Abstract
In France, the reference concept for the geological disposal of high-level waste (HLW) consists of horizontal micro-tunnels, containing carbon steel casing and overpacks (around a stainless steel container containing vitrified waste). The overpacks and casing will be exposed to an environment that will evolve over time: from a hot and humid atmosphere containing oxygen, to an anoxic environment saturated with water at the geothermal temperature. Lots of corrosion experiments have been performed in representative environments to study steel corrosion modes (general corrosion, pitting and crevice corrosion, stress corrosion cracking, hydrogen embrittlement…) while taking into account the influence of temperature, radiation, mechanical stresses and microorganisms. Some key results have influenced the design of the HLW disposal cell, including the carbon steel grades and dimensions of the casing and overpacks, or the addition of an alkaline grout material between the casing and the host rock.
This paper is part of a supplement on the 6th International Workshop on Long-Term Prediction of Corrosion Damage in Nuclear Waste Systems.
French HLW disposal concept
Andra is in charge of the Meuse/Haute-Marne underground research laboratory implemented in a clayey rock, in the East of France. The construction of the Laboratory began in 2000 onwards. It is located 500 m below the surface, in the Callovo-Oxfordian claystone layer (about 130 m thickness and 155 million years old). If agreed, the future deep geological disposal will be built nearby.
Cigéo (Centre Industriel de stockage GEOologique) is the name of the deep geological disposal facility for high and intermediate level long-lived radioactive wastes (HLW and IL-LLW) to be built in France.
Cigéo will consist of two main surface facilities: one to receive and prepare waste packages before their shipment underground via a 10% incline ramp and the other one with shafts to go underground and perform the excavation work of the repository. The Cigéo project plan (Figure 1) is a view of the repository after 100 years in operation. Dimensions are approximately 3 by 5 km.
Possible architecture of the Industrial Centre for Geological Disposal Cigéo.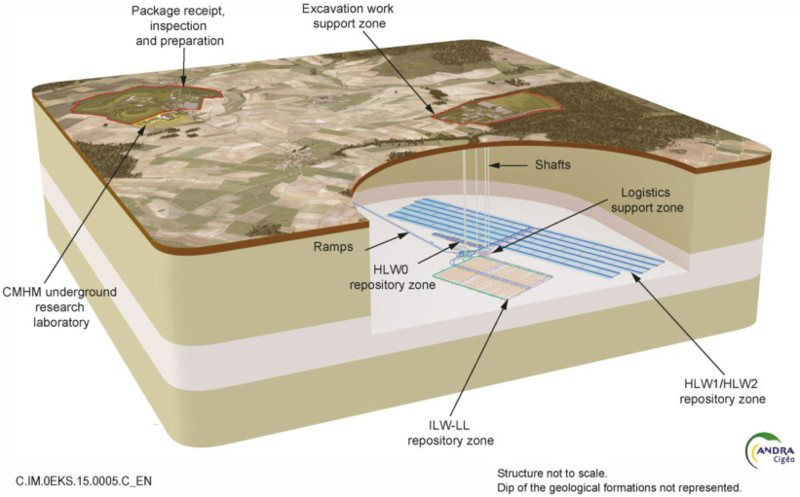
The high-level waste disposal concept must meet various safety requirements at different stages of the disposal facility lifetime: it must enable packages to be disposed of and potentially retrieved over a period of at least 100 years (‘reversibility’ requirement) and ensure that radioelements are fully confined for several centuries. To meet these requirements, two metal components have been specified in the French HLW disposal concept:
an overpack (Figure 2), containing the stainless steel primary canister, that ensures short-term functions (handling safety for primary packages) as well as confinement of the vitrified waste matrix with the absence of water during the thermal period (e.g.: corresponding to a temperature of vitrified waste matrix above 50 or 70°C depending on the type of waste) and radioactive decay phase. This containment phase lasts several centuries, with a minimum of 500 years. The overpack is composed of a carbon steel body and lid (forged steel P285NH since 2014) with a wall thickness of 65 mm (55 mm initially in 2005), and ceramic pads to help waste packages to be stored and potentially removed. Body and lid are electron beam welded. a microtunnel casing (carbon steel API 5L X65 grade with a fine ferrite microstructure), which must enable waste packages to be disposed of and potentially removed during the reversibility period. The cells (microtunnel) are in drifts (Figure 3). The cell length is about 100 m with a 70 cm diameter. R7-T7 vitrified waste container in disposal package (overpack). Cells (microtunnels) in galleries.
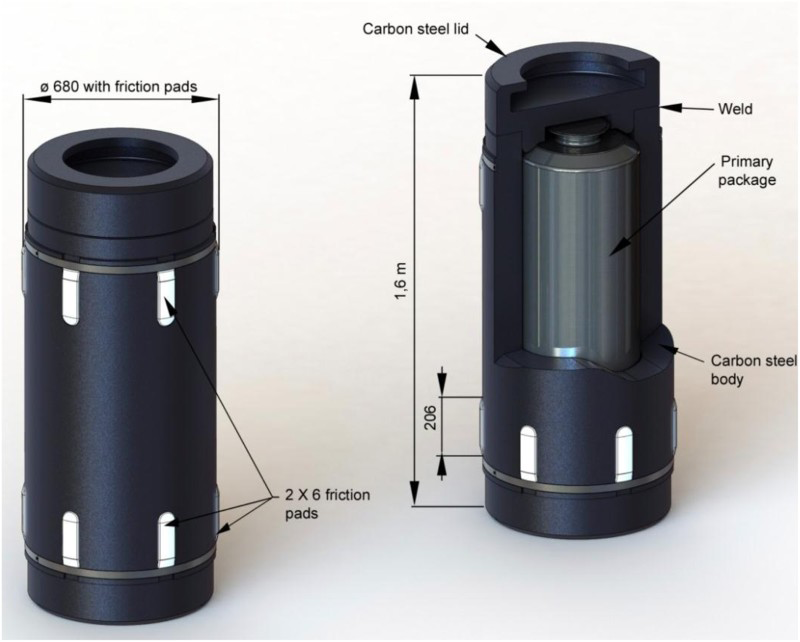
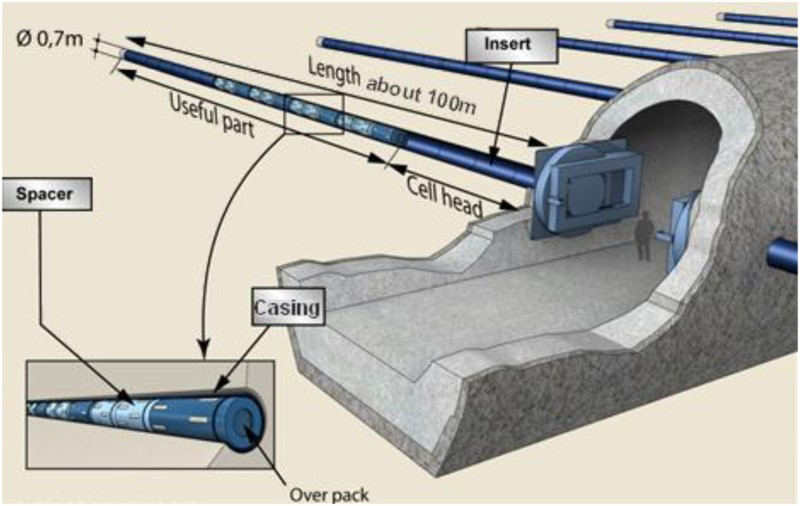
Environmental evolution of carbon steel components
The overpacks and casing will be exposed to an environment that will evolve over time: from a hot and humid atmosphere containing oxygen, to an anoxic environment saturated with water at the geothermal temperature of the Callovo-Oxfordian over the medium and long term. Between these two extreme states, the disposal cell will be subject to various hydraulic, thermal, physical–chemical and mechanical transients. Figure 4 presents the environmental evolution of the two steel components after four key situations: cell excavation, waste package insertion, cell closure and access drift closure.
Environmental evolution of carbon steel components.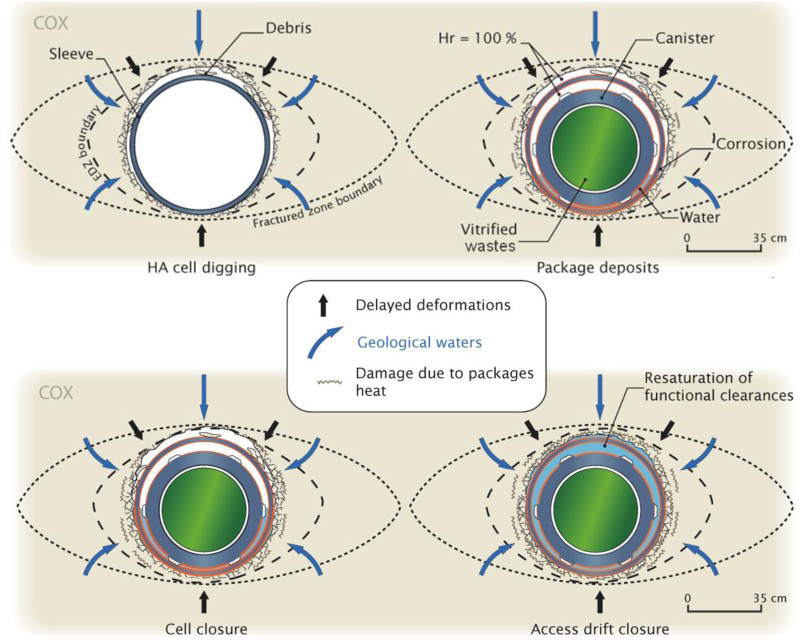
Corrosion experiments in various representative environments
Steel corrosion modes were therefore studied for various hydraulic (liquid porewater, steam), chemical (oxidising or anoxic environment, partial CO2 pressure), and geochemical (clay water, mixtures of water and unstructured argillite, compact argillite) environment conditions, while taking into account the influence of temperature, radiation, mechanical stresses and microorganisms. Data acquisition is based on considerable additional work performed by Andra's partner laboratories as well as in the Meuse/Haute-Marne URL.
Experiments in surface laboratories
The corrosion behaviour of carbon steels in conditions representative of a geological repository has been studied by a large number people [1–5]. To supplement and verify these results in the anticipated conditions of the repository concept proposed by Andra, long-term tests (several years) are performed at 90 and 50°C in order to determine the kinetics and the corrosion products of carbon steels in clay porewater or argillaceous suspensions, as well as in contact with compact claystone. These tests allow particularly characterisation of the nature and protective role of the corrosion products formed in these various conditions over long periods. Short-term tests (up to a few months) are also carried out to gain a clearer understanding of the behaviour of carbon steels in transient conditions (slightly acidic, etc.). Various steels are used for these tests, some of which comprise a weld bead. For example, to study the general corrosion of carbon steels in compact clay, three types of experimental set-ups were developed:
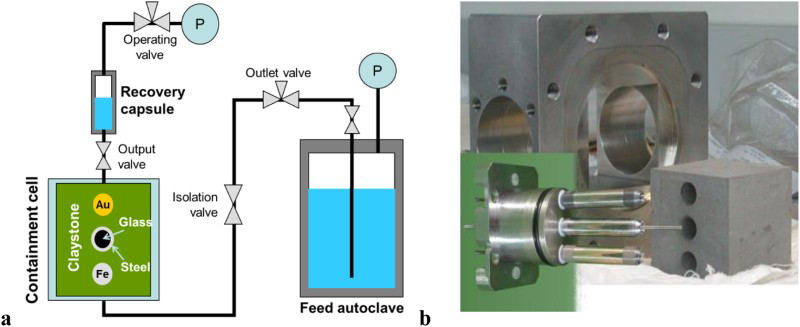
cells for studying the evolution of the instantaneous corrosion rate in clay porewater, argillaceous suspensions or claystone, by electrochemical impedance spectroscopy [6] (Figure 5); for the long-term tests, banks of cells containing steel specimens coated in claystone in anoxic conditions [7]; centimetric mock-ups representing an overpack inside a tube closed at its ends and drilled with calibrated holes to simulate penetrating defects in the lining. (a) General schematic diagram of the Arcorr test (CEA). The clay water, placed under a gas blanket in the autoclave, is injected into the heated test cell. The cell contains a brick of claystone where three cylindrical electrodes are inserted. (b) electrodes for EIS measures and machined COx claystone.
As carbon steels can be susceptible to stress corrosion cracking (SCC) in environments containing carbonates, a specific programme is being run to study this type of corrosion, in terms of crack initiation and crack propagation. The means used for this programme involve: (i) slow strain rate tests, at the corrosion potential or an applied potential; (ii) crack initiation tests under constant load on smooth or notched tensile specimens, or with applied strain in U-bend testpieces; (iii) crack propagation tests on fatigue pre-cracked fracture mechanic specimens.
The influence of γ-irradiations and microorganisms [8] on corrosion processes is also studied.
Experiments in the Andra's underground research laboratory
Meanwhile, a number of long-term experiments are being run in the Meuse/Haute-Marne URL:
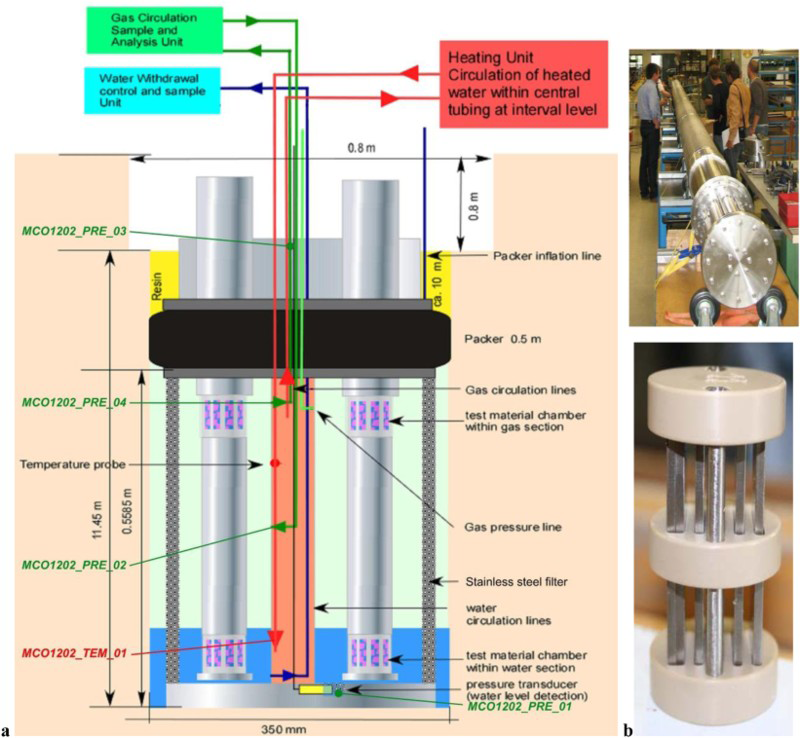
Investigation of the corrosion behaviour of various steels in clay porewater under anoxic conditions at 90°C, without direct contact with the claystone (Figure 6 representing long-term conditions of the gap between the casing and the overpack. Monitoring of the corrosion kinetics by gravimetric measurements and characterisation of the layers of corrosion products); Investigation of the corrosion behaviour of the same steels in the anoxic vapour phase at 90°C, before saturation of this gap (Figure 6); Monitoring of the corrosion rate of carbon steel samples in direct contact with the claystone, at 90°C, by electrochemical measurements (electrochemical impedance spectroscopy); ‘Stand alone’ tests at 50°C, common to several materials (glass, steels, clays) providing data on the kinetics and the corrosion products of carbon steel in contact with the claystone and over long periods (up to 20 years). The characterisations are performed after removal of the samples from several equipped boreholes, which will be dismantled at different specified times. Underground corrosion experiment: (a) instrumentation of the MCO 1202 drilling; (b) photographs of the complete system and steel coupons on the sample holder.
In all these experiments, an analysis of the microorganisms, their activity and their role in the corrosion process is also carried out.
Archaeological analogues
The investigation of archaeological artefacts is a means of accessing significant long-term duration. The corrosion products formed on tools buried for several centuries in an anoxic environment on the Glinet site (16th century steelmaking site located in Haute-Normandie, France) were thus characterised using sophisticated analysis techniques such as micro-Raman, nano-SIMS and STXM [9–12].
Modelling of general corrosion in clay environments
A robust and reliable prediction of the aging and interactions of engineered barriers in a repository situation necessarily involves modelling of the corrosion behaviour. A mechanistic approach was adopted several years ago and led to the development of thediffusion poisson coupled model [13] and the production of the Calipso corrosion code [14]. This code was recently coupled with a geochemistry-transport code, Kirmat [15]. This makes it possible to give a realistic representation of both the corrosion behaviour of iron as well as the alteration of clay. Another model [16,17] has also been developed to predict the evolution of the corrosion rate of carbon steel under a protective deposit.
Development of knowledge and main results concerning steel corrosion in the Callovo-Oxfordian claystone
Many environments lead to low uniform corrosion rates (less than 10 µm/yr). For example, it is the case with humid atmospheres where the particularity of disposal is that there are no wet/dry cycles, as well as different anoxic clay environments resulting in increasingly uniform corrosion states with low corrosion rates thanks to the formation of protective corrosion products (oxides, carbonates and iron silicates).
As it corrodes, iron generates alkalinity coming from water reduction, as well as the reduction of protons supplied by HCO3 − dissociation. The latter act as a reservoir of available protons in media containing CO2. The pH at the metal/electrolyte interface gradually increases and leads to the formation of siderite, thermodynamically stable under the pH and redox potential conditions of the environment. If the reaction system is supplied with CO2, the quantity of siderite is high and the pH stabilises below 8. If not, the siderite is limited to the available alkalinity in solution.
As corrosion of iron proceeds, the gradual increase in pH at the metal surface tends to stabilise the formation of siderite or chukanovite which acts as a protective deposit; in addition the formation of a fine magnetite or maghemite layer close to the metal surface occurs, which somehow protect the metal surface as a passive layer.
A comparable mechanism was proposed to demonstrate general corrosion of carbon steels in CO2 containing environments frequently met in oil and gas production [18–22].
However, recent results have revealed that possible chemistry transients of clay porewater may trigger significant corrosion rates of carbon steel leading to potential risks of premature failure of metal components.
High corrosion rates measured in situ following an oxidising/acidic transient period
An in situ experiment was conducted in the Meuse/Haute-Marne URL to investigate the corrosion of carbon steel coupons in the COx claystone. It consisted of a vertical and descending borehole 12 m deep, equipped with a 2-phase test chamber (porewater + gas produced by the COx claystone) containing steel coupons immersed in each medium. The results have shown that oxidation of minerals (pyrite) present in the COx claystone, caused significant pH drop of the environment in contact with the casing. Indeed, partial dehydration of clay as well as oxidation of pyrite (FeS2) in clay during the drilling phase of the borehole led to the formation of acidity (H2SO4) which promoted the dissolution of carbonates (CaCO3) and formation of CO2 in the test chamber. Then, dissolution of CO2 in water increased acidity and the CO2 partial pressure increased as the system was closed.
This acidic transient reached a pH of 4.5. Meanwhile, high corrosion rates of several hundred micrometers per year were measured, lasting beyond the acidic transient when the environment re-established neutral conditions thanks to the buffer effect of carbonates which was efficient when the CO2 partial pressure reached a steady state. In this case, carbon steel surfaces were covered by a non-protective layer, mainly made of a complex chloride compound (β-Fe2(OH)3Cl) underneath an iron carbonate layer, maintaining active dissolution of the metal surface even in near neutral pH [23,24].
Stress corrosion cracking
SCC tests in argillaceous suspension performed by CEA [25,26] showed that micro-cracks can initiate on a ferrite-pearlite and banded microstructure of carbon steel, under static load equal to 0.8 yield strength. However, these cracks remained relatively short and did not exceed 200 μm in length (Figure 7). In addition, crack propagation investigation revealed limited propagation of a transgranular crack [27] under a stress intensity factor of Micro-cracks initiated on a carbon steel (P265) exposed for 8000 h at 90°C in a 50-50 (wt-%) mixture of water and clay, under a stress equal to 80% the yield strength [25].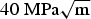 .
.
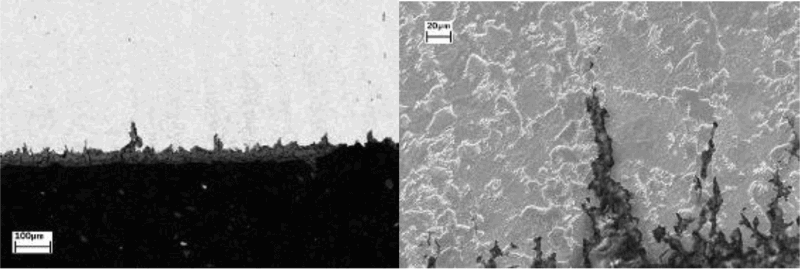
Metallographic observations clearly revealed the banded microstructure (ferrite + pearlite) of the base metal as well as the presence of many inclusions. For the weld, the hardness in the heat affected zone (HAZ) was far too high (>22 Hv), therefore promoting environmentally assisted cracking (EAC). Overall, the first SCC tests in clay environment have demonstrated the importance of the microstructure with respect to crack initiation and propagation. Therefore, optimised carbon steels have been selected for the following SCC studies, with a particular emphasis on the importance of the microstructure.
Influence of γ-irradiations on corrosion
The radiolysis of humid air or water in the vicinity of the overpack and casing enhances the production of oxidising species (such as H2O2) or chemical acid species (such as HNO3). This results in a rise of the general corrosion rates of carbon steels, as well as localised corrosion such as pitting.
These processes are partially known in simplified media (tests in pure or slightly mineralised water), at room temperature, in the presence of air and for high dose rates (often well beyond 100 Gy/h). It has been shown that the effect of the radiolysis of water on corrosion could be significantly attenuated in the absence of oxygen [28].
Furthermore, these tests are generally performed over very short periods (less than a few days) which means that the long-term effects on corrosion cannot be observed, especially a possible attenuation of corrosion rates.
Andra launched with CEA and SUBATECH a study programme on this subject in order to obtain data corresponding to the environment anticipated in a claystone disposal repository: in a gaseous atmosphere initially expected under repository condition, before resaturation (formation of nitric acid), and in an argillaceous, aqueous, anoxic environment with the influence of temperature, more relevant to long-term repository conditions. Corrosion tests were carried out under gamma radiation for 1 year with dose rates similar to those expected with different overpack thickness (several dozen Gy/h).
The first corrosion results with a dose rate of 80 Gy/h revealed an increase of the corrosion rates in comparison to the case with the absence of radiation. No influence of humid air or water radiolysis was observed around 20 Gy/h (anoxic conditions and residual oxygen content). Experiments with oxygen are currently in progress. A study performed by SKB [29] also showed that in the presence of a dose rate of 11 Gy/h, the kinetics of corrosion were comparable to those observed without radiation after 10 months of testing (passivation phenomenon).
Evolution of disposal concepts and choices of materials linked to corrosion processes
Some key results obtained with the corrosion tests have influenced the design and materials involved in the HLW disposal cell [30]. The main changes are highlighted as follows:
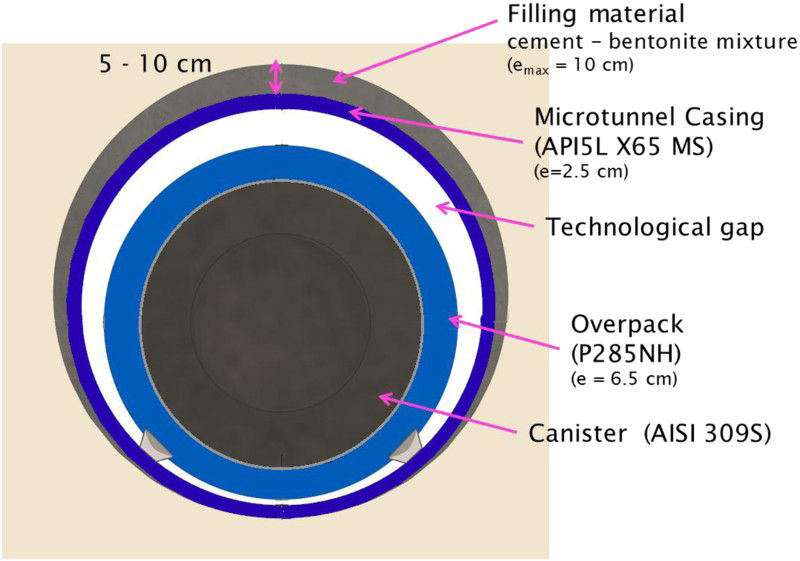
The unfavourable influence of oxygen on general and localised corrosion led to a slight change of the HLW disposal concept in 2014. An alkaline grout material will be injected between the casing and the host rock (Figure 8). This is expected to neutralise the residual acidity for the entire disposal cell and limit the oxygen ingress from the ventilated drifts as much as possible during the operating phase. This will likely promote slow uniform corrosion rates from the casing emplacement onwards. At this stage, the selected grout material is cement-based with the addition of bentonite without any organic additives (superplasticiser). Considering the possible role of hydrogen in SCC mechanisms, steel grades, offering the best possible resistance to hydrogen embrittlement have been selected and these steels undoubtedly also offer improved resistance to intergranular SCC that can be feared in a more alkaline environment: API 5L X65 MS used in the petrochemical industry, which has a fine-grain even ferritic microstructure and strong mechanical properties (for the casing) and P285 NH (forged steel) with an impurity-controlled composition (for the overpacks). After welding the overpack lid with the electron beam technique, stress-relief treatment will be applied to eliminate steel internal residual stresses and limit the risk of SCC in the weld (particularly the HAZ). The thickness of the overpack will be determined so as not to exceed a dose rate of more than 10 Gy/h on its external surface. Filling material between the casing and clay (cement–bentonite mixture).
R&D programme
A R&D programme has been defined and launched to investigate the influence of the cement-based grout material on casings and overpacks (from corrosion and mechanical behaviour point of view). All experiments are carried out in surface and underground research laboratories, on samples taken from full-scale mock-ups including welding and heat treatment of the weld. The main issues are described herein:
Experiments are carried out to determine the corrosion kinetics as well as the risks of localised corrosion in the expected environments: casings and overpacks will be exposed in direct contact with the cement-based grout or in seepage water percolating through the cement-based grout. These experiments have taken into consideration the role of residual oxygen, radiations and microorganisms. Some experiments have been dedicated to study the risk of localised corrosion emanating from passivation or environmental heterogeneity (for instance, inside the casing, with an environment containing water and gas or at the base of the casing in mixed contact with cement and clay). The risk of EAC has been investigated with various experiments focusing on crack initiation under constant load, crack propagation for a range of stress intensity factor, as well as slow strain rate tests under potentiostatic conditions promoting EAC. In addition crack velocity monitoring by using the electric method is under progress. Corrosion modelling will continue on the role of protective layers and passive layers, but also on corrosion/geochemistry and corrosion/mechanical coupling. The mechanical behaviours of the overpacks and casing will be modelled taking into account both the anisotropic mechanical loading of the COx as well as the different types of corrosion in repository conditions [31].
