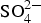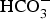Abstract
Stress corrosion cracking of P235 and P265 steels is investigated through slow strain rate and constant deformation tests on samples exposed in a cell reproducing the underground conditions of the French deep geological nuclear disposal, at 25 and 90°C. The samples were taken from the base metal, the heat affected zone and the fusion zone of a P265 steel weld joint. Tests were performed at the free corrosion potential and under polarisation in the cathodic or anodic domains. Long-term constant deformation tests were also carried out to evaluate the behaviour of the material under static loading in hydrated clay environment. The results show a significant sensitivity of the tested materials to environmental cracking in the experimental conditions, with an enhancement of the effect of the environment for the weld metal. A mechanism of hydrogen embrittlement is proposed to be responsible for the cracking phenomenon. Susceptibility to environmental assisted cracking observed during these tests indicates the need to optimise welding parameters and steel chemical composition and metallurgy, particularly regarding the presence of inclusions in the base metal and stress relief of the weld metal.
This paper is part of a supplement on the 6th International Workshop on Long-Term Prediction of Corrosion Damage in Nuclear Waste Systems.
Introduction
The concept of geological disposal developed by Andra for the high-level nuclear waste is based on the use of a multi-barrier system, in which the vitrified nuclear waste is contained first in a stainless steel canister, which is in turn encapsulated in a second, external carbon steel canister, the overpack [1,2]. One of the reasons for the use of carbon steel for the external canister is that it is less sensitive than some passive materials to localised corrosion, such as pitting and crevice corrosion, particularly in the presence of chloride ions contained in the geological environment (clay) and at 90°C, the temperature expected to be maintained during the first several decades by the residual radioactivity of the waste [2,3]. However, even if the behaviour of carbon steel is well documented and makes it a good candidate material for this application, the risk of stress corrosion cracking (SCC) and hydrogen embrittlement has to be taken into account [4,5].
Chemical composition of the waters used in the studies on iron corrosion in geological disposal and on SCC of buried pipelines.

aThe composition of the water is the result of a thermodynamic calculation based on the mineral composition of the rock and the thermodynamic database of the BRGM.
bThe composition of the water was given as a medium composition from solutions sampled in different sites where failures has been reported.
In chemical conditions considered representative of pipelines environments, two mechanisms leading to SCC have previously been identified [6]. The first is observed in low pH solution and is associated with transgranular cracks (TGSCC), the second occurs in alkaline pH and is associated with intergranular cracks (IGSCC). TGSCC occurs at open circuit potential in dilute carbonate/bicarbonate solutions and it is thought to be promoted by hydrogen stress cracking (HSC) or hydrogen enhanced localised plasticity (HELP). IGSCC occurs at a potential just below the transition active/passive behaviour of the steel in alkaline pH. This process, due to the presence of a pseudo-passive layer of magnetite is generally proposed to be related to film formation/fracture mechanism. Both IG and TGSCC are promoted by dynamic stresses, but under constant stress, such as residual stress, crack initiation and propagation is possible. Concerning the role of steel metallurgical parameters on SCC, the authors agree that there are large differences between the steel grades. These differences are mainly due to the microstructure and its effects on the material's strength, microchemistry and inclusions. Thus, there are many grades that are sensitive to SCC [8,9]. Furthermore it is known that heterogenic microstructures and non-metallic inclusions enhance the risk of failure by SCC [10].
Materials and methods
Materials
Chemical composition of the P235 and P265 carbon steels (in wt-%).

The metallographic analysis of the external canister leads us to subdivide the P265 component into three zones: base metal (BM), heat affected zone (HAZ) and fusion zone (ZF). The P265 BM (Figure 1(a)) and the P235 metal show a biphasic microstructure with ferritic grains, alpha iron, and pearlitic bands, a mixture of alpha iron and iron carbide, as a result of the forging process.
Optic micrographs of the P265 base metal (a - BM) and of the P265 heat affected zone (b - HAZ) after an exposition of in a 2.5% natal solution in order to reveal perlite which appears in black bands.
The microstructure of the P265 HAZ (Figure 1(b)), in the vicinity of the fusion zone shows a heterogeneous structure of pearlite and micrometric grains of ferrite. The P265 fusion zone (FZ) can be further subdivided into three zones as shown in Figure 2: (i) an external fusion zone (EFZ) which corresponds to the solidification of a mixture of gamma iron (austenite) and liquid metal, (ii) a medium fusion zone (MFZ) with large (1-2 mm long) austenitic grains that grow following the heat gradient, and (iii) an axial fusion zone (AFZ) with equiaxial austenitic grains. During cooling, in the initial austenitic grains three phases have precipitated: (i) ferrite at the grain boundaries, and two hard phases in the grain core, (ii) acicular ferrite and (iii) globular bainite.
False colour micrograph of the P265 Fusion zone (FZ) from optic micrograph after an exposition of a 2.5% nital solution. Ferrite appears in light grey (or in red), acicular ferrite in dark grey (green) and bainite in black (blue). EFZ refers to external fusion zone, MFZ to medium fusion zone and AFZ to axial fusion zone.
The hardness of the BM (both P235 and P265) is between 130 and 180 Hv, while in the P265 HAZ up to 250 Hv has been measured and the hardness of the fusion zone (P265 FZ) has been measured between 190 and 310 Hv.
Experimental procedures
The sensitivity of stress corrosion cracking of P235 and P265 steels in storage environment has been studied using slow strain rate tests which were conducted on cylindrical samples (diameter 4 mm, length 16 mm). Samples were at open circuit potential (OCP), at imposed potentials in the cathodic and anodic domains, at 90 and 25°C, in a cell containing the synthetic interstitial representative Bure water (Table 1). At 90°C, experiments were performed under 50 bars with 50 mbar of CO2 leading to a pH of 8. At 25°C, the pH was maintained at 7 through bubbling a mixture of 5 ppm CO2 in nitrogen (1 bar). Uniaxial tensile tests were conducted at a slow strain rate ranging from 10−6 to 10−7 s−1.
The initiation of stress corrosion cracking under conditions representative of a storage environment has been investigated with static mechanical solicitations. Only P265 steel samples have been tested under static loading at 90°C in the Bure clay synthetic water (Table 1) and also in a mixture of 50% by weight of water and Bure clay (‘argilite’) for a period of 1 year in both cases. Two solicitations have been investigated: 0.8 and 1.2 Rp0.2%.
Results and interpretation
Sensitivity to stress corrosion cracking
As illustrated in Figure 3 for P265 BM, the stress–strain curves obtained at 25°C in Bure representative water (pH = 7) at various imposed corrosion potentials show an important decrease of the elongation when compared to the curve obtained in air (5.10−7 s−1), meaning a susceptibility of the P265 to stress corrosion cracking. The imposed potential of −750 mV/SCE (Saturated calomel electrode) corresponds to the free corrosion potential. Similar results have also been obtained for the P265 at 90°C and for P235 at 25°C.
Stress–strain curves obtained at 25°C in Bure representative water (pH = 7) with P265 BM at various imposed corrosion potentials compared to the curve obtained in air/solicitation: 5.10−7 s−1.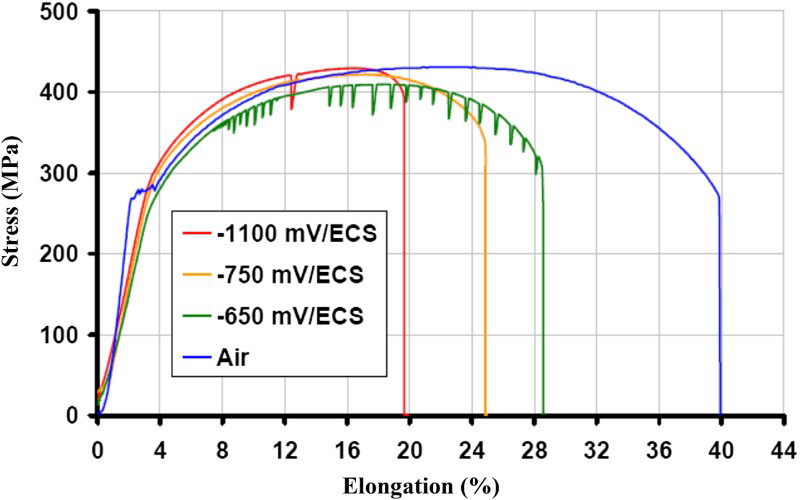
To quantify the susceptibility to SCC, a specific indicator, I SCC introduced by Torres-Islas [11,12], was used. Initially, the percentage of reduction of area (RA%) after a slow strain rate tests is defined by the following equation:
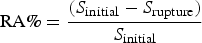
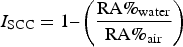
With P235, studies have been mainly performed at 25°C and have given similar results as with P265 BM, even if the P265 BM is slightly more susceptible to SCC than P235. For instance, at the free corrosion potential in Bure representative solution and for a solicitation of 10−6 s−1, the percentage of the reduction area, RA, is 64% for P265 BM (77% in air) and 74% for P235 (81% in air), leading to respective I SCC of 0.17 and 0.09. For low cathodic potentials (−1100 mV/SCE) in the same solution and with the same solicitation, RA is 45% for P265 BM and to 51% for P235, which corresponds to a cracking criteria equal to 0.42 for P265 BM and 0.37 for P235. An example of a crack observed in the plastic deformation zone is shown in Figure 4 for P235 steel that was exposed at the open circuit potential or polarised at −1100 mV/SCE (the same morphology is observed in both conditions).

Regarding P265, the fusion zone (FZ) shows significant loss of ductility in Bure representative water, which is a greater loss than the BM or the HAZ at temperatures of either 25°C (pH = 7, 10−6 s−1) or 90°C (pH = 8, 10−7 s−1), as illustrated in Figure 5. This behaviour seems to follow the same trend than the evolution of the hardness, as expected and as discussed above.
Susceptibility to SCC of the P265 different weld domains in Bure representative solution (90°C, open circuit potential, pH = 8, 10−7 s−1). BM: base metal; HAZ: the heat affected zone; MZ: the melting zone.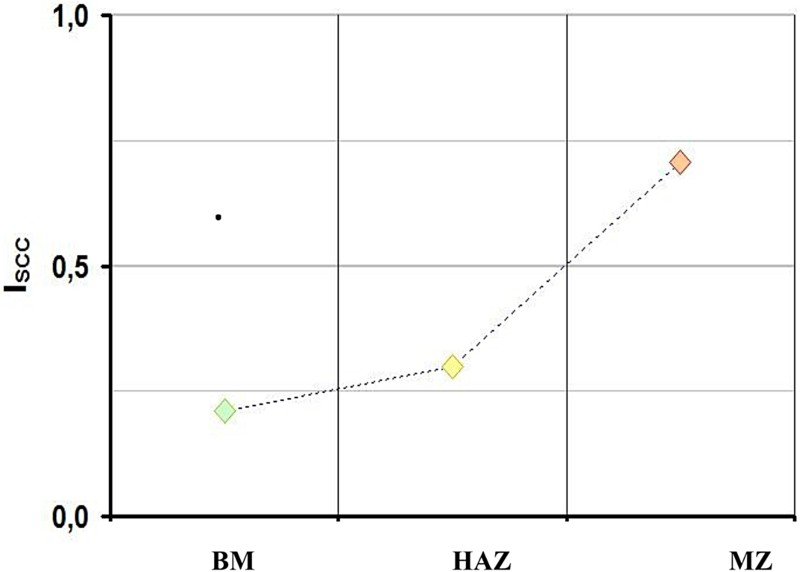
The susceptibility of the P265 steel to SCC is increased by slow strain rate and is clearly promoted by cathodic potential, −1100 mV/SCE (Figure 5) which in turn is clearly correlated to hydrogen embrittlement [13]. Furthermore, from 90 to 25°C, the I SCC factor increases significantly from 20 to 50%. This behaviour clearly illustrates that hydrogen induced cracking (HIC) could occur in the fusion zone at the range of temperatures tested (25-90°C). In the BM, the effect is maximal at room temperature (i.e. 25°C). Figure 6 also shows that even though there is no SCC at anodic potential below the potential of hydrogen stability, there is still a susceptibility of the fusion zone to SCC.
Influence of the imposed potential on the susceptibility of P265 to stress corrosion cracking in Bure representative water at 25°C (pH = 7) and at 90°C (pH = 8) and for the base metal (MB), the heat affected zone (ZAT) and the melting or fusion zone (ZF) at three different solicitations (10−6, 5. 10−7 and 10−7 s−1).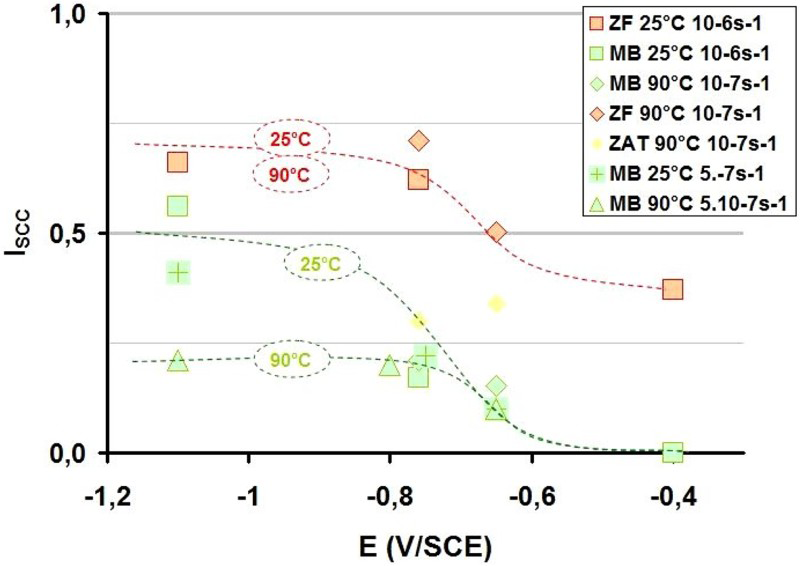
Initiation of stress corrosion cracking
Constant load tests were performed using specimens of P265 steel (BM, HAZ and MZ) exposed to a 50% mixture of clay and water under two constant loads: 0.8 and 1.2 Rp0.2%, at 90°C over the period of one year. After 1 year of exposure, multicracking was observed on BM, HAZ and molten zone (MZ), as illustrated in Figure 7. On BM, cracks seem to be localised to pearlite bands and could be the result of preferential chemical attacks due to the galvanic coupling between the pearlite and the ferrite. this highlights the deleterious effect of band structures. On welded zones (HAZ and MZ), transgranular cracking is observed and may be attributed to a hydrogen embrittlement phenomenon caused by the excessive hardness of the welded zone (up to 310 Hv), as it is recommended that hardness should not exceed 250 Hv for these type of carbon steels [14,15].
Observations of cracks on P265 specimens exposed during 1 year in a 50% clay-water mixture at 90°C at constant loads. BM: base metal; MZ: molten zone.
Conclusion
The results of this study show a significant sensitivity of the tested P235 and P265 carbon steels to environmental cracking in the testing conditions (Bure representative water, at 25 and 90°C), under slow strain rates and also under constant loads, with an enhancement of the stress corrosion cracking phenomena for the weld metal. A mechanism of hydrogen embrittlement is considered to be responsible for the cracking phenomenon. Susceptibility to environmental assisted cracking observed through these tests illustrates the need to optimise the steel chemical composition and metallurgy (particularly regarding the presence of inclusions in the BM), the welding parameters (to avoid the formation of harmful phases such as bainite) and the stress relief by heat treatment of the weld zone. Our findings therefore suggest that it is important to avoid bands structures and to reduce the hardness of the weld area.