Abstract
Although the epidemiology of first-episode psychosis (FEP) is poorly understood due to a lack of systematic studies, it represents the underlying basis for our understanding of the disorder and forms part of the rationale for many clinical, biological, therapeutic, and long-term outcome studies [1]. FEP subsumes a broad range of clinical diagnoses: schizophrenia, schizophreniform disorder, schizoaffective disorder, brief psychotic disorder, delusional disorder, bipolar disorder I with psychotic features, major depressive disorder with psychotic features, substance-induced psychotic disorder and psychotic disorder not otherwise specified.
Diagnostic classification of FEP patients is complex because clinical features may change over time and overlap between diagnostic categories [2], and application of diagnostic hierarchies may mask complex patterns of comorbidity. This fluidity in clinical presentation can lead to diagnostic misclassification when diagnosis is based solely on hierarchical cross-sectional assessment early in the phase of illness. For this reason, ascertainment of an unselected and representative FEP cohort is preferred because it does not require the use of ill-fitting diagnostic schema and it allows us to track the natural history of FEP over time.
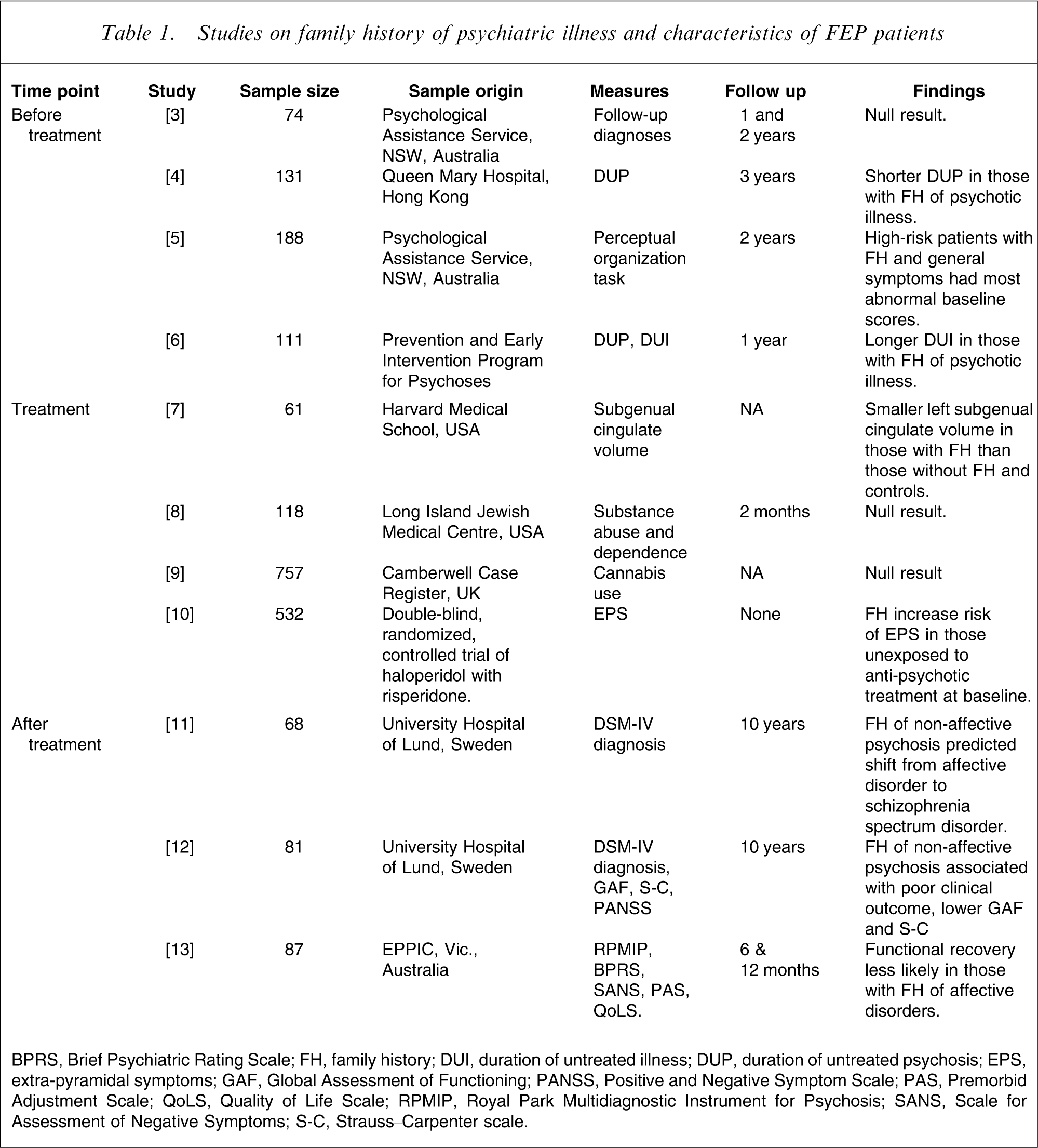
Family history is a major risk factor for the development of psychosis but the relationship between family history and the course of psychosis, especially FEP, has received very limited attention (Table 1). Familial morbid risk for schizophrenia is almost 10 [14], which means that there is a nearly 10-fold increase in lifetime risk of developing the same disorder for first-degree relatives. First-degree relatives of patients with bipolar disorder also have a greatly increased risk of developing bipolar disorder and other psychotic illness [15]. Consequently, it is important to consider the role of family history not only as a risk factor for the development (onset) of disorder but also in the course of illness.
Studies on family history of psychiatric illness and characteristics of FEP patients
BPRS, Brief Psychiatric Rating Scale; FH, family history; DUI, duration of untreated illness; DUP, duration of untreated psychosis; EPS, extra-pyramidal symptoms; GAF, Global Assessment of Functioning; PANSS, Positive and Negative Symptom Scale; PAS, Premorbid Adjustment Scale; QoLS, Quality of Life Scale; RPMIP, Royal Park Multidiagnostic Instrument for Psychosis; SANS, Scale for Assessment of Negative Symptoms; S-C, Strauss–Carpenter scale.
Eleven studies have investigated the utility of family history for predicting either premorbid status or transition from premorbid state to threshold disorder, admission characteristics while still naive to anti-psychotic drugs or psychiatric and functional outcomes over 6 months–10 years (Table 1).
Among FEP patients, family history of psychotic illness has been found to predict a shorter duration of untreated psychosis (DUP) compared to patients with no family history of psychiatric illness or those with a family history of only non-psychotic illness [4]. Norman et al. could not replicate this finding, but found that duration of untreated illness (DUI; time from onset of any psychiatric symptoms to treatment) was significantly longer among patients with a family history of psychotic illness [6]. This was due to these patients experiencing a longer period of time between onset of early signs and onset of psychosis.
Transition to psychosis among patients with at-risk mental states (as defined by Yung et al. [16]) was not predicted by family history of psychiatric illness [3], but diagnostic stability may be related to family history. Jarbin and von Knorring found that FEP patients who were initially diagnosed with affective disorder and who developed a schizophrenia spectrum disorder approximately 10 years later were significantly more likely to have a positive family history for non-affective psychosis, compared to patients diagnosed with affective disorder at both time-points [11].
Poor outcome among FEP patients appears to be predicted by family history of psychiatric illness. Jarbin et al. reported that patients with schizophrenia or schizoaffective disorder and a family history of non-affective psychosis were significantly more likely to have a poor functional outcome at 10 year follow up [12]. Conus et al. found that family history of affective disorder was associated with a reduced likelihood of recovery of function at 12 months in patients experiencing their first episode of psychotic mania [13].
To date, no studies investigating family history of psychiatric illness have found an association with substance use. Sevy et al. found no difference in family history of schizophrenia among patients with first-episode schizophrenia or schizoaffective disorder, with or without a personal history of substance misuse [8]. A recent study focusing on cannabis use in patients with first-episode schizophrenia also found no association with family history of schizophrenia [9].
Honer et al. found that among FEP patients not previously exposed to anti-psychotic medication, family history of severe mental illness significantly increased the risk of extra-pyramidal symptoms and signs (EPSS) [10]. This association was not observed among patients who had undergone anti-psychotic therapy. This suggests some familial (possibly genetic) predisposition to movement disorders in FEP patients that is obscured by anti-psychotic therapy.
Previous FEP studies that have examined family history have three major limitations. First, with the exception of the Boydell et al. and Honer et al. studies [9], [10], all recruited fewer than 200 subjects (Table 1) thereby limiting the statistical power of many reported analyses. Second, there was wide variation in the definition and assessment of family history. Family history has been variably defined as any psychiatric disorder [5], [10], affective versus non-affective disorder, or psychotic versus non-psychotic disorder [4], [6], [11], [12]. Some previous studies have used structured measurement instruments with known properties, such as the Family Interview for Genetic Studies (FIGS) [3] or the family history component of the Retrospective Assessment of the Onset of Schizophrenia [4], [6]. Others gathered family history information from unstructured discussions with patients, medical records, or interviews with social workers and carers [7], [9], [11], [12]. These different collection methods will, of course, yield different degrees of validity and reliability but the properties of unstructured family history data collection are currently unknown.
Third, no previous study that examined family history in an FEP sample ascertained an unselected and representative FEP cohort. All previous studies have applied a variety of exclusion criteria including specific FEP diagnosis [6], [8–10], [13], recent substance use [8], current substance use disorder (SUD) [7], [10], past SUD [7], any other psychiatric comorbidity [10], onset of psychosis after 19 years of age [11], [12], low IQ [7], [10], [13], a neurological condition or history of head trauma [4], [7], [10], left-handedness [7], a history of special school attendance [4], and pregnancy, lactation or inadequate contraception [10]. Others initially ascertained a sample deemed to be at ultra-high risk for psychosis based on specified criteria [3], [5].
The aim of the present study was to characterize the utility of family history data for predicting psychiatric and functional status during the pre-treatment phase of illness, at first presentation, and at 18 months follow up. The sample consists of a large, unselected and representative treated FEP cohort attending the Early Psychosis Prevention and Intervention Centre (EPPIC) in Melbourne, Australia.
Methods
Setting and sample
The First-Episode Psychosis Outcome Study (FEPOS) is a clinical file audit study conducted at EPPIC in Melbourne, Australia [17], [18]. EPPIC provides a comprehensive early intervention programme for people aged 15–29 years experiencing FEP, as defined by the World Health Organization [19], [20]. The catchment area for EPPIC covers the north-western suburbs of Melbourne, which at the time of the file audit had a population of approximately 800 000. The treated incidence of FEP among 15–29-year-olds in the catchment area is 16.7 per 10 000 person years for male subjects and 8.1 per 10 000 person years for female subjects [19]. The absence of other psychiatric facilities or private psychiatrists for the target population, as well as limited, if any, leakage to private facilities outside the catchment area, provides EPPIC with a non-selected FEP cohort and the FEPOS with a treated epidemiological sample [19].
Between 1 January 1998 and 31 December 2000, 786 patients were treated at EPPIC following the Australian clinical guidelines for early psychosis [21]. Treatment spanned an average of 18 months for most patients but for those who entered the programme at age 15 or 16, treatment was extended until 18 years of age. After exclusion due to discharge to other services, or a non-psychotic diagnosis at treatment end-point, 661 patient files were included in the file audit [18]. A clinical file audit tool, the Early Psychosis File Questionnaire (EPFQ) was used to extract information from consecutive medical files and has been described in detail elsewhere [17], [18].
In brief, the EPFQ records (i) premorbid adjustment, duration of prodrome (DP), DUP, psychiatric history, and family history of psychiatric disorder; (ii) psychiatric status at entry to EPPIC, including DSM-IV diagnosis [22], severity of illness, functional status and degree of insight; (iii) psychiatric status during treatment including comorbid conditions, adherence to medication, and service engagement; and (iv) psychiatric status at discharge or disengagement, including final DSM-IV diagnosis, symptomatic and functional outcomes and severity of illness. The North Western Health, Melbourne ethics committee granted approval to access data from medical records.
Measures
Psychiatric diagnoses
A consensus diagnosis following the DSM-IV [22] was determined for all patients via an intensive diagnostic and treatment process. Diagnoses were assigned by clinicians and case managers within 6 weeks of admission to EPPIC (baseline diagnosis) and on the basis of all information gathered in the file over 18 months (final diagnosis). Research psychiatrists assessed all information available in charts with respect to baseline and final diagnoses, conferring with the case manager of patients if there were any queries regarding diagnosis [23]. The validity of the FEPOS diagnoses was assessed by comparing the EPFQ diagnosis with the Structured Clinical Interview for DSM-IV (SCID-I/P) diagnosis [24] for 114 randomly selected patients given the SCID-I/P within 6 weeks of admission to EPPIC. Very good agreement was found for a diagnosis of any psychotic illness (κ = 0.80) and any comorbid substance abuse (κ = 0.74).
Clinical assessments
Premorbid functioning was defined as the best Global Assessment of Functioning (GAF) [22] level in the year preceding illness onset. GAF was also used to assess adjustment at both entry and discharge from EPPIC. Severity of illness was assessed at entry to, and discharge from EPPIC by the Clinical Global Impressions-Severity of Illness scale (CGI-S) [25]. The interrater reliability (κ) in this sample for the CGI-S was 0.87 and for the GAF 0.88 [17]. Concurrent validity was also high; the correlation between scales was r = 0.78 [18].
Onset of sustained positive psychotic symptoms was rated by clinicians using the Duration of Untreated Psychosis scale [26], [27]. DUP was defined as the number of days between onset of sustained positive psychotic symptoms and treatment [28]. DP was defined as the time from the first clinically significant deviation from the patient's premorbid personality to manifestation of first psychotic symptoms.
Definition of family history
Family history was collected by clinicians in an unstructured fashion with the aid of a family tree. Clinicians interacted with patients and their families over the entire 18 month treatment period and could therefore revisit patient files to update information when necessary. Family history was defined in reference to first- and second-degree relatives combined as (i) no family history of any clinically significant psychiatric disorder; (ii) a positive family history of a psychotic disorder, with or without a positive family history of other psychiatric disorders; and (iii) a positive family history of a non-psychotic disorder, with no family history of psychosis. Because a structured instrument was not used to collect the data, and patients’ relatives were not formally assessed for evidence of psychiatric illness, we did not break down family history into specific psychiatric diagnoses.
To evaluate the convergent validity of our family history data, we compared clinician assessments of psychiatric family history with that obtained from a structured instrument, the FIGS [29]. Data were drawn from an independent sample of 46 FEP patients from EPPIC involved in a study on relapse prevention [30]. The FIGS was administered to all study participants, and clinician-collected family history was extracted from their medical records. There was substantial agreement between clinician and FIGS-rated family history of psychosis, with concordance of 87% and κ = 0.69 (95% confidence interval (CI) = 0.41–0.86). But agreement for non-psychotic family history was comparatively low, with 69.6% concordance and κ = 0.28 (95%CI:= − 0.03 to 0.52). Disagreement about the presence of a non-psychotic family history typically reflected positive histories identified by the FIGS but not clinicians. In 11 of 14 cases where there was discordance, there was a negative clinical rating but a positive FIGS rating. The agreement for the three-category measure of family history is therefore intermediate between these two values at κ = 0.54 (95%CI = 0.21–0.77).
Data analysis
Variables were summarized as means (with SD) and proportions (with SE) by family history both for the total sample and separately for each sex. The three family history subgroups were compared on psychiatric diagnosis, severity of illness and functioning (GAF and CGI), DP, DUP, SUD and all other comorbid disorders. For continuous data that did not conform to the normal Gaussian distribution, log10 transformations were used prior to analysis. Measures of functioning and illness severity at discharge from EPPIC were adjusted for GAF and CGI-S score at admission and time in EPPIC.
Comparisons of continuous dependent variables (GAF, CGI, DP, DUP) were made using one-way analysis of variance (ANOVA) or one-way analysis of covariance (ANCOVA). For categorical dependent variables, χ2 tests were used. ANOVAs that yielded a significant F-statistic were followed by a series of post-hoc comparisons using the Games–Howell test, which takes into consideration unequal sample sizes [31]. For significant χ2 tests, standardized residuals (z) were examined to determine which cells contributed to the significant χ2 [32] (only ps associated with the z-statistic are reported in the text). Two-tailed tests were used to judge the significance of statistical tests at α = 0.05. All analyses were conducted with Stata version 9.2 (StatCorp, College Station, TX, USA) and SPSS version 15.0 (SPSS, Chicago, IL, USA).
Results
Sample characteristics
The total FEPOS sample consisted of 661 patients. The eligible sample for this study consisted of 635 patients due to missing family history data on 26 patients. Of the eligible sample, 271 (27.7% female) had no family history of psychiatric disorder; 190 (40.5% female) had a family history of non-psychotic disorders only; and 174 (36.8% female) had a family history that included psychotic disorders. There was no association between family history and age at treatment entry (average 21 years) or duration of treatment (average 69 weeks). There was a significant association between gender and family history ( (9.04), p = 0.01), with male subjects less likely to have a family history of psychiatric illness and female subjects more likely to have family history of non-psychotic illness. Subsequent analyses were therefore conducted both for the total sample and stratified by gender.
Pre-treatment assessments
There was no association between pre-treatment GAF and family history, or DUP and family history, in the full sample or the male and female subsamples (Table 2). There was a significant difference in mean DP (log-transformed) between the family history subgroups. Examining the total sample (F(2,571) = 3.65; p = 0.03; η2=0.013), those with no family history of psychiatric disorders had a shorter mean DP than patients with a family history of non-psychotic psychiatric illness (p = 0.04). When stratifying by gender, a significant difference in mean DP was found only in male subjects (F(2,378) = 3.18; p = 0.04; η2=0.017). Among male subjects, the mean DP was significantly shorter in those with no family history (383.5 days) compared to those with a family history of psychotic psychiatric illness (481.4 days; p = 0.03).
Pre-treatment assessments, by family history type and gender
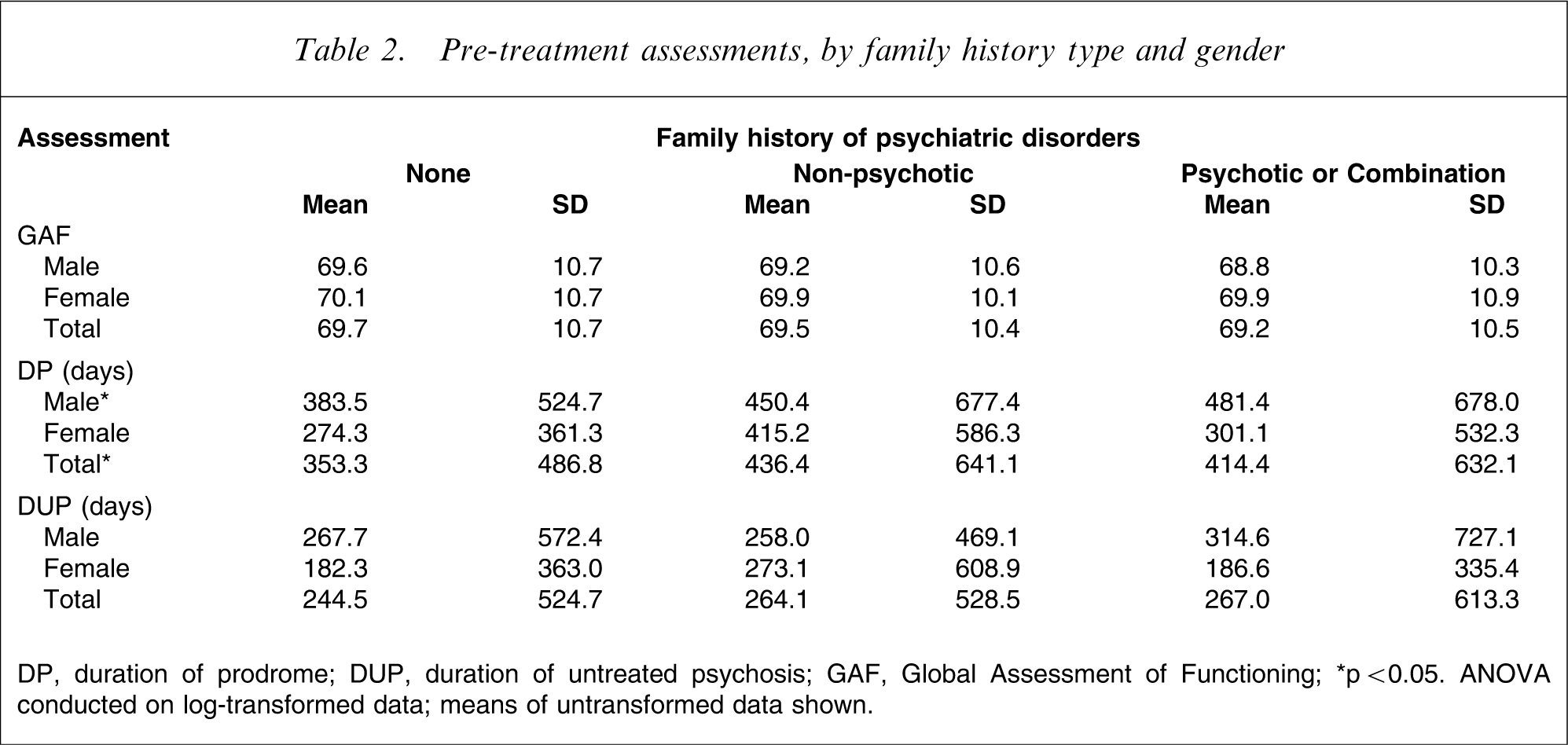
DP, duration of prodrome; DUP, duration of untreated psychosis; GAF, Global Assessment of Functioning. ∗p < 0.05. ANOVA conducted on log-transformed data; means of untransformed data shown.
First presentation assessments
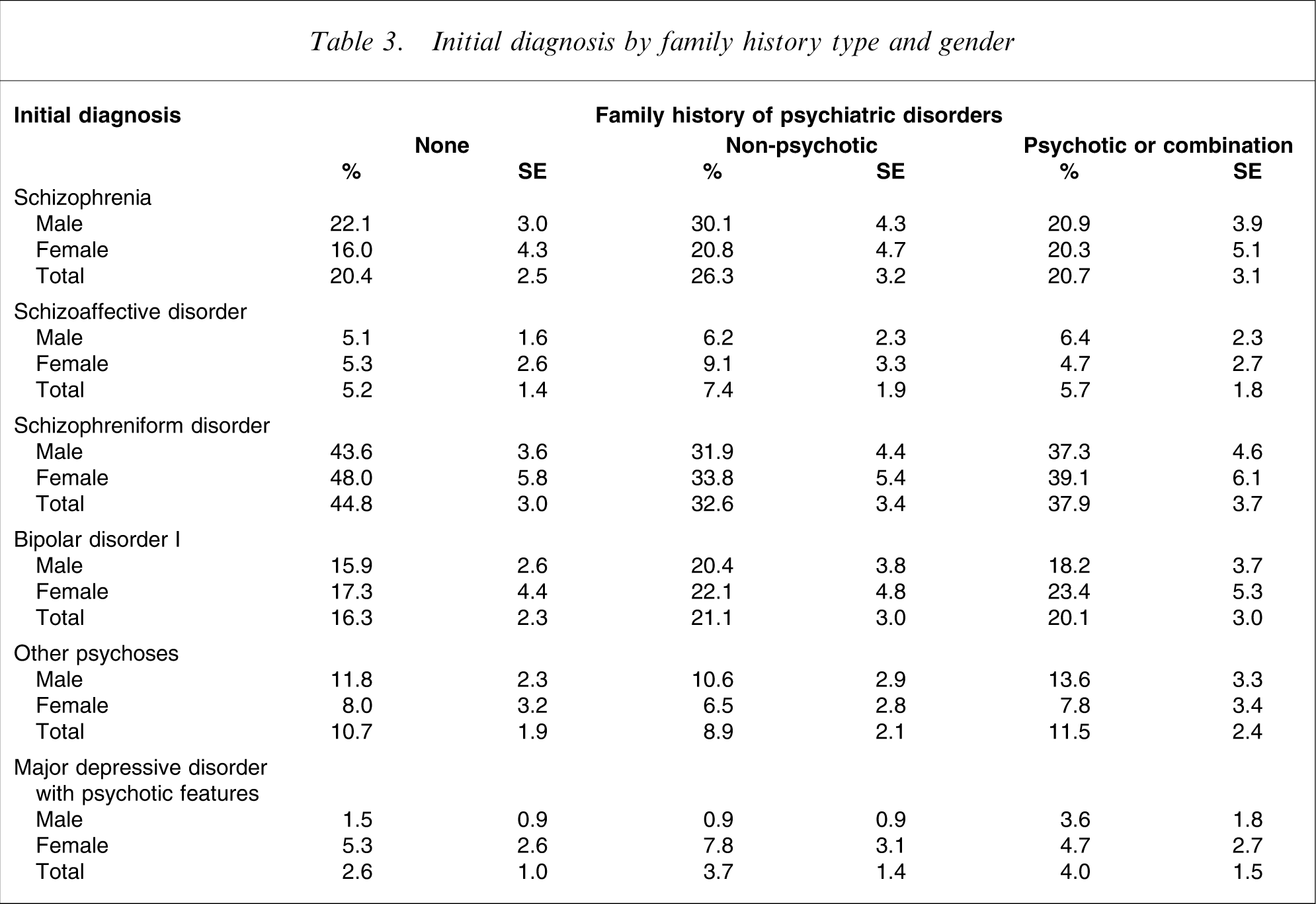
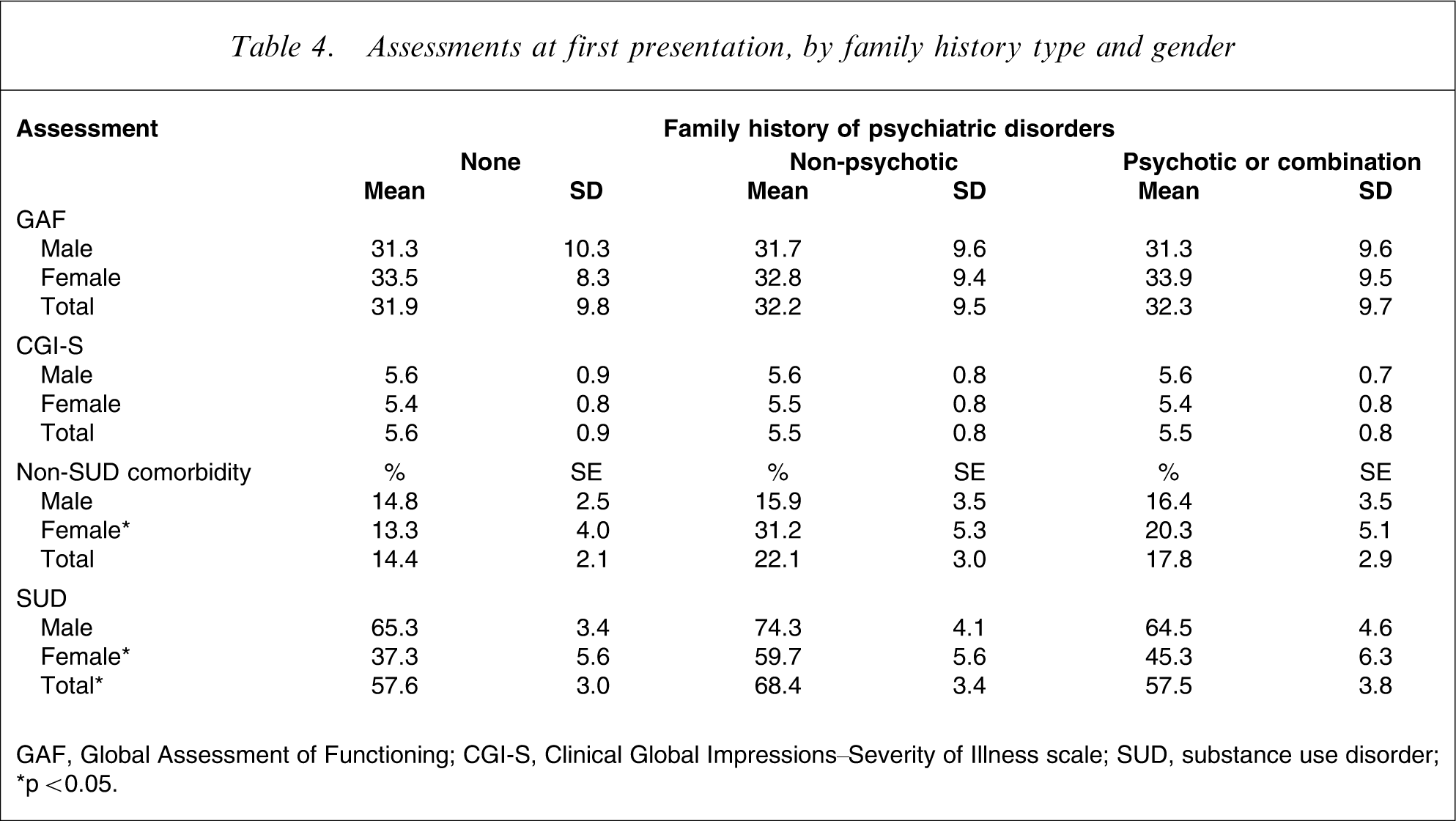
The association between family history and DSM-IV diagnosis, severity of illness (CGI-S), and functional status (GAF) was non-significant for the total sample and stratified by gender (Tables 3 and 4).
Initial diagnosis by family history type and gender
Assessments at first presentation, by family history type and gender
GAF, Global Assessment of Functioning; CGI-S, Clinical Global Impressions–Severity of Illness scale; SUD, substance use disorder;
∗p < 0.05.
At entry to EPPIC 17.6% of subjects had one or more comorbid psychiatric disorders (excluding substance abuse/dependency). Diagnoses included personality disorder (7.2%, n = 46), depression (6.8%, n = 43), eating disorders (2.4%, n = 15) and mental retardation (2.5%, n = 16). No association between family history and non-SUD comorbidity at treatment admission was observed in the full sample. After stratifying by gender there was a significant association between non-SUD comorbidity and family history in female patients ( (7.21), p = 0.03, n = 216). Those with no family history of psychiatric illness were less likely to have a comorbid disorder at entry, while those with a family history of non-psychotic illness were more likely to have a comorbid disorder. The number of patients with non-SUD comorbidities was too small to investigate associations with specific disorders.
Abuse of, and dependency upon licit and illicit substances (excluding tobacco) was common in this sample, with 60.8% of patients diagnosed with a substance use disorder (67.5%, male; 47.7%, female). SUD at first presentation was positively associated with a family history of non-psychotic disorders ( (6.63), p = 0.04, n = 635, Table 4). After stratifying by gender the association was significant only in female patients ( (7.85), p = 0.02, n = 216). The association between family history and SUD was primarily driven by cannabis abuse/dependence, which has a prevalence of 46.6% in the total sample (48%, male; 36.1%, female). Cannabis SUD was significantly associated with family history in the total sample ( (11.64), p = 0.003, n = 635), and in male and female patients ( (7.70), p = 0.02, n = 419; (7.96), p = 0.02, n = 216, respectively). This association was mainly due to a greater prevalence of cannabis SUD among patients with a family history of non-psychotic illness (56.8%) compared to those with no family history (43.2%) and those with a family history of psychotic illness (40.8%).
Outcome assessments
Family history was not significantly associated with severity of illness (CGI-S), functional status (GAF) (Table 5), or diagnosis at outcome (data not shown).
Outcome assessments, by family history type and gender
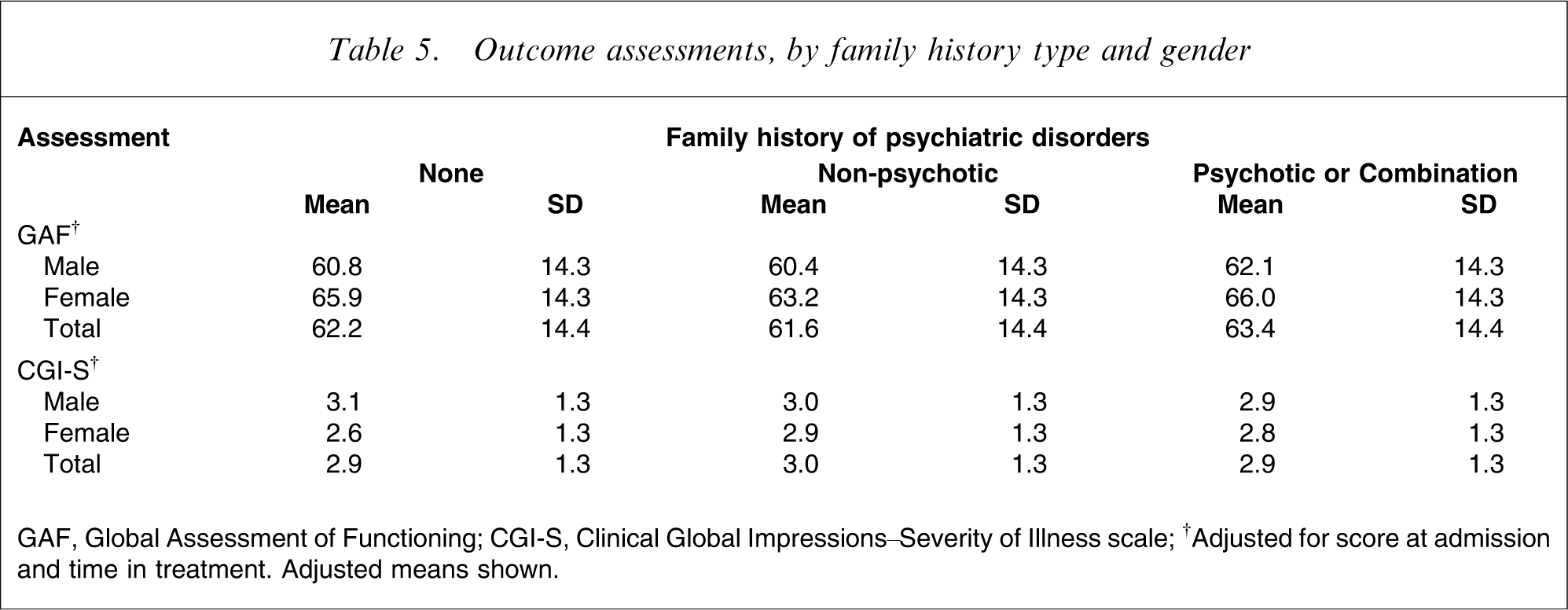
GAF, Global Assessment of Functioning; CGI-S, Clinical Global Impressions–Severity of Illness scale; †Adjusted for score at admission and time in treatment. Adjusted means shown.
Discussion
This study investigated the clinical correlates of positive family history for psychotic and non-psychotic disorders in a large unselected and representative FEP cohort. We replicated some, but not all, of the associations that have previously been found between family history and clinical measures in other FEP samples. In the FEPOS sample there was no association between family history and measures of functioning and severity of illness at any time point (before treatment, at first presentation, or at discharge). This contrasts with the results of Jarbin et al. [12] and Conus et al. [13], who found that family history of psychiatric disorders was related to patient recovery.
Our findings also contribute to the existing research on lag time to identification of psychotic illness. Like Norman et al. [6], but in contrast to Chen et al. [4], we did not find an association between family history and DUP. But Norman et al. did find an association with duration of untreated illness (DUI), which they defined as time from onset of any psychiatric symptoms to treatment [6]. We identified an association between family history and DP. DP differs from DUI in that DP is concerned only with time until onset of psychotic symptoms, not treatment. We found that among male patients DP was longest in those with a family history of psychotic illness, which is congruent with the finding of Norman et al. [6], that those with positive family history had the longest DUI.
Norman et al. suggest that the difference in time to treatment between those with and without a family history of psychotic disorder may not be due to family experience with psychotic illness, but to some underlying biological factor affecting the manifestation of psychosis [6]. The present findings lend some support to this hypothesis because family history differentiated patients only on time to onset of psychotic symptoms; no difference was found in time to treatment once psychotic symptoms were displayed. But further research is needed before any strong conclusions can be drawn in this area, especially because the definition of prodrome and other time-to-illness measures is a research focus in and of itself [33].
Women with a family history of non-psychotic disorders were more likely to have a non-SUD related comorbid condition at first presentation. Women with a family history of non-psychotic disorders were also significantly more likely to abuse, or to be dependent on cannabis at first presentation. We could not locate any previous reports investigating comorbid disorders and family history in an unselected FEP sample, although two previous studies of patients experiencing their first episode of schizophrenia or schizoaffective disorder did not find any association between family history and substance use [8], [9].
The gender differences we observed in the associations between family history and measures of DP and comorbidities (both SUD and non-SUD related) provides additional support for the hypothesis, suggested by recent research [34], that psychotic illness may manifest differently in male and female subjects. Gender may thus represent an important, and simple, tool for assisting clinical diagnosis and determination of treatment in FEP patients.
Additionally, the present finding that cannabis SUD may be related to a family history of non-psychotic disorders, and potentially to genetic risk factors, warrants further investigation. The prevalence of cannabis abuse/dependence in this sample is substantial compared to the general population (46.6%, total sample; 48%, male; 36.1%, female), but comparable to that found in other samples of psychosis patients [35]. The association between cannabis use and psychosis is well documented [36], and research suggests that genetic factors may interact with cannabis use to precipitate the development of psychosis [37], [38]. Family history of psychiatric illness may serve as a proxy for genetic risks that can be easily collected by clinicians and incorporated into decisions about diagnosis and treatment.
This study is limited by the fact that the data used were taken from a file audit sample. Data from this type of study can have problems with data quality, reliability, and validity. But as detailed here and elsewhere [18], [39], every effort was made to investigate the validity and reliability of the measures collected. There was poor agreement between clinician and FIGS ratings of non-psychotic family histories. The pattern of disagreement suggests that clinicians fail to identify many positive family histories of non-psychotic disorders. This result is not unexpected given that patients and their families are probably more likely to recognize and report psychotic than non-psychotic illness in family members, and that clinicians may focus more on eliciting information about relatives with psychotic illness when assessing patients with psychosis. We recommend that FEP services use structured instruments to assess psychiatric family histories. Although family history of psychosis is an important risk factor for FEP patients, results from the FEPOS suggest that family history of non-psychotic disorders may also predict important patient characteristics, such as comorbidity, which have important consequences for long-term outcomes in these patients.
The present findings regarding the validity of the family history data have implications for the associations we identified with psychiatric comorbidity. In this sample, associations between family history and comorbidity were primarily related to the prevalence of SUD and non-SUD conditions in patients with family histories of non-psychotic illness. Given that reports of family history of non-psychotic illness in this sample are less reliable than reports of psychotic illness, these results require replication in an independent sample. We present them here to enable comparison with other studies that have differentiated between family history of psychotic versus non-psychotic illness.
Overall, the present analysis of family history data and clinical measures from a large, epidemiologically representative sample of FEP suggests that the relationship between family history of psychiatric illness and clinical characteristics, such as time to symptom onset and comorbidity, merit further investigation. The present research also indicates that knowledge of patient family history may also be useful in treatment determination for FEP patients. This finding suggests a role for genetic information in FEP diagnosis and treatment, and provides support for molecular genetic investigations of FEP.
Footnotes
Acknowledgements
The authors thank the patients for their participation in the study, and Roy McKenzie for his assistance with patient medical records. KIM is supported by the Clinical Neurosciences Platform funded by Neurosciences Victoria and an NHMRC Public Health Fellowship (520452). SLM and DLF are supported by Colonial Foundation. The original FEPOS data collection was supported by Eli Lilly Company Australia for the purpose of comparing olanzapine and risperidone treatment in FEP (to ML).
