Abstract
In the context of the French repository for high-level wastes, carbon steel (C-steel) materials are currently considered among the most cost effective and reliable materials for the prediction of long-term corrosion behaviour. An in situ experiment was conducted at the Meuse/Haute Marne underground research laboratory to investigate the corrosion behaviour of different grades of C-steel up to a maximum temperature of 85°C. C-steel coupons were placed in a two-phase test chamber containing liquid and gas emanating from the Callovo-Oxfordian host rock, at given times and for specific durations, under anoxic conditions. After their exposure in the test chamber, various characterisations were carried out. Overall, after 36 months, the results highlight a potential acidic transient. This could promote high corrosion rates of C-steel. Therefore, an alternative concept has been considered since 2014, which involves the addition of a cement–bentonite-based grout in order to buffer the possible acidic transient.
This paper is part of a supplement on the 6th International Workshop on Long-Term Prediction of Corrosion Damage in Nuclear Waste Systems.
Introduction
Carbon steel (C-steel) materials are foreseen to be used as structural materials in many repositories that will be implemented in clay soils (Switzerland, France) [1,2]. In the French repository (so-called Cigéo ‘Centre industriel de stockage géologique’) for intermediate and high level long-lived nuclear waste, it is planned to use a multi-barrier system emplaced at a depth of 500 m in an argillaceous (45% clay, 25% carbonates and 30% silts) rock called the Callovo-Oxfordian (COx) claystone [3]. High level waste (HLW) will be cooled in a glass matrix emplaced in a stainless steel container to form the primary package. It will be inserted into an overpack made of C-steel that will essentially ensure the protectiveness of the glass matrix during the thermal phase (T ≤ 70°C). The overpack will be introduced into a disposal cell, consisting of a horizontal borehole 80 m long with a diameter of 0.9 m, cased with C-steel [3].
In the present study, the corrosion processes of different C-steel grades (P235, P275 and SA516) were investigated, by applying gravimetric measurements, surface characterisation and assessments of microbial activity. Overall, the investigations lasted for 3 years with a variety of changes made to the thermo-hydro-chemical conditions in the test chamber.
Materials and method
A vertical and descending borehole was drilled under N2 down to 12 m in the COx claystone. One week after the drilling phase, equipment with a test chamber and a packer ensuring air tightness were introduced and heated to 85°C using a heating system located in the surface gallery.
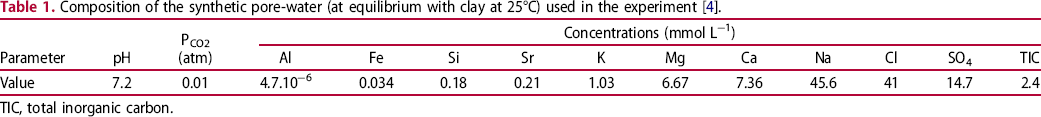
The test chamber contained two environments: liquid (initially synthetic pore-water (see Table 1 [4]), which was gradually replaced by water seepage) and gas emanating from the COx host rock. In addition, water circulation was possible because of a pressure gradient between the test chamber (at 3 bars) and the host rock (up to 40 bars), and also between the test chamber and the surface gallery at atmospheric pressure. A water analysis unit, consisting of a cabinet equipped with an Eh redox potential electrode, a pH electrode, a conductivity electrode and water cell vials, was used to sample water and study the chemistry and microbial activity of the liquid phase. Three steps were considered in this study; each one corresponded to the introduction of C-steel coupons with a ferrite–pearlite microstructure (see Table 2 for their chemical composition and Figure 1 for a representative microstructure), at a given time into specific environmental conditions in the test chamber: t = 0 for step 1, t = 644 days for step 2 and t = 1040 days for step 3. For each step, the coupons were removed after different exposure times.
Microstructure of P235 grade C-steel coupon perpendicular to the rolling direction, after etching in a 10% nital solution. Composition of the synthetic pore-water (at equilibrium with clay at 25°C) used in the experiment [4]. TIC, total inorganic carbon. Chemical composition of the low-alloy carbon steel used in the corrosion experiment (wt-%).

Once removed from the test chamber, the coupons were rinsed with ethanol and dried to prevent post-removal oxidation. Gravimetric measurements were performed on some of them which had been weighed before the experiment. In addition, they were weighed before and after being soaked in a solution of 50wt-% H2O/50wt-% HCl (37wt-% HCl concentration) containing 5 g L−1 hexamethylenetetramine ((CH2)6N4) as an inhibitor of iron dissolution under acidic conditions. This was repeated three times and the weights of the coupons were measured using a high accuracy balance (MS205DU, Mettler Toledo, resolution 0.01 mg) to allow an estimate of the average general corrosion rate (CR). The damaged thickness, thcor (µm), was calculated according to Equation (1).
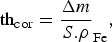
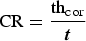
Some coupons were kept so that the corrosion layers formed at the metal surface could be characterised using microscopic, micro-spectroscopic and micro-diffractometric techniques on cross sections. µ-Raman spectroscopy (LabRAM HR, Horiba), scanning electron microscopy (SEM, JEOL JSM7000-F, Jeol) and X-ray diffraction with a 2D detector (GE Healthcare image plate) were used.
Microbial investigation was carried out on water sampled from the test chamber and coupons (mainly SA516 grade) that were swabbed. The most probable number technique was used to determine the number of anaerobic fermenting bacteria, strict aerobic bacteria, sulphate-reducing bacteria (SRB) and thiosulphate-reducing bacteria (ThRB) present per mL of solution or per cm2 of the coupon surface [5].
Results and discussion
Environmental evolution
Different parameters were monitored online or measured at given times. The temperature was gradually increased from ambient to 85°C (at 77 days) and then maintained at 85°C except when two failures of the heating system occurred at 709 days and 1440 days, respectively. The former resulted in a loss of air tightness in the test chamber, enhancing a decrease of pressure down to atmospheric pressure and therefore a loss of pressure gradient necessary for continuous outflow-rate and monitoring of the pH, Eh redox potential and conductivity.
The value of Eh evolved throughout the experiment. During the first 300 days, it was positive (from 300 mV to 0 mV/SHE), revealing an initial oxidising episode. Beyond 709 days, the measurements were not relevant anymore as they were only performed on stagnant water, located in the lines of the water unit surface, with no circulation.
Figure 2 presents the evolution of the values of pH and Fe dissolved in solution. It shows an acidic pH transient, reaching a minimum value of 4.5 during the first 300 days, coinciding with the oxidising episode. Figure 2 also highlights an increase in the concentration of Fe dissolved in the liquid phase, which reached 960 ppm (1.7 × 10−2 M). The pH transient can be explained by the initial exposure to air, which oxidised the pyrite present in the COx claystone as expressed in Equation (3). Consequently, H+ and sulphate were released, which increased the acidity. From 300 days onwards, the pH returned to near-neutrality, as shown in Figure 2. This can be explained by the buffer effect of carbonates contained in the COx claystone, which can neutralise acidity by the reactions expressed in Equations (4) and (5). Figure 3 shows the evolution of the partial pressure of CO2, with an increase during the oxidising and acidic pH transient followed by a steady-state period, consistent with the neutrality of the test chamber. Once the partial pressure of CO2 had become relatively stable, the buffering effect of the carbonates became efficient and allowed a return to near-neutral pH values.
Evolution of the pH and Fe content dissolved in solution. Evolution of PCO2 overtime at room temperature (dashed line) and 80°C (plain back line).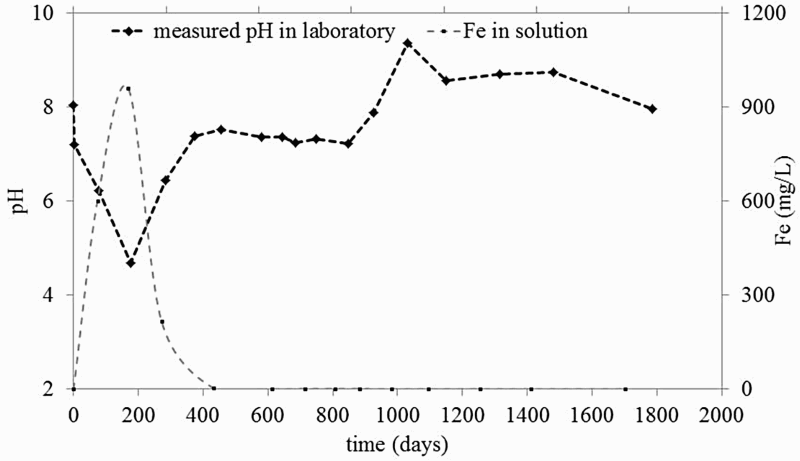
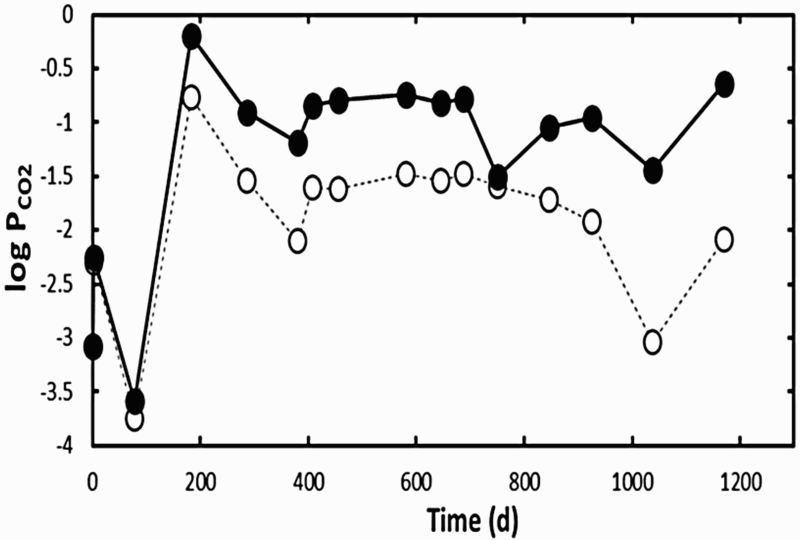
Figure 4 shows the evolution of key species such as chloride, sulphate and sodium. It highlights an increase in their concentrations during the first 300 days, which coincides with the oxidising episode. Concentrations of dissolved Cl− ([Cl−]aq) and Na+ ([Na+]aq) increased from 1330 mg L−1 (3.8 × 10−2 M) to 2750 mg L−1 (7.8 × 10−2 M) and from 930 (4.0 × 10−2 M) to 3100 mg L−1 (0.13 M), respectively. Then the concentrations decreased down to the reference value for [Cl−]aq and 7.8 × 10−2 M for [Na+]aq. From t = 900 days, the concentrations increased again to reach 2520 mg L−1 (7.2 × 10−2 M) and 4330 mg L−1 (0.19 M) for [Cl−]aq and [Na+]aq, respectively, at t = 1172 days.
Evolution of the concentrations for chloride, sodium and sulphate.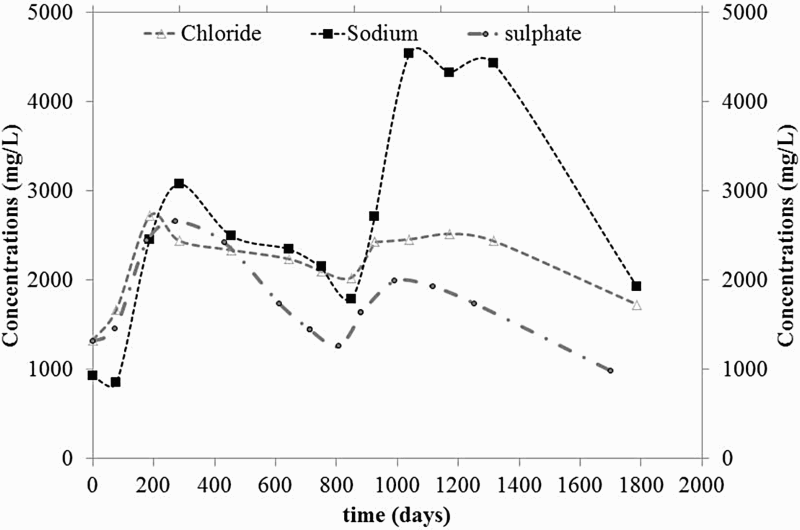
The initial increase in sulphate is a consequence of pyrite oxidation, as expressed in Equation (3). Regarding chloride and sodium, it is well established that the drilling process dries the walls of the borehole. In other words, salts precipitate and when exposed to water, they dissolved thereby increasing the concentration in the pore-water [6]. The decrease in concentration after 300 days is just a dilution effect, due to water seepage. The increase in concentration shown from 800 days is a consequence of the water level variations that occurred following the first heating system failure, which caused further leaching of the wall boreholes containing salts.
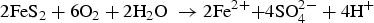
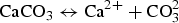
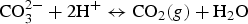
Corrosion processes
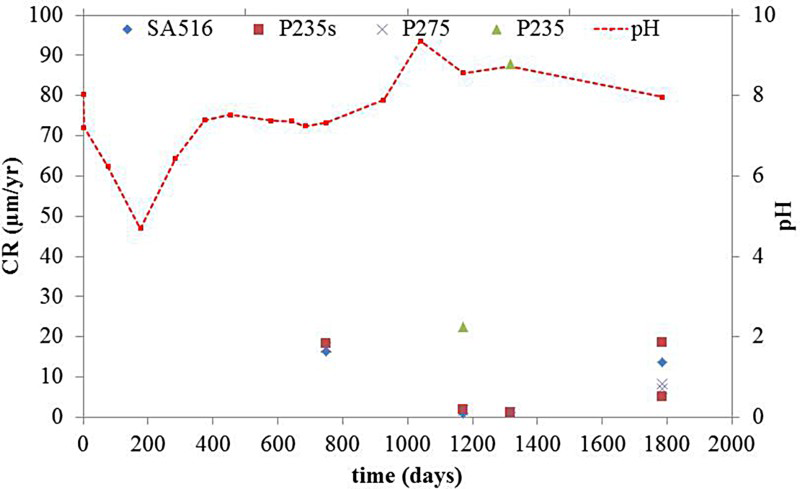
The steel grade did not influence the CR, either in the gas or liquid phase, where non-uniform general corrosion was observed for all the coupons. In the gas phase, the CR was very low, with a mean value of 2 µm/year and a maximum value of 12 µm/year for the coupons subjected to an immersion episode after the breakdown of the heating system. XRD and Raman analyses showed that the metal surface was essentially covered by magnetite. On the other hand, the coupons exposed to the liquid phase revealed different corrosion behaviours depending on the pH of the medium to which they were exposed. For instance, coupons from step 1 were subjected to the acidic pH transient and exhibited a high CR up to 250 µm/year (Figure 5). The corrosion layers were composed of β-Fe2(OH)3Cl at the metal surface, as well as mackinawite (Fe(1-x)S(x) with 0<x<0.1) and siderite (FeCO3) in the outer layer. The coupons, introduced later in the test chamber (steps 2 and 3) and exposed to near-neutral pH conditions, revealed a much lower CR with an average of 10 µm/year and a maximum of 30 µm/year (Figure 6). Their surfaces were covered with Fe-silicate (Cronstedtite (Fe3.5Si1.5O5(OH)4) and greenalite ((Fe3Si2O5(OH)4) and/or magnetite (Fe3O4) when maintained at 85 °C. However, the coupons exposed to near-neutral conditions and subjected to temperature variations revealed traces of β-hydroxychloride (β-Fe2(OH)3Cl) at the metal surface. The results obtained by gravimetry and surface characterisation clearly indicate the role of the acidic pH transient in promoting active corrosion, while near-neutral pH conditions led to a protective film developing on the metal surface. On the one hand, it seems that magnetite and Fe-silicate tend to protect the metal surface (coupons from step 2 and step 3 exposed at 80°C). On the other hand, β-hydroxychloride (β-Fe2(OH)3Cl) inhibits its protectiveness and favours active corrosion (coupons from step 1) [7,8]. When present simultaneously with magnetite and Fe-silicate, corrosion seems to be limited (coupons from step 3 subjected to temperature variations).
Evolution of the CR in the liquid phase: coupons from set 1 exposed initially (acidic conditions). Evolution of the CR in the liquid phase: coupons from sets 2 and 3 exposed from t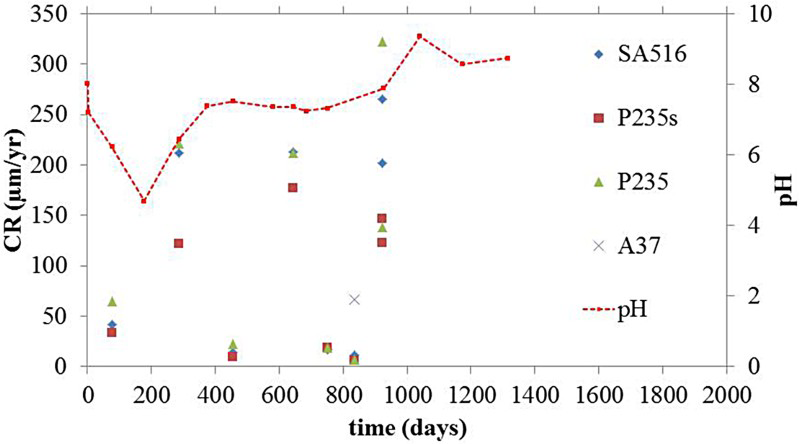
Mesophilic and thermophilic sulphidogenic bacteria, such as SRB and ThRB, were detected during the whole experiment. The results revealed no clear relationship between the CRs and bacteria content. However, the coupons exposed for a longer time revealed a higher content of bacteria (up to 104 bacteria/cm2). The presence of mackinawite in the corrosion layers of some coupons probably results from the metabolism of SRB, which reduce sulphate to sulphide and therefore enhance Fe-sulfide production, as expressed in reaction (6). This assumption is consistent with the decrease in sulphate content, despite water seepage that introduces sulphate continuously.
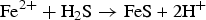
Conclusions
The exposure of C-steel coupons was spread over three steps involving different time durations, temperatures and hydrological conditions, allowing significant investigation of the corrosion processes in media composed of pore-water and gas. Overall, most coupons suffered from non-uniform general corrosion. The coupons from step 1, which were exposed to the acidic environment, revealed a high CR, reaching 250 µm/year, that was sustained beyond the acidic transient. The corrosion layers were essentially composed of β-Fe2(OH)3Cl in direct contact with the metal surface, as well as mackinawite and siderite in the outer layer. In comparison, coupons exposed to near-neutral conditions seemed to be significantly less corroded, with an approximate CR of 10 µm/year; for these coupons, magnetite and Fe-silicate were widely observed on the surface. Nevertheless, β-Fe2(OH)3Cl was also present on the coupons subjected to temperature variation under near-neutral conditions. This observation has revealed the need to investigate the mechanisms involved in β-Fe2(OH)3Cl formation. Regarding the coupons exposed to the gas phase, they showed a low CR with a mean value of 10 µm/year, as well as magnetite in the inner corrosion layer.
This study has shown that an acidic and oxidising transient occurs in COx claystone environments, which should be taken into consideration for the design of future HLW disposal cells, as it may have an impact on the design of the metallic barriers foreseen in the future French repository. Following this study, the current French reference concept for HLW has evolved slightly and it is proposed to inject a cement-based, alkaline grout material between the host rock and the casing in order to neutralise the potential acidic transient.
Footnotes
Acknowledgements
The authors thank Solexperts AG (Switzerland) for their contribution to the experiment and the interesting discussions on the experimental set-up, and Hydroisotop GmbH (Germany) for the chemical analyses.
Disclosure statement
No potential conflict of interest was reported by the authors.
