Abstract
Due to industrialization and globalization, habitual diets are increasingly dominated by energy-dense, nutrient-poor foods at the expense of high-fibre, unprocessed foods [1]. There is now some evidence to suggest that diet and nutrition play a role in the aetiology of depression [2–6], but there is a paucity of data concerning this relationship to date. Depressive illness is influenced by genetic, hormonal, immunological, biochemical, and neurodegenerative factors; nutrients and habitual diet modulate each of these factors and, as such, these large-scale dietary changes have the potential to affect prevalence rates of depression within the community [7].
The mineral magnesium may be of particular interest in depressive illness. Depression is associated with systemic inflammation [8–11], which is also associated with low intakes of magnesium [12, 13]. Serum magnesium deficiency has been shown in depressive disorders [14–16] but these findings are not uniform, with one study showing higher serum magnesium in patients with major depressive disorder compared to healthy controls [17]. A magnesium-deficient diet has been shown to increase depression- and anxiety-related behaviour in mice [18], while magnesium treatment has demonstrated antidepressant- and anxiolytic-like effects in animal models [19, 20]. Moreover, magnesium treatment improves symptoms of depression in chronic fatigue syndrome [21] and in patients with premenstrual syndrome [22], while two open-label studies showed a beneficial effect of magnesium treatment on symptoms of mania [23, 24]. Additionally, biological pathways involved in the pathophysiology of depression, as well as important transduction pathways, are modulated by magnesium [25].
The aim of the present study was thus to examine the cross-sectional association between magnesium intake and depression and anxiety in a large sample of community-dwelling middle-aged and older adults. We hypothesized that lower intakes of magnesium would be associated with higher levels of depressive and anxiety symptoms and case-level illness.
Methods
Study population
The Hordaland Health Study was conducted from 1997 to 1999. A subsample of this study, consisting of 9187 participants from four selected communities and born in the years 1925–1927 or 1950–1951, were invited to participate in a dietary substudy. Of these, 7074 (77%) agreed to participate and underwent a health examination that included comprehensive questionnaires regarding diet, mental health, and other lifestyle and medical parameters. In total 6140 subjects completed the dietary assessments (87%). Individuals with diagnosed kidney disease (n=2) were removed from the analyses, because kidney disease is known to alter magnesium status. Individuals with very low (<2000 kJ, n = 41) or very high (>15 000 kJ, n = 152) estimated daily energy intakes were excluded from the analyses, as were those with missing data on relevant dietary variables and covariates (n = 237). Participants with complete data on at least 13 of the 15 variables used were included in all analyses using pairwise exclusion. These adjustments resulted in a final sample of 5708 individuals (male, n = 2461; female, n = 3247). All participants gave written, informed consent to participate. The study was granted ethics approval by the Western Norway Regional Committee for Medical Research Ethics and by the Norwegian Data Inspectorate.
Exposure: magnesium intake
Dietary intakes were assessed by utilization of a self-administered, optically readable food frequency questionnaire (FFQ) [26], originally developed at the Department of Nutrition, University of Oslo [27], and designed to obtain information on usual food intake during the past year. This comprehensive questionnaire consisted of questions regarding 169 food items, and included frequency alternatives (from once per month to several times per day), the number of food units eaten and the portion sizes. It also assessed the number and timing of meals per day. The foods were grouped in order to reflect normal Norwegian meals. From this information, dietary intakes of magnesium were calculated using a database and software system based on the Norwegian Food Composition table [28].
Outcome: Hospital Anxiety and Depression Scale
In this study, symptoms of depression and anxiety were self-reported using the Hospital Anxiety and Depression Scale (HADS) [29]. The HADS is a self-administered questionnaire consisting of 14 4-point Likert-scale items, seven for anxiety (HADS-A subscale) and seven for depression (HADS-D subscale), encompassing the past week. The HADS is a widely used instrument designed to assess both caseness and symptomatology of depression and anxiety in epidemiological research and specialist care [30]. In the present study, scores on HADS-A and HADS-D were summed to give a total continuous score for each subscale that had the potential range of 0–21, and these scores were scaled so that 1 unit represents the distance between the 25th and 75th percentiles [31]. For those participants with fewer than three missing values on the HADS, scores were replaced with individual means. In additional analyses, case-level depression was defined as a HADS-D score ≥8, while case-level anxiety was defined as a HADS-A score ≥8 [31]. Both depression and anxiety symptomatology and case-level depression and anxiety were examined as outcomes in testing the hypothesis.
Covariates
Covariates were identified a priori, based on previous literature. These included total energy intake, gender, age group (47–49 years or 70–74 years), waist–hip ratio (WHR), body mass index (BMI), blood pressure (BP), income, education, physical activity (PA), current smoking and alcohol use. Folate intake was initially included as a possible confounding variable, due to its previously identified association with depression [2, 6]. Due to the high correlation, however, between folate and magnesium intake found in the present study (Pearson's R = 0.855, p < 0.001) and issues of collinearity, we decided not to include folate in further analyses [32]. Income consisted of nine categories ranging from zero to ≥500 000 Norwegian kroner per annum. Current smoking was categorized as individuals reporting the smoking of cigarettes, cigars or pipes on a daily basis (yes/no). PA consisted of four categories: no PA or light PA < 1 h week−1; light PA ≥ 1 h week−1; hard PA < 2 h week−1; hard PA ≥ 2 h week−1. Education included the categories of basic schooling, finished high school, 1–3 years of university and ≥4 years of university. Alcohol use was measured as units of alcohol consumed per fortnight, and categorized into three levels: no drinking, drinking 1–15 units per fortnight, and drinking >15 units per fortnight. In all analyses, diabetes and cardiovascular disease (CVD) were also checked as possible mediating factors.
Statistical analysis
The association between magnesium intake and both depression and anxiety scores was evaluated using linear regression analyses. Multivariate models were developed using magnesium intake (mg day−1) as the exposure variable and depression or anxiety scores as the dependent variables. Effects were reported with adjustment for energy intake, followed by adjustments for gender and age, then WHR, BMI and BP, with the final model adjusted for education, income and health behaviours: smoking, PA and alcohol consumption.
All analyses were replicated with case-level anxiety or depression as the outcome variables, using logistic regression models to estimate odds ratios with 95% confidence intervals. In each set of analyses, model 1 included magnesium intake (mg day−1) adjusted for energy intake; model 2 included additional adjustments for gender and age; model 3 included WHR, BMI and BP; model 4 further adjusted for education, income and health behaviours: smoking, PA and alcohol consumption. In all statistical analyses the level of significance was α = 0.05. In validating the models, interaction terms were tested for significance.
Results
Subject characteristics are summarized and presented in Table 1. Because there were no age or gender interaction in the association of interest (all interaction tests p > 0.05), and no a priori reason to expect any such interaction, further statistical analyses were conducted for the entire study population. Univariate associations between each of the covariates and magnesium intake as the outcome variable were examined (Table 2), and all covariates except WHR, BP and age, were related to magnesium intake. Higher education, higher income, higher PA levels, higher alcohol consumption, lower BMI and non-smoking were all associated with higher magnesium intake, adjusted for energy, age group and gender. Women had a higher energy-adjusted magnesium intake, whereas there were no differences between age groups in energy-adjusted magnesium intake.
Characteristics of the Hordaland Health Study participants
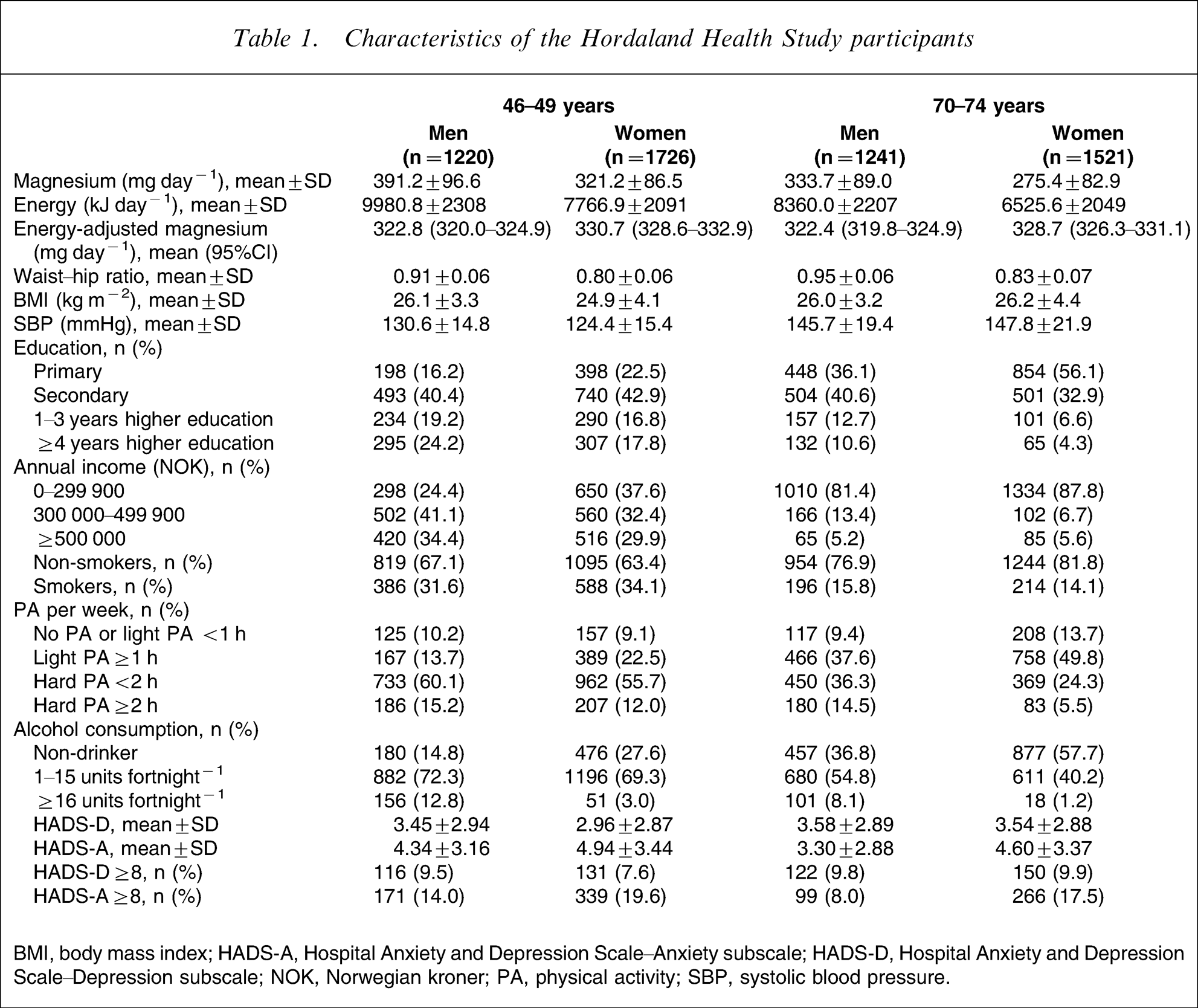
BMI, body mass index; HADS-A, Hospital Anxiety and Depression Scale–Anxiety subscale; HADS-D, Hospital Anxiety and Depression Scale–Depression subscale; NOK, Norwegian kroner; PA, physical activity; SBP, systolic blood pressure.
Association of magnesium intake and covariates: Hordaland Health Study
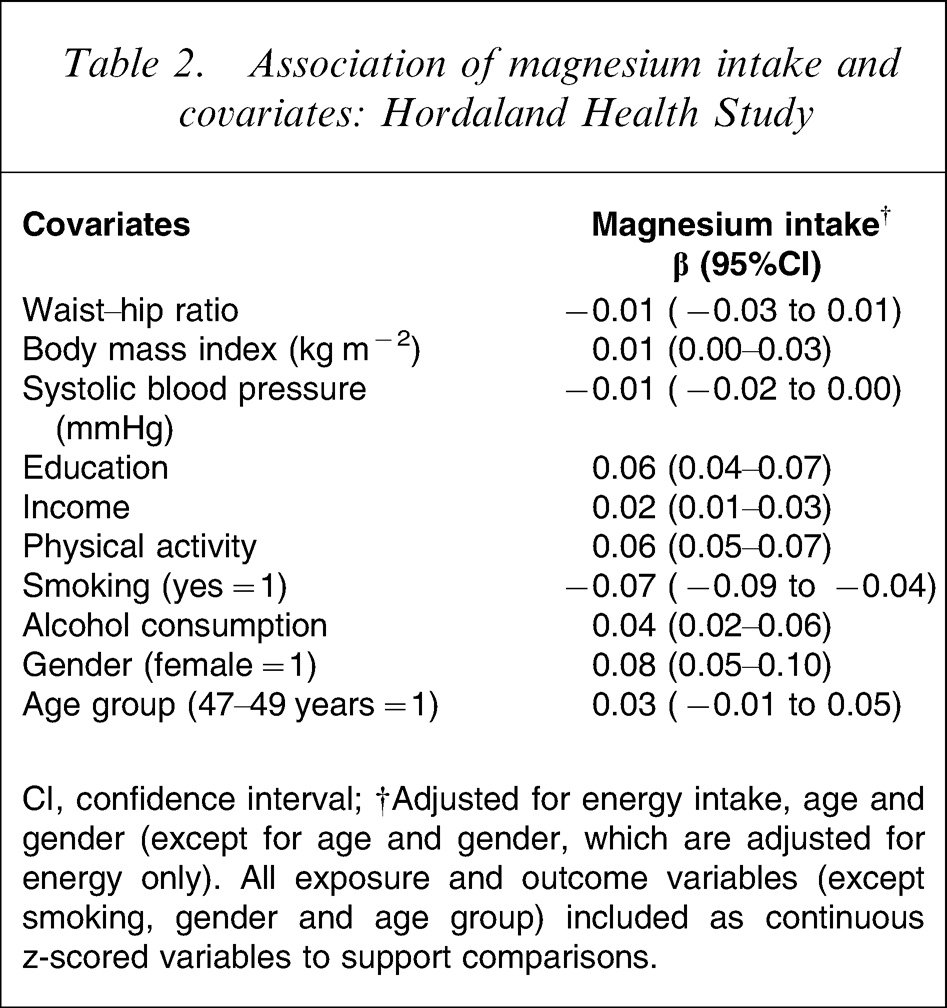
CI, confidence interval; †Adjusted for energy intake, age and gender (except for age and gender, which are adjusted for energy only). All exposure and outcome variables (except smoking, gender and age group) included as continuous z-scored variables to support comparisons.
In analyses with continuous standardized scores as the outcome, there was an inverse association between magnesium intake and depression scores (Table 3). This relationship was not confounded by age group, gender, body habitus or BP. It was somewhat attenuated, but remained statistically significant, after further adjustments for socioeconomic and lifestyle variables. Because both depression and magnesium were standardized scores, the effect size for this association was a 0.17 SD (adjusted for energy only) or 0.11 SD (fully adjusted) decrease in symptoms of depression for every energy-adjusted standard deviation increase in magnesium intake. For symptoms of anxiety, we found a similar but weaker association. While the association adjusting for energy intake only was not significant, further adjustments for gender, age group, WHR, BMI and systolic BP strengthened the association, which became significant. In the fully adjusted model, however, the association was no longer significant (Table 3). We further examined the association of both education and PA, as established risk factors for depression, to depression scores in order to compare the effect size of the magnesium association. After adjustments for age group and gender, there was a 0.08 SD decrease in symptoms of depression for every standard deviation increase in education and a 0.14 SD decrease in symptoms of depression for every standard deviation increase in PA.
Relationship between magnesium intake and depression and anxiety scores†: Hordaland Health Study.
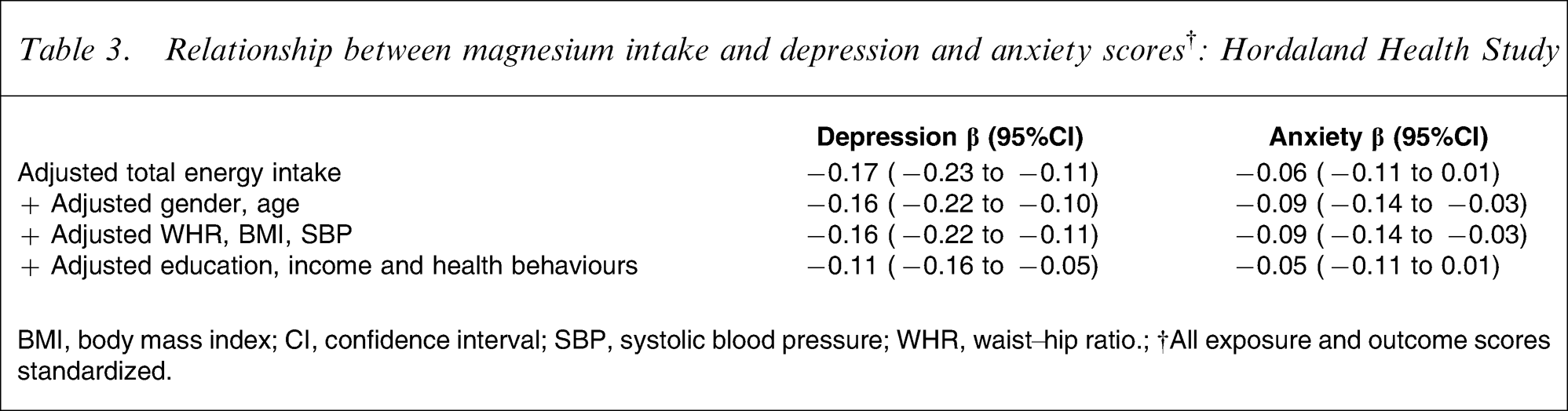
BMI, body mass index; CI, confidence interval; SBP, systolic blood pressure; WHR, waist–hip ratio.; †All exposure and outcome scores standardized.
In the present study 517 people (9.1%) met criteria for case-level depression (HADS-D ≥8) and 874 (15.3%) for case-level anxiety (HADS-A ≥8). In the logistic regression analyses, with case-level anxiety or depression as the outcome variables, magnesium intake was related to case-level depression across initial levels of adjustments, but the association was attenuated when adjusted for socioeconomic and lifestyle factors (Table 4). The association between magnesium and case-level anxiety was weaker and borderline significant throughout adjustments, and non-significant in the fully adjusted model (Table 4). Neither diabetes nor CVD mediated the observed relationships.
Association of magnesium intake† and case level depression and anxiety
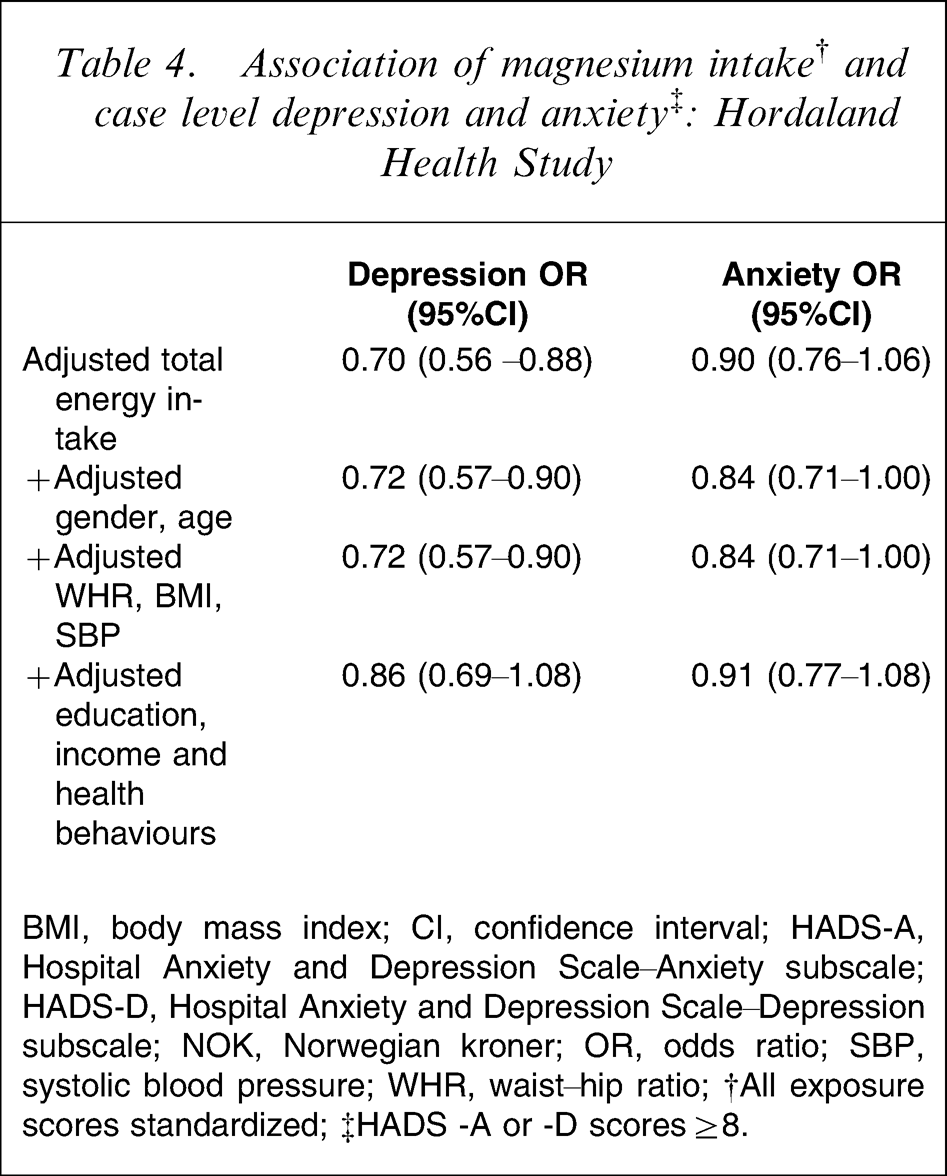
BMI, body mass index; CI, confidence interval; HADS-A, Hospital Anxiety and Depression Scale–Anxiety subscale; HADS-D, Hospital Anxiety and Depression Scale–Depression subscale; NOK, Norwegian kroner; OR, odds ratio; SBP, systolic blood pressure; WHR, waist–hip ratio; †All exposure scores standardized; ‡HADS −A or −D scores ≥8.
Discussion
The main finding of the present cross-sectional study is that of an inverse relationship between magnesium intake and depressive symptoms in community-dwelling middle-age and older adults; the associations persisted after adjustments for age, gender, body habitus, BP and socioeconomic and lifestyle factors. A similar trend was found for magnesium intake and anxiety symptoms, although the associations were weaker and not significant after adjustments. The strength of the association between magnesium intake and depression score was comparable to (and perhaps exceeding) that of low education and PA, both of which are established risk factors for depression [33–38].
While recognizing that the cross-sectional nature of this study prevents any determination of causality, these data do offer support for the hypothesis that the intake of magnesium is related to mental health in community dwelling adults. They are also concordant with a growing body of literature suggesting a role for other aspects of habitual diet in depression. For example, in most [3–5, 39], but not all [40], cross-sectional population studies, both long-chain omega-3 fatty acid and fish consumption have been shown to be inversely associated with depressive symptomatology, while two prospective population studies have demonstrated increased odds of developing clinically significant depression for those with low serum folate [6] and low folate intakes [2]. Magnesium and folate, however, are found in many of the same foods and it may be the case that unrecognized confounding by magnesium intake or status influenced previous studies identifying a relationship between folate and depression.
There are a number of important limitations to the present study: although the results are compatible with our hypothesis, the cross-sectional design of this study prevents any exposition regarding the direction of the relationship. It may be the case that a poor-quality diet is a result of mental health symptoms, rather than a causative factor. Although measures of socioeconomic status and health-related behaviours, as well as metabolic parameters that are related to both depressive illness and dietary patterns, were examined as covariates in the analyses and excluded as confounders, we cannot rule out the possibility that the present findings may be biased by unmeasured confounders or by residual confounding. The major strength of the present study lies in its large, population-based sample of both genders, including both middle-age and older participants. The FFQ utilized in the present study was of a very high quality, being comprehensive and well validated [27], and the size of the sample reduces the impact of measurement error. The data regarding relevant socioeconomic status and lifestyle covariates were also of a high standard.
Another important issue in studies of dietary variables as predictors of illness outcomes is that of ‘fishing’. It is essential in these studies that an a priori hypothesis exists before analyses are undertaken, to avoid the potential for simply reporting any statistically significant associations in large datasets such as the Health Study of Hordaland County Norway (HUSK). In the present study, a hypothesis was developed on the basis of the literature, and a study proposal developed for approval from the HUSK steering committee, months before the study was undertaken. In proceeding with analyses, only those variables identified as exposure, covariate and outcome variables in the proposal were examined. This is an important aspect of the study because it reduces the risk of the results being a type I error.
While being cognisant of the limitations of our study, there is some cause to hypothesize that the demonstrated relationship between habitual magnesium intake and depressive symptoms is mediated by biological mechanisms. Previous literature has demonstrated an association between low magnesium and increased inflammation in both rodents [41, 42] and humans [12, 13]; as well as between depression and inflammation in clinical and epidemiological studies [8, 9, 43–45]. This suggests that one mechanism behind our finding of an association between magnesium intake and depression may be related to the immune system. Studies in animal models have shown that induced magnesium deficiency in rats results in a systemic pro-inflammatory/pro-oxidant state [46], associated with elevations in circulating inflammatory cells and inflammatory cytokines, such as interleukin-6, tumour necrosis factor-α and positive acute phase proteins [41, 42] as well as increased neutrophil activity [47]. Thus, habitually inadequate intakes of magnesium may result in a chronic state of systemic inflammation, potentially contributing to increased depressive symptoms.
If our findings do reflect a role for magnesium in modulating symptoms of depression, these data from community dwelling adults in Norway may underestimate the impact of magnesium-deficient diets. Approximately 68% of US adults consume less than the American recommended daily allowance (RDA) of magnesium (420 mg day−1 for men, 320 mg day−1 for women [48]), with 19% consuming less than half of the RDA [12]. Moreover, intake of magnesium is known to be inadequate in many Western countries [49–53]. Intake of magnesium in Norway, however, is generally higher than that in other Western countries, due to a greater consumption of whole grains [54].
To our knowledge this study is the first to test the hypothesis that magnesium intake is related to anxiety and depression in an epidemiological setting. While being cognisant of the limitations of the study, the present findings of an inverse association between magnesium intake and depression may have both public health and treatment implications. In primary and specialist care settings, as well as in the public sphere, recommendations regarding lifestyle modifications are commonly made on the basis of reducing the risk of cancer, CVD or type II diabetes. In contrast, psychiatry lacks such prevention recommendations based on lifestyle factors. In common with both folate and omega-3 fatty acids, magnesium intake is likely to act as a proxy for a healthy diet because it is found primarily in foods that comprise many aspects of the Mediterranean diet [55]. If an evidence-based preventive health-care strategy incorporating dietary improvement can be developed for mental illness, it is likely to overlap heavily with existing strategies for other common, chronic, non-communicable diseases, with the hope of reducing the burden of illness and improving outcomes for people suffering from mental ill health.
Footnotes
Acknowledgements
The Hordaland Health Study was conducted from 1997 to 1999 as a collaboration between the National Health Screening Service, the University of Bergen in Norway, the University of Oslo in Norway and local health services in the Bergen region. F. Jacka was funded by the Australian Rotary Health Research Fund and a University of Melbourne Postgraduate Overseas Research Scholarship. R. Stewart is funded by the NIHR Biomedical Research Centre for Mental Health, South London and Maudsley NHS Foundation Trust and Institute of Psychiatry, King's College London. A. Mykletun is funded by the Norwegian Institute of Public Health.
