Abstract
The supercontainer is the reference concept for the post-conditioning of vitrified high-level radioactive waste and spent fuel in Belgium. It comprises a prefabricated concrete buffer that completely surrounds a carbon steel overpack. In this highly alkaline environment (pH ∼13.6) and under normal conditions (i.e. without the ingress of aggressive species), the carbon steel overpack will be protected by a passive oxide film, which is expected to result in very low uniform corrosion rates. Considering the long timescales applicable to waste disposal, determining accurate and reliable estimates of the uniform corrosion rate under anoxic conditions forms a very important part of the safety and feasibility strategy. On the other hand, the occurrence of localised forms of corrosion cannot be neglected and, therefore, it is also crucial to demonstrate that no other form of corrosion apart from uniform corrosion can take place. This paper gives an overview of the status of the research into the uniform corrosion, pitting corrosion and stress corrosion cracking behaviour of P355 QL2 grade carbon steel exposed to an artificial concrete pore water solution.
This paper is part of a supplement on the 6th International Workshop on Long-Term Prediction of Corrosion Damage in Nuclear Waste Systems.
Keywords
Introduction
The supercontainer (SC) is currently being studied as the reference design for the final disposal of vitrified high-level radioactive waste (VHLW) and spent fuel (SF) in Belgium. It is essentially a massive cylindrical prefabricated concrete block, named the buffer, into which a watertight cylindrical carbon steel container, the so-called overpack, holding either VHLW canisters or SF assemblies, will be inserted. In the currently preferred design option, the concrete block will be fitted into an outer stainless steel container, termed the envelope [1].
A Portland cement-based (PC) concrete has been chosen for the buffer, because it will provide a highly alkaline chemical environment, in which the external surface of the overpack will be passivated, and it is expected only to be prone to slow uniform corrosion (passive dissolution) [2 -9]. Considering the long timescales in waste disposal, determining accurate and reliable estimates of the uniform corrosion rate under anoxic conditions becomes very important.
On the other hand, however, metals that are protected by a passive oxide film are known to be susceptible to localised breakdown if the right environmental conditions arise [10–13]. This can result in accelerated dissolution of the underlying metal, which could lead to premature penetration of the carbon steel overpack. Therefore, it is crucial to demonstrate that no form of corrosion other than uniform corrosion can take place. Considering the environmental conditions that the overpack is expected to be exposed to during the disposal period, pitting corrosion and stress corrosion cracking (SCC) are considered to be potentially the most detrimental forms of attack.
Experimental details
Chemical composition of the reference P355 GL2 grade carbon steel.

Apart from the pitting corrosion experiments, carried out in saturated Ca(OH)2 solution (pH ∼12.5) and in saturated Ca(OH)2 solution + NaOH (pH ∼13.5) at 25°C, all experiments were conducted in artificial concrete pore water solution, which is a simplified version of the pore water expected for the PC-based buffer material in the SC concept, known as young cement water (YCW). The test solution had a chemical composition of 0.14 M Na+, 0.37 M K+, 2.4 mM 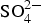 and a calculated pH value of 13.6.
and a calculated pH value of 13.6.
Anaerobic uniform corrosion studies
The hydrogen gas liberated during the anaerobic corrosion process was measured by using either (1) a barometric gas cell technique or (2) sealed autoclaves equipped with pressure transducers. In the barometric gas cell technique, the testpieces were placed in zirconia crucibles that were inserted in gastight glass cells. Zirconia crucibles were used, because of their high resistance to chemical and radiochemical degradation in alkaline conditions. Any gas produced in the gas cell caused a pressure increase, which resulted in an increase in the height of a mercury column in a glass manometer, which was connected to the glass cell. By measuring the height of the mercury, it was possible to calculate the volume of gas produced and hence to derive the corrosion rate (based on the formation of magnetite as the final corrosion product). The results were corrected for temperature and external atmospheric pressure fluctuations [14–16].
Carbon steel wire (1017 grade) with a diameter of 0.4 mm was cut into a predetermined length giving a total exposed surface area of 0.1 m2. Before testing, the steel wires were pickled under anoxic conditions in 10% HCl, in order to remove any residual air-formed oxide, then rinsed in deionised water and dried. Preparation of the test solutions and filling of the test cells was conducted in an argon-purged glove box to ensure initial anoxic conditions in the cells [15].
Gas cell experiments were carried out to study the effects of temperature (25 and 80°C), gamma irradiation (0 and 25 Gy h−1, with 25 Gy h−1 being the expected dose rate at the surface of the 3-cm thick carbon steel overpack), chloride concentration (0 and 100 mg L−1), sulphide concentration (0 and 500 mg L−1) and thiosulphate concentration (0 and 100 mg L−1). Experiments were also conducted in mixed 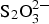 , S2− and Cl− solutions to investigate synergistic effects.
, S2− and Cl− solutions to investigate synergistic effects.
Pitting corrosion studies
Passivity breakdown of the protective oxide film was studied using the potentiodynamic polarisation technique. A typical three-electrode electrochemical cell was used. The test specimens (working electrodes) were machined from a 3-cm thick plate into cylinders of 1 cm diameter and 2.5 cm length. The peripheral area of the cylinders was masked using Kapton polyimide film, leaving a total exposed surface area of 0.785 cm2. The specimens were mechanically wet-ground, with successively finer SiC papers, down to 1200 grit, then cleaned with acetone and double-distilled water and finally dried under flowing N2 gas. A Pt plate was used as counter electrode, and a commercial Ag/AgCl (sat. KCl) electrode, connected to the cell via a Luggin probe, was used as the reference electrode. All potentials are reported versus the Ag/AgCl (sat. KCl) scale. The potentiodynamic experiments were recorded at a sweep rate of 0.1667 mV s−1 [17].
Stress corrosion cracking studies
Stress corrosion cracking susceptibility was examined via the slow strain rate testing (SSRT) technique. Tests were performed in YCW under anoxic conditions at 140°C, at a constant strain rate of 5 × 10−7 s−1 at open circuit potential (OCP). The SCC susceptibility was evaluated using the results of SSRT in Ar (as an inert reference environment) at the same temperature and strain rate. The effect of the presence of sulphide (100 and 500 mg L−1) was investigated.
Three welding processes are considered to be potential techniques for the final closure of the carbon steel overpack: submerged arc welding (SAW), metal active gas (MAG) welding and reduced pressure electron beam (RPEB) welding. Tensile test specimens were prepared from as-welded 3-cm thick plates (no heat treatment was applied). The specimens were machined entirely (in the longitudinal direction) from the base metal, heat-affected zone (HAZ) and weld (Types I, II and III, respectively). Specimens whose gauge section included the weld, HAZ and, in some cases, part of the base metal were also machined (Type IV). The SSRT tests were carried out on cylindrical, notch-free uniaxial tensile specimens 55 mm long and 5 mm in diameter (with a gauge length of 10.5 mm and a gauge diameter of 2.5 mm, giving a cross-sectional area of the gauge section of 4.9 mm2).
The fracture surfaces were analysed using optical microscopy (OM) and scanning electron microscopy (SEM).
Experimental results
Anaerobic uniform corrosion studies
Figure 1 shows an example of the anaerobic corrosion rates (assuming magnetite as the corrosion product), for unirradiated (closed symbols) and irradiated conditions (open symbols). Very high initial corrosion rates were observed that dropped rapidly to very low values, of the order of <0.1 µm yr−1. These low values were also confirmed by weight loss measurements.
Corrosion rate evolution for 1017 grade carbon steel in YCW (determined by measuring the H2 production by means of barometric glass gas cells) as a function of temperature and chloride concentration under unirradiated (closed symbols) and irradiated conditions (25 Gy h−1) (open symbols). Expansion of the Y-axis at low rates (right graph) illustrates the low long-term corrosion rates of the order of <0.1 µm yr−1.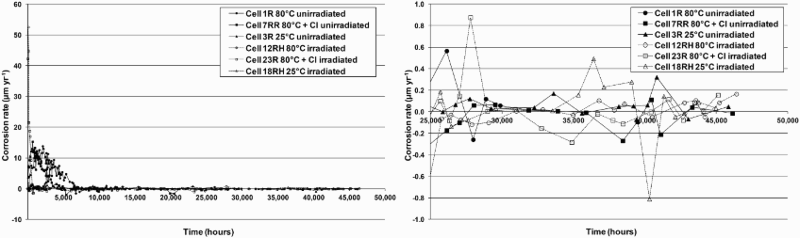
No significant deleterious effect of radiation on the long-term corrosion rate, at the dose-rate tested (25 Gy h−1), has been determined. The anaerobic corrosion rates were not significantly increased by the presence of 100 mg L−1 Cl−, 500 mg L−1 S2− or a mixture of 500 mg L−1 S2−, 100 mg L−1 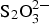 and 100 mg L−1 Cl−, at 25 or 80°C.
and 100 mg L−1 Cl−, at 25 or 80°C.
The anaerobic corrosion rate was also measured by monitoring the hydrogen evolution using sealed autoclaves that were equipped with pressure transducers. From these tests, anaerobic corrosion rates as low as 0.03 µm yr−1 were obtained (Figure 2). This corrosion rate corresponds to a loss of 3 mm over an exposure time of 100,000 years, attesting to the great general corrosion resistance of carbon steel in highly alkaline solution under anaerobic conditions.
Corrosion rate evolution for 1017 grade carbon steel in YCW (determined by measuring the H2 production by means of pressure transducers).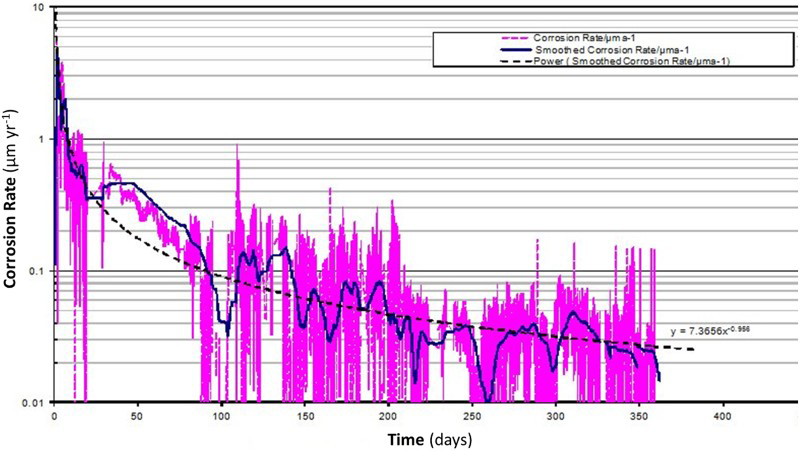
Samples of the corrosion product from a number of cells were analysed by SEM/EDX and Raman spectroscopy. These examinations showed that the corrosion product was predominantly magnetite at 80°C, with distinct crystalline deposits, which are assumed to form upon a protective barrier layer. At 25°C, a spinel structure was observed and the corrosion product layer was a uniform black layer with no distinct crystals visible in OM.
Pitting corrosion studies
As seen from the data in Figure 3, when the potential is swept in the positive direction, the polarisation curve exhibits a wide passive range, which is largest for the lowest chloride concentration (left graph), followed by a sharp increase in the polarisation current density signifying passivity breakdown at a critical pitting potential. In some cases, such as for a 0.1 M chloride concentration at 25°C (right graph), a sharp increase in the current density was observed at potentials higher than 0.7 V vs Ag/AgCl(sat. KCl), which is due to the onset of the oxygen evolution reaction rather than passivity breakdown. The passivity breakdown potential (E b) shifted towards more active (negative) values with increasing chloride concentration (left picture) and increasing temperature (right picture). The potentiodynamic polarisation plots exhibited a number of short-period current spikes before the critical breakdown potential was reached. These current spikes are due to breakdown of the passive film followed immediately by repassivation (i.e. due to metastable pitting) [17].
Potentiodynamic scans performed on P355 QL2 grade carbon steel in (i) saturated Ca(OH)2 at 85°C as a function of chloride concentration (left graph) and (ii) in saturated Ca(OH)2 + 0.1 M NaCl as a function of temperature (right graph) [17].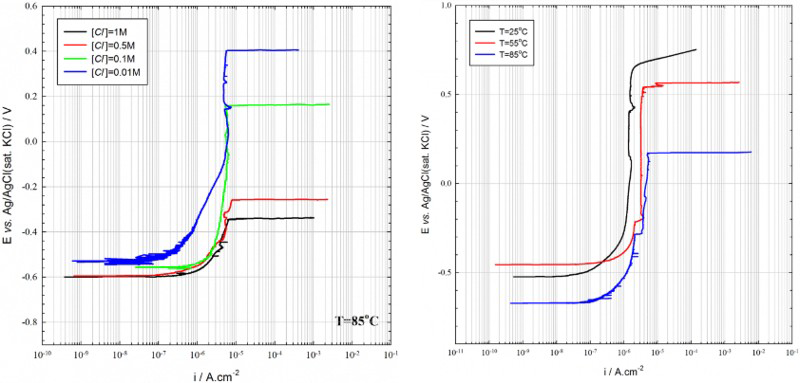
Potentiodynamic polarisation experiments were typically repeated 15-20 times (for each condition) to obtain a statistical distribution of the breakdown potential, E b (Figure 4). A newly polished specimen was used for each run. The distribution in E b followed a near-normal distribution. The satisfactory correlation between the experimental and calculated distribution in E b confirms the validity of the point defect model (PDM) for describing the breakdown of the passive film on the grade of carbon steel tested [17].
Calculated cumulative probabilities (solid lines) in E b for carbon steel in deaerated sat. Ca(OH)2 with different chloride concentrations at 85°C compared with the experimentally determined distribution (marked points) [11].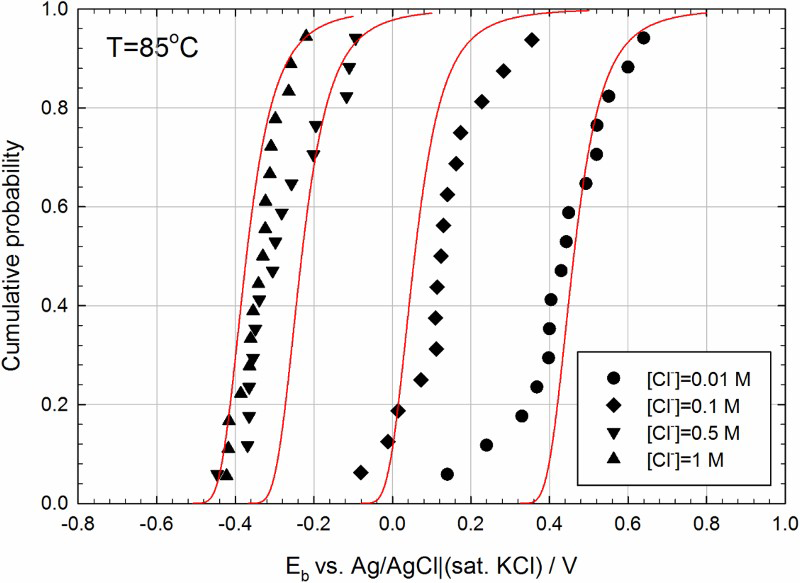
The data from the potentiodynamic polarisation measurements were interpreted in terms of the PDM with the aim of extracting values for important fundamental parameters. The effects of chloride concentration, temperature and scan rate, as used in the potentiodynamic polarisation determinations of the critical E b, were studied. The polarisability (α) of the film/solution interface was determined to be ∼0.16 and was found to be insensitive to temperature. The critical areal concentration (ξ) of condensed cation vacancies at the metal/film (m/f) interface that leads to passivity breakdown, as predicted by the PDM, was calculated from the experimental dependence (Figure 5) of the critical E b on the square root of the voltage sweep rate (υ 1/2) as being <1.09 × 1016 cm−2. This value is in excellent agreement with that calculated from the unit cell dimensions of the base metal (ξ ≈ 1.53 × 1016 cm−2) and the barrier oxide layer (Fe2O3) (ξ ≈ 6.89 × 1015 cm−2) for vacancy condensation upon the metal lattice or upon the cation sublattice of the film, respectively [17]. The ‘less than’ operator precedes the experimentally determined value of ξ, because the passive current density, which is used in its calculation, is not due to the migration of cation vacancies alone, but is dominated by the migration of metal interstitials. Thus, only a very small fraction of the passive current density at breakdown can be attributed to the migration of cation vacancies, which is responsible for passivity breakdown.
Measured E b for carbon steel in deaerated sat. Ca(OH)2 +1 M NaCl solution at 25°C as a function of scan rate [11]. The open circles represent the results from three different tests conducted under the same conditions.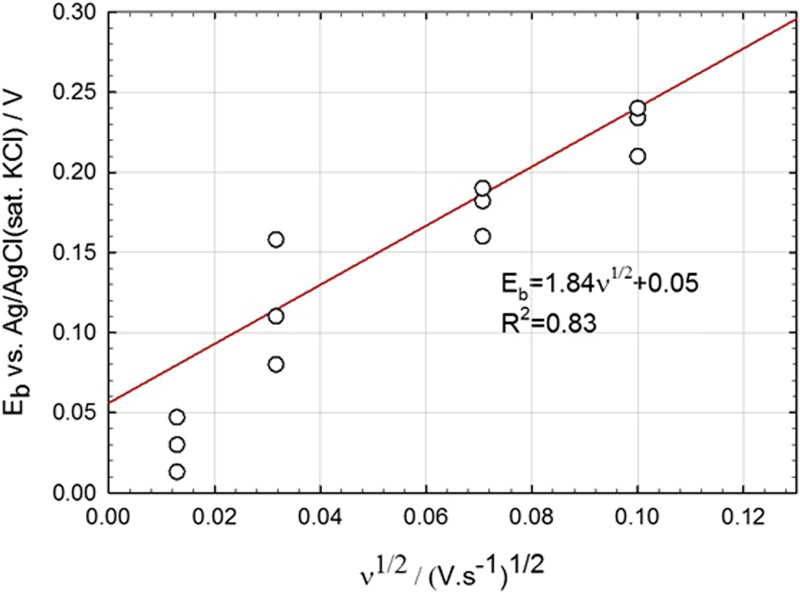
Stress corrosion cracking studies
Figure 6 shows typical plots of the stress–strain behaviour of welded tensile specimens of the P355 QL2 grade carbon steel tested in YCW with various additions of sulphide (100 and 500 mg L−1) under anoxic conditions. All tests were conducted at 140°C under OCP. The stress–strain curves of the tensile specimens tested in argon (reference environment) are plotted as black curves for comparison. The strain to failure of the MAG-, SAW- and RPEB-welded tensile specimens tested in the sulphide-containing solutions (100 and 500 mg L−1) was comparable to that of the specimens tested in the solutions without sulphide and that in Ar.
Stress–strain curves for MAG (left), SAW (middle) and RPEB (right) welded tensile specimens machined entirely (in longitudinal direction) from the weld (type III).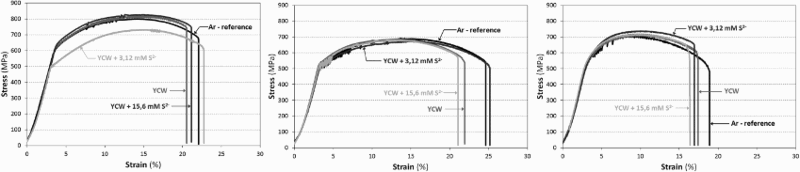
SEM images of the fracture surfaces of MAG-welded tensile specimens are shown in Figure 7. The micrographs in the top row were taken at 45× magnification, showing the entire fracture surface. The micrographs in the bottom row were taken at a higher magnification (1000×), showing a zone representative of the entire fracture surface. At this higher magnification, no differences in appearance between the fracture surfaces of the specimens tested in Ar and those of the specimens tested in YCW (with and without sulphide) were observed. The entire fracture surfaces of all specimens were found to consist only of ductile dimples, which are indicative of a purely mechanical fracture; in other words, the presence of sulphide did not change the SCC behaviour of the P355 QL2 grade carbon steel under the tested conditions.
Analysis of the fracture surfaces of MAG-welded tensile specimens machined entirely in the longitudinal direction from the weld (Type III specimens).
Conclusions
The SC is the reference concept for the post-conditioning of VHLW and SF in Belgium. It comprises a prefabricated concrete buffer that completely surrounds a carbon steel overpack. In the SC design, the carbon steel overpack is expected to be protected by a passive oxide film, which is postulated to result in very low uniform corrosion rates. Considering the long timescales applicable to waste disposal, determining accurate and reliable estimates of the uniform corrosion rate under anoxic conditions forms a very important part of our safety and feasibility strategy. On the other hand, the occurrence of localised forms of corrosion cannot be neglected and, therefore, it is also crucial to demonstrate that no other form of corrosion apart from uniform corrosion can take place.
This paper provides an overview of the status of research into the uniform corrosion, pitting corrosion and SCC behaviour of P355 QL2 grade carbon steel in artificial concrete pore water solution (representative of the PC-based buffer material in the SC).
In general, very good agreement was found between the anaerobic corrosion rate data obtained using different measuring techniques (e.g. weight loss and hydrogen evolution measurements). This has increased our level of confidence in estimating reliable values for the anaerobic corrosion rate of carbon steel in high pH concrete. The long-term anaerobic corrosion rate is <0.1 µm yr−1 and continues to decrease with time (based on the measurement of the amount of H2 gas using barometric glass gas cells). Using pressure transducers, anaerobic corrosion rates as low as 0.03 µm yr−1 were measured. This corrosion rate corresponds to a loss of 3 mm over an exposure time of 100 000 years, attesting to the great general corrosion resistance of carbon steel in highly alkaline solution under anaerobic conditions.
All the observations and results presented in this paper provide convincing evidence for the validity of the PDM for modelling passivity breakdown on carbon steel in Belgium's HLW disposal concept. At ambient temperature (and at low chloride concentration), the critical pitting potential lies within the O2 evolution region, but the critical pitting potential rapidly shifts in the negative direction with increasing temperature.
The presence of sulphide (up to 500 mg L−1) does not seem to lead to an increased risk of SCC of carbon steel in artificial concrete pore water solution under anoxic conditions.
Footnotes
Disclosure statement
No potential conflict of interest was reported by the authors.
