Abstract
Given the high cost of mental illness, both to the heath-care system and the social welfare system it is appropriate that considerable attention be given to the policy and health service needs of these conditions. Governments are increasingly using economic information such as cost-effectiveness to inform policy decisions and for the allocation of health-care resources.
The February 2006 press announcement from the Council of Australian Governments (COAG) of an action plan including significant new investment by all governments to improve mental health services has been met with cautious optimism [1]. As pointed out by Andrews, key policy questions remain: what is the cost, and is it cost-effective to increase the coverage of people with mental disorders [2]. Accurate information on expenditure and the value of different treatments is necessary for designing cost-effective interventions. But there continues to be some debate within the mental health care sector about the ‘best bets, best buys’ in terms of where to allocate additional resources.
The economic consequences of schizophrenia are considerable. Health-care costs for patients with schizophrenia are high in relation to its prevalence and comprise up to 1.5–3% of the total health service budget [3–5]. Additionally, health-care spending is highly skewed, with no or few expenses for many people and large expenses for a minority [6]. There is no reason why schizophrenia is different.
The social costs of schizophrenia are similarly high. The Australian Low Prevalence Disorders Study (LPDS) found that people with schizophrenia experience high levels of unemployment and social isolation and are socioeconomically disadvantaged [7]. In recent years a major effort has been directed towards quantifying the burden associated with various conditions. The measure of disability-adjusted life years (DALYs) lost was developed as a global measure of health status [8]. Schizophrenia was ranked as the 16th (male) and 18th (female) most common cause of disability burden in Australia in 1996, accounting for 1.5% of the total burden of disability [9]. It was ranked sixth for men and fifth for women in the burden attributable to mental disorders in the more recently conducted Australian Burden of Diseases study [10].
Andrews et al. present a theoretical model that estimates the relative efficiency of current treatment programmes for 10 different mental disorders [11]. In this analysis schizophrenia was associated with a small reduction in burden of illness (13%) at a relatively high cost (AUD$200 000 per DALY avoided). The authors go on to assert that ‘it would be important to examine the detail of this expenditure to determine whether the same clinical outcome could be achieved with less use of high-cost elements, giving greater efficiency overall’. As such, real-world data are required to both test the conclusions generated from the Andrews et al. model, and to provide more information on the likely best buys in mental health service provision in Australia.
The purpose of the present paper was to describe the direct and indirect costs associated with schizophrenia. We present the Schizophrenia Care and Assessment Programme (SCAP), a prospective, longitudinal, study of global health outcomes for 347 people with schizophrenia. Although the SCAP study was funded by the pharmaceutical company Eli Lilly, it was not a drug trial but rather an observational study of symptomatic, functional, humanistic and economic effects.
Direct costs are those associated with the provision of health care, and include the costs of hospitalization, medical consultations, diagnostic procedures, medications, patient transportation, counselling services etc. Indirect costs are those associated with loss in productivity because of illness, which can be due to temporary or permanent disability or premature death. Indirect costs can be incurred by both the person with the illness and/or their carers.
There are two ways of evaluating the cost of treatment for a disorder such as schizophrenia. The first is a bottom-up approach, in which individual resource consumption data are collected and then the costs are calculated for each participant by multiplying the frequency of each resource unit used by the cost per unit. This was the approach used in the present study. The second option is a top-down approach, in which where total expenditure is calculated by cost category for certain health-care areas and then divided by the number of the affected population.
The costs of health care for people with schizophrenia in Australia have been investigated using both top-down and bottom-up approaches [10, 12–18]. However, most studies completed to date have used data collected some time ago [13, 15]; economic modelling techniques based on imputed data [14]; or limited cross-sectional service use data collected using self-report. [15]. Estimates of the cost of care for people with schizophrenia collected in a prospective manner are less common and few studies have had the capacity to relate these expenses to clinical outcomes.
In the past few years several observational schizophrenia research trials in the USA have used insurance claims data for costing purposes [19–21]. In the UK, patients’ medical records, pharmacy records and community nurse records were used as an alternative source of objective service utilization information [22]. Self-reported service use data are still being used in naturalistic schizophrenia studies in Europe and Australia [15, 23, 24].
Treatments provided to patients with schizophrenia have evolved substantially over recent years. To some extent there has been a shift of the site of care of patients from long-stay hospitals to the community [25, 26]. There has also been alterations in the prescription patterns of antipsychotic medication from the older typical (first generation) to the newer and more expensive atypical (second generation) medications [27]. These trends are likely to have resulted in significant changes to the costs associated with health-care provision for people with schizophrenia. Results from this study should provide greater insight into the both the direct and indirect costs associated with schizophrenia at a time when considerable changes are occurring in the management of this condition.
Methods
The Australian SCAP was a prospective longitudinal observational study of patients with schizophrenia and related disorders conducted at the Dandenong Psychiatry Research Centre in Victoria, Australia between December 1997 and May 2003. Parallel studies were undertaken in the USA (2400 patients from six sites) [28], and the UK (600 patients from six different sites) [29]. Full details on the methodology of SCAP are given in Kulkarni et al.[30].
Participants
The present participants consisted of 347 patients who were recruited as part of the SCAP study.
Resource data collection
Resource utilization data and costs were collected from several payment and activity systems, patients’ medical records as well as from questionnaires (Table 1). Participants consented to grant access to information held within a variety of national and state-based electronic databases, described in the following sections.
Summary of direct costing methods used
AN-DRG, Australian National Diagnostic-Related Groups; DAMHS, Dandenong Area Mental Health Service; HIC, Health Insurance Commission; MH-CASC, Mental Health–Classification and Service Costs; MRAF, Medical Record Abstraction Form; OTC, over-the-counter; PBS, Pharmaceutical Benefits Scheme; PRISM, Psychiatric Record Information System Manager; SCAP, Schizophrenia Care and Assessment Programme.
Public hospital psychiatric service costs
The Psychiatric Record Information System Manager in Victoria (PRISM) was a database containing records on individual psychiatric care (hospital and outpatient) provided by state-based services to outpatients, including length of stay and diagnosis for all hospitalizations, and detailed information on the type, duration and setting of hospital services provided in community settings.
Admission/discharge data were presented as the number of new hospitalizations within each medical record abstraction form (MRAF) period. MRAFs spanned the 6 months prior to each of seven study phases (baseline, 6, 12, 18, 24, 30 and 36 months). If the hospitalization continued into the following MRAF period, no admission was registered during the second MRAF period. Similarly, the average length of hospital stay was calculated per admission and length of an admission in any one study period could thus exceed the 6 months. Costs of hospitalization were calculated per day in hospital during each period and reported as the cost by study period.
Community medical service costs
Data regarding non-hospital-based medical outpatient services, community medical services and Medical Benefits Scheme (MBS)-funded services were collected from the Health Insurance Commission (HIC) database. This provides a central record of all Medicare-funded outpatient private specialist and general practitioner medical services and diagnostic procedures.
Medication costs
Pharmaceutical Benefits Scheme data (community)
Records of medications available via the Pharmaceutical Benefits Scheme (PBS) dispensed by community pharmacies throughout Australia were collected.
Hospital-provided medications (inpatient and community)
A separate hospital pharmacy information system provided details of all medications dispensed at discharge from hospital or from the outpatient and community services provided by hospital-based services.
Individual data were obtained from each of the aforementioned information systems for the entire study period. Private insurance or health-care records were not accessed for the present study. The majority of participants (>90%) held some form of health-care concession card, meaning that most participants were eligible for the highest level of subsidized service without having to reach a predetermined safety-net threshold. Thus, data extraction from the various electronic registries (HIC and PBS) was very complete.
Costing methods
Direct costs
Individual direct costs were calculated for each patient by multiplying the frequency of each resource unit used by the corresponding cost per unit. Annual cost calculations were based on the total 6 monthly costs per subjects in the study, excluding patients with missed visits. Actual costs were calculated for hospital pharmacy medications, Medicare services and PBS medications.
Hospitalization
For the costing of hospitalizations, Australian National Diagnosis-Related Groups (DRG) costs for public hospitals in Victoria were used [31]. For psychiatric DRG codes, the pharmacy cost component was subtracted from the total cost and replaced with the costs of the actual psychotropic medications used by each patient. Non-psychiatric DRG codes included cost of medications within the pharmacy cost component. Estimated per-day DRG hospital costs were applied to the number of days in hospital by the relevant DRG code. Data provided by the admission/discharge system include same-day separations as well as emergency separations. Costs attributed to each DRG code incorporated emergency department costs along with all other components. The same unit costs were applied to long-term community care psychiatric units. Admission/discharge data collected in SCAP also included all admissions for non-psychiatric conditions including injury, poisoning, burns and trauma.
Transport
Involuntary admissions for psychiatric illness are associated with ambulance services. In the absence of exact data on use of ambulance, we have adopted the assumption used in the LPDS that 20% of involuntary admissions were by ambulance [32]. Victorian ambulance costs (AU$565.80, Metropolitan Ambulance Service 1999) were applied.
Community care
The unit costs of community services were calculated on a per hour basis specific to the staff type providing each service. The base year for the costing was 1999, which used salaries for 1998/99. Wages were inflated to current levels by adjusting by changes in average weekly earnings. Staff-related on-costs, patient-attributable time and office overheads were all considered in order to calculate cost per hour of staff time. These derived unit costs were then applied to PRISM data collected on the number of services recorded by each patient to obtain the total cost of community services.
Medications
Details of psychotropic medications taken in hospital over the study period were extracted from patients’ medical records. The average cost per mg, as reported by the hospital pharmacy information system, was applied to medications taken in hospital. Data on medications dispensed in the community were obtained from the PBS for the medications for which the HIC paid some benefit. All records were available for medications costing more than $3.50 for concession card holders and more than $22.40 for general patients. Medications costing less than $22.40 for general patients are not subsidized by and recorded in the PBS scheme. To estimate these costs, the median costs of medications under the PBS threshold per patient per year were used as an approximation for all SCAP patients who had a record of such a medication in the year 2000. The median cost was used because most of those patients were not considered to be heavy medication users, and using the average cost would have skewed the data. The year 2000 was chosen because this was the only year when all SCAP participants were enrolled in the study.
There was a group of general patients who received further subsidy under the PBS safety net threshold scheme. When a safety net threshold is reached in one calendar year, an individual may become a concession cardholder for the rest of the year. The general safety net threshold was AU$686.40 in 2002. The thresholds are updated annually but for simplicity the 2002 figures will be referred to. To estimate the cost of medications under the PBS threshold used by those patients when they were general patients, we used the mean cost of medications under the PBS threshold per patient per year for all patients who had a record of such a medication in the year 2000. The mean cost was used for those patients, because most were similarly heavy medication users.
Legal costs
Legal costs were estimated conservatively using the average expenditure per criminal case lodgement at a Magistrate's court. The cost of AU$127 (Australian Institute of Criminology 1999) was applied to the number of self-reported arrests.
Indirect costs
Productivity costs were estimated, using the human capital approach, as ‘the reduced future gross income due to mortality and/or morbidity’ [33] and valued by their opportunity cost. Hence, cost estimates appropriately represent losses in earnings that the patient would have achieved, adjusted for his/her likelihood of being employed, if he/she had not been affected by the illness [33]. This analysis considered morbidity-related patient productivity costs only. Productivity costs associated with premature mortality, or those incurred by carers, were not evaluated.
The main data source for productivity costs was a comprehensive patient-reported outcome questionnaire, the SCAP Health Questionnaire [34], which was completed every 6 months. Patients with morbidity-related productivity losses were identified and then categorized into two groups for each 6 month period. The first group included people who did not work for pay at all. On the basis of the human capital estimation method, the productivity costs were calculated assuming that, in the absence of illness, these patients would be employed at the same rates as the general population, and hence would earn the same average wage.
The second group consisted of patients who had been employed for some length of time but who had indicated that their income was less than their typical earnings. Those patients who reported working ≥35 h per week were excluded from this group because they were considered as fully employed. The productivity costs for this second group were also calculated following the human capital approach. In this case, however, the reported number of hours spent at work was deducted from the cost calculation. The productivity costs in this group can be regarded as the costs associated with the productivity losses of employed persons due to under-employment and absenteeism as a result of their schizophrenia.
Patients who reported that their main activities were ‘going to school, college or university’ were excluded from the estimation of productivity cost. For those patients included in the estimate, any time spent at school was deducted from the cost calculation. Gender- and age-specific data were used in the calculations. In order to reflect the demographics of the catchment area, data from three Victorian local government areas (Greater Dandenong, Casey and Cardinia), as reported in the 1996 Census, were used.
Transfer payments
Transfer payments include such monies as tax foregone, which represents lost tax revenue resulting from decreased earnings due to unemployment/underemployment. This may include the potential income tax foregone and the potential indirect (sales) tax foregone, but in the present study only the former was considered. To estimate the income tax foregone by patients, the average individual income tax rate for the general population in the catchment area in 1999–2000 was determined. The estimated income tax rate was then applied to the morbidity-related lost earnings incurred by the patient. In SCAP, the mean age of participants was 34 years at baseline. The 1996 Census reported that the average weekly income per person aged 34 was $368 in the catchment area. This associates a personal income tax rate of approximately 14%. The individual resident income tax rates were obtained from the Australian Tax Office. For other components of transfer payments, estimated figures from Carr et al. 2003 [32], were used.
Statistical analysis
Uniformly, the service utilization and cost data were not normally distributed, in that the majority of participants were within the low range of the scale and a few participants were using a very high number of services. Hence, the non-parametric Wilcoxon test was used to test the hypotheses of no difference between the distributions of the continuous variables between each study period.
Results
Participants
The details of participants included in the study have been described separately by Kulkarni et al.[30]. In brief, the sample for this analysis included 347 patients (215 male, 132 female) with a mean age 33.8±11.0 years. Data for a total of 267 (77%) patients were available for analysis for the full 3 years of the study. Medication use during the study period is summarized in Figure 1 and in greater detail elsewhere [30].
Proportion of participants by type of medication.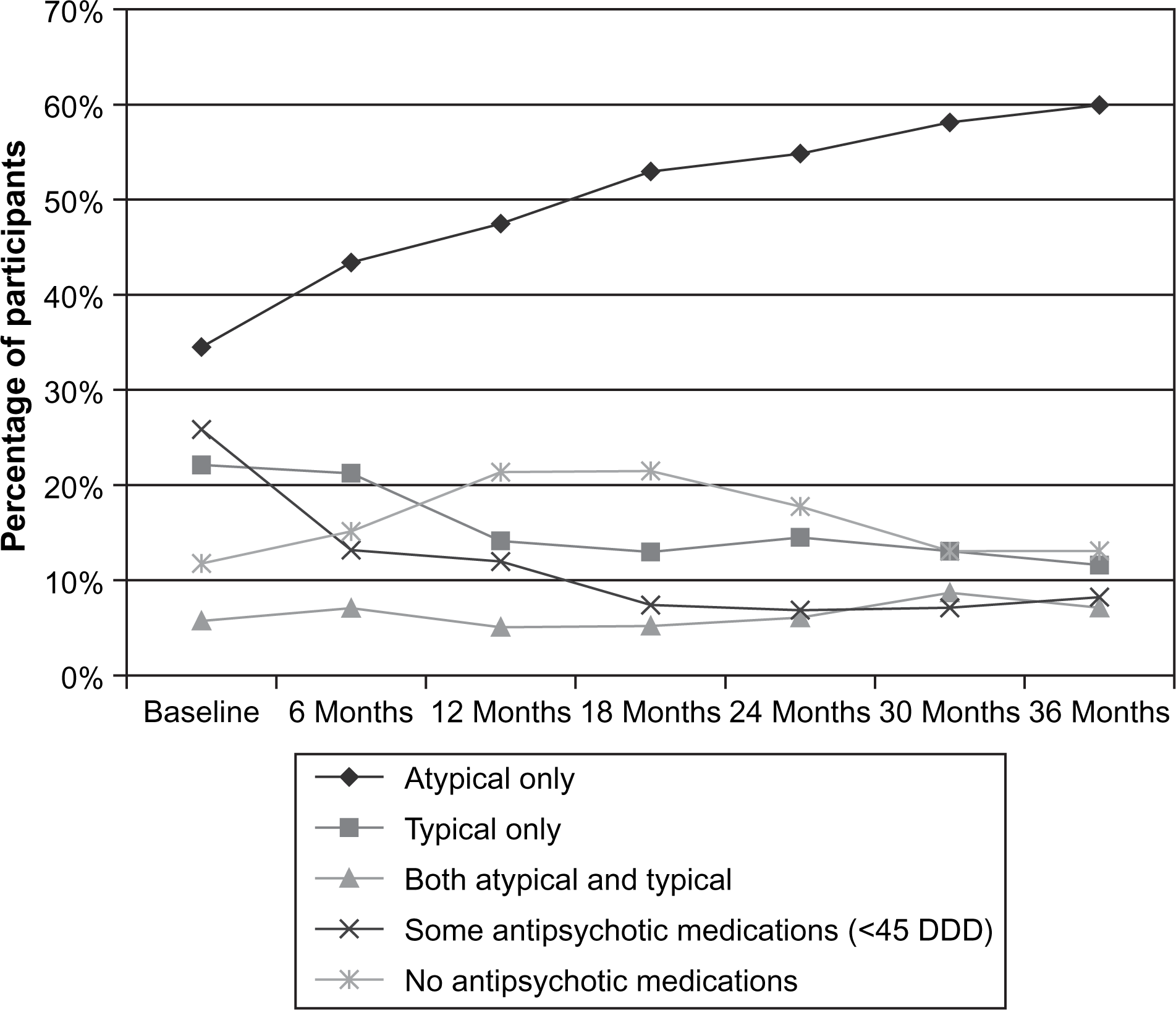
Service use
Outpatient and community health-care service use
The distribution of service use for each study phase is presented in Table 2. A total of 10 517 community psychiatric services (79% PRISM and 21% MBS) were used, by 304 participants (87.3%), at baseline. By 36 months a total of 5802 services (50% PRISM and 50% MBS) were used by 260 participants (97.4%). The reduction in service use across the study was significant (Z = − 14.1, p < 0.001). This was also significant between the 6 and 12 month visits (Z = − 4.3, p < 0.001). The proportion of participants who received 30 or fewer total services per phase increased from around 60% at baseline and 6 months, to approximately 75% at the 18 and 24 month visits, reflecting a change from hospital-provided to community-provided care.
Mean and range of all outpatient/community occasions of service per participant
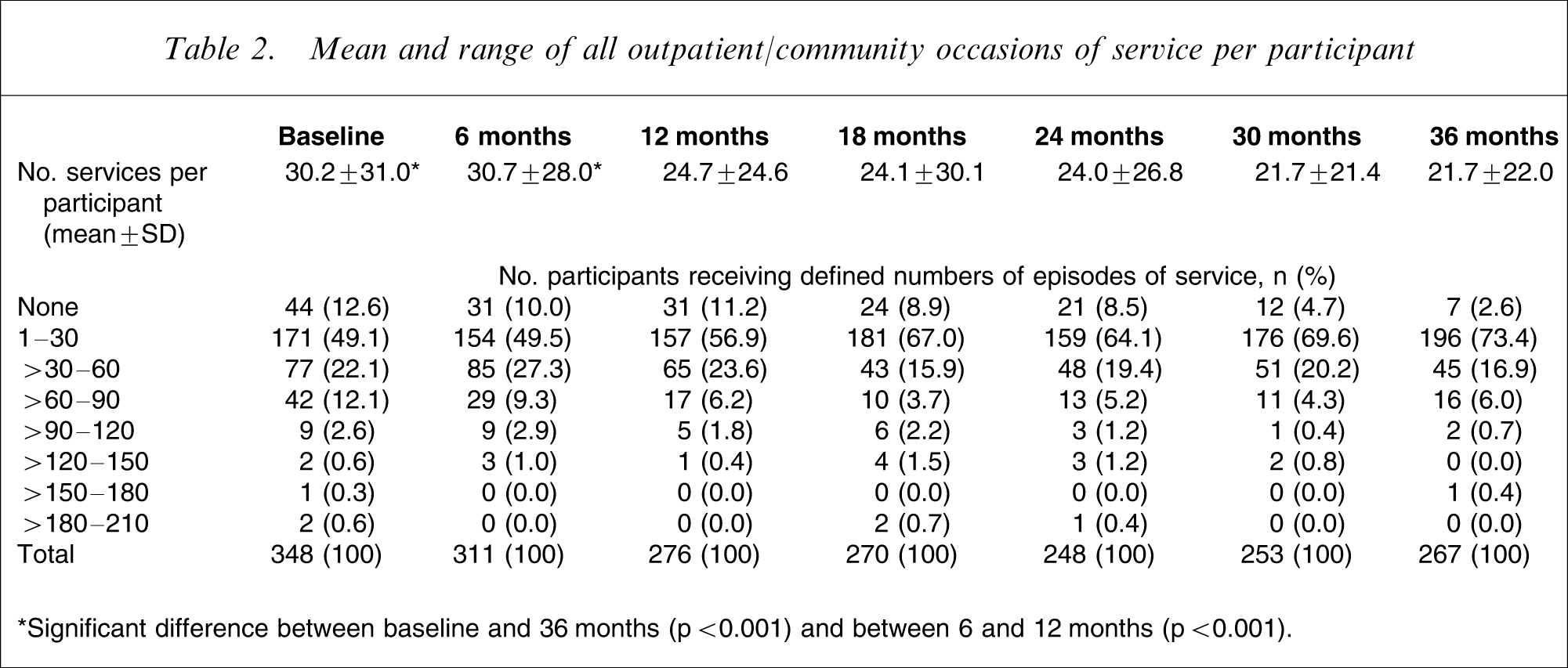
∗Significant difference between baseline and 36 months (p < 0.001) and between 6 and 12 months (p < 0.001).
Hospitalization
The mean number of days spent in hospital for the first 6 month period was 20.2±52.4 (median 0.5) and between 8 and 11 days for all other study periods (median = 0 each period). The higher number of days spent in hospital at baseline may be a result of a significant number of participants being recruited while inpatients. Indeed, a total of 50% of participants had been admitted to hospital in the 6 month period prior to enrolment, declining to a stable proportion of 20–22% at 18–36 months. By 36 months 9.7% of participants had one hospital admission in the previous 6 months, 6.4% had two admissions and 4.9% had three or more admissions. The duration of hospital admissions at 36 months ranged from 7.7 days up to 179 days.
Costs
Mean costs associated with patients with schizophrenia are shown in Table 3. The average annual cost of treatment was AU$32 160 from a societal perspective and AU$27 294 from a government perspective during the first year of the study, decreasing to AU$29 181 and AU$23 541, respectively, in the third year (Figures 2 and 3).
Average cost per patient from a societal perspective: first year. MBS, Medicare Benefits Scheme; PBS, Pharmaceutical Benefits Scheme. Average cost per patient from a societal perspective: third year. MBS, Medicare Benefits Scheme; PBS, Pharmaceutical Benefits Scheme.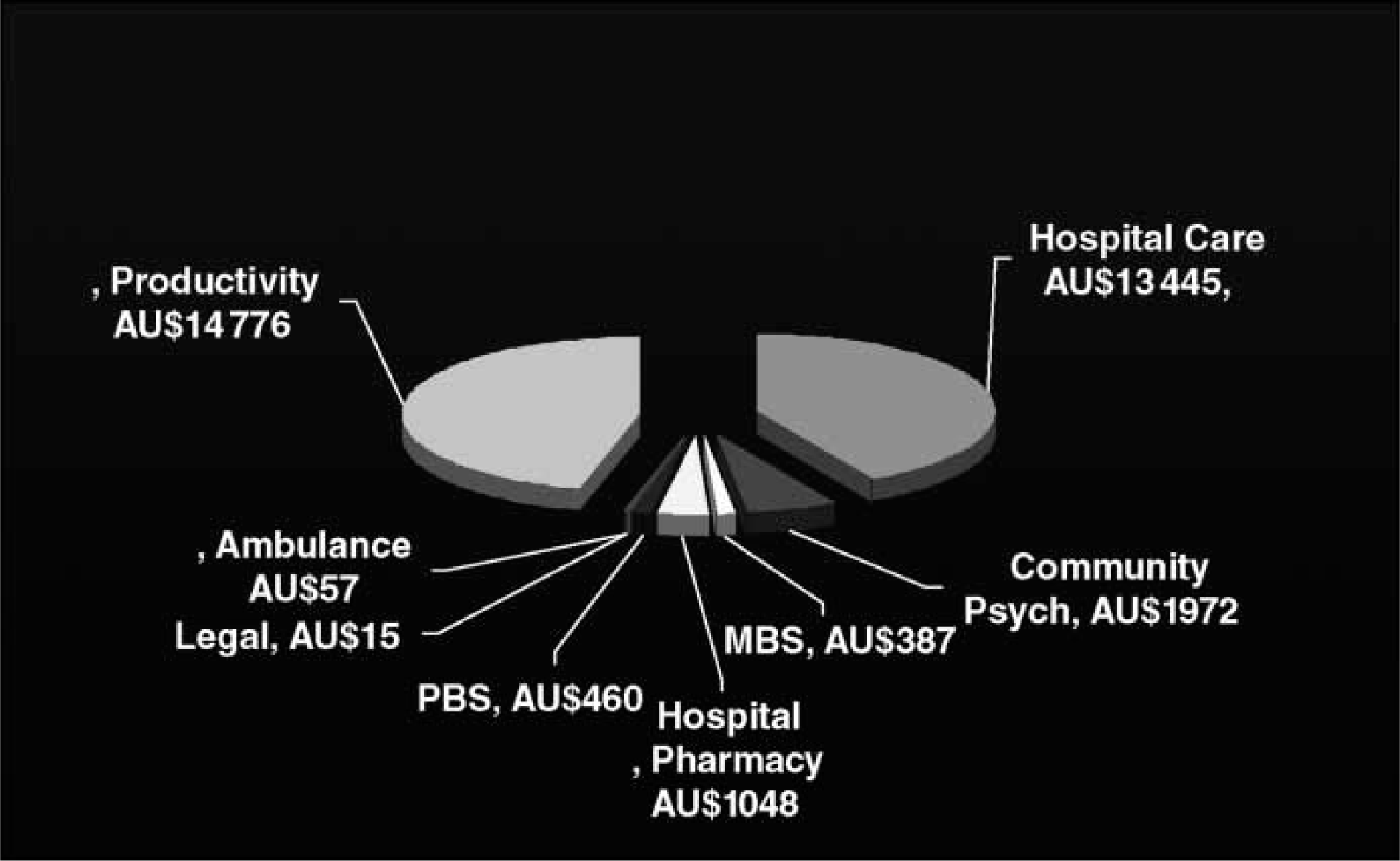
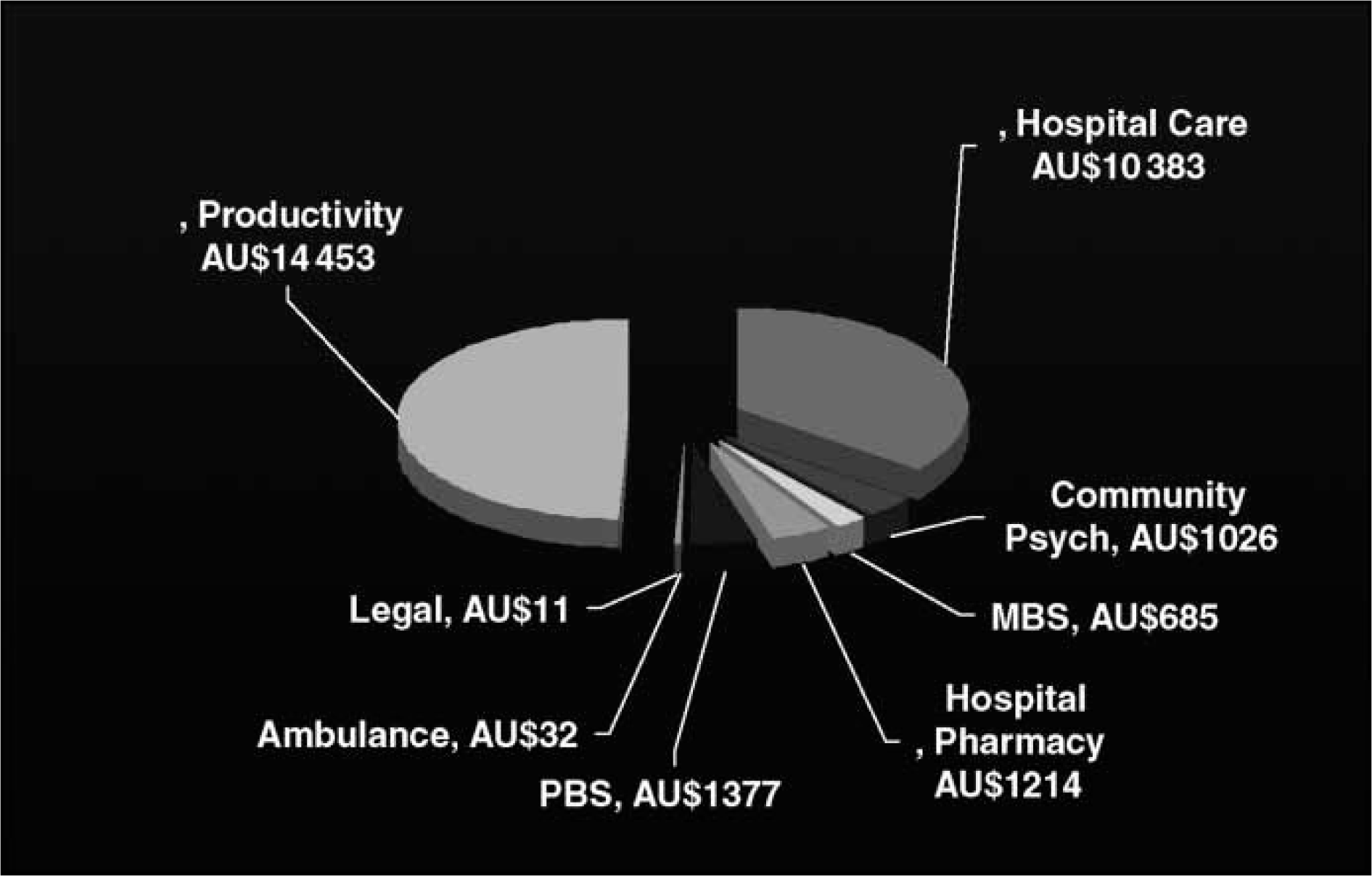
Average cost per patient from societal and government perspective (AU$)
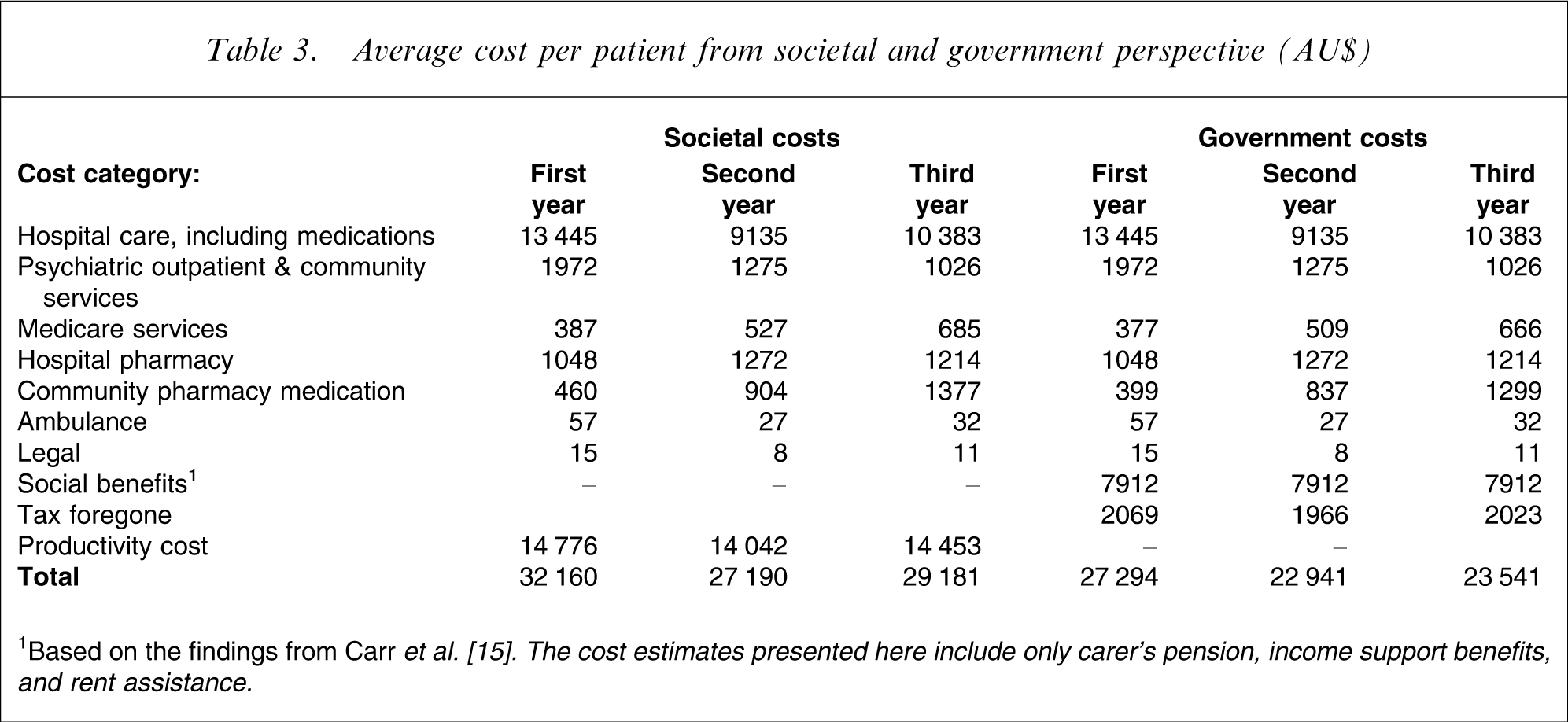
1Based on the findings from Carr et al. [15]. The cost estimates presented here include only carer's pension, income support benefits, and rent assistance.
Health-care costs
The total average cost of health-care services and medications used by participants over the first year was AU$17 383, decreasing slightly to AU$13 140 and AU$14 717 in the following 2 years. Across the study period there was a significant decrease in total costs (Z = − 2.8, p = 0.005), which was mainly due to reduced costs associated with hospitalization. This was significant between 6 months and 12 months (Z = − 5.4, p < 0.001). There was a shift in the overall distribution of health-care costs as shown in Figure 4. In the first year of the study 91% of the medical costs were attributed to services and 9% were medication costs, while 2 years later, medication costs accounted for 18%. The increase in the costs of community pharmacy medications (PBS medications) was the most pronounced; from AU$460 per person in the first year to AU$1377 in the third year of the study. Hospitalizations were the most expensive direct cost component. As seen in service usage data, fewer participants were admitted and for less time in the second and third year of the study (59% in hospital for an average 29 days in year 1, compared with 29% admitted for years 2 and 3 for an average 18 and 19 days, respectively). Thus, the average cost of inpatient treatment decreased (Z = − 3.4, p < 0.005), with the greatest fall in costs in the first 18 months (significant between 6 and 12 months (Z = − 4.8, p < 0.001).
There was a progressive increase in total medication costs over the study. This was significant overall (Z = − 8.7, p < 0.001) and between baseline and the 6 month visits (Z = − 5.8, p < 0.001) and 24 month and 30 month visits (Z = − 2.8, p = 0.005). The distribution of costs between the different sources of medication changed over the study period as more prescriptions were dispensed by community pharmacies, and less by hospitals. The annual cost of psychotropic medications taken while in hospital was AU$77 per study subject in the first year, AU$59 in the second year and AU$76 in the third year of follow up. Those costs were incorporated into the cost of hospitalizations. As outpatients, participants received medications at total average cost of AU$1508 per person in the first year, increasing in the following 2 years to AU$2176 and AU$2591, respectively.Figure 4
Distribution of the average medical costs per patient (ambulance costs are not shown because they are <1% in total). PBS, Pharmaceutical Benefits Scheme; PRISM, Psychiatric Record Information System Manager.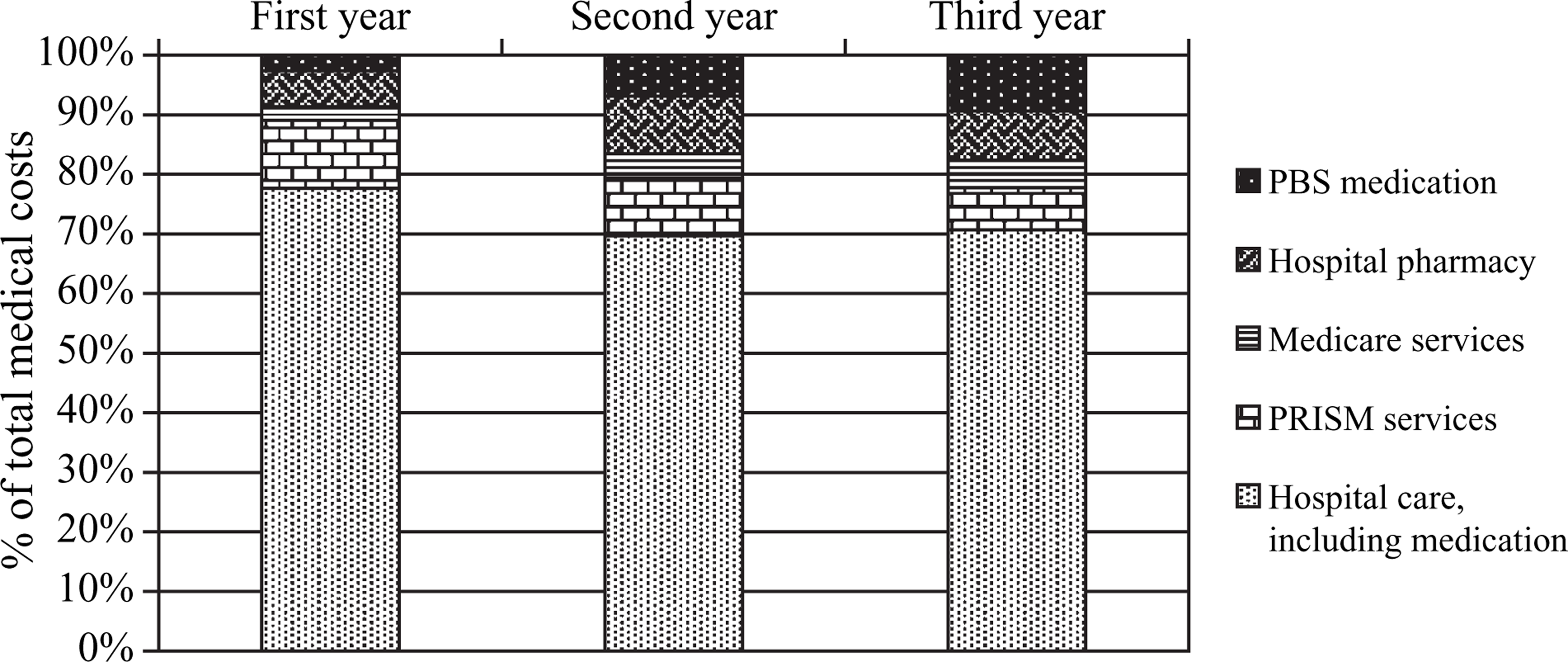
The average cost of community and outpatient psychiatric (PRISM) services was AU$1972 per patient in the first year, decreasing to AU$1275 and AU$1026 after that. This accounted for 11%, 10%, and 7% of health-care costs over the 3 years, respectively. The majority of the patients used some PRISM service in the 3 years (84%, 72% and 64%, respectively). The average number of services per participant decreased from 48 in the first year to 33 in the second year and 25 in the third year. Most of the services (70% in the first year and 76% in the next two years) were provided by psychiatric nurses. Only a very small percentage (1%) of PRISM services was delivered by psychiatrists, which did not change over time. Psychologists provided <2% of services.
The average annual cost of Medicare services per patient was AU$387 in the first 12 months, which increased to AU$527 and AU$685 in the following years. This includes all medical services used by participants, not just those related to their mental illness. In the first year 66% of participants used some medical services in the community. With greater numbers out of hospital in the next 2 years, this increased to 74% and 93%, respectively. The average number of services per person also increased from 13 to 21 over the 3 year period. Services performed by general practitioners account for just over half of all Medicare services, whereas <10% of participants were attended by a consultant psychiatrist. This relation did not change over time. The mean annual cost per participant ranged from AU$13 140 to AU$17 383 but the median cost per participant over the course of the study was AU$7819.
The distribution of total treatment costs, as shown in Figure 5, is markedly skewed, with 80% of participants accounting for 39% of the costs, averaging $5900 per patient per year. The remaining 20% of participants consumed the balance (61%) of resources, averaging AU$37 340 per participant per year.
Distribution of total treatment costs by number of subjects.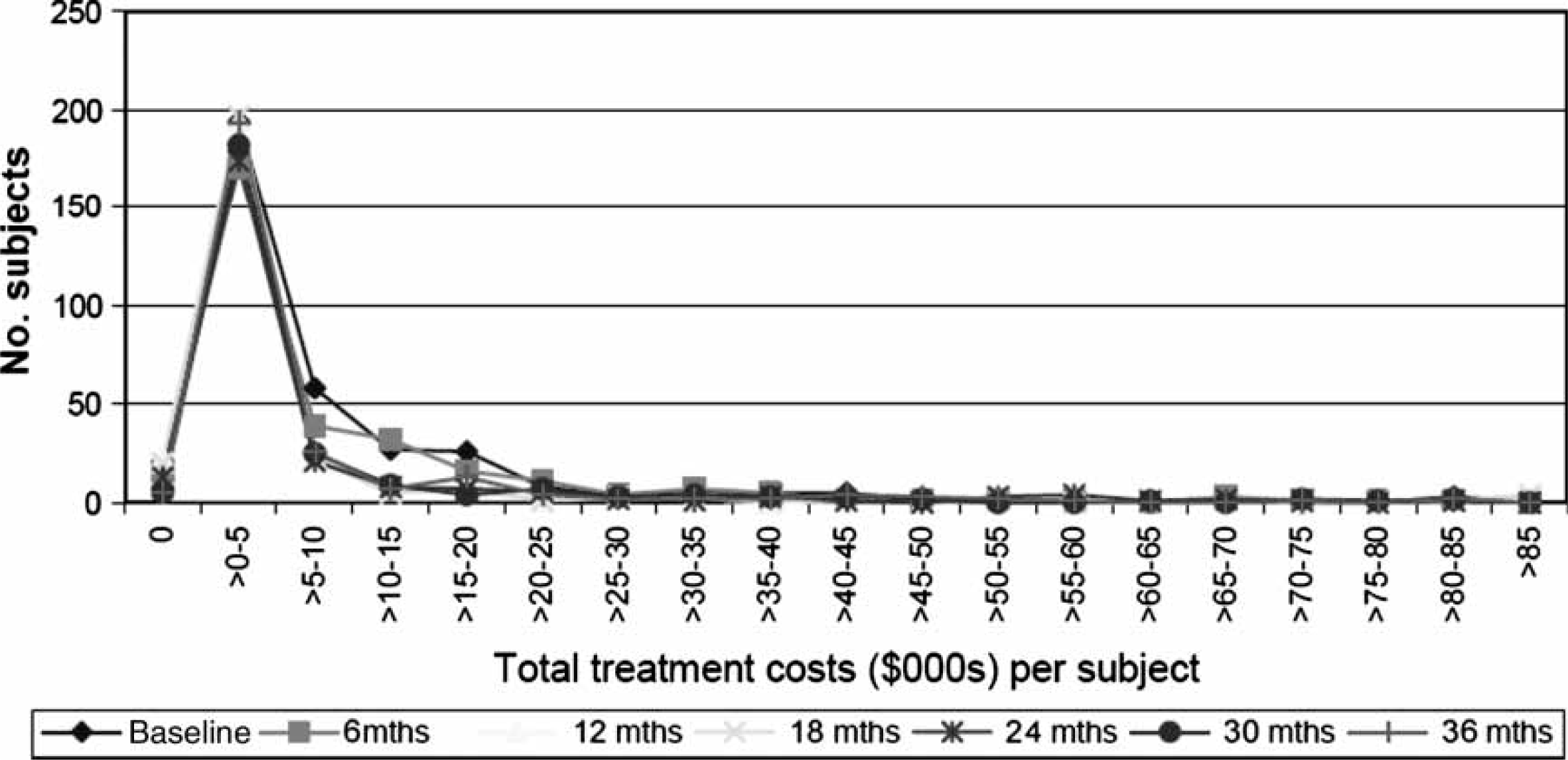
Indirect costs
The average productivity costs per patient were estimated at AU$14 776 in the first year, AU$14 042 in the second year, and AU$14 453 in the third year (Table 3). These costs include the morbidity-related productivity costs incurred by patients only. It was found that only 24% (9% full-time and 15% part-time) of men and 11% (2% full-time and 9% part-time) of women were in paid work, as shown in Table 4.
Proportion of patients employed over the 24 month period
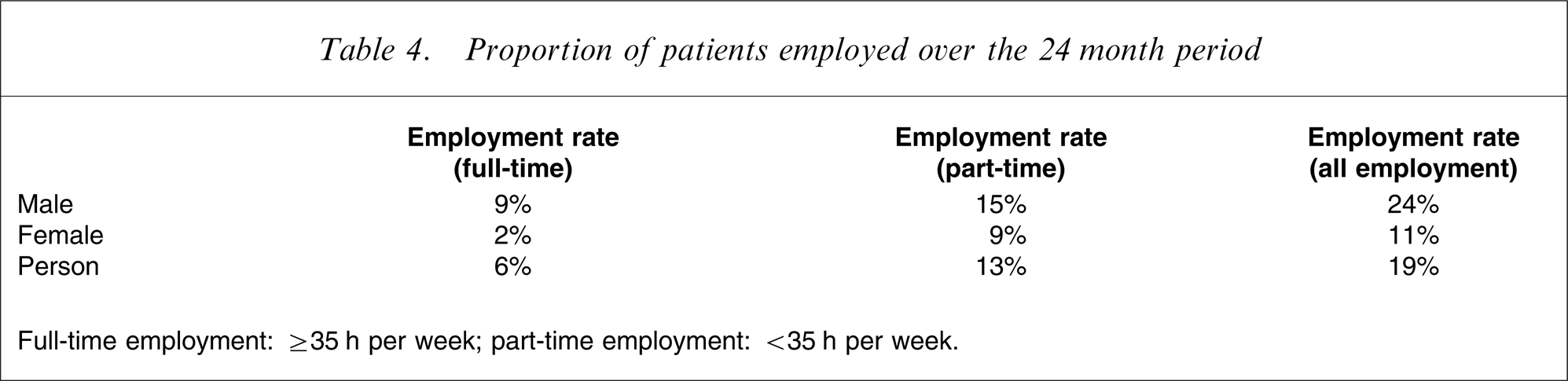
Full-time employment: ≥35 h per week; part-time employment: <35 h per week.
Discussion
This study is the first of its kind to prospectively and comprehensively document the costs associated with the treatment of individuals with schizophrenia as well as the ensuing indirect expenses. There was a reduction of the total average costs per patient in the second year of the study and a change in the distribution of costs. Indirect costs remained almost the same but due to a decrease in direct medical costs, the indirect costs accounted for a higher percentage of the total costs in the second and third year of follow up. As has been previously described [14, 15, 35], hospitalizations accounted for the largest proportion of treatment costs, at between 70% and 80% over the course of this study.
The pattern of shifting costs may be explained by a number of factors and is not limited to clinical improvement or other patient factors. For example, both inpatients and outpatients were recruited and therefore during the first year more than half of the patients were admitted to hospital. In the next 2 years the percentage of those hospitalized decreased and remained stable. As a result, the average cost of hospitalization decreased by 23% over the follow-up period. As such, the costs analysed in the second and third years of the study are more likely to accurately represent the hospital costs associated with schizophrenia. The proportion of patients using community-based psychiatry services, and the average number of services per patient, also decreased in the second and third year of the study, while there was a corresponding increase in the use of Medicare-funded services over this period. There was also an increase in medication costs. As illustrated in Figure 1, this is likely to reflect the shift towards the prescribing of atypical antipsychotic medications that occurred during this time period.
The prospective nature of the present study allows us to consider the pattern of service-related costs, how these change over time for patients with schizophrenia, and allows realistic judgments about the service utilization figures. A substantial majority (92%) of patients had some community psychiatric service, or were hospitalized, in the first year, whereas, 1 year later, this percentage decreased to 76% and further to 68% in the following year. Despite this 24% reduction of engagement with standard public psychiatric services, we were able to continue the collection of outcomes data. This avoids a major bias with shorter term studies that would predominantly evaluate the cost of treatment for patients who are current high service users and therefore would risk overestimating some of the service costs.
Few other studies have attempted to provide specific costs for the care of patients with schizophrenia in Australia. The major attempts to do this have been based on the cross-sectional LPDS, a two-phase census-based study conducted in four Australian metropolitan areas in 1997 and 1998 [35, 36]. Retrospective reports of service use were made by patients for the 12 months prior to study interview and cost estimates were based upon these. The total average societal cost reported by the LPDS was higher (AU$46 785) than that determined in the present study. Similar figures were reported by the Access Economics Report 2002 (approx. AU$50 000 per person with schizophrenia) [37]. However, the design and methods of those studies were very different from ours. The Access Economics Report, for example, was a top-down cost of illness evaluation, based on prevalence rates reported by the LPDS. The costs consist of 36% direct costs (AU$18,000) and 64% indirect costs (AU$32,000), including losses due to premature death.
The LPDS was a bottom-up study of patients with psychotic disorders, of whom 60% were patients with schizophrenia [35, 36]. Costs of treatment and medications were based on self-report, and costs per unit of service were estimated. The LPDS reported higher service use figures. For example, the LPDS patients had an average of 12 GP visits per patient compared to seven GP services per SCAP patient. Similarly, average length of hospitalization was 6 weeks for the LPDS patients compared with 29 days in SCAP. This may have been largely driven by differences in the service profiles of the treatment areas and the services made available to patients. The present study was conducted in Victoria where all long-stay psychiatric facilities were essentially closed from the mid-1990s. Within Dandenong Area Mental Health Service, a service covering an outer metropolitan urban and semi-rural region, there was provision for acute hospitalization but the bed resources for this were limited, resulting in a short duration of stay and a treatment programme based predominantly around community services. There was also quite limited access for patients to private psychiatrists. In contrast, the LPDS was based in a number of services around the country including several covering inner urban areas [36]. This included States in which longer term hospitalization was still a treatment possibility at the time of the LPDS and 10.1% of the study sample spent 12 months in hospital. The total number of days spent in hospital was not provided but 51.6% had at least one admission with an average length of stay of 13 weeks (median 6 weeks), which is obviously much greater than the average 20 days spent in hospital per year by patients in the present study.
In addition, some of the costing assumptions used in the LPDS were not supported by the SCAP findings. For example, the LPDS patients reported only the identity of pharmaceuticals they used and an assumption was made that each medication was taken for 12 months during the year. Our experience with the SCAP medication data does not support such a supposition. On the contrary, we consistently found that over time and across patients, a number of different drugs, in some cases from different therapeutic classes, were dispensed to the same patient. Another important point to be made is that in SCAP most of the antipsychotic medications were dispensed through the hospital pharmacy. Those medications are usually provided at no cost to the patients and their cost to the State government is substantially lower compared to the PBS costs used in the LPDS. Further, in SCAP the overall average costs per service were lower. For instance, the cost per outpatient and community service was approximately AU$40, which was less than half of the estimated cost per service used in the LPDS. Finally, Carr et al. estimated and costed the use of non-government and voluntary agency input to patient care, which was not included in the present study [35].
It is commonly asserted that indirect costs should be included in economic evaluation, arguing that improved productivity in the future as a result of treatment will provide more resources for health care in the long run. In regards to indirect costs, this study clearly supports the hypothesis that schizophrenia is associated with substantial productivity losses. Only 24% of male patients and 11% of female patients were employed on average over the 3 year study period. In contrast, a total of 69% of men and 50% of women in the general population of Greater Dandenong, Casey and Cardinia were employed [Australian Bureau of Statistics: personal communication]. Even when only the morbidity-related patient productivity costs were included in the analysis, the average productivity cost per patient was estimated as around AU$14 000 each year. In addition, it was shown that schizophrenia generates substantial cost to government in the form of transfer payments. These payments, such as benefit payments and tax forgone, represent redistribution of money within the society and do not represent real economic costs. However, from the government's perspective, transfer payments are important because they represent important budgetary implications of the illness.
There were a number of limitations in the design and execution of the SCAP study. First, because the information systems accessed focused on publicly financed services, we underestimated cost associated with the usage of private health-care resources. However, only 2.6% of the SCAP subjects reported having private health insurance, limiting this effect. Underinsurance of patients with schizophrenia has been reported in other counties as well [38]. In addition, we did not collect data on the use of non-government support and rehabilitation agency services although these were relatively limited in the area at the time of the study. In this regard, it will be critical for future research to assess the costs of alternative housing provision for patients, such as supported residential programmes and hostels, especially with the shift away from longer stay hospital care.
In conclusion, SCAP offers an insight into the health-care component costs for patients with schizophrenia in Australia. Accurate estimates of the costs of psychiatric illness are critical for the planning of services. The data we have gathered, and the comparison it offers to the LPDS, indicate that this should be done on a relatively local level integrated with an understanding of the breadth and limits of local service provision. The highly skewed nature of the costing data demonstrate that for a significant proportion of patients the health-care costs associated with the treatment of schizophrenia are modest. Identification of those factors contributing to high costs should provide insights into potential interventions to improve the efficiency of overall service delivery. Because health-care costs are driven to such a marked degree by rates of hospitalization, the availability of beds and the system structure will play a major role in the determination of the costs of care. This remains the case even within services that have shifted, such as the one in the present study, to a predominantly community-based method of treatment.
Footnotes
Acknowledgements
We would like to thank all the subjects who participated in the study and the staff of Dandenong Area Mental Health Service who facilitated the study. The study was funded by Eli Lilly. We would also like to thank Natasha Marston for assistance with manuscript preparation.
