Abstract
Substance abuse is highly prevalent in schizophrenia and associated with numerous negative consequences. While studies have regularly reported more severe depressive symptoms in addicted schizophrenia patients relative to non-abusing patients, some studies have not corroborated this finding. The current meta-analysis was performed to quantify the relative severity of depressive symptoms in dual-diagnosis schizophrenia. A search of the literature using computerized engines was undertaken. Studies were retained in the analysis if (i) they assessed depressive symptoms using validated scales specific to depression (e.g. Hamilton Depression Rating Scale); and (ii) groups of schizophrenia patients were divided according to substance use disorders (alcohol, amphetamines, cannabis, cocaine, hallucinogens, heroin and/or phencyclidine). According to the inclusion criteria, 20 studies were available for mathematical analysis. A small, positive and significant effect size estimate (n =3283; 1680 dual diagnosis; 1603 single diagnosis; adjusted Hedges's g =0.292; p =0.003) was obtained, within a random-effect model, suggesting that some dual-diagnosis patients experience more severe depressive symptoms than single-diagnosis patients. This significant difference was found only for studies using the Hamilton Depression Rating Scale but not for other depression scales. The results of the present meta-analysis suggest that addicted schizophrenia patients experience more severe depressive symptoms compared to non-abusing patients, but that the difference is smaller than commonly assumed. The meta-analysis also shows that the significance of results is related to the scale used to measure depressive symptoms. These results have methodological implications for future studies of depressive symptoms in dual-diagnosis patients, and potential implications for the prevention and treatment of depressive symptoms in schizophrenia.
The lifetime prevalence of substance use disorders (SUD) in schizophrenia is close to 50% [1, 2]. Substance abuse in schizophrenia is associated with numerous negative consequences, including psychotic relapses criminality, homelessness, unemployment, treatment non-compliance, and health problems [3, 4]. Among the consequences of substance abuse in schizophrenia, depressive symptoms have been regularly reported [5–8]. This observation is on par with an extensive literature showing that depressive symptoms are common features associated with long-term use of psychoactive substances (PAS) in non-psychosis patients, whether it is alcohol, cannabis, cocaine or heroin [9–12]. However, some studies have not corroborated the observation of an increased severity of depressive symptoms in dual-diagnosis schizophrenia (DD) [13, 14]. Thus the relative severity of depressive symptoms in DD, compared to non-abusing schizophrenia patients (SCZ), remains to be determined.
Despite variability of results among studies, the prevalence of depression during the longitudinal course of schizophrenia is at least 25% [15, 16]. Depressive symptoms in schizophrenia are of clinical concern because these symptoms are linked to substantial morbidity, including suicide attempts [17, 18]. Suicidal thoughts and attempts are frequent in schizophrenia, and suicide accounts for approximately 10–20% of patient deaths in schizophrenia [19, 20]. In this context, the identification of factors contributing to depression in schizophrenia may have implications for the prevention and treatment of these symptoms.
The objectives of this meta-analysis were to (i) verify that addicted schizophrenia patients suffer from more severe depressive symptoms, relative to non-abusing patients; (ii) to quantify the relative severity of depressive symptoms in DD; and (iii) to identify factors contributing to heterogeneity of results among studies published so far.
Methods
Searches
The current search is an update of a previous search of computerized literature databases (PubMed and PsycInfo) [21], using the following keywords: ‘schizophrenia’, ‘alcohol’, ‘amphetamine’, ‘cannabis’, ‘cocaine’, ‘hallucinogens’, ‘heroin’, ‘marijuana’, and ‘phencyclidine’. Studies were also identified by cross-referencing of included studies. A consensus was reached between authors on the studies retained or discarded, based on the following inclusion and exclusion criteria.
Study selection
Inclusion criteria were: (i) patients with a schizophrenia spectrum disorder: schizophrenia, schizoaffective disorder, and schizophreniform disorder; (ii) schizophrenia patients with and without a comorbid SUD (abuse or dependence; current or lifetime); (iii) PAS: alcohol, amphetamines, cannabis, cocaine, hallucinogens, heroin, or phencyclidine (PCP); (iv) depressive symptoms measured with validated scales specific to depression (e.g. Hamilton Depression Rating Scale, HDRS [22]; Calgary Depression Scale for Schizophrenia, CDSS [23]; Beck Depression Inventory, BDI [24]); and (v) studies published or available on MedLine before September 2006.
Exclusion criteria were (i) affective (bipolar/unipolar) psychotic disorder and/or toxic psychoses; (ii) schizophrenia with tobacco or benzodiazepine addition; and (iii) depressive symptoms measured with psychiatric interviews or scales not specific to depression (e.g. Positive and Negative Syndrome Scale, PANSS [25]; Brief Psychiatric Rating Scale, BPRS [26]).
Data extraction and quantitative data synthesis
Two reviewers (SP & AAS) independently extracted data; disagreements were resolved by discussion. Using Comprehensive Meta-Analysis (CMA) [27], effect size estimates of the differences in depressive symptoms between DD and SCZ patients were calculated. Effect size estimates were calculated from means and standard deviation (depressive total score) for each group of subjects: schizophrenia patients with and without SUD. Within a random effect model, effect size estimates were derived using Hedges's g [28], which provides effect sizes adjusted for sample size. Random-effect models, being more stringent than fixed-effect models, allow population-level inferences [29]. The direction of the effect size was positive if DD patients had a higher score on the retained depression scales (a higher score means more severe depressive symptoms) than SCZ patients. To control for age and symptoms, an independent two-sample T test was performed. For sex, a χ2 test was applied.
Results
Study characteristics
The resulting studies (n = 1348) were screened, 1324 were discarded based on the evaluation of the abstract or the article (for more details, see [21]). Twenty-five articles corresponded to the search criteria. Out of the yielded 25 articles, scores for depressive symptoms were incomplete or unreported for three studies (after contacting authors for missing data) [30–32]. Moreover, one study was excluded because it involved affective and non-affective psychosis patients [33]. In addition, the Scheller-Gilkey et al. (2004) study [34] involved patients already tested in Scheller-Gilkey et al. (2003) [35], the Margolese et al. (2006) study [36] involved patients from Margolese et al. (2004) [37], and the Van Mastrigt et al. (2004) study [38] involved patients from Addington and Addington (2007) [39]. After exclusion of the aforementioned studies 18 articles were available for meta-analysis. Noteworthy, the Addington and Addington article counted as two studies because cannabis and alcohol abusers were separately compared to healthy controls [39]. Thus, 19 studies were available for meta-analysis. Last, we have added to this group of studies an unpublished study from our group responding to the inclusion criteria of the current meta-analysis [Potvin et al. unpublished data, 2007]. In the end, 19 articles (20 studies) were included in the meta-analysis for a total sample size of 3283 schizophrenia patients (1680 DD and 1603 SCZ). The 19 articles/20 studies included in the meta-analysis measured depressive symptoms with well-validated scales for depression, and involved schizophrenia patients abusing alcohol, cocaine or a mixture of PAS (current and/or lifetime; Table 1).
Cross-sectional studies of depressive symptoms in single- and dual-diagnosis schizophrenia patients
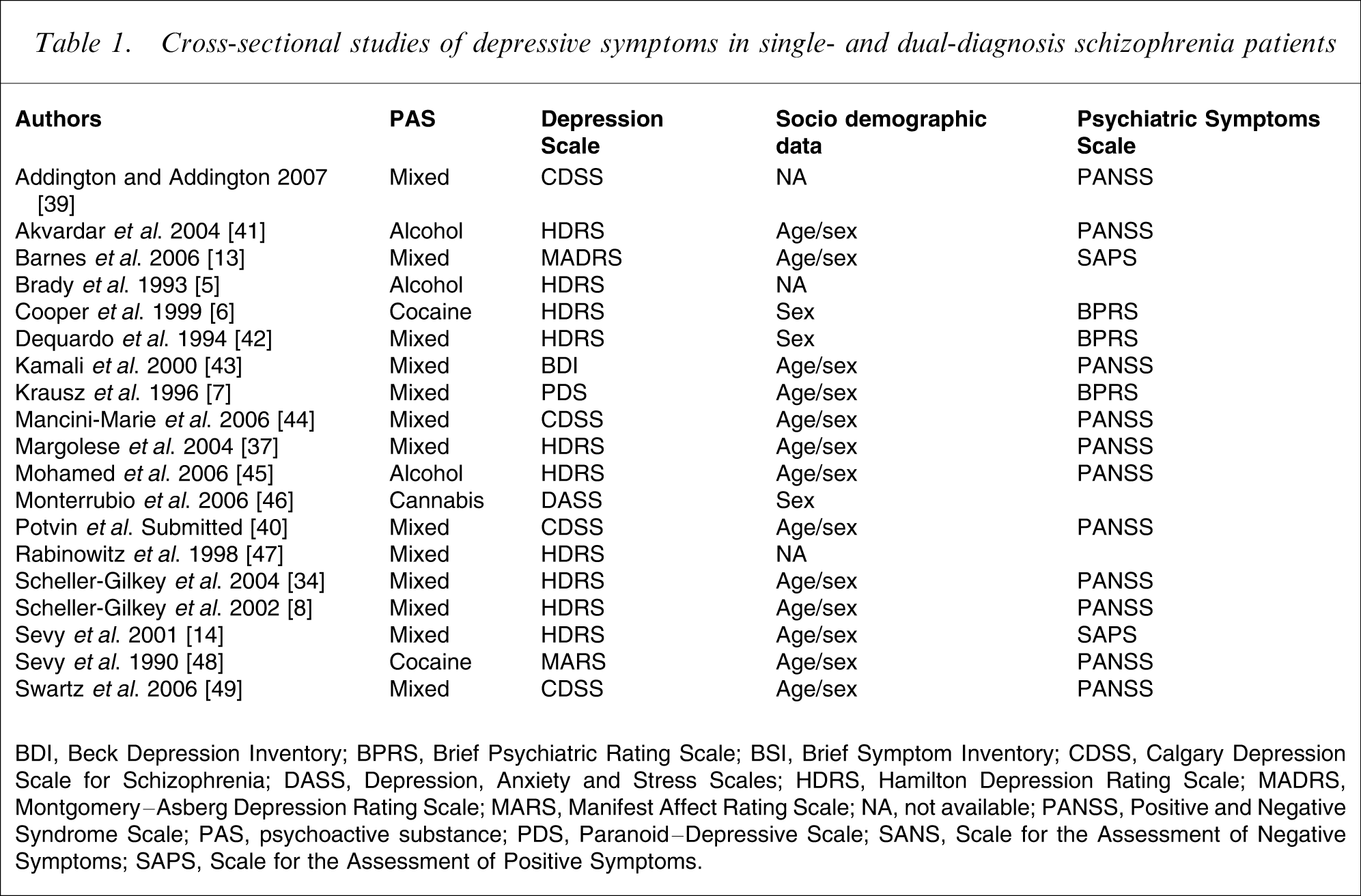
BDI, Beck Depression Inventory; BPRS, Brief Psychiatric Rating Scale; BSI, Brief Symptom Inventory; CDSS, Calgary Depression Scale for Schizophrenia; DASS, Depression, Anxiety and Stress Scales; HDRS, Hamilton Depression Rating Scale; MADRS, Montgomery–Asberg Depression Rating Scale; MARS, Manifest Affect Rating Scale; NA, not available; PANSS, Positive and Negative Syndrome Scale; PAS, psychoactive substance; PDS, Paranoid–Depressive Scale; SANS, Scale for the Assessment of Negative Symptoms; SAPS, Scale for the Assessment of Positive Symptoms.
Quantitative data synthesis
Twenty studies were included in the composite analysis. Within a random-effect model, a small, positive and significant effect size was obtained, suggesting that some DD patients experience more severe depressive symptoms than SCZ patients (Figure 1).
Forrest plot of the effect size estimates of the differences in the severity of depressive symptoms between schizophrenia patients with and without substance use disorders. The direction of the effect size is positive if dual diagnosis patients have a higher score on the retained depression scales (a higher score means more severe depressive symptoms) than non-abusing schizophrenia patients.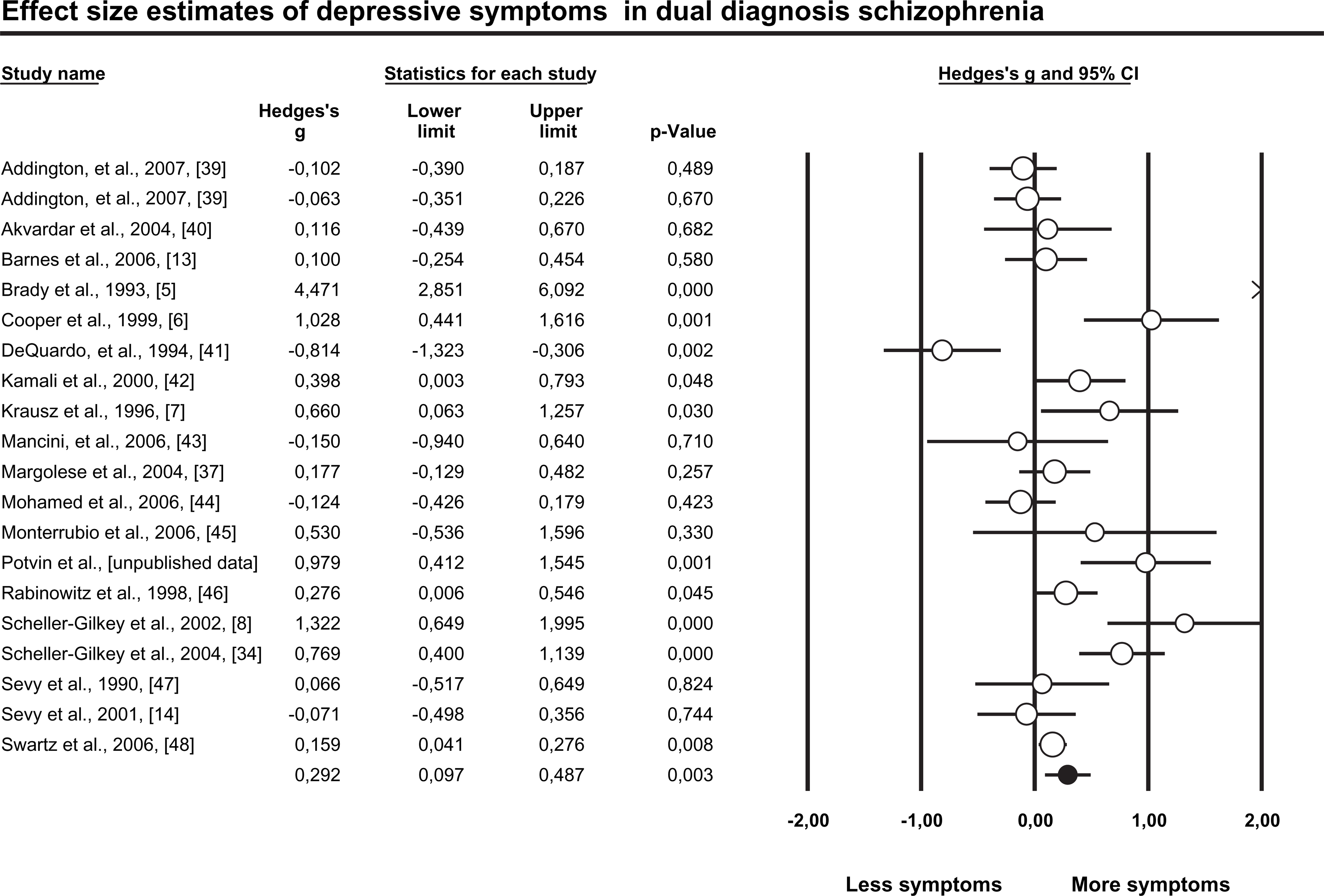
Secondary analyses
Secondary analyses were performed based on the type of PAS abused by patients and the depression scale used to measure depressive symptoms.
When studies were subdivided according to PAS, non-significant effect size estimates emerged for alcohol, cannabis and cocaine. For mixed PAS, a small, positive and significant effect was found (Table 2).
Secondary analyses
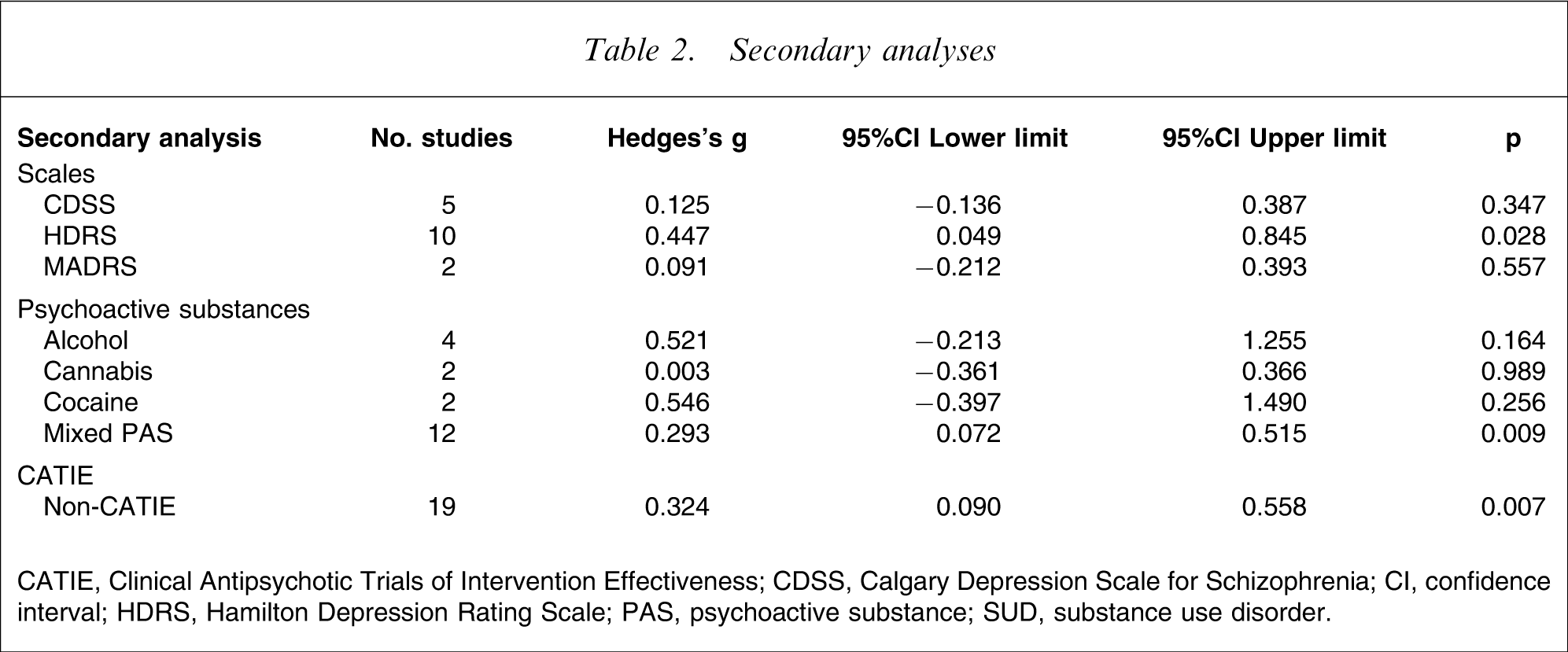
CATIE, Clinical Antipsychotic Trials of Intervention Effectiveness; CDSS, Calgary Depression Scale for Schizophrenia; CI, confidence interval; HDRS, Hamilton Depression Rating Scale; PAS, psychoactive substance; SUD, substance use disorder.
Among the 20 studies included in the meta-analysis, 10 studies screened depressive symptoms with the HDRS; five with the CDSS; and two with the Montgomery–Asberg Depression Rating Scale (MADRS). A moderate and significant effect size estimate was produced for studies using the HDRS, while small and non-significant effect size estimates emerged in the case of studies using the CDSS and the MADRS (Table 2). Noteworthy, one of the five studies using the CDSS, namely the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study, involved 1123 patients, out of the 3283 included in the meta-analysis. Thus, we performed a separate analysis excluding the CATIE study (non-CATIE), and it produced a small, positive and significant effect size estimate (Table 2).
Psychopathology
To control for symptoms, PANSS-positive and PANSS-negative scores were separately pooled, for the DD and SCZ groups of available studies. Out of 20 studies included in the meta-analysis, 12 studies reported psychopathology scores measured with the PANSS (Table 1). Using this scale, no differences in positive symptoms were identified between DD and SCZ groups (DD group: PANSS-positive score = 18.6±2.7; SCZ group: PANSS-positive score = 17.2±2.4; t = 1.331; p = 0.197). Similarly, no differences in negative symptoms emerged between the two groups (DD group: PANSS-negative score = 18.5±2.8; SCZ group: PANSS-negative score = 19.6±4.0; t = − 0.751; p = 0.461).
Sociodemographic data
It has been repetitively reported that DD patients are younger and more frequently male than SCZ patients [50, 51]. To control for such confounding factors, age and sex data were separately pooled for the DD and SCZ groups of available studies.
Age
Thirteen out of 20 studies reported the age of patients (Table 1). No differences in terms of age were observed between the DD and SCZ groups (DD group: 35.0±8.1 years; SCZ group: 37.9±8.3 years; t = − 0.895; p = 0.380).
Sex
Sex ratio was provided in 16 studies (Table 1). Relative to the SCZ group, patients in the DD group were significantly more likely to be male (DD group: 945 men (83.9%) vs 249 women; SCZ group: 796 men (71.5%) vs 544 women; χ2=114.5; p = 0.0001). Considering this sex difference between DD and SCZ patients, we performed a meta-regression analysis using CMA, with male sex ratio of DD patients (each study) as the regressor and the effect size estimate (each study) as the independent variable. Male sex ratio of DD patients (expressed in percentage) did not emerge as a significant predictor of effect size estimates (β = − 0.004; p = 0.641).
Discussion
The aim of this study was to perform a meta-analysis of cross-sectional studies measuring depressive symptoms in single- and dual-diagnosis schizophrenia patients using validated scales for depression. The meta-analysis was carried in order to verify the assumption suggesting that DD patients experience more severe depressive symptoms, compared to abstinent schizophrenia patients. Twenty studies (19 articles) were included in the meta-analysis. Within a random effect model, a small, positive and significant effect size estimate was obtained for DD patients relative to SCZ patients. As such, these results show that some DD patients experience more severe depressive symptoms than SCZ patients, as it has been widely assumed in the literature [5, 51, 52]. However, these results also show that the difference in depressive symptoms between the two groups is small. An effect size estimate is a measure relative to pooled standard deviations (σ) [Effect size=((MeanDD – MeanSCZ)/σ); where σ = √(SDDD 2+SDSCZ 2)/2)] [53]. Here, an effect size estimate of 0.292 signifies that there is 20% of non-overlap (approximately) in depressive symptoms between the two groups [54]. Thus, approximately 20% of DD patients experience more depressive symptoms than SCZ patients. It must also be considered that the studies (n = 3) for which data was not available (qualitatively, not statistically) reported no differences in depressive symptoms between DD and SCZ patients [30–32].
Secondary analyses were performed based on the type of PAS abused by patients and the depression scale used to measure depressive symptoms. When studies were subdivided according to PAS, non-significant effect size estimates emerged for alcohol, cannabis and cocaine. For mixed PAS, a small, positive and significant effect size estimate was produced. Therefore, it seems premature to conclude about the differential impacts of specific PAS on depressive symptoms in schizophrenia.
A secondary analysis was also conducted based on the scale used to measure depressive symptoms. A moderate, positive and significant effect size estimate was produced for the 10 studies using the HDRS; a small, positive and non-significant effect size estimate emerged in the case of the five studies using the CDSS; and a small, positive and non-significant XXX (effect size)was produced with the two studies using the MADRS. Thus, the scale used to measure depression seems to be one of the reasons why some authors have found differences in depressive symptoms between DD and SCZ and others have not. The measure of depression in schizophrenia raises methodological concerns because there is an overlap between depressive, negative and extrapyramidal symptoms (EPS) [55]. Most depression scales (including the HDRS and the BDI) consist of items (anhedonia, psychomotor retardation etc.) common to depressive, negative symptoms and EPS. Therefore, the finding of increased depressive symptoms in DD patients with studies using the HDRS (Hedges's g = 0.447; p = 0.028) may reflect a measure problem. That is, the observed difference may not be specific to depressive symptoms. Alternatively, the CDSS was developed precisely to avoid items that overlap with negative symptoms and EPS. Although preliminary, some studies have shown that the CDSS, compared to the HDRS or the BDI, is more efficient in the discrimination between depression, negative symptoms and EPS [56]. Thus, the small and non-significant effect size obtained may offer a more valid picture of the clinical reality than results obtained with the HDRS.
Noteworthy, one of the five studies using the CDSS, namely the CATIE study, involved 1123 patients, out of the 3283 included in the meta-analysis. Thus, we performed separate analyses for the CATIE study and non-CATIE studies. For the CATIE study and non-CATIE studies the effect size estimate was of similar magnitude (small and small-to-moderate, respectively). As such, these results highlight no major discrepancy between the CATIE study and non-CATIE studies.
Controls were also performed in order to rule out the potentially confounding effects of sociodemographic data and psychiatric symptoms. No differences emerged between DD and SCZ patients in terms of age, positive and negative symptoms. However, we found that DD patients were more likely to be male relative to SCZ patients. Although we found no relationship between male sex ratio and the amplitude of effect sizes, it must be considered that among schizophrenia patients, male subjects present more depressive symptoms than female subjects [10–12]. Thus, the increased depressive symptoms in DD patients may be related to the confounding effect of sex, not to substance abuse per se.
To our knowledge this is the first meta-analysis to investigate depressive symptoms in addicted schizophrenia patients, despite 20 years of clinical research on this topic. Also of interest, the meta-analysis was performed on a large sample size (3283 patients), using well-validated depression scales. In addition, the finding of increased depressive symptoms in DD patients may have implications for the prevention and treatment of depression in a subgroup of substance-abusing patients. Of interest, the small magnitude of the effect size estimate suggests that the difference in depressive symptoms between DD and SCZ is smaller than what has been assumed in the literature. Also noteworthy, the meta-analysis highlighted two factors, depression scale and sex, which may account for the difference in depressive symptoms observed between the two groups.
The results of the present meta-analysis must be interpreted cautiously. It could be tempting to conclude that PAS use exacerbates depressive symptoms in a subgroup of schizophrenia patients, because long-term use of alcohol, cannabis and cocaine is associated with depressive symptoms [10–12]. However, the cross-sectional design of the studies included in the meta-analysis does not permit to draw inferences about causality, and the reverse explanation cannot be ruled out. That is, severe depressive symptoms may lead patients to use PAS, as proposed by the self-medication hypothesis [57]. Alternatively, uncontrolled factors such as psychiatric medication, may lie behind the difference in depressive symptoms observed between DD and SCZ patients.
In conclusion, the results of this meta-analysis show that DD patients experience more severe depressive symptoms than SCZ patients, even though this result seems to be limited to a small subgroup of substance-abusing patients. This result may have potential implications for the prevention and treatment of depression in a subgroup of schizophrenia patients. However, the effects of depression scales and sex must be carefully controlled before a firm conclusion can be reached. In the future, the comparative reliability/validity of the HDRS and CDSS in DD will need to be assessed. The ability of both scales to discriminate between depressive, negative and extrapyramidal symptoms will also need to be compared in DD. Ideally, depressive symptoms would be measured both during the active phase of substance abuse and after a period of drug withdrawal, to determine whether the observed differences reflect the acute effects of PAS or more stable traits. Compared to the HDRS, the CDSS is more efficient in discriminating between depressive, negative symptoms and EPS in SCZ patients. A replication of this finding in DD patients would imply that the HDRS may have overestimated the differences in depressive symptoms between DD and SCZ patients, due to overlapping symptoms. A closer attention to the matching of male sex ratios of DD and SCZ patients is also warranted. The meta-analysis showed that in the available body of literature, DD patients were more likely to be male, relative to SCZ patients. Given that male schizophrenia patients are known to present more depressive symptoms than female patients, the differences in sex ratios between DD and SCZ patients may have contributed to the differences in depressive symptoms observed between the two groups. To sum up, the current meta-analysis showed that DD patients experience more depressive symptoms relative to SCZ patients. However, this difference was smaller than commonly assumed in the literature. Further, the choice of depression scale and sex ratio emerged as two potential confounding factors that will need to be controlled with greater caution in future studies on the topic.
Footnotes
Acknowledgements
ES is the holder of the Eli Lilly Chair in Schizophrenia from the University of Montreal. SP is the holder of a postdoctoral scholarship from the Fonds de la recherche en Santé du Québec.
