Abstract
An unexpected dose related increase in oral squamous cell carcinomas was observed in a standard 2-year carcinogenicity study with a novel calcium channel blocker, in which Wistar rats received daily doses of 0, 1.5, 7, 20, or 40 mg/kg of the compound mixed with a standard diet containing fibers from barley. This finding was associated with an increased incidence of severe (destructive) periodontitis and the formation of oro-nasal fistulae at the 2 highest doses. Five assays of the compound for genotoxicity were negative indicating that a genotoxic effect was highly improbable. To investigate the underlying pathogenic mechanisms a second 2-year study in the same strain of rats was initiated and the influence of the diet and/or a possible local irritancy by the drug was assessed. In this second study the compound was administered by oral gavage at daily doses of 0, 7, or 40 mg/kg (later reduced to 20 mg/kg due to systemic intolerance) to rats maintained either on the standard diet or on a low fiber diet assumed to be less aggressive in terms of inducing periodontal lesions. Dose dependent gingival overgrowth (a class-related effect) was observed in the incisor and molar teeth area of all treated groups but was independent of the diet used. No oral tumors were found in the standard diet or low fiber diet controls and all treatment groups fed the low fiber diet, whereas in the high-dose group fed the standard diet a total of 8 oral squamous cell carcinomas were detected in association with an increased incidence of severe periodontitis. These results indicate that the increased incidence of squamous cell carcinomas observed upon chronic administration of the compound is not due to a direct tumorigenic effect of the drug. Tumor formation is attributable to severe periodontal disease favored by the diet and class related gingival overgrowth.
Keywords
Introduction
Spontaneous squamous cell carcinomas of the oral cavity are relatively rare in rats (Brown and Leininger, 1994). Chronic ingestion of diet containing fibers from oat and barley and the ensuing severe chronic periodontitis are reported to be predisposing factors leading to their formation(Madsen, 1989). A novel calcium channel antagonist, mibefradil dihydrochloride, was associated with an unexpected increase in the number of squamous cell carcinomas of the oral cavity in a standard 2-year dietary admixture carcinogenicity study in rats, in the presence of increased periodontitis. To investigate the underlying pathogenic mechanisms and to assess the influence of the diet and/or a possible local irritancy by the drug a second 2-year study in the same strain of rats was initiated.
Material and Methods
Compound
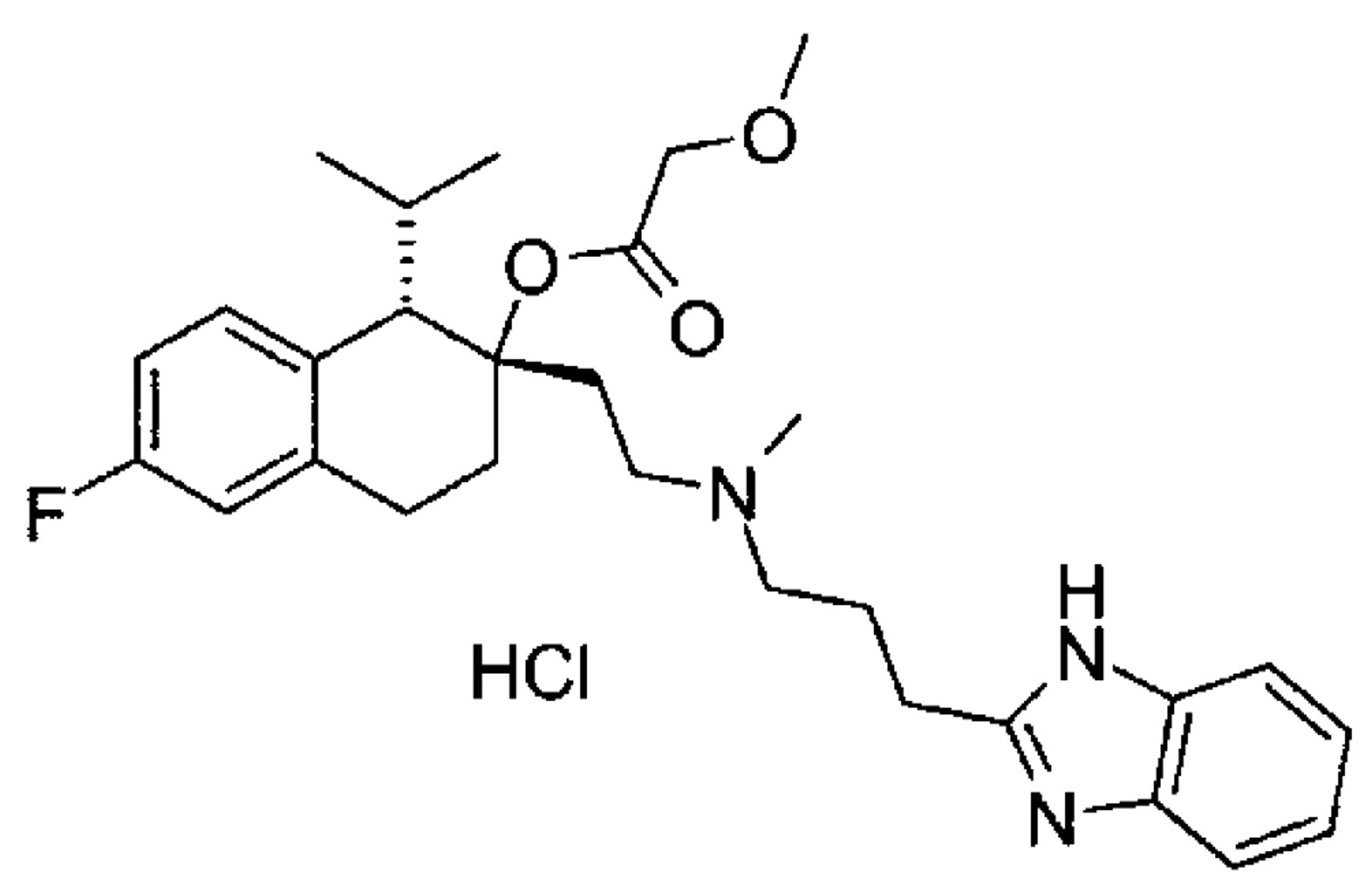
Mibefradil dihydrochloride (1S,2S)-2-[2-[[3-(2-benzimi-dazolyl)propy]methylamino]ethyl]-6-1,2,3,4-tetrahydro-1-isopropyl-2-naphthylmethoxyacetate dihydrochloride), CAS RN: 116666-63-8, referred to hereafter as “mibefradil,” has the chemical structure shown below:
Animals and Housing Conditions
The standard carcinogenicity study was performed at F. Hoffmann-La Roche, Ltd., Switzerland. The investigatory study was carried out at Covance Laboratories, Harrogate, England. In both studies male and female Wistar rats (strain HanIbm:Wist) obtained from Biological Research Laboratories Ltd. (Fuellinsdorf, Switzerland) were used. At start of dosing the animals were 7–8 weeks old. The animals were housed in air-conditioned rooms in which the temperature was maintained at approximately 22°C with a daily 12 hours light/dark cycle. Two rats were housed in each cage in the first study and five rats in each cage in the second study. Water and diet were supplied ad libitum. All animals were provided human care in compliance with the Swiss regulation on animal welfare (standard carcinogenicity study) or the provisions of United Kingdom National Law, in particular the Animals (Scientific Procedures) Act, 1986 (investigatory 2-year study).
Diet
Two different powdered rodent diets were used, that is, a standard diet which is manufactured from, among other materials, barley and a special diet that is not derived from barley. Fiber content of the diets was assessed subjectively under the microscope and far more fibers were seen in the standard diet. The special diet was therefore thought to be less aggressive in terms of the induction and promotion of periodontitis in rats (Robinson, 1985). The composition of each diet is given in Table 1.
Study Designs
In the standard carcinogenicity study mibefradil was administered by feed admixture in daily doses of 1.5, 7, 20, or 40 mg/kg for 2 years (Table 2). In addition 2 control groups were used. Each group consisted of 50 male and 50 female rats and all rats were fed the standard diet. Routine examinations included clinical observations, body weight, food consumption and hematology. Plasma level determinations for the parent compound were made in months 3, 6, 12, 18, and at the end of the study using a reverse phase HPLC method with fluorescence detection.
In the subsequent investigatory 2-year study the drug was dosed as an aqueous solution by oral gavage (for study design see Table 3). It was given in daily doses of 7 and 40 mg/kg at a constant dose volume of 5 ml/kg/day. The high dose (40 mg/kg/day) for mibefradil treated females was reduced to 30 mg/kg/day in Weeks 27–70 and for mibefradil treated males it was reduced to 7 mg/kg/day in Weeks 67–70. From Week 71 to 2 years both sexes in these groups were dosed at 20 mg/kg/day. These changes were necessary in view of increased mortality associated with high plasma drug levels when compared to rats of the standard carcinogenicity study (Plasma Analysis). Three control groups were used. The groups consisted of at least 65 males and 65 females. For toxicokinetic evaluation additional 5 males and 5 females were used in control group I and 10 males and 10 females in each of the mibefradil treated groups. To evaluate reversibility additional 50 males and 50 females were treated at 40 mg/kg/day for 44 weeks and thereafter set on recovery until completion of the study (week 101–104). The standard diet was fed to 1 control and 1 high-dose group. In the other groups the special diet was used. Routine examinations included clinical observations, body weight and food consumption. Plasma level determinations for the parent compound were made at the beginning of the study and in months 3, 7, and 18.
Necropsy and Histopathology
All animals, including those prematurely dead or killed as well as the toxicokinetic animals (investigatory study), were necropsied. Sections of all major tissues and organs and samples of necropsy findings were fixed in 10% neutral buffered formalin. Bones were decalcified in an aqueous solution of 5% formic acid and 5% formaldehyde (37%). In the standard carcinogenicity study all tissues were embedded in paraffin wax, sectioned at about 4 μm and stained with hematoxylineosin for routine histological examination, whereas in the investigatory study only the head and masses from the head were processed for histological examination. The sections through the oral and nasal cavities were performed according to Figures 1 and 2. For each animal in the first study three levels of the nasal cavity and the upper jaw were examined. In the second study four sections of the oral and nasal cavities, including the lower jaw (Figure 2, sections 2, 3, 4, 5) and, for most animals, an additional section from the base of the mandibular incisors (Figure 2, section 1) were examined. When necessary, serial sections were prepared in both studies to elucidate the nature and extent of the lesions.
Results
Standard Dietary Carcinogenicity Study
In controls and animals at 1.5 and 7 mg/kg/day body weight development was similar throughout the study. In the 20 mg/kg/day dose group a slight decrease in body development (about 10%) occurred starting at the 18th month of treatment. At 40 mg/kg/day body weight development was lower from the beginning and final body weights were approximately 20% lower than those in controls. Average food intake was similar in the control groups and the 7, 15, and 20 mg/kg/day dose groups. At 40 mg/kg/day there was a slight reduction (approximately 10%) in food intake. At 20 and 40 mg/kg/day several animals had swollen and reddened gums around the incisors, however this finding was not followed up due to a technical error. A small number of animals developed nodules in the molar regions of the upper and lower jaws (first observed in week 40), which clinically resembled dental abscesses. Most of these nodules were diagnosed histologically as squamous cell carcinomas (Figures 3 and 4). The remaining nodules showed hyperplastic features typical of the so-called drug-induced gingival overgrowth (Hassell and Hefti, 1991) the well known nonpremalignant class-effect of calcium channel antagonists (see below. In contrast to the second study in this study gingival overgrowth was diagnosed in individual rats only and is therefore not listed in Table 2. The reason was that in this study only the upper jaw and only the molar area was assessed and—as known subsequently from the investigatory study—this change is more pronounced on the lower jaw. Furthermore the marked inflammatory process made it difficult to distinguish between drug-induced and reactive hyperplasia). In addition to the large macroscopically visible tumors, microscopic tumors were also detected.
The incidence of the lesions is summarized in Table 2. Squamous cell carcinomas were found in 13/100 animals treated with 40 mg/kg/day and in 5/100 animals treated with 20 mg/kg/day. Controls and lower dose groups had comparable incidences (1/100 animals treated with 7 mg/kg/day, 2/100 animals treated with 1.5 mg/kg/day and 1/100 in one of the 2 control groups). The tumors grew very aggressively and invaded adjacent tissues, including the nasal cavity. Varying degrees of tumor differentiation were seen, ranging from well-differentiated keratinizing to poorly differentiated non keratinizing tumors.
An enhanced incidence of severe periodontitis was noted at the highest doses (for 20 mg/kg/day see footnote b in Table 2) in association with the high tumor incidence. Periodontitis of varying degrees, essentially in the area of the molar teeth, was detected in both control and treated animals. It was often associated with entrapped foreign bodies (i.e., food particles, hairs) (Figure 5). The inflammation varied from mild (grades 1–2 on a scale from 1–5) to moderate to severe (grades 3–5 on a scale from 1–5). In advanced cases (grades 3–5) it often extended to the alveolar bone resulting in destruction of alveolar bone, leading—in its most extreme form—to the development of oro-nasal fistulae (Figure 6). In mibefradil-treated animals an increased incidence of destructive periodontitis was observed and at the high-dose (40 mg/kg/day) this finding was associated with a high incidence of oro-nasal fistulae (for 20 mg/kg/day see footnote b in Table 2). In some animals of the high-dose group the fistular epithelium showed varying degrees of dysplasia, and squamous cell carcinomas seemed to arise from the epithelium of the fistulae (Figure 7).
Investigatory (Oral Gavage) Carcinogenicity Study
Control animals fed the standard diet tended to gain more weights than the special diet fed animals in the rapid growth phase although overall body weight gains were similar. During the second year of the study high-dose animals tended to gain less body weight than the controls, irrespective of the diet they were fed. Control males ate similar quantities of food whether fed the standard or the special diet, however control females fed the standard diet ate slightly more than those fed the special diet. Overall the pattern of food consumption was inconsistent between the sexes and the diets, i.e., high-dose males fed the standard diet ate more than the controls and high-dose females fed the standard diet ate less than the controls. As observed in the first study, swollen/reddened gums in the immediate vicinity of the incisors were seen in high-dose group animals.
This finding was first observed in week 34 in males and week 36 in females. The incidence was comparable at this dose level irrespective of the diet. The lesion was diagnosed as overgrowth of the gingiva of the incisors. Gingival overgrowth was also seen in the molar area and was more prominent in the lower jaw (Figures 8 and 9).
The animals were affected to a variable extent and, in some rats the hyperplastic gingiva had a diameter of more than 5 mm (Figure 10). The typical histological feature of this lesion was a distinct proliferation of the fibrovascular tissue of the lamina propria. In addition, slight hyperplasia of the squamous epithelium was seen, partly with elongated rete pegs projecting downwards into the lamina propria. Mixed inflammatory cells occurred to a varying extent, but were generally rather sparse. None of the animals in either study demonstrated a direct progression from hyperplasia to squamous cell carcinoma, i.e. there was no evidence of progression from the hyperplastic gingiva to squamous cell carcinomas. Reversibility of the gingival overgrowth was demonstrated in the group placed on recovery after 44 weeks of dosing (Table 3, High III).
The histopathological findings from the investigatory study are summarised in Table 3. Eight oral squamous cell carcinomas were detected in the high dose group rats fed the standard diet (High II), whereas no tumors were found in any of the groups maintained on the low fiber diet (Low and High I). Seven carcinomas arose from the molar gingiva and one arose from the incisor gingiva. The tumors were small and were detected only upon histopathological examination. The incidence of advanced periodontitis was again clearly increased in group High II, however to a somewhat lower extent when compared with the previous study. There was some increase in the incidence of periodontitis in the high dose group (High I) fed the special diet but to a lesser degree than that fed with the standard diet. Complete oro-nasal fistulae occurred in the standard diet high-dose group only (three animals) and the incidence of incomplete oro-nasal fistulae was also increased in this group (High II) when compared to the group receiving the same dose, but fed the special diet (High I).
Plasma Analysis
Table 4 compares some pharmacokinetic data (Cmax, Cmin, AUC 0–24 hours) for both studies. Systemic exposure to the parent compound was 2–3 times higher in rats dosed by oral gavage at 40/30 mg/kg/day when compared with those receiving the same dose by dietary admixture. When gavage doses were adjusted to 20 mg/kg/day higher exposures than those observed in the dietary study, though less marked, were still apparent. The plasma levels were not affected by the diet. In the first study there was no obvious difference in exposure levels between rats treated with 20 mg/kg/day and rats treated with 40 mg/kg/day.
Discussion
Mibefradil induced gingival overgrowth, the class related, systemic effect of calcium channel antagonists, which has been described in man, monkeys, dogs and rats (Nyska et al., 1990; Hassell and Hefti, 1991; Nishikawa et al., 1996; Hsiao et al., 1999). The pathogenesis of this lesion is still unclear (Hassell and Hefti, 1991), but, in general, it is reversible after cessation of treatment (Seymour et al., 1993). Malignant changes in the gingival tissues are extremely rare. In man there have been isolated case reports that have shown SCCs arising in drug-induced gingival overgrowth secondary to cyclosporin or phenytoin therapy (Varga et al., 1991; McLoughlin et al., 1995; Saito et al., 1999). In such cases the effect of both drugs on aspects of the immune response may be significant in the pathogenesis of the malignant change. Furthermore, other risk factors for oral malignancy (smoking and alcohol consumption) cannot be ruled out in these cases.
To date, there has been no case reported of malignant change occuring in gingival overgrowth secondary to medication with a calcium channel blocker. The increased incidence of oral squamous cell carcinomas in the standard 2-year rat carcinogenicity study was, therefore, an unexpected finding. Five assays of mibefradil for mutagenicity (Ames test with 8 bacterial strains, HPRT gene mutation assay with V79 Chinese hamster cells, chromosome aberration assay with human lymphocytes, in vitro unscheduled DNA synthesis assay with rat hepatocytes, in vivo micronucleus test in mouse bone marrow) and 4 assays (Ames, V79/HPRT, chromosome aberration assay and in vivo micronucleus test) with the main human metabolite were performed. Results were uniformly negative indicating that a genotoxic effect was highly improbable. In addition, the development of an epithelial tumor as a result of mibefradil-induced gingival overgrowth is most unlikely because the cell type principally reacting to the drug is the fibroblast, which makes up the bulk of the hyperplastic nodules (Seymour et al., 1996).
The above carcinogenicity studies demonstrated that the principal factor in the development of gingival squamous cell carcinomas was marked, or frequently severe destructive (bone-eroding) periodontitis resulting from the use of a standard diet containing fibers from barley, and that gingival overgrowth per se did not progress to neoplastic lesions. Squamous cell carcinomas were only detected in rats maintained on the standard diet, although (as the second study showed) systemic plasma exposure to the parent compound and gingival overgrowth were comparable in groups fed either on this diet or the low fiber diet. The standard diet induced a high incidence and severity of periodontal disease, including partial and complete oro-nasal fistulation, and a low background incidence of oral squamous cell carcinomas (1/345 control rats from the combined studies). Rats fed the low fiber diet had a relatively low incidence of severer forms of periodontitis, and no squamous cell carcinomas were found in these animals. The association between chronic inflammation and oral squamous cell carcinomas in rats has been previously reported (Madsen, 1989). However, our studies showed that, when the standard diet was fed, treatment with mibefradil enhanced both the severity of periodontitis and the incidence of these carcinomas. The incidence was increased, moreover, independently of the method of administration (dietary admixture or oral gavage). However, the incidence of both periodontal disease and squamous cell carcinomas was highest when the drug was given by feed admixture.
It is believed that the drug-induced gingival overgrowth provides an increased opportunity for entrapment and penetration of foreign bodies (i.e., food particles, hairs) because the swollen gingiva is more susceptible to mechanical injury. An additional factor is probably the intrinsic irritant property of the drug, evidence of local irritancy was seen after gavage administration in preclinical studies (unpublished data). This would explain why the highest incidence of periodontitis and squamous cell carcinomas was observed in rats treated by feed admixture, despite the fact that the systemic exposure to the parent compound and the degree of gingival overgrowth were consistently lower in these rats when compared to rats administered the same (or even lower) doses by oral gavage. The fact that many of the tumors were much larger in the feed admixture study could also be an indication that tumor development was enhanced by mibefradil’s intrinsic irritant properties. However, one has also to consider, that overall in the first study severe periodontitis including fistula formation was more pronounced both in drug-treated and control groups. We have no obvious explanations for this difference, but, as the studies were performed in separate laboratories, effects related to minor differences in the experimental conditions cannot be entirely excluded.
The extrapolation of these findings to man is inappropriate for several reasons. The pathogenesis of periodontal destruction in rats is multifactorial (Page and Schroeder, 1982). Gram-positive bacteria are essential to initiate the gingival inflammatory response. Subsequently dietary factor, hair impaction, housing conditions and the strain of rat used can all affect the rate of periodontal destruction. Periodontal disease in the rat is accompanied by gingival distortion and a subsequent splitting of the gingival tissues into junctional epithelium (which remains attached to the tooth surface), and a displaced stratum corneum. As periodontal destruction progresses following impaction of food and other debris between the teeth, a relatively large surface area is exposed to the oral environment increasing the opportunity for further entry of food and other debris. The increasing cycle of tissue destruction and repair affords an increased opportunity for genetic errors during mitosis and thus tumor induction. The development of periodontal disease in humans is related to bacterial plaque (Listgarten, 1986). Periodontal destruction is accompanied by loss of connective tissue to the root surface and the subsequent apical migration of the junctional epithelium. This results in periodontal pocket formation, which has minimal contact with the oral environment. Dietary factors, in particular the nature and consistency of food, are not involved in the pathogenesis of periodontal disease in humans (Rugg-Gunn, 1993).
Gingival overgrowth occured in rats and is a well-recognised unwanted effect of calcium channel antagonists and some other drugs in man. However, this condition is not a predisposing factor for the development of malignant changes in this tissue in man, nor does it render the patient more susceptible to periodontal destruction per se (Seymour et al., 1996). Primary squamous cell carcinomas of the gingival tissue in man is a rare malignant lesion of the oral cavity (McClatchey and Zarbo, 1996), despite the fact that periodontitis is a very common condition and calcium channel antagonists have been very widely used for many years. Most importantly, the role of periodontal disease per se in the development of squamous cell carcinomas of the gingiva in man has not been established.
It is concluded that there is no evidence that systemic exposure to mibefradil alone induced the gingival squamous cell carcinomas found in the 2-year rat studies. The increase in the numbers of such carcinomas only arose secondarily to diet associated with severe periodontal disease favoured by class-related gingival overgrowth and, in the dietary admixture study, a possible local irritating effect of the compound. The studies described suggest that care should be taken in selecting the diet for carcinogenicity studies, particularly if previous toxicity studies have identified the oral cavity as a target organ. In addition, the mode of administration (dietary admixture versus oral gavage) of a drug with known irritant properties should be considered carefully for such studies.
