Abstract
Epidemiological research indicates that the life expectancy for people with schizophrenia and other psychotic disorders is approximately 20 years less than their age-matched counterparts in the general population [1]. Mortality rates due to coronary heart disease (CHD) among people with psychotic disorders are around twice that seen in the general population [1]. The leading cause of excess death among people using mental health services in Australia is CHD [2], with at least one-third of people with schizophrenia also experiencing a coronary heart condition [3]. Internationally, CHD occurs more frequently and accounts for more premature deaths than suicide among people with schizophrenia [4]. People with schizophrenia also have much higher rates of CHD risk factors such as obesity, dyslipidaemia, hypertension, diabetes and smoking, and less access to medical care than people without schizophrenia [5]. The use of some antipsychotic medication has a documented association with CHD risk factors, such as weight gain, glucose and lipid abnormalities and cardiac side-effects [1, 5]. The exact nature of this interaction is not clear, but given the very high prevalence of high-fat, low-fibre diets, lack of exercise and smoking among people with severe mental disorders, it has been argued that these unhealthy behaviours are the likely causes of the majority of CHD among this group of people, irrespective of medication and socioeconomic deprivation [6]. Unhealthy lifestyles and lower CHD knowledge provide a focus for more comprehensive CHD interventions among people with severe mental disorder [6]. Problematic alcohol and other drug (AOD) use is also highly prevalent among people with severe mental disorders, contributing to overall CHD risk, and it has been recommended that problematic AOD use should be assessed and managed along with other CHD risk behaviours [7].
Given that a large number of these CHD risk factors have an environmental origin [8], it is arguable that they may respond to psychological intervention. Practice guidelines on the management of psychotic disorders recommend that clinicians can play an important role in screening for CHD risk factors, and that attention should be paid to these secondary conditions as well as to treatment for mental health problems [1, 5]. Unfortunately, CHD risk factors remain poorly detected and treated among people with psychotic disorders [9]. Lifestyle-type interventions with this group have typically not been as aggressively addressed as their complex psychiatric problems, despite the enormous impact of these factors on health and well-being, long-term morbidity/mortality and treatment compliance [5]. A small body of research, however, is beginning to emerge, suggesting that the psychological treatment of some of these risk factors may be feasible and effective. For example, interventions for smoking [10–12], weight loss [13–15] and physical activity [16] have been developed and implemented. While some experts argue that trying to stop smoking, change diet and increase the amount of physical activity might result in a patient failing all three [10], a recent Cochrane review concluded that an increased focus on improving several lifestyle activities can assist a person to stop smoking [11].
The present pilot study aimed to test the feasibility and short-term impact of a multi-component risk factor intervention for reducing (i) CHD risk; (ii) smoking; and (iii) weight, among smokers with psychosis. Secondary dependent variables included physical activity, unhealthy eating, substance use, psychiatric symptomatology, treatment retention, general functioning, and quality of life.
Methods
Participants and procedure
This was a feasibility study, utilizing a pre–post-treatment design with no control group. Participants were 43 smokers with a non-acute psychotic disorder who were recruited from Sydney and the Newcastle region of New South Wales, and Melbourne, Victoria, Australia. Referrals were received from community health agencies, general practitioners, and psychiatric rehabilitation services. Inclusion criteria were: (i) aged ≥18 years; (ii) smoking at least 15 cigarettes per day; (iii) body mass index (BMI) ≥27; and (iv) an ICD-10 diagnosis of a psychotic disorder. Exclusion criteria were (i) medical conditions that would preclude treatment (with nicotine replacement therapy (NRT) or overall; e.g. uncontrolled diabetes) and (ii) brain injury.
All participants provided written informed consent and were assessed before treatment and again a mean of 19.6 weeks later. The treatment consisted of nine individual 1 h sessions of motivational interviewing (MI) and cognitive behaviour therapy (CBT) plus NRT, in addition to treatment as usual. A letter was sent to the participant's general practitioner, case manager and/or treating psychiatrist to notify them of the involvement of their patient. Treatment sessions were conducted at the research centre or, if the subject preferred, at their local community clinic. The pre-treatment assessment session was of 1.5–2 h duration and the post-treatment assessment session lasted approximately 1 h. Research assistants who had not been involved in the delivery of the treatment programme conducted post-treatment assessments. Participants were reimbursed $AUD20 for their time, travel and participation on each assessment occasion. This research was approved by relevant regional and university ethics committees.
Diagnostic measures
Diagnosis was determined using the Diagnostic Interview for Psychosis (DIP) [12], a semi-structured interview that confirms diagnosis using the Operational Criteria for Psychosis [13] and produces a diagnostic classification in accordance with ICD-10. In addition to providing a clinical diagnosis, the DIP gathered information on sociodemographic characteristics, premorbid adjustment, current symptoms and medication and social functioning.
Outcome measures: primary dependent variables
Coronary heart disease risk
Estimated CHD risk scores were derived using the modified Framingham risk score [14] and an age- and sex-specific percentile score derived from the 1989 Risk Factor Prevalence Survey conducted by the National Heart Foundation [15]. Percentile scores >80 indicate higher CHD risk relative to other Australians of a similar age and gender who do not have those risk factors.
Smoking
Outcome measures were: continuous abstinence; point prevalence abstinence; number of cigarettes smoked per day; and smoking reduction status (whether or not the participant had reduced their daily consumption of cigarettes by ≥50%, including abstinence) relative to baseline. Continuous abstinence refers to the proportion of participants who reported not smoking at all from the nominated quit date to the post-treatment assessment point. Point prevalence abstinence refers to the proportion of participants who had been abstinent for the 7 days preceding the follow-up assessment [16]. Current abstinence from smoking was confirmed using a Micro 11 Smokerlyser (Air-Met Scientific Pty. Ltd., Melbourne, Victoria, Australia), which assesses breath levels of carbon monoxide (CO). A CO level <10 p.p.m. signified that the participant was unlikely to have smoked in the last 8 h. Nicotine dependence was measured by the Fagerstrom Test for Nicotine Dependence [17], in which scores range from 0 to 10, with scores >3 suggesting dependence. Nicotine withdrawal symptoms were measured using the Minnesota Withdrawal Scale [18], with higher scores indicating greater withdrawal symptoms. Additional questions on smoking history were also included.
Weight
Measures were: weight in kg (participants wore light indoor clothing, without shoes for measurement, with no adjustment to weight measures for clothing); BMI (calculating weight and height using standard methods); waist and hip circumference and ratio; and the perceived impact of weight, using the Impact of Weight on Quality of Life scale (IWQOL-lite) [19].
Outcome measures: secondary dependent variables
Physical activity
Participants were asked two questions about physical activity from the Smoking, Nutrition, Alcohol and Physical Activity guidelines [20]: number of times the person engaged in 20 min of vigorous physical activity and 30 min of moderate physical activity per week. A total activity score for each participant was calculated by adding the number of times per week each participant had exercised according to the categories of vigorous and moderate physical activity. A score of ≥5 occasions of exercise per week was categorized as adequate.
Unhealthy eating
An overall unhealthy eating index was created, with 1 point given for an answer to each question that indicated unhealthy eating habits. The index ranged from 0 to 12, with higher scores indicating more unhealthy eating habits. Unhealthy eating habits included: non-optimal serves per day of each of the five food groups (e.g. fruit, vegetables, breads, lean meats, and dairy); high-fat or high-sugar foods; choosing non-wholegrain products; consumption of full-sugar soft drinks or cordials; missing breakfast; adding salt to food; using full-fat dairy products; and consuming meat with visible fat.
Biological measures
Blood pressure, blood cholesterol, and blood sugar levels were measured. In two sites these measures were collected by a pathology laboratory and analysed. In the other two sites the therapists administered these tests using an Omron Automatic Blood Pressure Monitor (Omron Electronics, Sydney, New South Wales, Australia), and finger-prick blood tests for blood sugar and cholesterol.
Alcohol, cannabis and other substance use
Levels of alcohol consumption and cannabis use were assessed in relation to the month prior to interview via the Opiate Treatment Index [21]. Alcohol consumption was classified as ‘hazardous’ if it exceeded recommended guidelines for drinking (on average no more than four standard drinks for men and two for women per day). The number of caffeinated drinks per day was also recorded.
Psychiatric symptomatology and quality of life
Psychiatric symptomatology was assessed using the 24-item Brief Psychiatric Rating Scale [22] and the Beck Depression Inventory II [23]. General functioning was measured using the 12-item Short Form survey (SF-12) [24], which produces Mental Component Scores (MCS) and Physical Health Component Scores (PCS), with lower scores indicating greater disability.
Therapy
The MI, CBT and NRT intervention was delivered individually to participants by a trained therapist, who followed a treatment manual. Regular supervision with a psychologist was provided. Up to 42 mg of NRT was provided per day as per the protocol described by Hughes et al. [25]. The intervention consisted of six weekly sessions of 1 h duration followed by three booster sessions at fortnightly intervals (nine sessions in total). An outline of the content of each session is provided in Table 1.
Outline of healthy lifestyle intervention

MI, motivational interviewing, NRT, nicotine replacement therapy.
Statistical analysis
Data were analysed using SPSS for Windows (version 14.0; SPSS, Chicago, IL, USA). Paired t-tests were used to examine the difference from before to after treatment. A small number of regression analyses were also conducted to examine the contribution of treatment and illness factors to change scores on the primary outcome measures. Because this was a small pilot study, no adjustments were made for the number of statistical tests conducted.
Results
Subject characteristics
Figure 1 shows the recruitment and attrition profile for the project. There were 60 referrals of whom 48 people (80.0%) were recruited into the study. Five people did not complete the post-treatment assessment and were not included in the analysis. The final sample consisted of 43 participants. The mean age of subjects was 36.3 years, just over half were male (58%), and most were single and had never married (81%). Twenty-eight per cent were employed full time or part-time and 90% received welfare support. The most common diagnoses were schizophrenia (53.5%) or schizoaffective disorder (25.6%), followed by bipolar disorder (13.9%) and non-organic psychotic syndrome (7%). All participants had experienced more than one episode of psychotic disorder as follows: multiple episodes, good recovery (18.6%); multiple episode, minimal recovery or deterioration (32.6%); chronic course with little deterioration (11.6%); and chronic course with clear deterioration (37.2%).
Recruitment and attrition profiles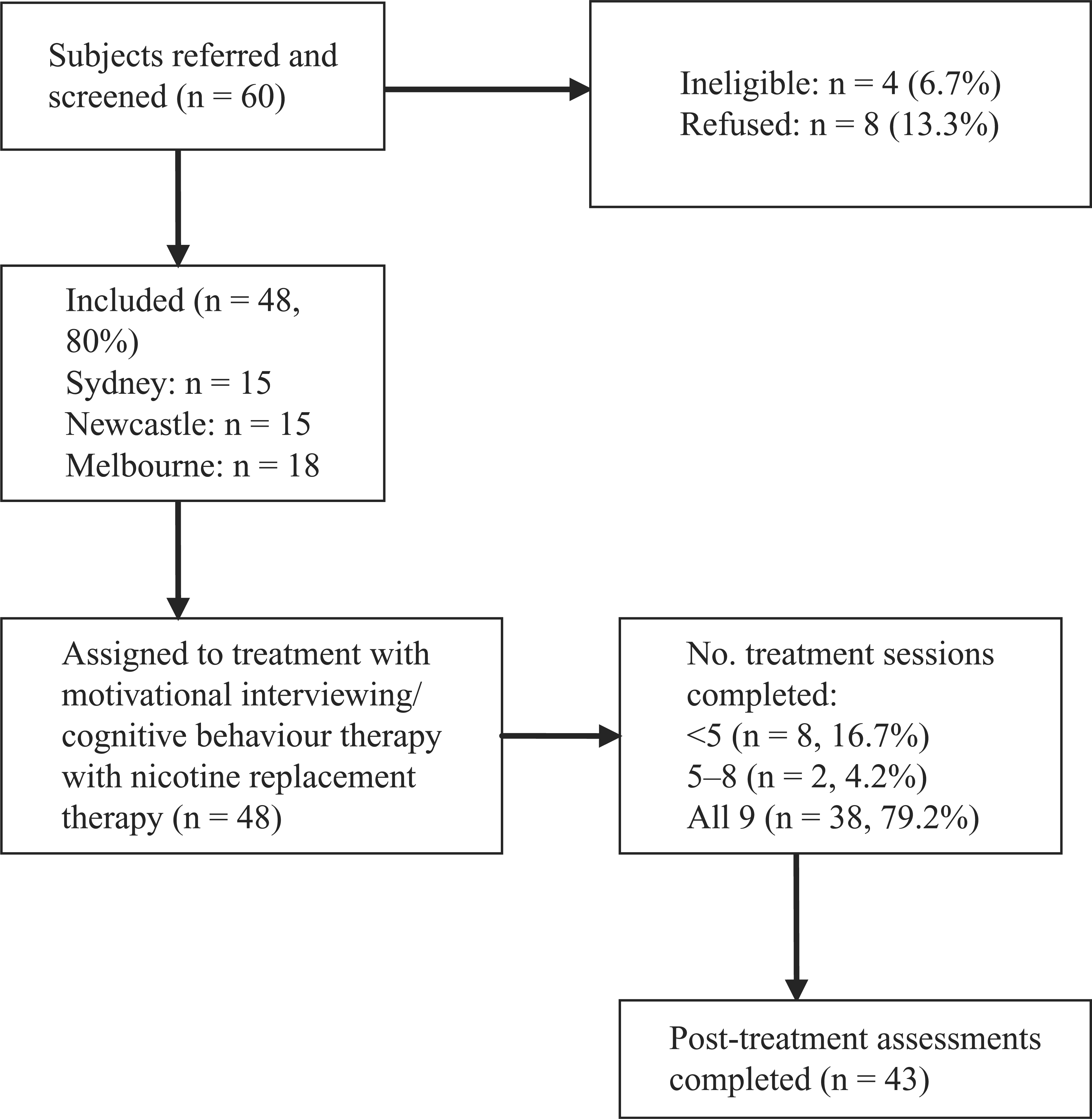
Differences between the participants who completed all nine sessions of the treatment programme (n=36) and those who completed fewer sessions (n=7) were analysed. There were no significant differences between groups on key demographic variables except for gender, with significantly more men (n=24, 96.0%) completing treatment than women (n=12, 66.7%; χ2 (1)=6.61, p<0.01). On average, non-completers had attended two sessions (SD=2.3, range=0–5).
Primary outcomes
Coronary heart disease risk
There was a significant reduction in overall CHD risk percentile scores (Table 2).
Outcomes on continuous measures (n=43)
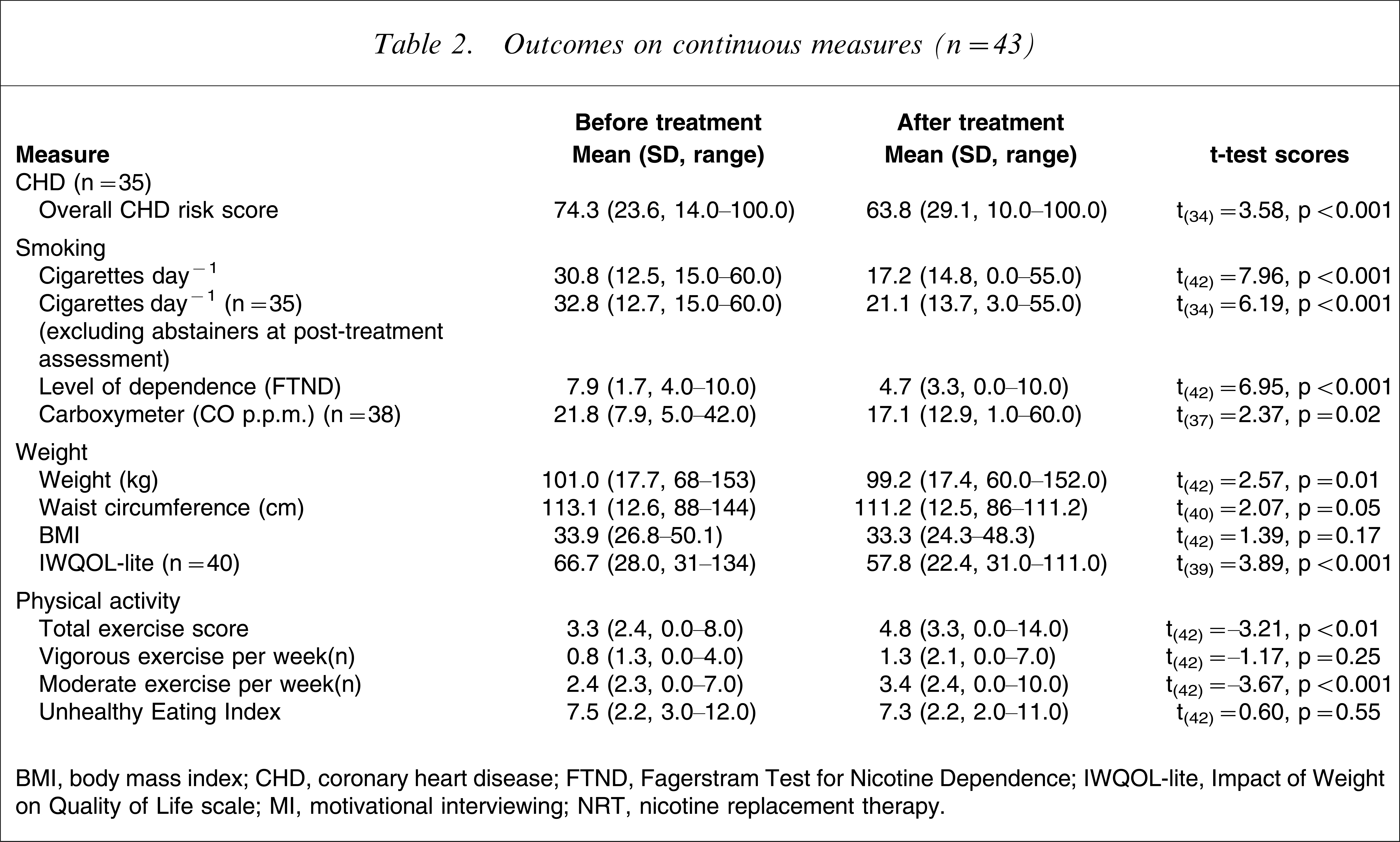
BMI, body mass index; CHD, coronary heart disease; FTND, Fagerstram Test for Nicotine Dependence; IWQOL-lite, Impact of Weight on Quality of Life scale; MI, motivational interviewing; NRT, nicotine replacement therapy.
Smoking
The mean age of commencing daily smoking was 17.1 years (SD=4.7), with an average of 2.8 (SD=1.5) serious quit attempts. Two participants were using NRT on entry to the study. At post-treatment assessment 11.6% of the sample reported being continuously abstinent from their quit date and 18.6% had been abstinent in the week prior to post-treatment assessment. Table 2 shows significant reductions in smoking in terms of cigarettes per day, level of dependence and expired carbon monoxide. Almost half of the sample (48.8%) reduced their smoking by at least 50%; 34.9% reduced their smoking by <50% and 16.3% showed no change in their smoking or worse smoking. Self-reported withdrawal symptoms reduced significantly from before treatment (mean=15.0, SD=7.8) to after treatment (mean=12.7, SD=7.7; t(42)=1.67, p=0.02). Likewise, observed withdrawal symptoms also reduced significantly from before treatment (mean=3.4, SD=3.3) to after treatment (mean=2.4, SD=2.6; t(40)=2.54, p=0.02).
Weight
As shown in Table 2 there were significant reductions from before to after treatment in weight, waist circumference, and in scores on the IWQOL-lite. There was no significant change in BMI (Table 2) or in participants’ waist/hip ratio (before treatment: mean=0.98, SD=0.08; after treatment: mean=0.97, SD=0.97; t(40)=1.61, p=0.12). Overall, approximately half of the sample achieved some weight loss, with 32.6% achieving a reduction of <5% of their pre-treatment weight and 20.9% achieving a reduction of at least 5%.
Secondary outcomes
Nutrition
There was a small non-significant improvement in the overall unhealthy eating index score (Table 2).
Physical activity
There was a significant increase in levels of physical activity as measured by the total activity score (before treatment: mean=3.3, SD=2.4; after treatment: mean=4.8, SD=3.3; t(42)=−3.21, p<0.01). The increase was evident in levels of moderate physical activity (before treatment: mean=2.4, SD=2.3; after treatment: mean=3.4, SD=2.4; t(42)=−3.67, p<0.001), as opposed to vigorous activity levels (before treatment: mean=0.84, SD=1.3; after treatment: mean=1.3, SD=2.1; t(42)=−1.17, p=0.25).
Substance use
There were no significant reductions in alcohol consumption (before treatment: mean=1.4, SD=3.5; after treatment: mean=1.0, SD=1.9; t(38)=1.25, p=0.22); cannabis use (before treatment: mean=0.92, SD=3.0; after treatment: mean=1.4, SD=4.4; t(42)=0.16, p=0.159), nor consumption of caffeinated drinks (before treatment: mean=7.1, SD=4.7; after treatment: mean=6.3, SD=4.2; t(43)=1.08, p=0.29).
Biological measures (blood sugar and cholesterol)
There were no significant changes in blood sugar from before to after treatment: 32.6% were at <4.4 mmol and 53.5% between 4.4 mmol and 8 mmol before treatment, with corresponding figures of 23.3% and 65.1% after treatment. Likewise, cholesterol levels did not show a significant change: 72.1% had a cholesterol level ≤5.5 mmol before treatment and 18.6% had a cholesterol level >5.5mmol; and after treatment 65.1% had a cholesterol level of ≤5.5 mmol and 20.9% had a cholesterol level >5.5 mmol.
Psychopathology
There were no significant before–after changes in SF-12 general functioning (MCS: from mean=40.8, SD=11.6 to mean=43.6, SD=13.3, t(42)=−1.54, p=0.13; PCS: from mean=45.5, SD=9.7 to mean=45.4, SD=10.4, t(42)=0.13, p=0.89), nor any change in levels of depression (from mean=14.2, SD=9.5 to mean=14.3, SD=12.6, t(42)=−0.06, p=0.96) or psychotic symptoms (from mean=40.7, SD=13.2 to mean=41.9, SD=12.2, t(42)=−0.67, p=0.51).
Contribution of treatment and illness factors to change
As a preliminary investigation of the potential factors influencing change, we examined associations between treatment completion, course of illness and the primary outcome measures, while controlling for gender, level of depression and substance abuse. There were no significant associations with change in CHD risk scores or number of cigarettes smoked per day. Two variables were significantly associated with weight loss: treatment completion status (partial correlation=−0.41, p=0.01) and course of psychotic disorder (partial correlation=0.38, p=0.02). Participants who had not completed treatment lost an average of 5.41 kg, compared to an average loss of 1.1 kg among those who completed treatment. Course of psychotic disorder was associated with greater weight loss, averaging 3.23 kg among those with a chronic illness with clear deterioration, 2.1 kg among those with a chronic condition but little deterioration, 1.5 kg among those with multiple episodes and partial recovery between episodes, and an average gain of 0.7 kg among those with good recovery between episodes.
Discussion
The major finding of the present study was that a multi-component CHD risk factor intervention consisting of MI/CBT and NRT among smokers with a psychotic disorder was associated with significant reductions in CHD risk scores, smoking and weight. A significant improvement was also reported in level of moderate physical activity, and a small change in the unhealthy eating index was reported. These findings are consistent with the view that behaviour change across several domains is possible [11], including among people with psychotic disorders. But no improvement in biological measures (cholesterol and blood pressure) was evident and a longer follow-up period may be needed to gauge changes in these measures.
Smoking results were comparable to or better than our previous smoking cessation treatment [26]. In the present study, at post-treatment assessment 11.6% of the sample reported being continuously abstinent from their quit date, 18.6% had been abstinent in the week prior to post-treatment assessment and 48.8% reported reducing their smoking by at least half. In the previous study, at post-treatment assessment the continuous abstinence rate for the treatment condition was 10.9% whereas for point prevalence abstinence it was 15.0%, while 43.5% of the sample reduced their smoking by at least half [26]. The reduction in the number of cigarettes smoked per day in the present study (from a mean of 31 cigarettes day−1 to a mean of 17 cigarettes day−1) compares favourably with the previous study (from a mean of 31 cigarettes day−1 to a mean of 23 cigarettes day−1 among the treatment condition subjects), and may be associated with the monitoring of nicotine withdrawal symptoms and the flexible delivery of NRT, as well as the more holistic lifestyle focus of the intervention.
The number of participants who completed all intervention sessions (n=36/43, 83.7%) suggests that the intervention represents a feasible approach to improving lifestyle and reducing CHD among people with psychosis. Further, the absence of any worsening in psychiatric symptomatology suggests that this type of lifestyle intervention and associated behaviour changes is tolerable among people with a psychotic illness. Interestingly, more men completed the intervention than women, suggesting that healthy lifestyle interventions may be especially appealing to men. Those who discontinued the intervention prematurely, however, were more likely to have lost weight than those who continued, possibly suggesting that this subgroup gave a higher priority to weight loss than to broader lifestyle interventions. Chronicity of illness course was also associated with greater weight loss, but not with a greater reduction in smoking, possibly indicating that those with a more chronic course had rarely been targeted for broader lifestyle interventions. Alternatively, a stepped care approach may be more suited to this subgroup, addressing weight first, and building upon this success with other behaviour changes as acceptable to the individual.
Because this was a pilot study of the feasibility of a multi-component CHD risk factor intervention, there are several methodological limitations of the study, including the absence of a control group and no longer-term follow up. To our knowledge, however, this is the first study among people with psychosis with multiple risk factors for CHD, including smoking and weight, to demonstrate the effectiveness of such an intervention. A randomized controlled trial, extending the length of this intervention in order to encourage further dietary changes, and comparing this intervention with a control condition, is warranted.
Footnotes
Acknowledgements
This study was funded by the Australian Commonwealth Department of Health and Ageing. GlaxoSmithKline provided NRT for the study. Thanks to the patients who participated in the study and the services from which they were recruited. Melinda Carrington provided valuable advice on CHD risk scores. Thanks to Anthony de Castella for assisting with the running of the trial, and to the therapists, Rachel Taylor and Di Harris. Thanks also to Louise Thornton for assistance in preparation of the manuscript.
