Abstract
Oral cancer is the seventh most common malignancy worldwide, and lifestyle factors participate in its development. Rodent studies can help identify substances that contribute to its development and provide information on the early stages of carcinogenicity. The National Toxicology Program (NTP) has conducted more than 500 short-term and 2-year toxicology and carcinogenicity studies in rodents, and some of the tested compounds resulted in oral cancer. Our goal was to review the NTP carcinogenic studies to describe those chemicals that have oral carcinogenic outcome in rodents. For this project, we reviewed the results from all NTP carcinogenicity studies and a board-certified veterinary pathologist reviewed the slides from all neoplasms in the oral cavity that were considered treatment related. We have identified 26 chemicals with an adverse effect in the oral cavity. Fourteen chemicals demonstrated clear evidence of carcinogenicity in the oral cavity. We provide information on the carcinogenic findings in rodents together with a detailed description of the morphologic aspects of the oral cancers and speculate that the carcinogenic effects can be induced by different pathological modes of action. The findings reviewed here provide indicators for potential oral carcinogenesis processes in rodent models, which can be further investigated in future mechanistic studies.
Introduction
Oral cancer is the seventh most common cancer type in humans, and it constitutes about 50% of all head and neck cancers. 1 Oral cancer rates are twice as common in males than in females. 2 Ninety percent of the cancers in the oral cavity are squamous cell carcinoma (SCC), and its incidence is on the rise in recent years. 3,4 The morbidity and mortality from oral cancers are high, and treatment options are limited, resulting in only 50% 5-year survival rate for oral SCC. 5 Many factors contribute to the development of oral cancer, but exposure to lifestyle agents is the major contributor. The most common life style agents contributing to oral SCC development include alcohol and tobacco use, betel quid chewing, and the presence of the human papillomavirus. 6 –8
The process of oral carcinogenesis is multistep, starting with hyperplasia, followed by dysplasia, carcinoma in situ, and finally, invasive carcinoma.
9
Comprehensive morphological description of the pathological findings of this multistep process into invasive cancer can provide valuable information for the development of effective treatments and preventive methods. Previous studies showed that long-term animal bioassays evaluating carcinogens in rodents, mainly rats and mice, had been correlated with human cancer as reliable indicators.
Methods Used in the NTP Studies
The NTP has used short-term and 2-year rodent bioassays to assess the overall toxicity and carcinogenicity of around 550 chemicals. We reviewed the NTP studies database on rodents in order to identify chemicals that can induce carcinomas of the oral mucosa (Supplemental Table 1). In the NTP studies, rodent models were exposed to chemicals through various routes including gavage, drinking water, or feed. The standard bioassays mainly included male and female B6C3F1 mice and F344/N rats. Harlan Sprague Dawley rats and Swiss Webster mice were occasionally used. Complete necropsies were performed on all the rodent models using standardized methodology. 12 The oral cavity and the tongue were examined grossly. The oral mucosa, while not a protocol required tissue for histological examination, was present in sections collected for the nasal cavity. All gross lesions were collected and examined histologically. Most of the tumors of the oral cavity, including tongue, represent gross lesions identified at necropsy. The tissues were fixed in formalin, and in the case of the nasal sections with oral mucosa and teeth, decalcified in nitric acid decal solution 5% and then cleaned and trimmed before being sectioned into 5-mm-thick sections and stained with hematoxylin and eosin. The maxilla was trimmed into 3 sections: posterior to the upper incisors (section I), midway between the incisors and first molar at the anterior surface of the incisive papilla (section II), and at the middle of the first molar (section III; Figure 1). The slides were examined, by a board-certified veterinary pathologist, and the results underwent rigorous review by the standard NTP pathology review process, culminating in a Pathology Working Group, which evaluated examples of treatment-related neoplasms. 13 The slides produced from the NTP studies were reviewed for the current project; no new slides were prepared. The Kaplan-Meier estimator was used by the NTP for the estimation of probability of survival. 14 Differences in the incidences of oral cavity neoplasms between groups were evaluated statistically by the poly-3 test. 15,16
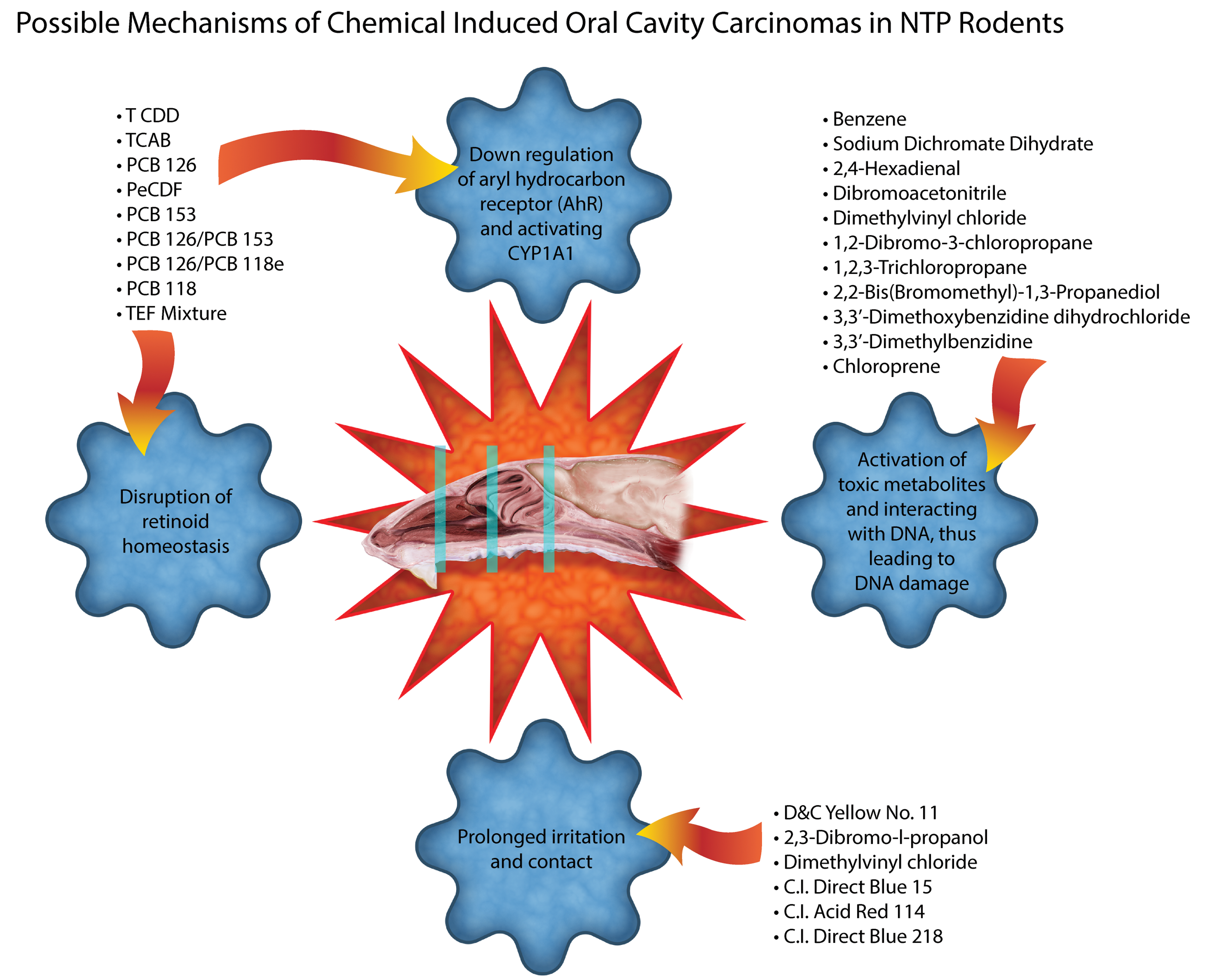
Possible mechanisms of chemical-induced oral cavity carcinoma in National Toxicology Program (NTP) 2-year rodent studies. The trimming locations of the basal turbinates in rodents are also indicated. See more details in https://www.niehs.nih.gov/research/resources/visual-guides/guides/l1-post/index.cfm.
The NTP’s level of evidence criteria for carcinogenic activity were positive or negative in studies performed in 1981 or prior; positive, negative, or equivocal in studies performed in 1982 and 1983; and clear, some, equivocal, no or inadequate in studies performed in 1984 and later (NTP Annual Report on Carcinogens, 1984).
Results
Summary of the Histopathological Characteristics of the Oral Findings in Rodents
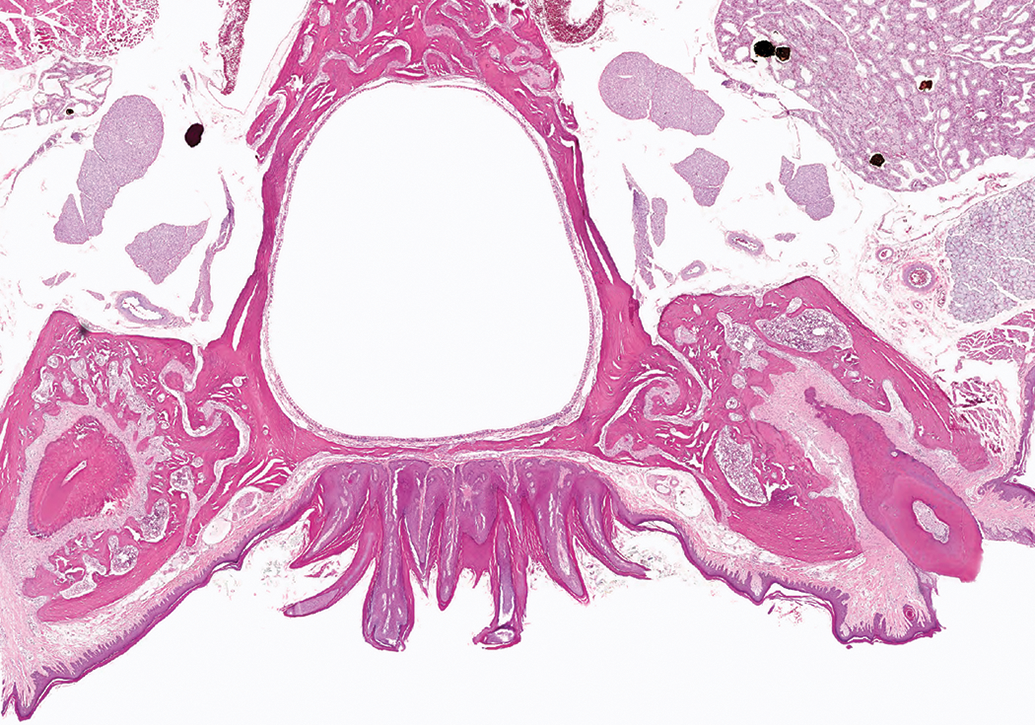
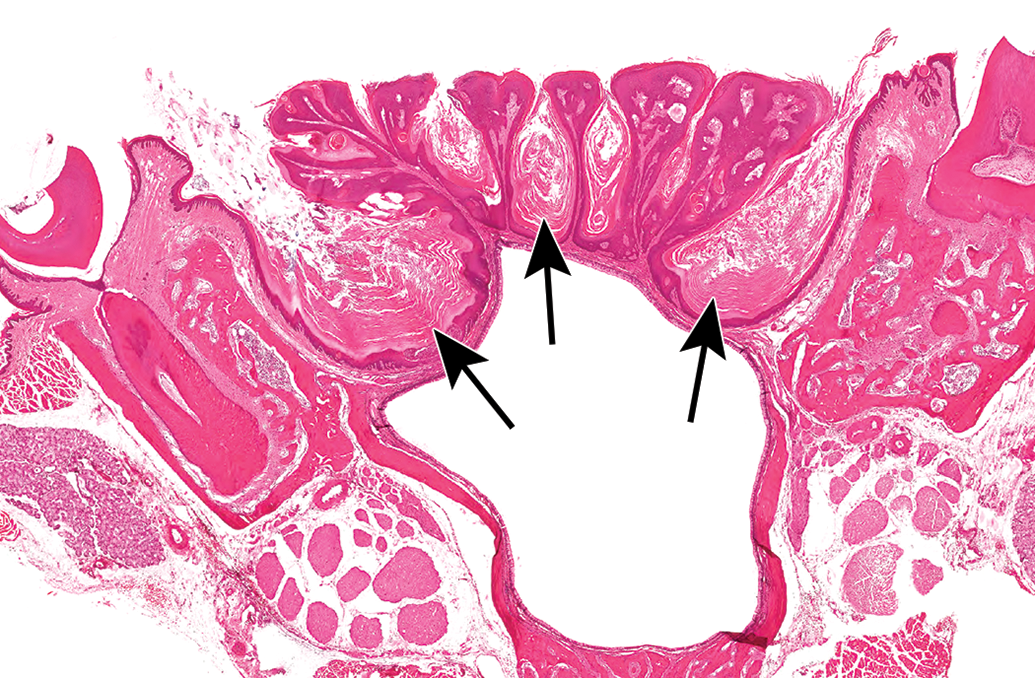
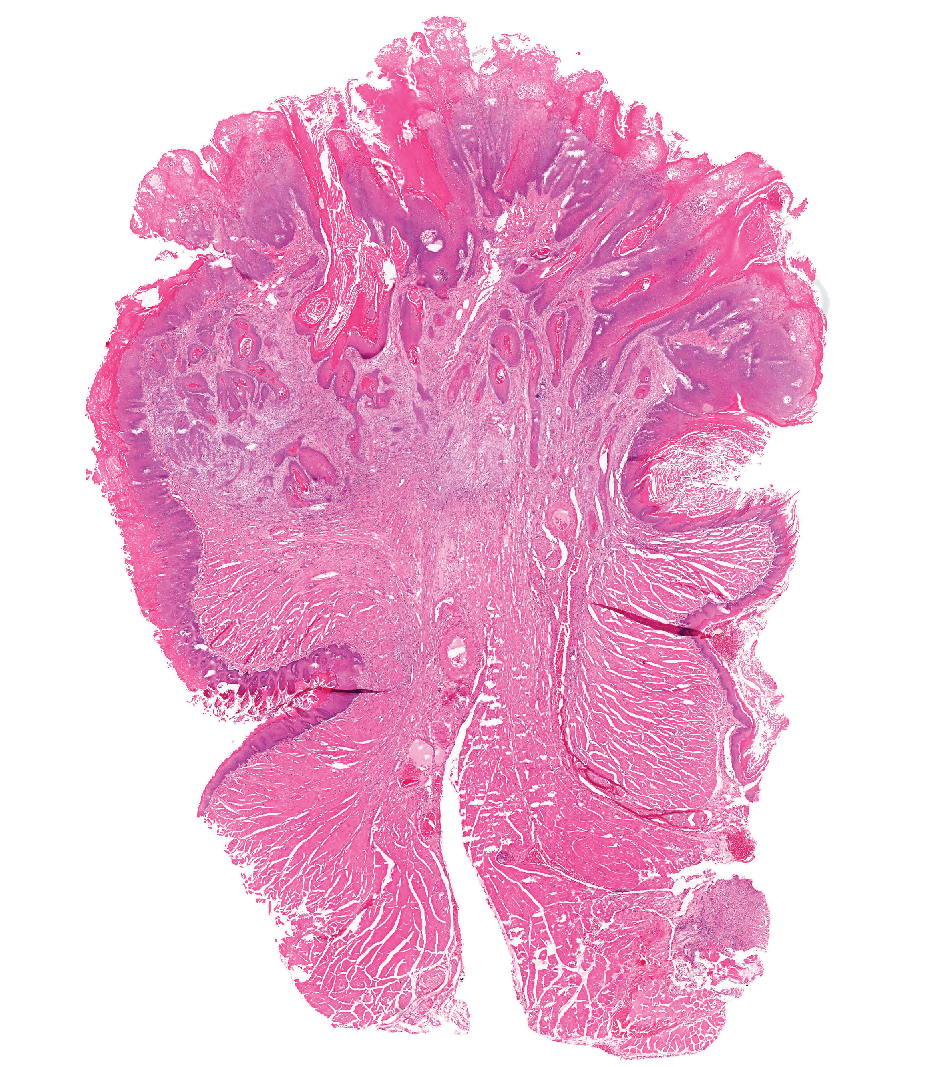
Squamous cell papillomas (SCPs) were frequently found on the tongue and palate. Many of the SCPs observed in the oral cavity were pedunculated exophytic growths with long finger-like projections on narrow fibrovascular stalks or mushroom-shaped polypoid growths, while others had a broad base, almost the width of the tumor (Figures 2 and 3). A few were characterized by a focal thickening in the epithelium; these were easier to confuse with hyperplasia, but neoplasms were more florid and had more mitoses than one would expect with hyperplasia, although not as numerous as in the SCC. Sometimes the benign tumors formed large cavitations filled with keratin (Figure 4), but they maintained an intact basement membrane and did not invade into the underlying tissue (Figure 5). Squamous cell papillomas were composed of well-differentiated squamous epithelial cells, which displayed the normal orderly progression of stratified squamous epithelium, with rounder, more basophilic basal cells to flattened, brightly eosinophilic cells at the outer layers. Squamous cell papillomas were typically keratinized, had a low mitotic rate, and did not display the cellular pleomorphism and anisokaryosis that were observed in SCC (Table 1).
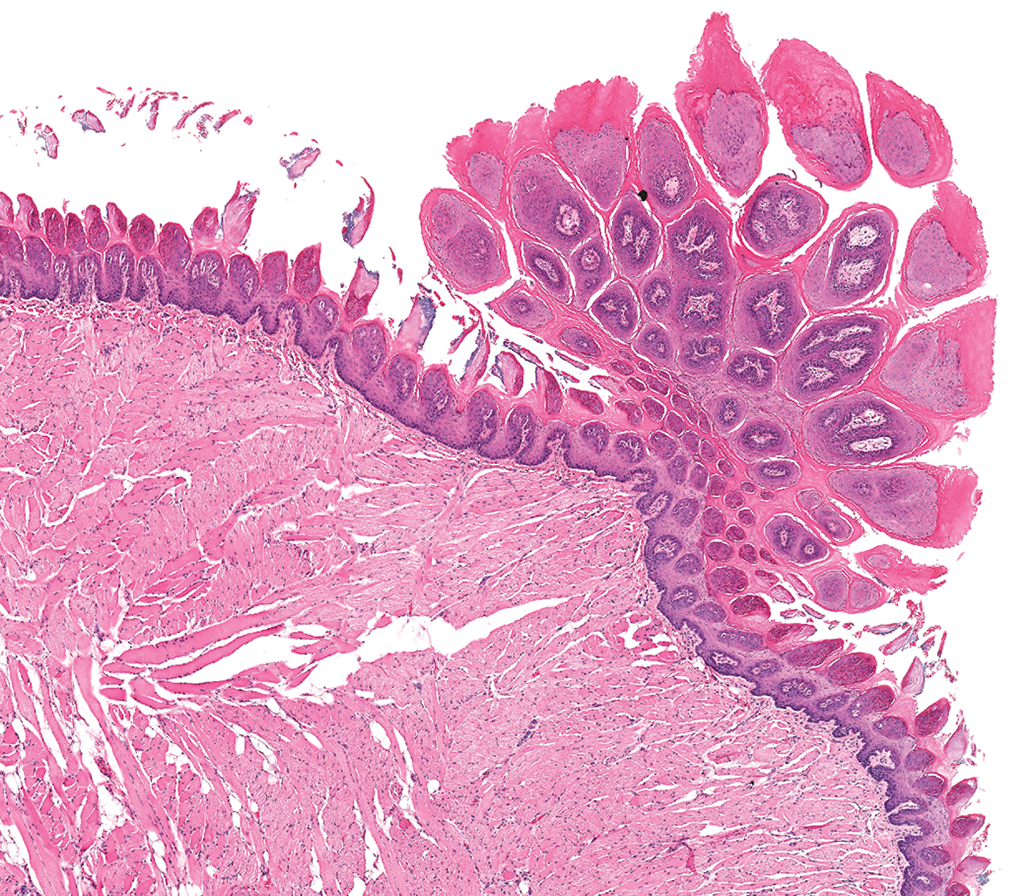
A squamous cell papilloma on the tongue of a female rat is composed of finger-like projections that are well keratinized and is attached to the underlying epithelium by a stalk that is much narrower than the tumor.
A squamous cell papilloma on the hard palate of a male rat is composed of long finger-like projections similar to those in figure 2, but the tumor is attached to the underlying epithelium by a very broad base.
Large, cyst-like areas filled with keratin (arrows) developed between the exophytic projections of a squamous cell papilloma on the palate of a female rat.
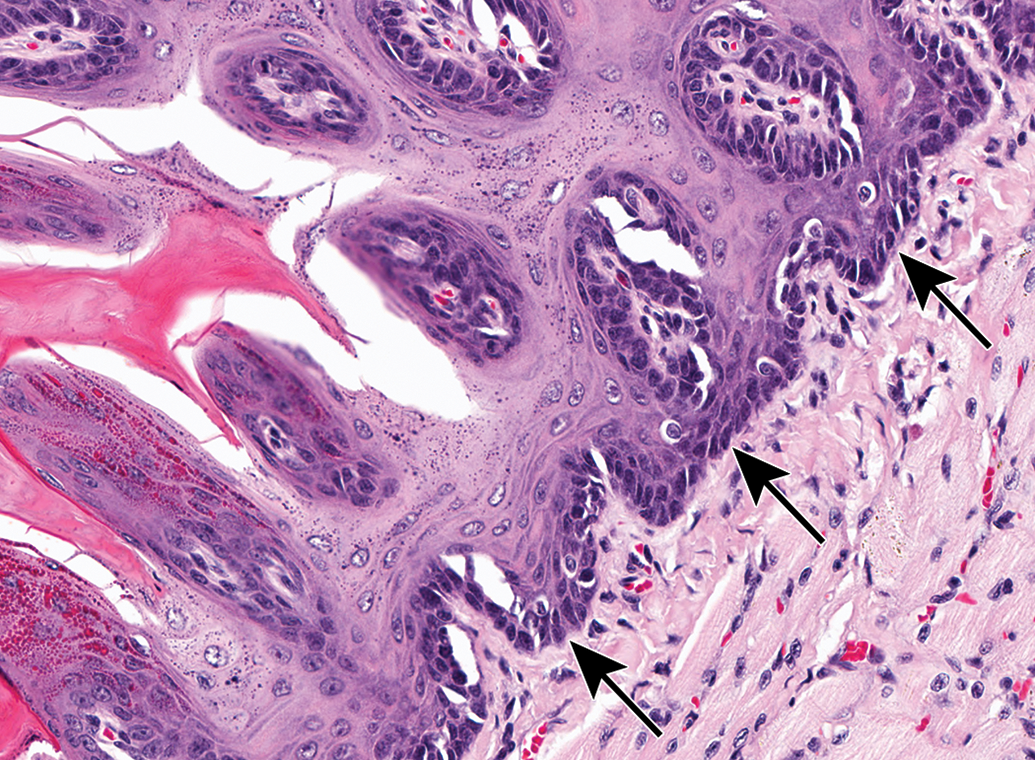
A high magnification of the base of a squamous cell papilloma from a female rat reveals an intact basement membrane with no invasion into the underlying tissue (arrow).
Features of Proliferative Squamous Lesions of the Oral Cavity.
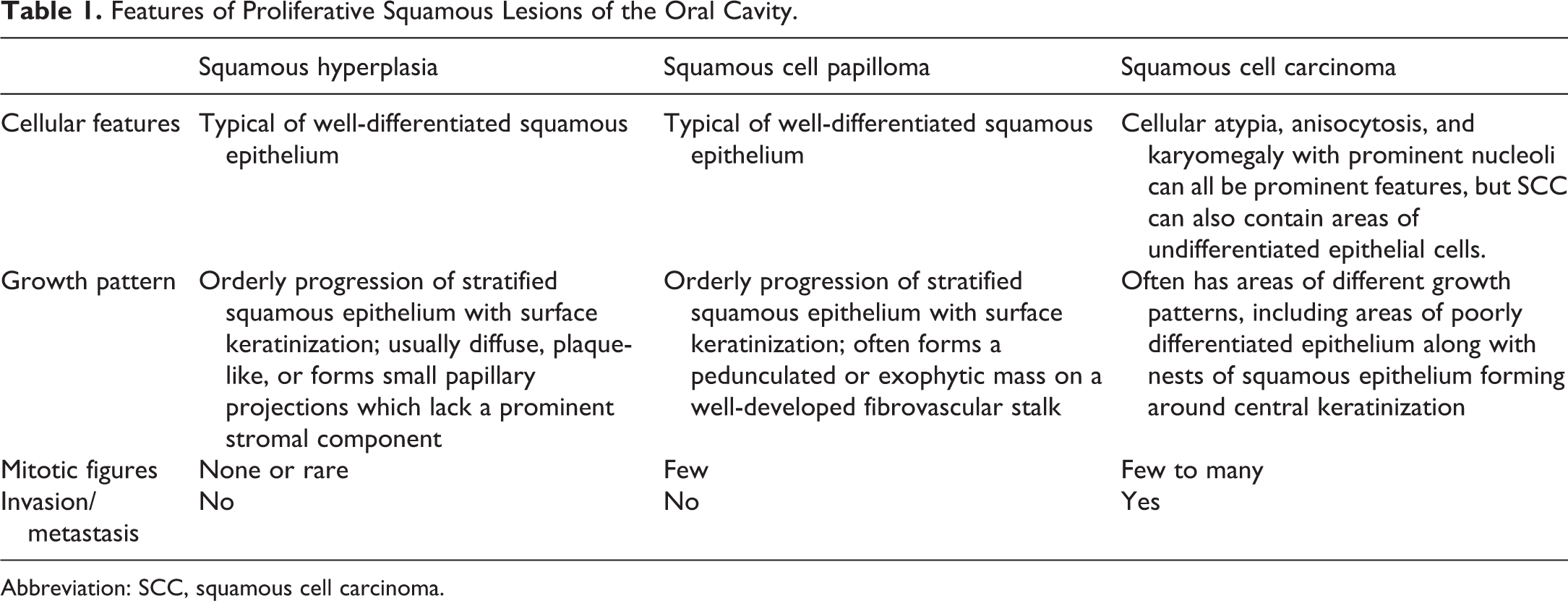
Abbreviation: SCC, squamous cell carcinoma.
Squamous cell carcinomas were commonly observed arising from the gingival mucosa, on the palate, and in the tongue, although they could occur anywhere in the oral cavity, and even in the esophagus. Squamous cell carcinomas originating in the gingival mucosa often appeared to arise from, or at least involve, the gingival mucosa adjacent to the tooth in nasal section III (Figure 6). In some animals, a large cavity, lined by thickened squamous epithelium and filled with keratin, would form adjacent to the tooth. Nests of epithelium appeared to drop-off and invade the surrounding tissue, often inciting a scirrhous reaction and destroying bone. Squamous cell carcinomas in this location tended to grow up into the nasal cavity and compress and invade into the nasopharyngeal duct (Figures 7 and 8). The surface of SCCs was often ulcerated and had associated inflammation. Sometimes pockets of suppurative inflammation (abscesses) formed in the deeper tissue, especially at the root of a tooth. Squamous cell carcinomas of the palate invaded into the nasopharyngeal duct and nasal cavity, like those that originated in the gingival mucosa near the teeth. Squamous cell carcinomas in the tongue were often large lesions (Figure 9) with infoldings and invaginations into the underlying muscle layers. In some tumors, there were large, cyst-like central areas filled with keratin and lined by a thick irregular layer of squamous cells (Figure 10). Solid cords and nests of squamous cells invading through the muscle characterized other tumors, or other areas of the same tumor. These cords and nests might contain central accumulations of keratin (Figure 11). Some tumors had thin, anastomosing cords of squamous cells which went deep into the muscle layers (Figure 12).
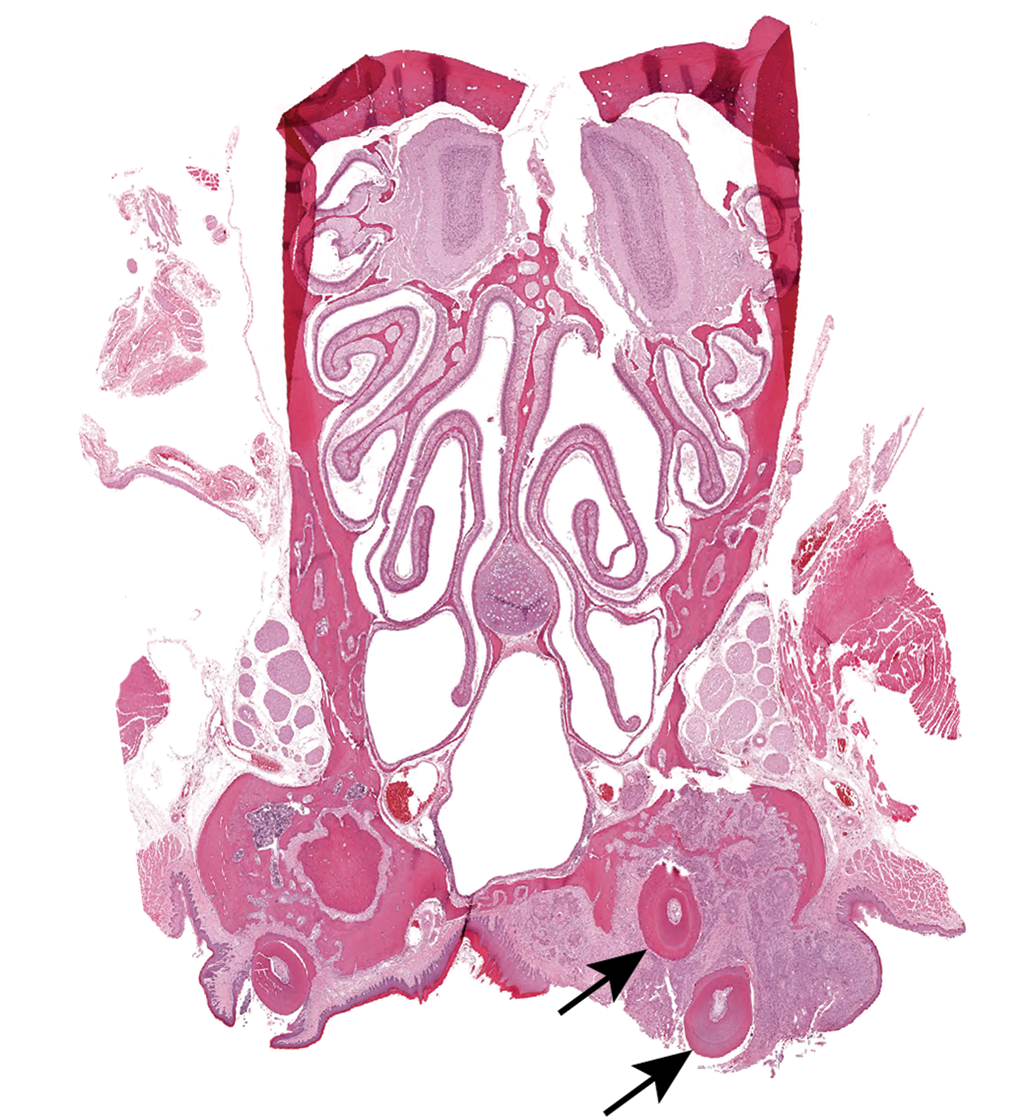
A squamous cell carcinoma originating in the gingival mucosa of a male rat reveals the roots of the molar tooth in the middle of the tumor (arrow).
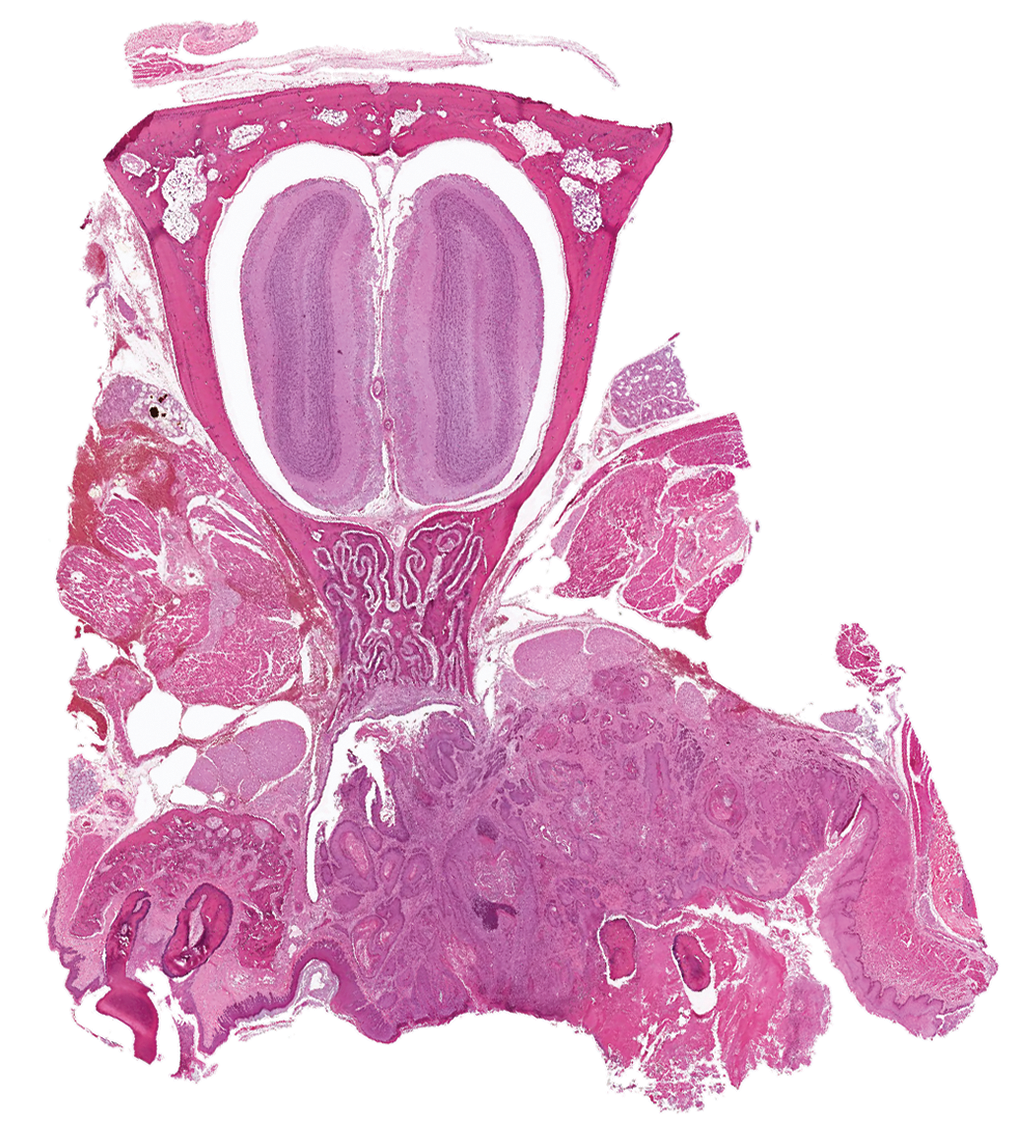
This squamous cell carcinoma arising in the oral cavity of a female rat has invaded through the bone and into the nasal cavity.
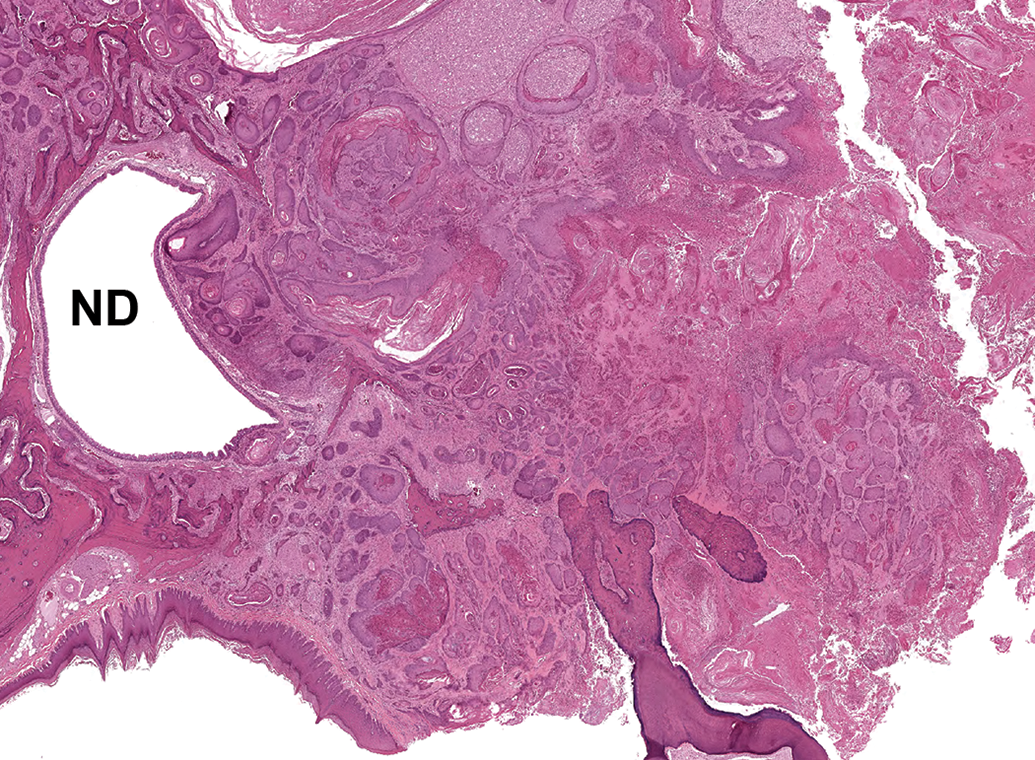
A squamous cell carcinoma arising in the oral cavity of a female rat is invading through bone and other tissue into the nasal cavity and compressing the nasopharyngeal duct. ND indicates nasopharyngeal duct.
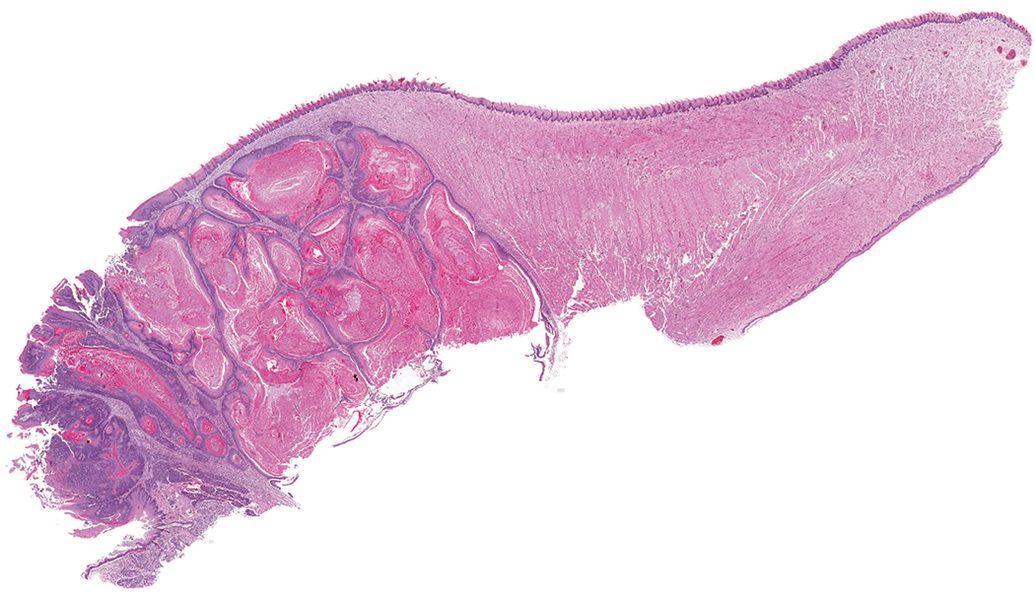
A large squamous cell tumor in the tongue of a male rat.
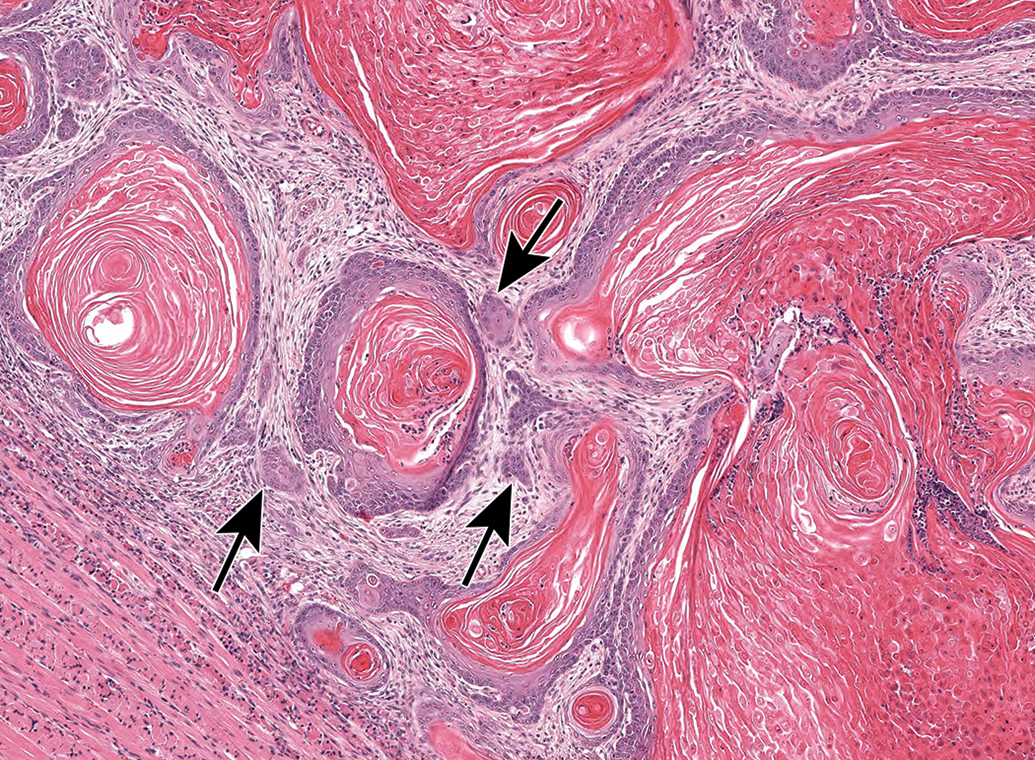
Large, dilated areas in a squamous cell carcinoma are filled with keratin and cell debris. These cyst-like areas are lined by layers of squamous epithelium that give rise to nests of cells invading into the surrounding tissue (arrows).
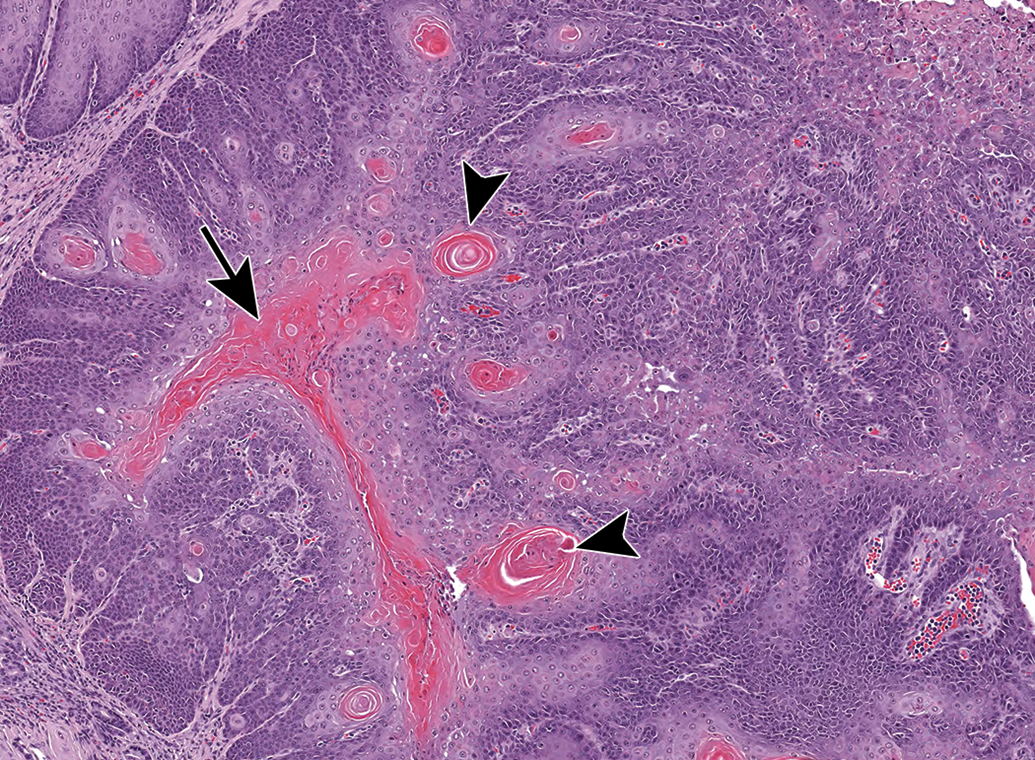
Thick fingers and sheets of neoplastic epithelium within a squamous cell carcinoma from the tongue of a male rat shows a central accumulation of keratin (arrow), and keratin whorls (arrowheads).
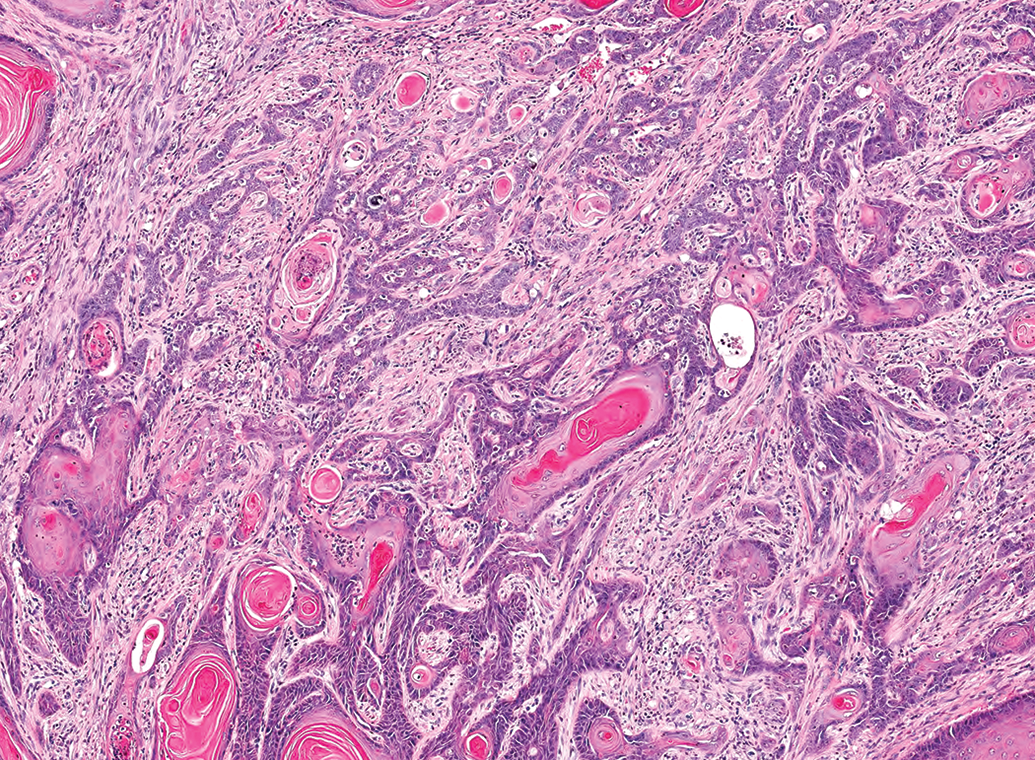
Some tumors contained long, thin anastomosing cords of squamous cells that invaded deep into the underlying musculature.
Squamous cell carcinomas often incited a scirrhous response, with nests or individual clusters of neoplastic squamous cells embedded in a background of fibroplasia (Figure 13). The surface of SCCs often had papillary projections (Figures 14 and 15) and areas of ulceration. Surface ulceration was associated with a mixed inflammatory cell reaction (Figure 16) that was also visible within deeper tissue. Large SCCs involved almost the entire section of tongue (Figure 17). Some of the smaller SCCs appeared to have started out as hyperplasia or squamous papilloma and become invasive (Figure 19). Cells in SCCs ranged from medium-sized closely packed, angular to oval cells with scant to moderate amounts of basophilic cytoplasm to very large round cells with copious amounts of lightly basophilic to brightly eosinophilic cytoplasm (Figure 19). These larger, eosinophilic cells were often associated with keratin formation, and formed whorls and nests of cells with a center filled with concentric layers of keratin, or small keratin pearls. Karyomegaly was common, and nucleoli were large and prominent. Mitotic figures were frequently seen, with many ×20 fields having more than 5 mitotic figures.
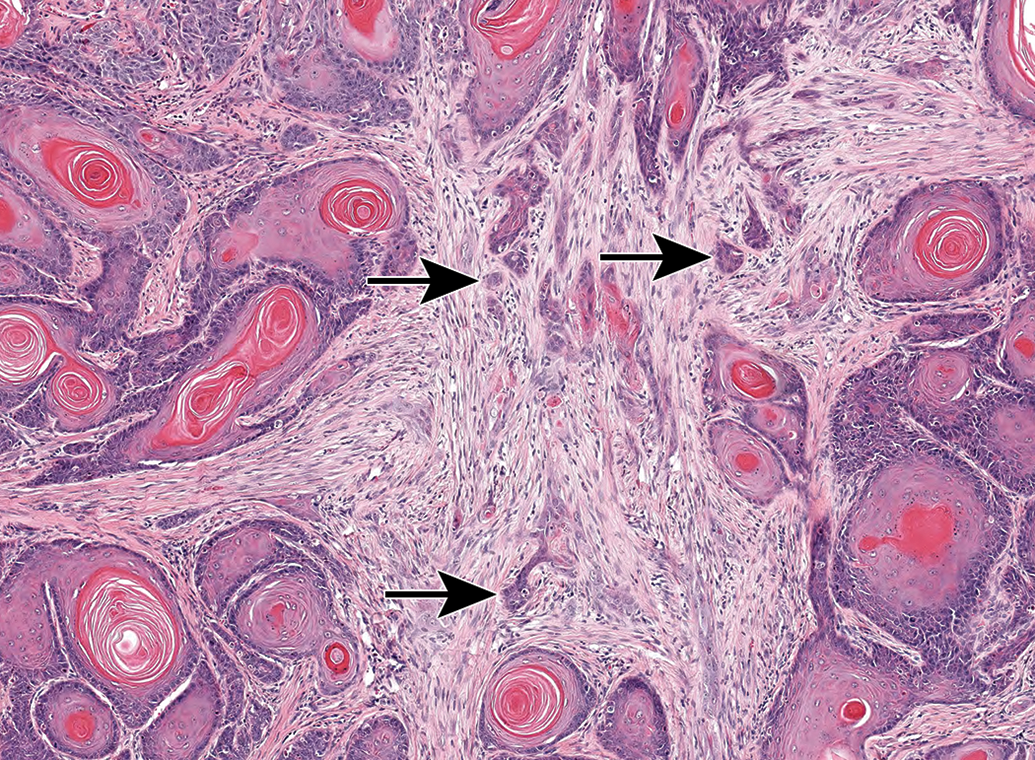
Squamous cell carcinomas frequently elicited a scirrhous reaction, with nests of malignant epithelial cells (arrows) in a fibroproliferative background.
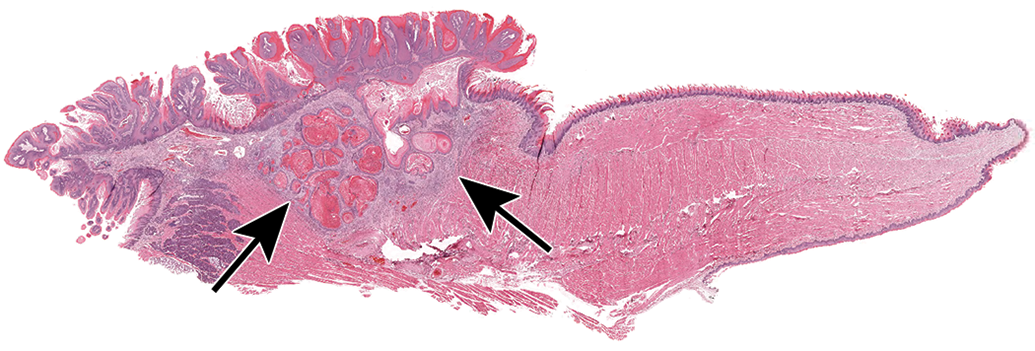
A squamous cell carcinoma in the tongue of a male rat has exophytic papillary projections on the surface, similar to what would be seen in a papilloma, but the tumor invades deep into the underlying muscle (arrows).
A squamous cell carcinoma in the tongue of a male rat has exophytic projections on the surface and invasion into the underlying muscle.
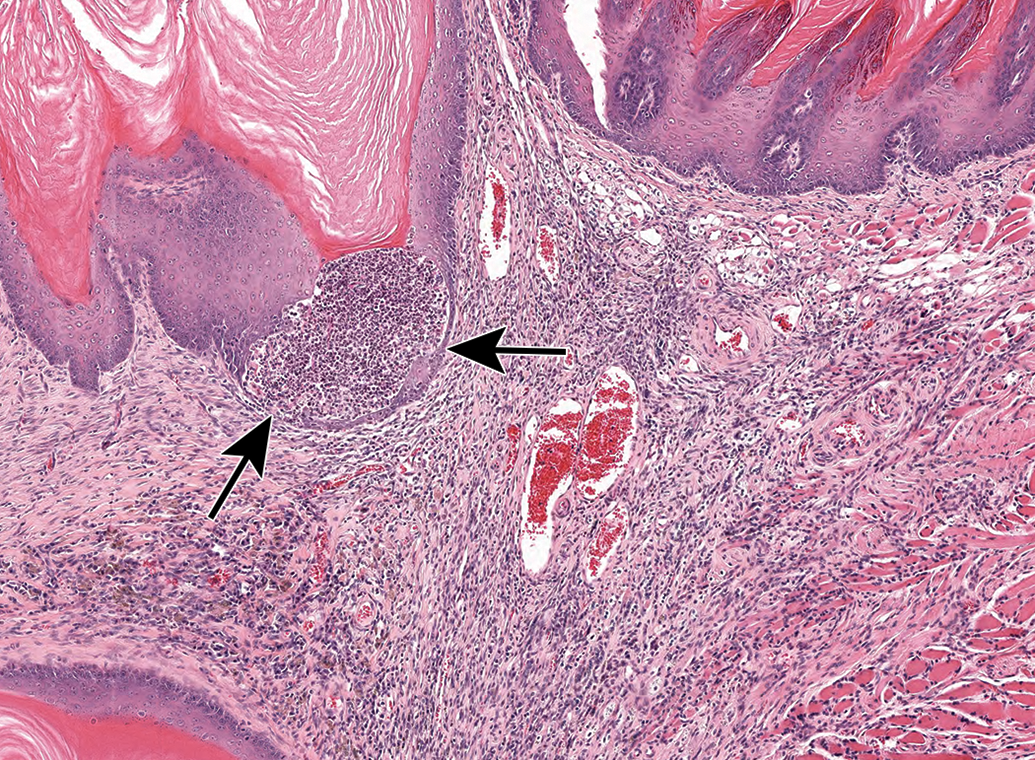
Pocket of inflammation within the epithelium (arrows) of a squamous cell carcinoma on the tongue of a rat.
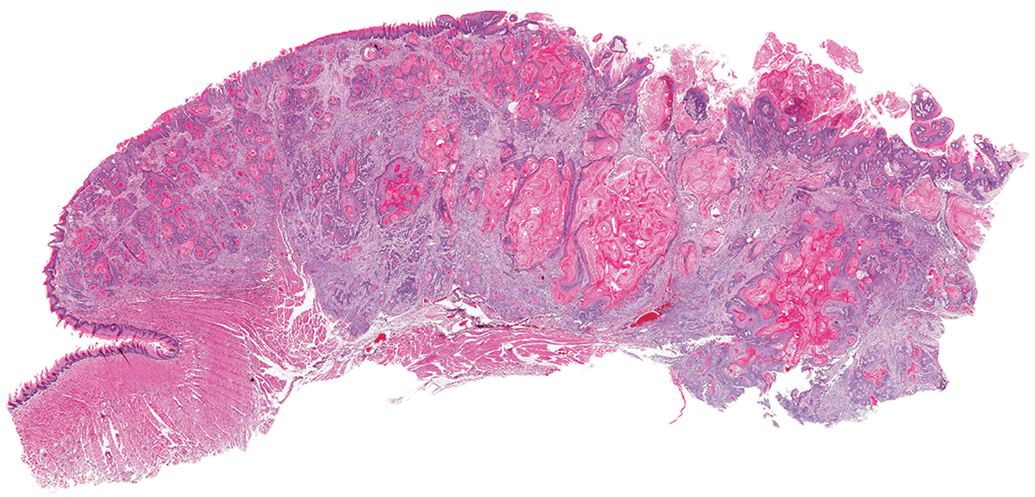
This large squamous cell carcinoma involved almost the entirety of the tongue of the male rat in which it was present.
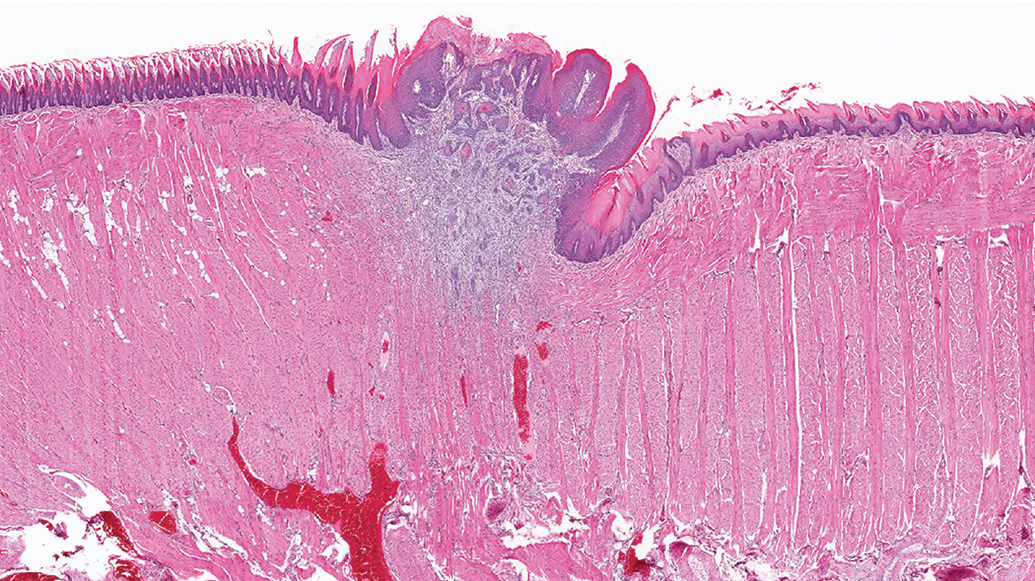
A small squamous cell carcinoma from the tongue of a male rat that may have started out as a focal hyperplasia or as a papilloma and became invasive.
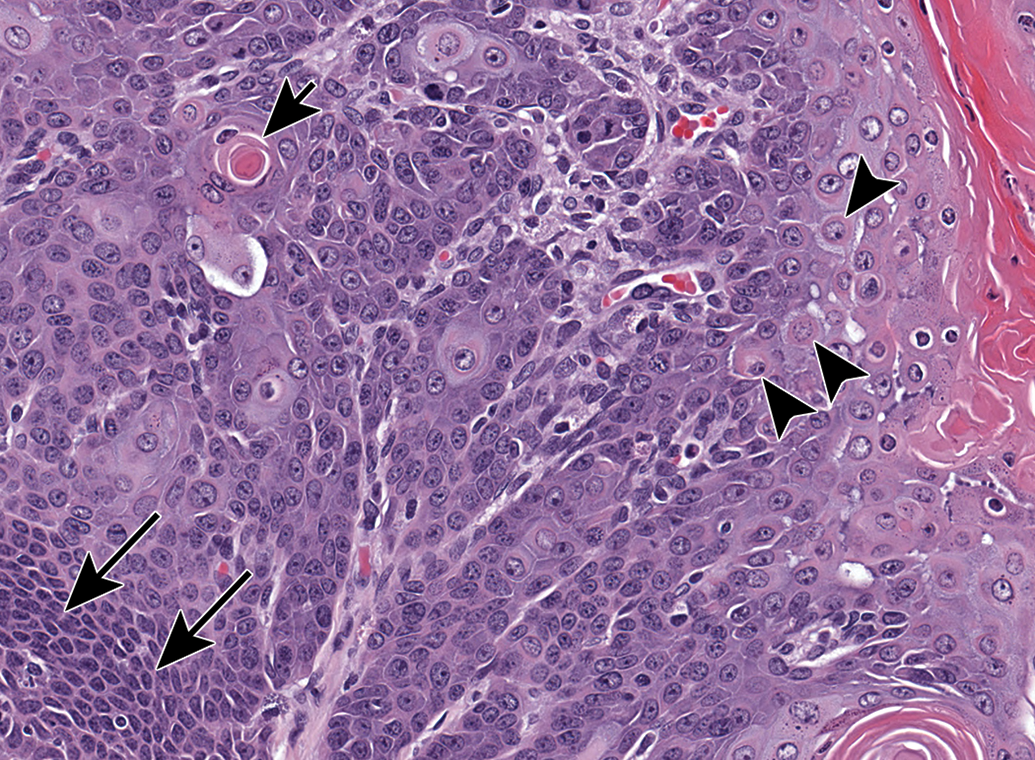
The cells in this squamous cell carcinoma in the tongue of a male rat range from closely packed angular to oval basophilic cells (long arrows) to large round cells with eosinophilic cytoplasm (arrowheads). A keratin pearl is present within a cell (short arrow).
No other types of oral cavity tumors were found to be caused by the tested compounds or as spontaneous lesions.
Chemicals That Induce Oral Cancer in Rodents in NTP Studies
In the current section, we review the pathological findings related to oral cancer in the specific compounds that were examined. Additional background information on the different compounds can be found in the relevant technical reports and in the accompanying Supplementary Material.
Benzene (NTP TR 289)
In a 2-year NTP study in F344/N rats, significantly increased incidence of SCPs and SCCs of the oral cavity (including palate, lip, and tongue) in the low, mid, and high doses (P < .05, relative to vehicle controls) were observed in both males and females. This study concluded that chronic, high-dose administration of benzene to rats produces oral cavity tumors. 17 The cause was attributed to a systemic effect of benzene-reactive intermediates, which are probably required for benzene carcinogenicity. 17,18 In vitro studies suggest that activation of toxic metabolites of converted benzene interact with DNA and form covalent adducts that can result in DNA damage. However, no aromatic adducts were detected unequivocally with DNA samples from most tissues examined in vivo, although the oral cavity was not tested. 18 Another less likely explanation for oral exposure is by licking of benzene-reactive intermediates excreted on the rats’ skin. 17
Sodium Dichromate Dihydrate (NTP TR 546)
The 2-year NTP study exposed 50 female and 50 male rats to drinking water containing 4 different concentrations of sodium dichromate dihydrate—0, 14.3, 57.3, 172, or 516 mg/L (equivalent to 0, 5, 20, 60, or 180 mg/L chromium). 19
Sodium dichromate dihydrate caused dose-related oral cavity (oral mucosa or tongue) squamous neoplasms, including SCC and SCP, in exposed male and female F344/N rats at concentrations above 172 mg/L. Squamous cell carcinoma in the oral mucosa was significantly higher in male and female rats exposed to 516 mg/L compared with control rats. Squamous cell carcinoma and SCP in the oral mucosa or tongue (combined) were significantly higher in male and female rats exposed to 516 mg/L compared with control rats. 19
It is suggested that oxidative stress resulting from high concentrations of Cr (VI) is a possible event in the mode of action for oral mucosa neoplasm. Cr (VI) enters the cells and undergoes metabolic reduction to Cr (III), therefore inducing oxidative stress and leading to toxicity. 20 Reduced Cr reacts with protein and DNA and forms adducts with DNA, which can result in mutations. 21,22
D&C Yellow No. 11 (NTP TR 463)
In order to determine the toxic effects of oral exposure to D&C Yellow No. 11, 14-day and 13-week toxicity studies were conducted in F344/N rats and B6C3F1 mice in 1991. A 2-year feed study was conducted on groups of 60 male and 60 female F344/N rats that were given 0, 500, 1700, or 5000 ppm D&C Yellow No. 11.
The results of the study indicated an increased incidence of squamous cell neoplasms of the oral cavity in male F344/N rats and in female F344/N rats at concentrations above 500 ppm, including papillomas of the tongue and oral cavity. Therefore, it was concluded that there is an association with neoplastic proliferations of the epithelium in the oral cavity. 23 It is believed that these changes are the result of prolonged irritation and contact with the oral mucosa, which lead to epithelial neoplastic proliferations. 24
2,3-Dibromo-l-propanol (NTP TR 400)
To understand the toxic effects of the chemical, toxicology and carcinogenesis NTP studies were conducted. A short-term study was first conducted by topically applying 2,3-dibromo-l-propanol (approximately 98% pure) in ethanol to the subscapular area of the skin of F344/N rats and B6C3Fl mice of both sexes 5 days per week for 16 days and 13 weeks. In the long-term study, F344/N rats received dermal applications of 0, 188, or 375 mg/kg and B6C3Fl mice received dermal applications of 0, 88, or 177 mg/kg DBP 5 days per week for 36 to 39 weeks (males) or 39 to 42 weeks (females).
In male and female F344/N rats, increased incidences of SCPs and SCCs of the oral mucosa were observed, occurring in nearly all low- and high-dose males and in almost all high-dose and most low-dose females mostly on the dorsum of tongue and pharynx and also in the mucosa of the lip palate or gingiva (statistically significant by Fisher exact test: P ≤ .001). No evidence for a carcinogenic activity was seen in mice. 25
It is thought that oral exposure through skin absorption and grooming behavior play a role in increasing the incidence of carcinomas of the tongue and gingiva. 25,26
2,4-Hexadienal (NTP TR 509)
The NTP conducted a study in male and female F344/N rats and B6C3F1 mice in which they received 2,4-hexadienal (89% trans, trans; 11% cis, trans isomer) in corn oil by gavage at doses of 0, 22.5, 45, or 90 mg/kg (rats) and 0, 30, 60, or 120 mg/kg (mice), for 16 days, 14 weeks, or 2 years. In the 2-year gavage study, 2 male B6C3F1 mice had SCC of the tongue at a dose of 120 mg/kg. The NTP concluded that the occurrence of SCC of the oral cavity (tongue) in male B6C3F1 mice may have been related to the administration of 2,4-hexadienal. Single incidences of SCP or SCCs of the tongue were observed in most treated groups of males and female rats. Although SCP of the oral mucosa occurred in one male and one female vehicle controls, no SCC of the tongue occurred in male or female vehicle controls. There was no clear dose-related association with 2,4-hexadienal administration of SCC or SCP combined in the oral cavity. It is noteworthy to mention that there were increased incidences of gingival squamous hyperplasia (GSH), SCP, and SCC in the forestomach of rats and mice. The forestomach is lined by squamous epithelium like the oral cavity and esophagus. However, unlike in humans, rodents have a forestomach in which the food is temporarily stored before digestion. Therefore, the forestomach is exposed to the ingested material much longer than the oral cavity or esophagus. 27
A few possible pathways are believed to be responsible for induction of neoplasm after 2,4-hexadienal exposure, including reactive oxygen species formation leading to DNA damage, or direct interaction causing gene mutations, or by stimulating an inflammatory process, which results in mutagenesis and carcinogenesis. 28,29
Dibromoacetonitrile (NTP TR 544)
In 2010, the NTP conducted a study in male and female F344/N rats and B6C3F1 mice, exposing them to dibromoacetonitrile (98.6% pure) in drinking water for 2 weeks, 3 months, or 2 years. 30,31
Male rats that were exposed to 200 mg/L had a significantly increased incidence of SCCs and SCPs of the oral cavity (oral mucosa or tongue) compared to control male rats. In female rats, SCPs and squamous epithelial hyperplasia were observed, but the incidences were not significantly different from controls. No oral neoplasms were detected in mice. This could be due to species difference in the reaction of tissues with the chemical. 31
Although mechanism of the toxic effects of dibromoacetonitrile is unknown, it is postulated that genetic toxicity and oxidative stress could lead to carcinogenesis. 32
Dimethylvinyl Chloride (NTP TR 316)
Due to its structural similarity to vinyl chloride, it was selected as a potential carcinogen. Toxicology and carcinogenesis studies were conducted by administrating dimethylvinyl chloride (96%-98% pure) in corn oil by gavage to groups of 50 male and 50 female F344/N rats and B6C3F1 mice at doses of 0, 63, 125, 250, 500, or 750 mg/kg body weight, 5 days a week, for 13 weeks and at doses of 0, 100, or 200 mg/kg per day, 5 days a week, for 2 years. 33
Exposure to dimethylvinyl chloride for 2 years was associated with SCP in 4% of male rats who received 200 mg/kg. Squamous cell carcinoma occurred in 10% of male rats at 100 mg/kg and in 4% at 200 mg/kg. Squamous cell carcinoma occurred in 4% of the 100 mg/kg females and 2% of the 200 mg/kg females, while SCP occurred in 8% of the 200 mg/kg females. No squamous tumors were recorded in the oral cavity of control male or female rats. No neoplastic lesions were detected in the oral cavity of mice. 33
Dimethylvinyl chloride is metabolized into an active toxin, chloroacetaldehyde, and then remains localized in the exposed tissue and results in direct carcinogenic activity by interacting with purine and pyrimidine residues of DNA. 34,35
1,2-Dibromo-3-Chloropropane (NTP TR 206)
Since workers were exposed to airborne dibromochloropropane (DBCP), the NTP conducted a study by exposing inhaled DBCP, with trace amounts of epichlorohydrin and 1,2-dibromoethane, to groups of 50 F344/N rats and B6C3F1 mice of both sexes at concentrations of 0.6 or 3.0 ppm DBCP for 6 hours a day, 5 days per week, for 76 to 103 weeks. Significantly higher incidences of dose-related SCP of the tongue in rats of both sexes were observed. Combined incidences of SCPs and SCCs of the pharynx were observed in female rats when compared with controls. 36 The statistical analysis of this study provided strong evidence for carcinogenicity of inhaled DBCP.
It is believed that several metabolic pathways produce reactive intermediates including cytochrome P-450 oxidation and glutathione conjugation and thus induce cellular DNA damage. 37
1,2,3-Trichloropropane (NTP TR 384)
In the 2-year study, groups of 60 rats of both sexes received 0, 3, 10, or 30 mg and groups of 60 mice of both sexes received 0, 6, 20, or 60 mg/kg of 1,2,3-trichloropropane/kg body weight in corn oil by gavage 5 days per week for up to 104 weeks. In 10 and 30 mg/kg rats, the incidences of SCPs and carcinomas of the oral (pharynx and tongue) mucosa were significantly increased. The incidence of SCC of the oral mucosa was significantly increased only in 60 mg/kg female mice. 38
A few metabolic pathways have been proposed, including oxidation by mixed function oxidases in the liver, glutathione transferase catalyzed formation of glutathione conjugates in the liver and episulfonium ion formation, or by undergoing conversion of halogenated compounds via cytochrome P-450 microsomal metabolism leading to DNA adducts. It can also cause cell proliferation in several tissues through gavage or inhalation exposure. 39,40
2,2-Bis(Bromomethyl)-1,3-Propanediol (NTP TR 452)
The NTP study included groups of 60 males and females exposed to 2,2-bis(bromomethyl)-1,3-propanediol (78.6% pure) in feed for 13 weeks or 2 years. B6C3F1 mice received 0, 312, 625, or 1250 ppm 2,2-bis(bromomethyl)-1,3-propanediol and Fisher 344/N rats were exposed to 2,2-bis(bromomethyl)-1,3-propanediol at concentrations of 0, 2500, 5000, or 10,000 ppm. There were significantly higher incidences of SCP of the oral cavity of male rats. In female rats, there was a higher incidence of squamous cell neoplasms in esophageal mucosa, some at the posterior aspect of pharynx. Focal areas of squamous cell hyperplasia were observed in the tongue and palate of the pharynx. 41
The chemical interacts with the mucosal epithelium, and it is suggested that its genotoxic mechanisms may contribute to the carcinogenic activity. Another suggested mechanism may also be due to oxidative damage to DNA. 41,42
Chloroprene (NTP TR 467)
The NTP study included preliminary, shorter term studies (16 days and 13 weeks) followed by a 2-year inhalation study on groups of 50 male and female F344 rats and B6C3F1 mice exposed to 0, 12.8, 32, or 80 ppm chloroprene for 6 hours/day, 5 days/week. Male rats exposed to 32 ppm and male and female rats exposed to 80 ppm had significantly higher incidences of SCP or SCP and SCC (combined) of the oral cavity involving palate, pharynx, gingiva, cheek, and tongue. 43
The mutagenic mode of action of chloroprene is postulated to be mediated by metabolizing to epoxide intermediates and the formation of DNA adducts via its epoxide metabolite. 44
Glycidol (NTP TR 374)
The NTP conducted a study by gavage administration of glycidol (94% pure, containing 1.2% 3-methoxy-l,2-propanediol,0.4% 3-chloro-1,2-propanediol,2.8% diglycidyl ether, and 1.1% 2,6-dimethanol-1,4-dioxane) in water to groups of F344/N rats and B6C3F1 mice for 16 days (doses for groups of 5 rats or 5 mice of each sex ranged from 37.5 to 600 mg/kg), 13 weeks (doses for groups of 10 rats ranged from 25 to 400 mg/kg, and doses for groups of 10 mice ranged from 19 to 300 mg/kg), or 2 years. In the 2-year study, groups of 50 F344/N rats of each sex were administered 0, 37.5, or 75 mg/kg glycidol in distilled water by gavage. Groups of 50 B6C3F1 mice of each sex were administered 0, 25, or 50 mg/kg glycidol in distilled water. There was an increased incidence of SCP of the mouth and tongue in female rats in both dosed groups, and the increase was significantly different from controls in the 75 mg/kg group. In addition, one female rat in the 75 mg/kg had SCC. In male rats, SCP or SCC (combined) was seen in 4% in low-dose and in 10% of high-dosed rats. However, these findings were also seen in 6% of the control group male rats. 45
The carcinogenicity of glycidol is postulated to be due to genotoxic activity, which is probably a direct effect of the compound and not due to metabolic activation. 46,47
Benzidine dye
The Society of Dyers and Colourists reported the production of 89 3,3′-dimethoxybenzidine-based dyes in 1971, including pigment orange 16, direct blue 1, direct blue 15, direct blue 8, direct blue 76, direct blue 98, and direct blue 218. As part of the NTP’s Benzidine Dye Initiative, 5 chemicals were evaluated in 2-year studies for carcinogenicity and toxicity. 3,3′-dimethylbenzidine and 3,3′-dimethoxybenzidine were chosen as the primary benzidine congeners. A group of prototypical dyes derived from those amines were selected including C.I. Acid Red 114, which is a 3,3′-dimethylbenzidine-based dye; and C.I. Direct Blue 15 and C.I. Direct Blue 218, which are 3,3′-dimethoxybenzidine-based dyes.
3,3′-Dimethoxybenzidine Dihydrochloride (NTP TR 372)
Toxicology and carcinogenesis studies were conducted by administering 3,3′-dimethoxybenzidine dihydrochloride (greater than 97.5% pure) in drinking water to groups of F344/N rats of each sex for 14 days, 13 weeks, 9 months, or 21 months. In the 21-month study (which was originally intended to last 24 months but was terminated early because of declining survival due to neoplasia in dosed groups), the F344/N rats received 3,3′-dimethoxybenzidine dihydrochloride in drinking water at doses of 0, 80, 170, or 330 ppm. In the NTP 21-month study, incidences of several neoplastic and nonneoplastic lesions were observed in exposed rats.
Significantly higher incidences of SCPs or SCCs (combined) of the tongue and palate were observed in males of the groups dosed with 80 ppm (18%), 170 ppm (13%), and 330 ppm (18%) when compared to the controls (2%). Squamous cell carcinomas occurred only in the 170 ppm (4%) and 330 ppm (3%) dosed female groups, with clear association with exposure to 3,3′-dimethoxybenzidine dihydrochloride. 48
3,3′-dimethoxybenzidine dihydrochloride is metabolized through conversion of aromatic amines by N-hydroxylation and formation of esters that are highly reactive electrophiles. This pathway contributes to DNA damage, resulting in carcinogenesis. 49 –51
3,3′-Dimethylbenzidine Dihydrochloride (NTP TR 390)
In the study conducted by the NTP, 3,3′-dimethylbenzidine dihydrochloride (approximately 99% pure) was administered in drinking water to groups of F344/N rats for 2 weeks (at doses ranging from 600 to 7500 ppm), 13 weeks (at doses of 300-4000 ppm), or 9 or 14 months (at doses of 30, 70, 150 ppm). The 14-month exposures were planned as 24-month exposures but were terminated early because of rapidly declining animal survival, primarily due to neoplasia.
Treatment with 3,3′-dimethylbenzidine dihydrochloride resulted in significantly increased incidences of SCP and SCC (combined) in the high-dose male and mid- and high-dose female groups when compared with the controls. In males, the incidence of SCP and SCC carcinoma (combined) was 5% in the mid-dose (70 ppm) group and 8% in the high-dose (150 ppm) group, while in females, the incidences were 12% and 22% in the mid- and high-dose groups, respectively. No squamous cell neoplasms of the oral cavity occurred in the vehicle control groups. 52
The metabolism process is believed to be similar to that of the related compound benzidine; conversion of aromatic amines by N-hydroxylation results in the formation of esters that are highly reactive electrophiles. This pathway contributes to DNA damage. 53
C.I. Direct Blue 15 (NTP TR 397)
C.I. Direct Blue 15 was chosen for evaluation due to the potential exposure by employees. Male and female F344/N rats exposed were to C.I. Direct Blue 15 in distilled drinking water for 14 days, 13 weeks, or 22 months (early termination was due to decreased survival and increased incidence of neoplasia). In the 22-month study, rats received 0, 630, 1250, or 2500 ppm C.I. Direct Blue 15.
There was a significantly increased incidence of squamous cell neoplasms of the oral cavity in dosed groups (630, 1250, and 2500 ppm) in male and female rats, both SCP and SCC, when compared with the control vehicles. 54
C.I. Acid Red 114 (NTP TR 405)
C.I. Acid Red 114 was chosen for a carcinogenesis study due to its potential for human exposure during the production of bisazobiphenyl dyes.
Desalted C.I. Acid Red 114 was administered in drinking water to groups of F344/N rats for 13 days, 13 weeks, 9 or 15 months, or 2 years. For the 2-year study, the doses given were 70, 150, and 300 ppm for males and 150, 300, and 600 for females. High-dose groups included 70 rats, 75 were in the mid-dose groups and 45 were in the low-dose groups. In females, there was a slight increase in SCPs and SCCs combined in dosed groups in comparison with control rats, but only the mid-dose female group showed a statistically significant increase (14%). Three SCPs occurred in dosed males (mid- and high-dosed groups). However, no SCCs were observed in male rats. 55
C.I. Direct Blue 218 (NTP TR 430)
Toxicology and carcinogenesis studies were conducted by administering C.I. Direct Blue 218 in feed to groups of male and female F344/N rats and B6C3Fl mice for 14 days, 13 weeks, and 2 years. Groups of 60 male and 60 female of both rats and mice were fed diets containing 0, 1000, 3000, or 10,000 ppm C.I. Direct Blue 218 for 2 years. Five treated male rats from the high-dose group (10,000 ppm) developed SCP of the pharynx, one male rat exposed to high-dose developed SCC and another developed basosquamous tumor. Squamous cell papilloma was observed in 1 (2%) of 50 of the female control group. The evidence was not considered strongly related to exposure to C.I. Direct Blue 218, since the incidence of SCC was not statistically increased. However, as other benzidine dyes have caused oral cavity neoplasms, the results observed suggest a relation to chemical exposure. 56
Dioxins and dioxin-like compounds
The dioxin toxic equivalency factors (TEF) evaluation included multiple 2-year rat bioassays to evaluate the relative chronic toxicity and carcinogenicity of dioxin-like compounds (DLCs), structurally related polychlorinated biphenyls (PCBs), and mixtures of these compounds.
3,3′,4,4′-Tetrachloroazobenzene (TCAB) (NTP TR 558)
The NTP performed a 2-year study on groups of 50 male and 50 female Harlan Sprague-Dawley rats and B6C3F1 mice. The rats were administered 10, 30, or 100 mg tetrachloroazobenzene (TCAB)/kg body weight in corn oil: acetone (99:1) by oral gavage. Mice were administered concentrations of 3, 10, or 30 mg TCAB/kg body also by gavage.
In the 2-year study, there was a significantly increased incidence of carcinogenicity in the oral mucosa of male and female rats administered TCAB when compared with the controls. Gingival SCC occurred in 10 mg/kg males and 100 mg/kg males and females. The occurrence of preneoplastic lesions (GSH and cystic keratinizing hyperplasia) in dosed groups of males and females were also significantly increased. 57
A possible mechanism may be related to the disruption of retinoid homeostasis induced by DLCs and causing vitamin A deficiency. Such disruption leads to altered growth and differentiation of the epithelium in the oral cavity, resulting in GSH that may develop into squamous neoplasms. 58,59
2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) (NTP TR 521)
The NTP conducted a chronic study with 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD), with interim terminations at 14, 31, and 53 weeks. Groups of 81 or 82 female Harlan Sprague-Dawley rats were administered 3, 10, 22, 46, or 100 ng TCDD/kg body weight in corn oil: acetone (99:1) by gavage for up to 2 years. After 2 years, SCC of the gingiva was observed in all the concentrations except 22 ng/kg and was significantly increased compared to controls in the 100 ng/kg.
2,3,7,8-Tetrachlorodibenzo-p-dioxin induces indirect alteration in retinoic acid receptor–dependent signaling pathway therefore affecting the epithelial differentiation of squamous epithelia. Another suggested mechanism includes the downregulation of aryl hydrocarbon receptor (AhR) after exposure to TCDD thus altering the differentiation and proliferation of the epithelium. 62,63
Polyhalogenated aromatic hydrocarbons comprise a large class of compounds including polychlorinated dibenzodioxins (PCDDs), polychlorinated dibenzofurans (PCDFs), PCBs, polychlorinated naphthalenes, and polybrominated diphenyl ethers. Certain PCDDs, PCDFs, and coplanar PCBs are able to bind to the AhR and show similar biologic actions to those of TCDD. They are commonly referred to as DLCs.
2,3,4,7,8-Pentachlorodibenzofuran (NTP TR 525)
The NTP performed a chronic study which included interim terminations at 14, 31, and 53 weeks. Groups of 81 female Harlan Sprague-Dawley rats were administered 6, 20, 44, 92, or 200 ng/kg 2,3,4,7,8-Pentachlorodibenzofuran (PeCDF) in corn oil: acetone (99:1) (97% pure) by gavage for up to 105 weeks. At 2 years, SCCs were seen in 2 of the 6 ng/kg dosed group, in 3 of the 200 ng/kg dosed, and one occurred in each of the vehicle control, 20 ng/kg, and 92 ng/kg groups. Gingival squamous hyperplasia occurred in all groups including the vehicle controls. Although the findings were not statistically significant, the lesions were considered to be exposure related. 64 2,3,4,7,8-Pentachlorodibenzofuran is believed to have similar mechanism of action as TCDD since it also requires initial binding to the AhR. 65
2,3′,4,4′,5-Pentachlorobiphenyl (PCB 118) (NTP TR 559)
The effects of 2,3′,4,4′,5-Pentachlorobiphenyl (PCB 118) alone were evaluated in the study conducted as part of the dioxin TEF evaluation. Groups of 80 female Harlan Sprague-Dawley rats were administered 100, 220, 460, 1000, or 4600 µg PCB 118/kg body weight in corn oil: acetone (99:1) by gavage, 5 days per week, for up to 105 weeks, with interim sacrifices at 14, 31, and 53 weeks. At 2 years, SCC was seen in the 220 μg/kg dosed group and 2 SCCs were seen in 4600 μg/kg dosed group. One SCC was also observed in the control group, suggesting that the occurrence of the neoplasms was less likely chemical related. 66
2,2′,4,4′,5,5′-Hexachlorobiphenyl (PCB 153) (NTP TR 529)
The NTP conducted a carcinogenicity study on PCB 153; prior to this, there had been no studies done on the carcinogenicity of PCB 153 alone. In order to evaluate the effects of PCB 153, groups of 50 female rats were exposed to PCB 153 dissolved in corn oil by gavage doses of 10, 100, 300, 1000, or 3000 μg/kg for 5 days a week for 2 years. Gingival squamous hyperplasia and SCC were observed in both control and dosed groups. 67
The mechanism of action is similar to TCDD which involves the binding of the compound with the AhR and activating CYP1A1, the gene encoding the protein cytochrome P4501A1, resulting in cell proliferation. 67,68
3,3′,4,4′,5-Pentachlorobiphenyl (PCB 126) (NTP TR 520)
Fifty female Harlan Sprague-Dawley rats were exposed to PCB 126 in corn oil with acetone by gavage with doses of 30, 100, 175, 300, 550, or 1000 ng/kg for 14, 31, or 53 weeks or 2 years. Apart from the groups administered 30 and 300 ng/kg, GSH occurred in all groups during the 2-year study. Gingival SCC occurred in all dosed groups at 2 years but only in the 1000 ng/kg study group was a significant increase compared to controls noted. 69
It is believed that the oral cavity squamous lesions may be related to the alteration in retinoid homeostasis that is known to be induced by PCB 126. 69
Since human exposure to DLCs has always occurred as a mixture, then NTP performed carcinogenicity assays on certain combinations of DLCs.
Binary Mixture of PCB 126 and 2,2′,4,4′,5,5′-Hexachlorobiphenyl (PCB 153) (NTP TR 530)
Groups of 53 female Harlan Sprague-Dawley rats were exposed to binary mixtures (constant and varying ratios) of PCB 126 and PCB 153 dissolved in corn oil through gavage, 5 days a week for 14, 31, or 53 weeks or 2 years. Daily doses were 10 ng/kg PCB 126/10 μg/kg PCB 153, 100 ng/kg PCB 126/100 μg/kg PCB 153, 300 ng/kg PCB 126/300 μg/kg PCB 153, or 1000 μg/kg PCB 126/1000 μg/kg PCB 153 in the constant ratio group, and 300 ng/kg PCB 126 with 100, 300, or 1000 μg/kg PCB 153 in the varying ratio group.
In the constant ratio groups, there were dose-related increased incidences of GSH in rats exposed to 100 ng/kg PCB 126/100 μg/kg PCB 153 and above. Incidences of gingival SCC of the oral mucosa were significantly increased in rats treated with 300 ng/kg PCB 126/300 μg/kg PCB153 and 1000 ng/kg of PCB 126/1000 μg/kg PCB 153. 70
Binary Mixture of PCB 126 and PCB 118 (NTP TR 531)
Female Harlan Sprague-Dawley rats in groups of 53, 54, or 66 were exposed to mixtures of PCB 126 and PCB 118 in corn oil by gavage, for up to 2 years. Daily doses were 10, 30, 100, 300, or 500 µg of PCB 118 along with 0.6% PCB 126, per kilogram body weight. Dose groups are referred to by the total levels of TCDD toxic equivalents (TEQ) provided by the PCBs in the mixture in each dose group. Groups of 81 female Harlan Sprague-Dawley rats were administered 7, 22, 72, or 216 ng TEQ/kg; a group of 86 female rats was administered 360 ng TEQ/kg.
Squamous cell carcinoma occurred in all groups except in the 216 ng TEQ/kg group; however, none of the incidences were significantly increased. The incidences in the 22 and 72 ng TEQ/kg groups exceeded the historical vehicle control range. Gingival squamous hyperplasia occurred in all groups including the control vehicle group, and incidences were significantly increased in all dosed groups except the 7 ng TEQ/kg group. 71
Mixture of 2,3,7,8-Tetrachlorodibenzo-p-Dioxin (TCDD), PeCDF, and PCB 126 (NTP TR 526)
The NTP study exposed Female Harlan Sprague-Dawley rats to a mixture of TCDD, PeCDF, and PCB 126 in corn oil: acetone (99:1) by gavage for 14, 31, 53 weeks, or 2 years. Groups of 81 female rats were administered 10, 22, 46, or 100 ng TEQ/kg body weight in corn oil: acetone (99:1) by gavage.
Exposure to the mixture caused significantly increased incidences of GSH, observed in all groups. Single incidences of gingival SCC occurred in vehicle control and 10 ng TEQ/kg groups and 2 were seen in the 100 ng TEQ/kg group. Thus, SCCs of the gingiva were not significantly increased over vehicle controls and may have not been treatment related. 72
Discussion
Oral SCC is one of the most common malignancies in humans worldwide and its incidence has been increasing recently. 73 It has been challenging to manage due to the lack of early detection markers. 74 In humans, long-term exposure to lifestyle-related factors, such as tobacco use and excessive alcohol consumption, viruses or chemicals, contribute to the development of oral cancer through genetic instability thus leading to carcinogenicity. 75 Consumption of tobacco in its various forms, such as snuff or chewing tobacco, and betel nut chewing, have become prevalent throughout the world, especially in the developing countries. 76 Chronic exposure to chewing tobacco/snuff induces local irritation and inflammatory reaction thus leading to damage to the epithelial cells of the oral mucosa. 77 Additionally, chewing tobacco leads to significant alterations in the oral microbiome. By disrupting the normal flora of the oral mucosa, local inflammation could pose an increased risk for oral cancer development. 78 The tumors associated with betel use are exophytic, unlike oral tumors in general, which are usually endophytic. This is believed to be due to the alterations in H-RAS gene mutations. 79,80
The presence of oral microbiome is also believed to contribute to carcinogen-induced tumorigenesis and progression in both rats and mice. 81
We analyzed the results of the carcinogenicity studies performed by the NTP. Among the nearly 550 chemicals in the NTP database, we have found 26 chemicals with a carcinogenic effect in the oral cavity. Fourteen of 26 of the studied chemicals demonstrated clear evidence of SCC and 3 chemicals showed clear evidence of SCP and SCC combined (based on NTP criteria of levels of evidence of carcinogenic activity in the NTP mouse and rat chronic cancer studies) in the oral cavity.
Although not all the studies were conducted in mice, 1,2,3-trichloropropane was the only NTP studied chemical that showed some or clear evidence of carcinogenic activity related to chemicals in mice. The administration of 2,4-hexadienal resulted in 2 incidences of SCC in male mice which were higher than in the vehicle controls.
It is unclear why rats were more prone to develop oral cancers than mice but rats are believed to be more susceptible to chemically induced tumors. 82
The overall incidence rates of SCP in the vehicle controls in F344 rats were 0.16% and 2.2% in female Harlan Sprague-Dawley rats. The incidence rates for SCC in F344 rats were 0.03% and 0.39% in female Harlan Sprague-Dawley rats. The incidence rates for oral tumors in the vehicle controls were not affected by the route of administration.
More than one mechanism is believed to be involved in chemical-induced carcinogenesis according to the studies performed in rodents. One of the suggested mechanisms is DNA damage and genomic instability, resulting in apoptosis. Such pathway alters the differentiation and proliferation of the epithelium. 83 Some chemicals such as benzidines and related aromatic amines induce neoplasms by the ability to N-hydroxylate the amine derivatives, thus activating the aromatic amines into reactive esters, acting as an ultimate carcinogen by causing DNA mutation. 84
Another suggested mechanism includes interaction with AhR. In the oral tissue of rodents and humans, AhR may be found in molar teeth buds and palatal epithelial cells of the gingiva. After the tissue is exposed to the chemical during morphogenesis of the palate, it becomes distributed throughout the palate and thus leads to downregulation of AhR, increased proliferation of the epithelial cells, and abnormal production of the stratified squamous epithelium. 85 However, humans have a different structure and composition of the amino acids of AhR than the rodents, and therefore, are 10-fold less sensitive to the chemicals. 86 An additional possible mechanism is the indirect disruption of the retinoid homeostasis in the liver by the carcinogenic chemical, leading to changes in the oral cavity. The chemical-induced changes in the retinoid action result in disruption of the normal differentiation and proliferation of the gingival epithelium. The epithelial cells differentiate into keratinized cells therefore developing squamous cell hyperplasia and neoplasia of keratinized squamous phenotype. 87 Another suggested mechanism is believed to be based on the continued and persistent irritation from contact with the chemical agent, leading to a hyperplastic reaction.
A review of the NTP database by Chandra et al also revealed that chemically induced neoplasms of the gastrointestinal tract are also relatively common. Of 290 carcinogenic agents, 19 chemicals caused intestinal neoplasia (14 in the rat and 5 in the mouse). Neoplasms occurred in both males and females, exclusively in the small intestine of the mouse and in the large intestine or both small and large intestine in the rat. 88 Similar to the suggested models of carcinogenicity in the oral cavity, also here proposed mechanisms for carcinogenicity include DNA damage or chronic irritation/inflammation. The possible mechanisms involved in oral carcinogenesis and the chemical compounds that activate them are summarized in Figure 1.
Here, we provided a detailed review of the histopathologic findings of carcinogenic changes induced by chemicals in the oral cavity of rodents. Such description can provide better insight into the carcinogenic process of oral cancers in general, and specifically, chemical-induced cancers. It can also be used to define better animal models for future in vivo studies, thus providing tools for the detection of oral cancers at an early stage and development of new specific treatments.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623321989956 - Chemical-Induced Oral Squamous Cell Neoplasms in Rodents: An Overview of NTP 2-Year Cancer Studies
Supplemental Material, sj-docx-1-tpx-10.1177_0192623321989956 for Chemical-Induced Oral Squamous Cell Neoplasms in Rodents: An Overview of NTP 2-Year Cancer Studies by Ruba Ibrahim, Amy Brix, David E. Malarkey, Abraham Nyska, Michal Asif and Yuval Ramot in Toxicologic Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
