coast [27]. This comparison is made because at this site the corrosion products also were found to have calcite and aragonite contents, unlike what is conventionally found in rust products for buried ferrous objects [28]. It is noted that the components of the rust products in soil environment show similarities with those from sea water immersion, particularly the presence of the chloride-bearing akaganeite. This suggests a degree of correspondence in corrosion mechanisms between these two environments that might be further investigated, noting that classical literature has very little to say about corrosion in calcareous soils [28].
XRD results Belgian North Sea chain link. 6 zones were distinguished from A to F. A is the closest to the remaining metal of the chain and F is the accretion edge. The XRD results are shown in % volume (indicative) Identification of the different corrosion layers around the Belgian North Sea chain link, compared to the model proposed by Neff et al. [27]. aFor ferrous objects buried in coastal, chloride-bearing soils near the coast.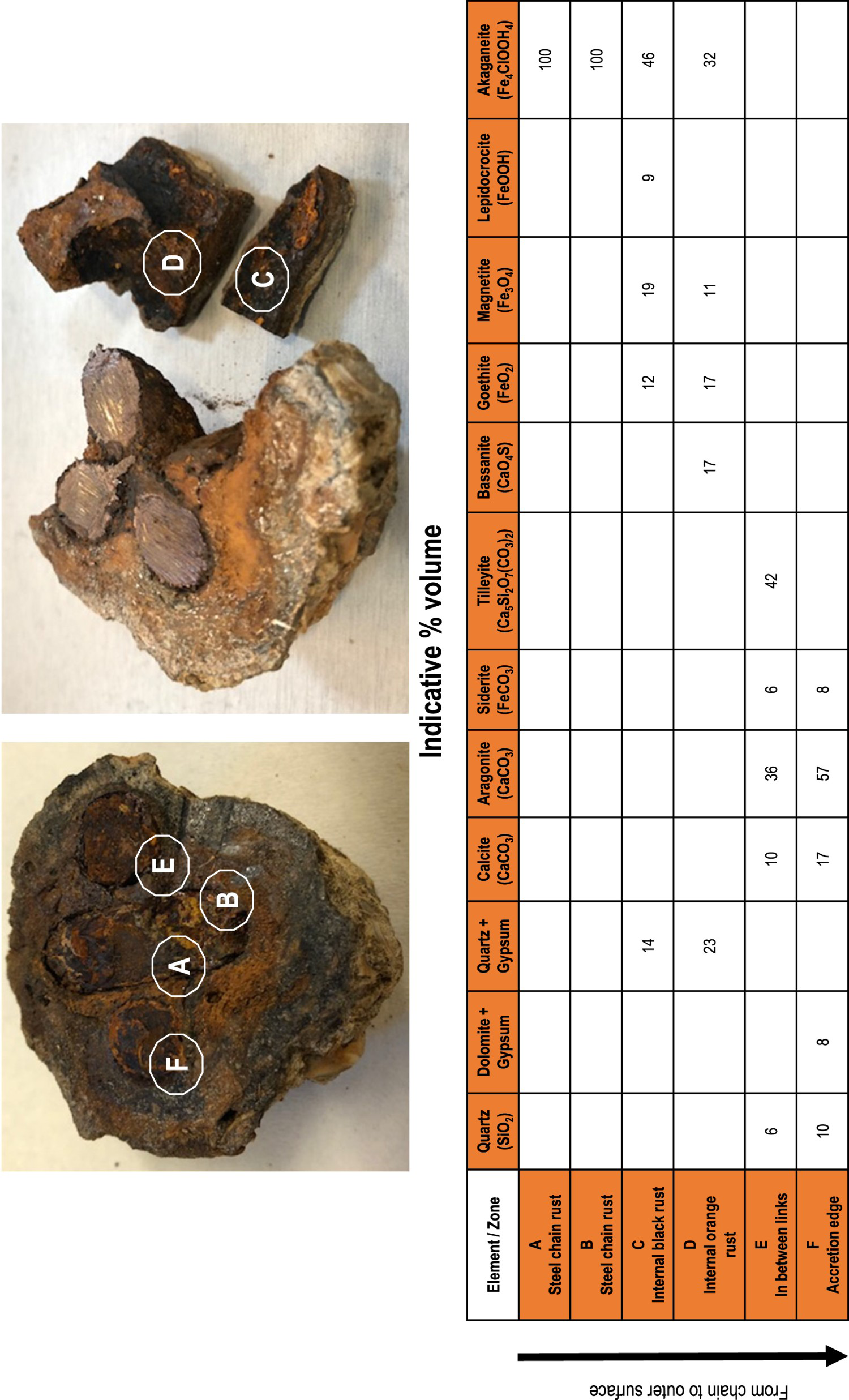

Discussion
The present investigation used observations on a number of shipwrecks with ages extending over a considerable period. It would be expected that the wrought irons and steels used for the construction of the ship hulls varied in composition. However, due to archaeological reasons, this could not be investigated. While there could be a small effect of steel composition on long-term corrosion, there is enough evidence in the literature that any such effect is small, both for shorter-term exposures and for longer-term [14,15]. This means that the difference between the long-term rate from the Melchers model and that observed in the Belgian part of the North Sea (BNS) cannot be explained in terms of steel (or wrought iron) composition.
The formation of a concretion layer on the surface of metal objects, such as described above, and as observed in the present cases, is known to initiate relatively soon, i.e. within months, after first immersion [22,26,29 31]. This concretion layer, initially [30] largely calcium carbonate, has been noted to become relatively impermeable to oxygen with increased exposure period, largely due to the formation of ferrous carbonate (FeCO3) and thereby tending to slow down the corrosion rate, at least for shorter periods of exposure [26,32]. The SEM-EDS and XRD measurements presented herein show that calcium carbonate also resides within the rust layers, and that FeCO3 also likely occurs there, consistent with earlier observations [26].
It is well-known that soon after first immersion of iron or steel in sea water the initial corrosion products are various iron hydroxides and that these are associated with elevated pH at the metal surface [26,27,33]. These regions are preferential for the deposition of any calcium carbonate precipitation in sea waters with a larger tendency for calcium carbonate to come out of solution, and to do so for long periods of time – i.e. throughout the exposure period.
Importantly, calcareous deposition observed in the present investigation and in some others is not universal. It does not occur, for example, in the northern part of the North Sea [34]. This indicates that there are very particular conditions in the BNS that tend to favour calcareous deposition. These are unlikely to be short-term effects such as caused by anthropological influences, since the shipwrecks are in some cases over 100 years old and water pollution in the North Sea has changed very considerably over that time [16]. One clue for the high calcareous deposition and the slower rates of corrosion rate lies in the presence of a number of limestone sands and geological formations in the region of the BNS, for example, the white cliffs of Dover. There is evidence that the BNS region and the sea water itself in this region is a significant repository of calcareous material. For example, Zintzen et al. [35], from an inventory of the epifauna of the shipwrecks Birkenfels and Bourrasque, noted that most organisms had calcium skeletons or exoskeleton. It is feasible that these could contribute to the formation of the outer calcium layers on any corroding object [30] and also to the calcareous content within the rust layers. Again, the precise mechanisms involved remain for further investigation.
Regarding the distribution of the different corrosion products and the carbonates through the rust layers (Table 3), it is noted that this is consistent with the observations reported by Neff et al. [27] for calcareous soils, noting that these had low chloride contents. The present observations also are largely consistent with those of North & MacLeod [30] for corrosion in sea waters off the Western Australian coast. Despite these consistencies, it is clear that improved explanations are required to fully account for the development of a steady state corrosion rate in highly calcareous sea waters, from first immersion to eventual long-term corrosion with the effect of calcium carbonate deposition within and on the external surfaces of the rust layers. That explanation should also allow for a range of calcium carbonate conditions, noting that corrosion products do not always show evidence of calcareous products either within or on the surface of the rusts.
Conclusion
The investigation of seven steel and wrought-iron shipwrecks buried in shallow depths along the Belgian North Sea with some exposed for more than 100 years produced the following results:
The long-term corrosion rate corresponding to phase 4 in the bi-modal model (2003) was found to be 0.016 mm y–1 with a precision (SE) of 0.0045, a rate much lower than predicted by the bi-modal model even though that was based on an extensive set of worldwide data, Field observations showed that there was extensive calcareous deposition on the outside surfaces of the rusts and SEM-EDS and XRD observations showed that besides the usual presence of magnetite within the rust layers there were also calcium and ferrous carbonates, The calcium and ferrous carbonates within the rust layers were found to be very dense and hard indicating a low degree of permeability for diffusion of oxygen and other ions, thereby reducing the rate of long-term corrosion, The high degree of calcareous deposition in the Belgian part of the North Sea where the shipwrecks are located is consistent with local observations of an unusually high presence of invertebrate fauna with calcium-based exoskeletons, The low long-term corrosion rate may have consequences for future conservation techniques and policies, including calculation of associated longevity and risk, as well as cost benefit decisions for prioritising management actions and the best use of budgets.
Footnotes
Acknowledgements
This project was partially funded by the DISARM SBO project funded by the National Science Fund in Flanders, Belgium, as well as by the Internal Projects ‘Modellering van corrosie van de omhulsels van de giftige munitie op de Paardenmarkt’ (Modelling the condition of the poisonous ammunition on the Paardenmarkt)’ and ‘Conservatie van historische Noordzeewrakken (Conservation of historical North Sea wrecks)’ at the Antwerp Maritime Academy. The authors thank Yves Van Ingelghem (Free University of Brussels) for his contributions to the technical discussions. Klaas Hoekman of Hoekman shipbuilding provided the construction details of the Sabrina II. All of the diving expeditions would have been impossible without the support of the Flanders Marine Institute. The authors also would like to acknowledge use of the facilities and technical assistance of Jenny Zobec and Yun Lin EMX Laboratory staff members of the University of Newcastle Australia.
Disclosure statement
No potential conflict of interest was reported by the author(s).
