Abstract
Attention-deficit hyperactivity disorder (ADHD) is the most common neuropsychiatric disorder of children, affecting approximately 5% of the school-age population [1]. It is characterized by developmentally inappropriate deficits in attentional performance, impulsivity, and motor restlessness or hyperactivity [2]. ADHD typically starts in early childhood and is associated with progressive functional impairment, including school dysfunction, problems with peer interaction, family conflict, poor occupational performance, injuries, antisocial behaviour, traffic violations, and accidents [1]. Prospective longitudinal studies indicate that up to 50% of subjects with ADHD continue to experience impairment from some symptoms of the disorder, if not the full syndrome, in adolescence and early adulthood [3]
Health-related quality of life (HRQL) is a multidimensional construct defined as the patient's ‘subjective perception of the impact of health status, including disease and treatment, on physical, psychological and social functioning’ [4, 5]. Instruments that assess HRQL can complement efficacy measures in clinical trials to provide a more thorough picture of the impact of disease and treatment on children's overall well-being [6]. HRQL instruments may be particularly relevant to research on treatment of ADHD because this disorder has a range of psychosocial outcomes that are not captured by clinical symptoms only. Western literature has demonstrated HRQL in clinic-based sample of ADHD children to be poorer than in the control population [7, 8]. ADHD has been found to be associated with marked decrements in quality of life, which improve when the disorder is treated [9]. Also the symptom severity and presence of comorbid psychiatric diagnosis are reported to be correlated with the parent report HRQL [10]. However, little information has been reported regarding the HRQL and associated clinical features in Asian ADHD children.
The present study used the Child Health Questionnaire 50-item Parent Form (CHQ-PF50) to evaluate the effectiveness of methylphenidate treatment by comparing treated Taiwanese ADHD children with healthy controls in the community. CHQ-PF50 is one of the HRQL instruments that may provide an estimate of the broader range of children's day-to-day functioning in multiple settings [11], and it has been proved to be a reliable and valid clinical instrument for assessing HRQL outcomes associated with treatments for ADHD [12]. In addition we collected various treatment-related and disease-related variables to analyse their relationship and predictive power with ADHD children's HRQL outcome.
Methods
Subjects
The ADHD subjects were 6–15-year-old children recruited from the outpatient service of the Department of Psychiatry of Kaohsiung Medical University–Chung-Ho Memorial Hospital in Taiwan. To enter the study, children were required to meet the following criteria: (i) diagnosis of ADHD on the basis of DSM-IV-TR criteria; (ii) currently receiving methylphenidate as the main treatment and did not take part in any concurrent behavioural programmes during the evaluation period; (iii) living at home with family and not in foster care or institution; and (iv) having a primary caregiver with at least 9 years of formal education so that the parental questionnaire could be successfully administered. The diagnosis of ADHD was made through the application of multiple data sources including (i) clinician interview; (ii) clinic observation of the child's behaviour; (iii) Werry–Weiss–Peters Activity Scale completed by parents and the Conners–Werry–Quay Activity Scale completed by the teacher. These two scales were adapted from Conners’ Activity Rating Scale [13] and have been applied extensively in clinical use in Taiwan [14]. Of the total 380 ADHD children referred to the clinic during the 12 month recruitment period (January–December 2006), 130 were eligible for entry into the study. Questionnaires were delivered to the parents of these 130 children and 119 were completed. Clinical information for each child was extracted from hospital notes for detailed characterization and they included (i) disease-related variables (e.g. ADHD subtype, age of diagnosis, items of core symptoms, intelligence quotients, comorbidity pattern; and (ii) treatment-related variables (e.g. age starting medication, duration of pharmacological treatment, treatment response as rated by a clinician).
Comorbidities of ADHD were recorded as the following four categories for statistical analysis: (i) externalizing disorder, which included oppositional defiant disorder and conduct disorder; (ii) internalizing disorder, which included mood disorder and anxiety disorder; (iii) neurological disorder, which included chronic tic disorder, Tourette's disorder, developmental language disorder and epilepsy; and (iv) school failure, which was defined as failure by a child, who was found to be normal according to the Wechsler Intelligence Scale for Children [15], in a school exam (in either Chinese, arithmetic or science). ‘Learning disabilities’ was not able to be diagnosed because standardized learning achievement tests were not available for every child.
The control group consisted of healthy children recruited from the community according to the process described here. Through written requests and mail-back questionnaires, we invited the willing parents of children in the same age range to participate. Parents were asked to fill out both the Child/Adolescent Psychiatry Screen (CAPS) and CHQ-PF50. CAPS is a preliminary screening tool for immediate and current psychiatric symptoms [16]. When the primary caregiver reported the child to have problematic symptom items in CAPS, thorough psychiatric assessment was done by trained clinicians as the next-step evaluation. In the present study efforts were made to recruit children who were reported by parents to be free of any psychiatric symptoms as screened by CAPS. However, DSM-based diagnostic interview was not performed on this group of community children. With a response rate of 90%, the total number of children screened to be free of psychiatric symptoms was 198 (95 boys, 103 girls). The final control group consisted of all the boys and 34 girls. Girls were downsized to make the gender ratio comparable between case and control groups. The process of female downsizing was done by random selection by a researcher blinded to the data. This study was approved by the Institute Review Board of the Kaohsiung Medical University Hospital. Informed consent was obtained from parents according to the guidelines of the Institutional Committee on Clinical Investigation. Final study subjects were 119 children with ADHD (96 boys, 23 girls, average age = 9 years 7 months±1 year 10 months) and 129 healthy control children (95 boys, 34 girls, average age = 10 years 1 month±2 years 3 months). There were no differences in age (p = 0.1) and gender (p = 0.23) between the ADHD and control group.
Measure
Child Health Questionnaire 50-item Parent Form
This instrument is a broad-based health outcomes measure designed to assess the physical and psychosocial well-being of children aged 5–18 years and was given to the parents to complete [11]. It contains 50 items measuring 12 domains of physical and emotional health: Physical Functioning, Behaviour, Self-Esteem, Role Limitations–Emotional/Behavioural, Role Limitations–Physical, Bodily Pain, Mental Health, General Health Perceptions, Parent Impact–Emotional, Parent Impact–Time, Family Activities, and Family Cohesion. Two summary scores, the Psychosocial Summary Scale and the Physical Summary Scale, are derived from weighted combinations of domain subscale scores. The 12 subscales are transformed to a range of 0–100, with 0 indicating worse health and 100 indicating better HRQL. The CHQ meets the criteria required for quality of life/functional health status instruments, with high indices of scale reliability and validity [17, 18]. This instrument has been psychometrically validated to be responsive to change in clinical status in ADHD children [12]. The Chinese version of CHQ-PF50 used was obtained from the publisher with appropriate standards met in the translation. The Psychosocial Summary Score provides an estimate of the children's overall psychosocial functioning across multiple contexts and is the main index used in statistical analysis. Subscales that contribute to this summary score include Behaviour, Mental Health, Self-Esteem, Role–Emotional/behavioural, and the two Parental Impact scales. Cronbach's alpha reliability coefficient for the CHQ-PF50 used in the present study was 0.86.
Clinical Global Impressions-ADHD–Improvement
The clinical global impressions-ADHD-improvement (CGI-I) is a single-item clinician rating of change in ADHD symptoms after treatment. A clinician rated each child's overall clinical improvement on ADHD core symptoms on 7-point Likert-style scales (7 = very much worsened, 1 = very much improved). Targeted symptoms being rated were the core ADHD symptoms listed in DSM-IV criteria (i.e. ‘hyperactivity’, ‘impulsivity’ and ‘inattentiveness’). The clinician rated the degree of change regardless of whether s/he believed that symptom change was due entirely to treatment drug. The change of scores as reflected in the Werry–Weiss–Peters Activity Scale (parent version) and the Conners–Werry–Quay Activity Scale (teacher version) provided information for the clinician to make a judgement on the CGI-I. According to CGI-I score, ADHD children were further divided into three groups for statistical analysis: no improvement (Likert scale score 3–7); somewhat improved (Likert scale score 2) and much improved (Likert scale score 1).
Analysis
CHQ PF-50 data from the ADHD group were first compared with data from the age- and gender-matched healthy children using independent t-tests. Effect sizes were calculated as the means of the control sample minus the means of the ADHD sample divided by the standard deviation of the control sample [19]. The effect size of 0.2 is considered small, 0.5 moderate and 0.8 large [20]. ADHD subjects grouped by different improvement status after methylphenidate treatment were compared on the Psychosocial Summary Scores of CHQ. Given that previous Western studies have demonstrated the link between clinical symptoms, comorbidity and HRQL among children with ADHD [9, 10], multiple regression using three different models was performed to investigate whether the clinician's judgement on ADHD core symptoms improvement predicted Psychosocial Summary scores of CHQ. Model 1 tested CGI-I as the independent variable alone. Model 2 tested CGI-I adjusted with children's current age, age at diagnosis, age starting and duration of methylphenidate treatment as covariants. Model 3 added ADHD comorbidity number (by type) as another covariant to be adjusted for investigating the power of CGI-I in predicting Psychosocial Summary scores of CHQ.
Results
Subjects
A total of 80.7% of the ADHD children were male, and the combined subtype was the most frequent presentation of the ADHD subtypes (54.6%). The average age to receive the initial ADHD diagnosis was 7 years 9 months, while the average age for starting methylphenidate treatment was 8 years 2 months. At the time the present survey was undertaken, the majority of children (69.7%) had received methylphenidate continuously for more than 6 months. The stimulant treatment response rate, defined as a CGI-I score of 1 or 2, was 68%. ADHD children were on average of normal intelligence with mean full-scale IQs of 94.69±15.57. A total of 27.7% of the ADHD children had comorbid externalizing disorders; 18.5% of the ADHD children had comorbid internalizing disorder; 26.9% of the ADHD children had comorbid neurological disorder, and 50.4% of the ADHD children had failures in school exams. Clinical data for the ADHD group are presented in Table 1.
Demographic and clinical characteristics of the children with ADHD (n = 119)
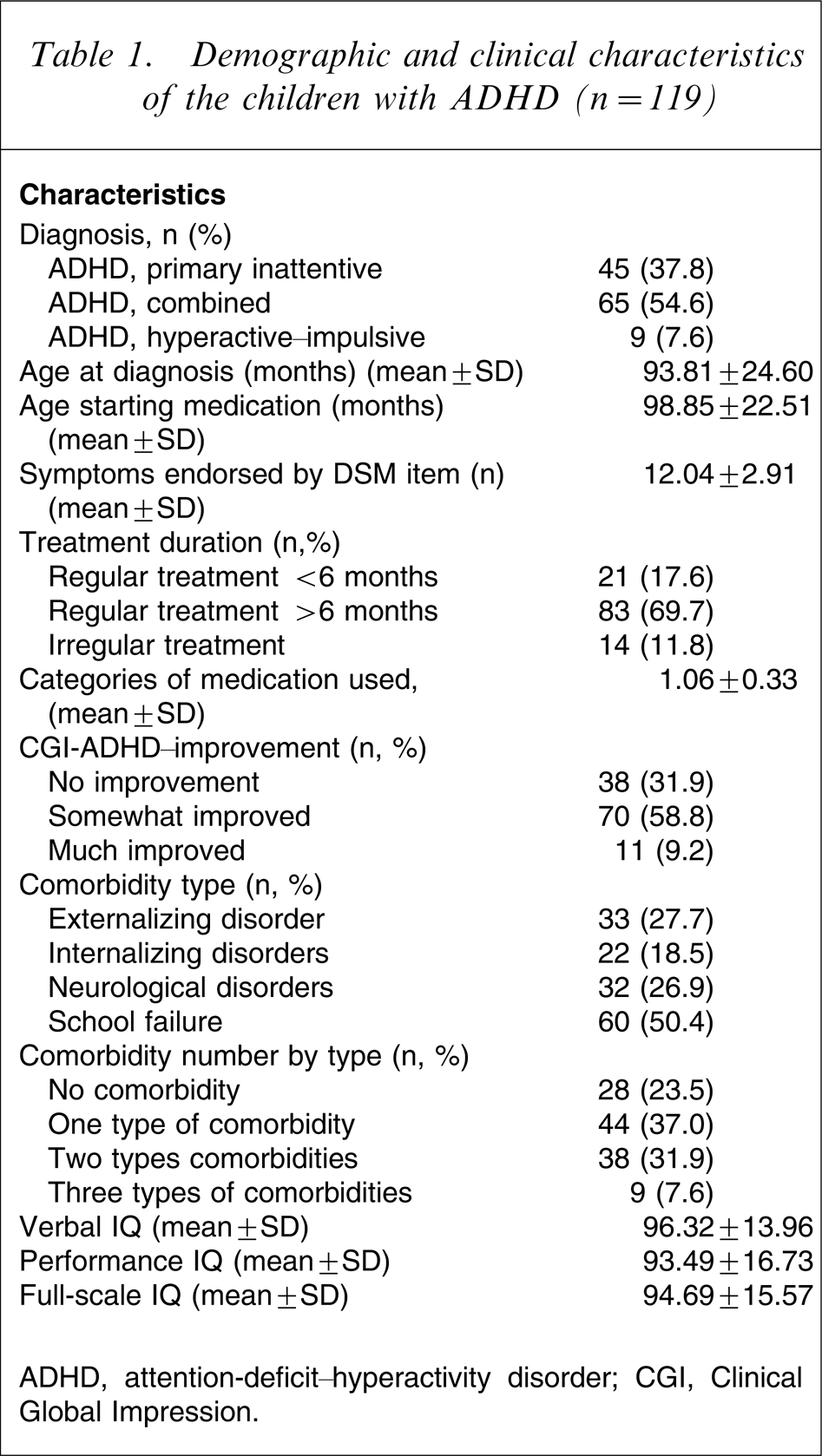
ADHD, attention-deficit–hyperactivity disorder; CGI, Clinical Global Impression.
HRQL of ADHD children compared with healthy community children
Parents reported ADHD children's HRQL to be significantly poorer than the healthy control group on all of the CHQ-PF50 psychosocial subscales (i.e. subscales of Behaviour, Mental Health, Self-Esteem, Role–Emotional/behavioural, two Parental Impact scales) and the Psychosocial Summary Score. In addition, the problems of ADHD children interfered with family activities and family cohesion significantly. A detailed presentation of the comparison between ADHD children and the healthy control group on CHQ-PF-50 is presented in Table 2.
CHQ-PF-50 scale and summary scores: ADHD and control samples
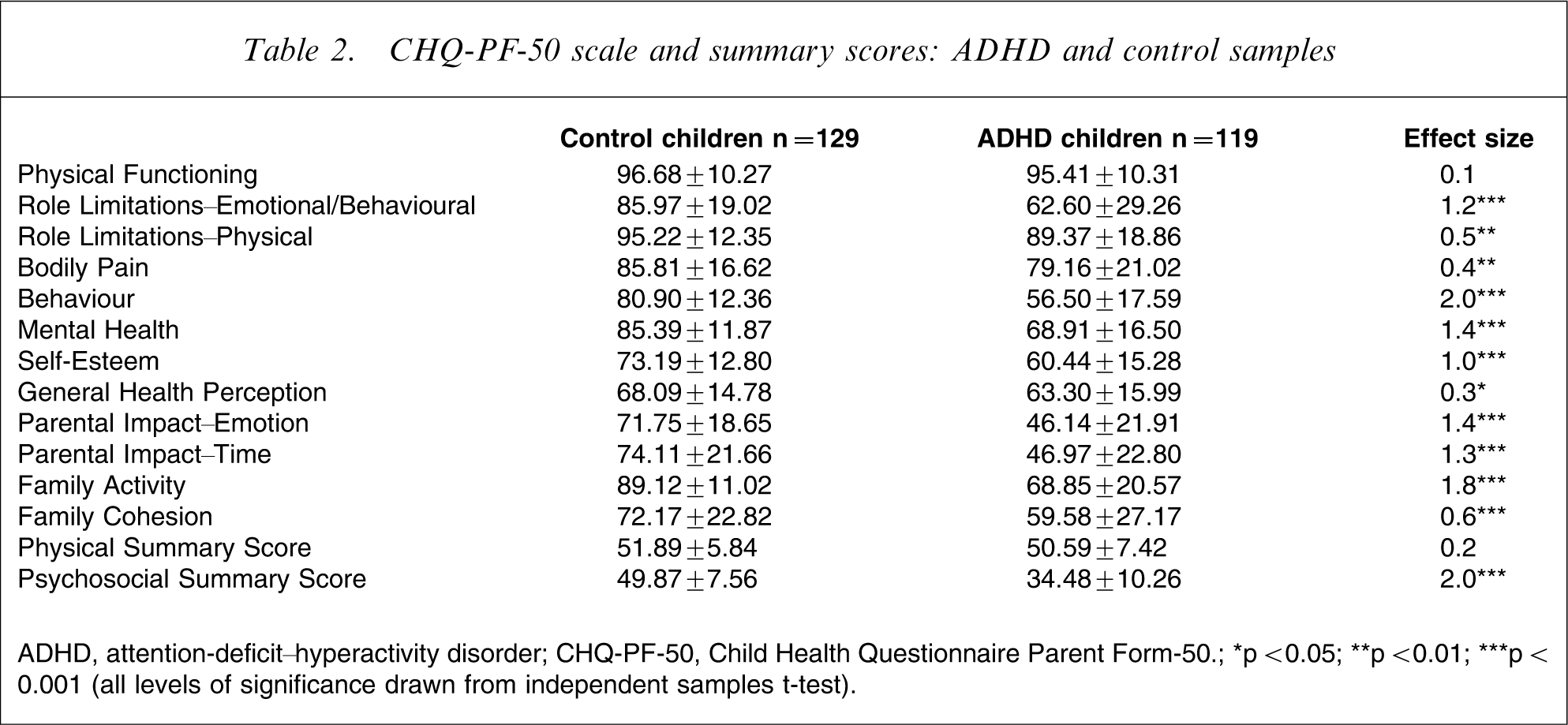
ADHD, attention-deficit–hyperactivity disorder; CHQ-PF-50, Child Health Questionnaire Parent Form-50; ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001 (all levels of significance drawn from independent samples t-test).
Predictors of HRQL
Analysis showed that Psychosocial Summary Score increased with the improvement of clinical symptoms after methylphenidate treatment. The effect size was 0.7 (p = 0.02) for the much improved group compared with the somewhat improved group; and was 0.9 (p = 0.008) for the much improved group compared with the no improvement group. Multiple regression using three different models was performed to investigate how the ADHD core symptoms improvement might predict Psychosocial Summary scores of CHQ. The results are shown in Table 3. Degree of ADHD core symptom improvement was positively related to Psychosocial Summary score of CHQ-PF50 as shown in model 1 (β1 = − 7.4, p1 = 0.025; β2 = − 9.3, p2 = 0.008). This effect was independent of children's age, age at ADHD diagnosis, age starting methylphenidate treatment and duration of treatment as shown in model 2 (β1 = − 7.6, p1 = 0.023; β2 = − 10.6, p2 = 0.005). However, if we added comorbidity number as one of the covariants to be adjusted in the multiple regression, the predictive power of CGI-I lost its statistical significance, as shown in model 3 (β1 = − 4.9, p1 = 0.16; β2 = − 7.2, p2 = 0.072). This implied that ADHD comorbidities have major influence on the relationship between improvement of ADHD core symptom and psychosocial domains of HRQL.
Clinician's rating on ADHD core symptom improvement predicting Psychosocial Summary score of HRQL

ADHD, attention-deficit–hyperactivity disorder; CGI-I, Clinical Global Impression–ADHD–Improvement; HRQL, health-related quality of life; †Adjusted for age, age at diagnosis, age starting treatment, duration of treatment; ‡Adjusted for age, age at diagnosis, age starting treatment, duration of treatment and comorbidity number.
Discussion
Symptoms and consequences of ADHD have a broad impact on individuals and family members, and reports on treatment outcome for ADHD used to focus primarily on symptom-relief efficacy and medication safety. Study after study in the developed countries have demonstrated that stimulants are effective in reducing the ADHD core symptoms in 65–75% of individuals with this disorder [21]. The present stimulant treatment response rate for clinical symptoms improvement was 68%, which is consistent with the Western data. In addition, the present study reported the extent of ADHD core symptom improvement to be positively associated with the Psychosocial Summary scores of CHQ-PF50, which was independent of children's current age, age receiving ADHD diagnosis, age starting methylphenidate treatment and duration of treatment. The Psychosocial Summary score incorporates a range of outcomes including behaviour in school, peer relations, family functioning, and psychological well-being. Hence the present results are consistent with previous Western reports indicating that improvements in ADHD symptoms have an impact on children's functioning across contexts [9]. The importance of medication was highlighted given that other clinical factors (such as age) had little impact on the relationship between symptom reduction and function across context as reported by our results.
The disease model of medicine has traditionally assumed that elimination of the symptoms of disease is in itself identical to functional remission. Nevertheless, studies have shown that symptoms and functioning are overlapping but not identical constructs in ADHD. Klassen et al. studied HRQL in 165 Canadian ADHD children using CHQ-PF50. Their results showed that children with ADHD, as compared with population samples, had comparable physical health but clinically important deficits in all psychosocial domains, family activities, family cohesion and psychosocial summary scores [10]. They also reported that children with two or more comorbid disorders differed significantly from those with no comorbidity in most areas. The present results for medication-treated Asian ADHD children having worse HRQL as compared with control children are in agreement with this Western report.
Many studies have already documented a significant rate of comorbid psychiatric conditions such as mood, anxiety, and disruptive behaviour disorders in children diagnosed with ADHD [22]. Most paediatric ADHD studies have found comorbidity rates of 50–90%, with ADHD typically presenting before other disorders manifest [23]. There is a complex interplay between ADHD and its comorbid psychiatric disorders because these comorbid conditions can affect the presentation, diagnosis, and treatment of ADHD, which further complicates the clinical presentations and developmental outcome [24]. The present results show that ADHD comorbidities had a major influence on the relationship between improvement of ADHD core symptom and the Psychosocial Summary Score of HRQL, which support the idea that ADHD comorbidities might influence severity, daily functioning, treatment and prognosis of ADHD [25]. Clinicians should be aware of the issue of comorbidity and its complexity in evaluating children of ADHD, and apply useful guidelines, such as the Texas Children's Medication Algorithm [26], for managing ADHD in the presence of comorbid conditions. Ideally, ADHD should be treated before other comorbid disorders develop, because ADHD is most responsive to treatment at this stage. Recent studies indicate a growing appreciation of the importance of early intervention, which might prevent the development of future comorbid disorders [24, 27–29].
The present study should be viewed in the light of certain limitations. First, the HRQL measure was based on parent report (CHQ-PF-50). The decision of whether to question children directly or to use parents as proxy responders is currently a focus of debate in paediatric research. One argument is that children of a younger age may not be able to provide reliable information on complex or abstract health-related constructs [30]. The average age of participants in the present study was 9 years 10 months, which made parent report a more appropriate choice for eliciting responses regarding abstract domains, such as emotional impact of illness. In addition, it has been demonstrated that parent and child ratings on HRQL have moderate to good concordance in children diagnosed with ADHD [31]. Second, the Psychosocial Summary Score used as the main index in the present study is contributed to by subscales of Behaviour, Mental Health, Self-Esteem, Role–Emotional/behavioural, and the two Parental Impact scales. Because the questions listed in the subscales of Behaviour and Mental Health assess the presence of mental health problems the same way that a childhood behaviour checklist does, lower Psychosocial Summary Score for the ADHD group may simply mean that the ADHD children have more mental health problem than the control children. However, comparison between the ADHD and control children on subscales scores for Self-Esteem, Role function and Parental Impact indicated significant differences. Also the problems of ADHD children were found to interfere with family activities and family cohesion significantly more than those in the control group. Hence we might say that the quality of life between these two groups of children really differed. Third, we did not make a systematic evaluation of parental psychopathology. Medication compliance was checked only by oral questioning of the parents. The history of ADHD in parents is known to affect parent–child interaction and all of the family members, hence the impact of parental psychopathology and medication compliance on the quality of life of the present subjects remains unclear. Also the present sample was drawn from a single institution and its community, so results may not be generalized to patients with ADHD followed by other centers. Finally, we used a single-item scale, CGI-I, to determine changes of ADHD core symptoms after methylphenidate treatment. Using a single-item measure might be considered to have inherent difficulty in establishing reliability according to some critics. However, CGI-I has been used extensively in psychopharmacology research [32] and has been regarded as acceptable given that other instruments were administered along with the clinician's judgement. In the present study the information provided by the Werry–Weiss–Peters Activity Scale (parent version) and the Conners–Werry–Quay Activity Scale (teacher version) contributed to the clinician-rated CGI-I. Hence, we think that it was acceptable to use CGI-I as an outcome efficacy measure.
Despite these limitations the present findings provide strong support for the notion that methylphenidate-treated ADHD children still have multiple problems with regard to broader health outcomes as represented by the concept of HRQL. Quality of life is an issue in Asian families with ADHD, as it is in Western cultures. It will be important to include scales measuring attributes other than clinical symptoms in trying to evaluate the effectiveness of ADHD treatment. Improvement of HRQL should be integrated in the overall treatment plan for children with ADHD, as previously advocated in many Western reports [8, 33].
Footnotes
Acknowledgements
This study was supported by a grant from the Kaohsiung Medical University Hospital 94-KMUH-ND-009. The authors thank the Statistical Analysis Laboratory, Department of Clinical Research, Kaohsiung Medical University Chung-Ho Memorial Hospital for its assistance.
