Abstract
The aim of the present paper was to critically examine evidence about the benefits of cognitive-behavioural therapy (CBT) for pediatric obsessive-compulsive disorder (OCD) from controlled and single group studies, including its benefits relative to medication are critically reviewed. Selected studies were categorized by study type and by risk of bias classification. Standardized mean differences (Hedges’ g or Cohen d) and, where appropriate, weighted mean difference (WMD) were calculated. All five comparison and 14 one-group studies showed a significant benefit for CBT within a wide range (ES = .78 to 4.38). Low risk of bias studies produced the lower adjusted effect sizes. The best available estimate of CBT efficacy relative to no treatment is about 1 standardized mean difference, equivalent to a treatment effect of 8 points on the Children's Yale-Brown Obsessive-Compulsive Scale. This represents a reduction in the risk of continuing to have OCD post-treatment of about 37% (95% CI 14% to 54%). Evidence from 3 studies indicates that the efficacy of CBT and medication do not differ significantly. CBT combined with medication is significantly more efficacious than non-active controls or medication alone but not relative to CBT alone. CBT should be regarded as a first line equivalent to anti-OCD medication with the potential to lead to better outcomes when combined with medication than medication alone can provide. Additional studies are needed to further clarify CBT's benefits and to investigate how it can be made more available as a treatment option for children and youth who suffer from OCD.
Keywords
Cognitive–behavioural therapy (CBT) is recommended as the psychotherapeutic treatment of choice for children and adolescents with obsessive–compulsive disorder (OCD) [1, 2]. For pre-pubertal children it is recommended as the first-line treatment, and for adolescents CBT combined with a selective serotonin re-uptake inhibitor (SSRI) is the recommended first-line approach. In addition to exposure with response prevention (ERP), CBT treatments include various other components such as psycho-education, cognitive training, anxiety management training and parental or family involvement. At the time, the reasons for the expert consensus recommendation in favour of CBT for children and adolescents with OCD were the demonstrated efficacy of CBT with ERP for OCD in adults [3], together with the belief that OCD in childhood is ‘virtually identical to the adult form’ [4], p.94]. ERP in particular also has a logically consistent and compelling rationale that presents a clear relationship between the obsessive and compulsive symptoms, their maintenance and the CBT treatment.
There are a number of issues, however, which indicate that the downward extension of CBT to pediatric forms of OCD may face unique problems. Firstly, a number of recent studies question the presumed developmental continuity between pediatric OCD and adult OCD. Geller and colleagues have described a bimodal age of onset for OCD, with one peak at about 10 years of age and another during puberty or early adulthood [5, 6]. While equally prevalent and similar in its clinical characteristics to the later-onset OCD, the childhood-onset disorder has a number of distinctive features that have implications for clinical management, including possible responsiveness to psychotherapeutic interventions. In particular, it is male-predominant and more strongly co-morbid with disruptive behavioural problems, and developmental disorders, including autistic disorder, depression and other anxiety disorders [7]. OCD in children also co-occurs with tic disorders, and has been noted as a part of a pediatric autoimmune neuropsychiatric disorder associated with streptococcal infections [8]. These co-morbidities may impact on the rationale for CBT in so far as they indicate differing mechanisms for symptom onset and maintenance. For example, the observation that OCD children with co-morbid tics have less well-developed cognitions triggering their compulsions suggests a lesser role for cognitive appraisals in the maintenance of the OCD than proposed by current CBT models [6]. Alternatively, co-morbidities associated with impulsivity may reduce the capacity of child and adolescent OCD patients to tolerate the discomfort involved in the exposure and response prevention component of CBT.
A second issue of extending CBT to pediatric forms of OCD concerns the more self-reflective cognitive techniques utilized with adults to enhance tolerance and adherence to ERP. Some of these techniques may be of limited value with younger children because they presume a level of meta-cognitive skill; for example, an ability to reflect on thoughts and emotions, not common until adolescence. In addition, CBT for OCD in children and adolescents generally requires support and therapeutic assistance from the family. Recent findings [9] show that parents of OCD children aged 8 to 14 years were less rewarding of the child's independence and less likely to promote positive problem-solving than parents of children with other types of anxiety disorders, externalizing problems or those with no problems. Barrett et al.[9] suggested significant limitations to parents' capacity to support their OCD child in undertaking self-directed ERP.
These potential difficulties make it doubly important to examine carefully the evidence base for the effectiveness of CBT for pediatric OCD. There are indications that the empirical evidence to support the use of CBT in children with OCD and adolescents with OCD is not of the highest quality [2], may be simply lacking [10] or is being misinterpreted. Abramowitz et al.[11] recently presented a meta-analysis of the effectiveness of treatments for pediatric OCD reviewing 18 studies using either CBT or medication. While they concluded that CBT with ERP was effective in reducing pediatric OCD symptoms, they recognized that their conclusions were based predominantly on uncontrolled trails. In addition, their meta-analysis did not consider risk of bias in the reviewed studies and used only pre–post differences to estimate effect sizes even for the controlled trails. A recent wide-ranging review of the status of CBT theory and the treatment for pediatric OCD [12] presented similar evidence concluding that CBT is an effective treatment without considering the impact of study design, study comparability and study quality on treatment effect sizes.
There are strong indications that the use of one-group prior to post comparisons artificially inflates effect sizes because of how meta-analysis deals with correlated scores and because of the confounding of maturation effects with treatment effects. Lipsay and Wilson [13] and Wilson and Lipsay [14] analysed the results of numerous meta-analyses of psychological, behavioural and educational treatments and found that pre–post estimates consistently overestimate the treatment effects relative to comparison designs. For some types of intervention the bias was large. They calculated that pre–post designs produced effects sizes on average 60% larger than those from comparison designs [13]. Although Abramowitz et al.[11] weighted individual studies’ effect sizes to take into consideration differences in precision due to sample size and design, their overall strategy of only considering pre–post effects even for comparison designs assumed that pre–post effects were unbiased estimates of the effect of the treatment for pediatric OCD. Consequentially, the pooled mean effect sizes for CBT and medication presented in that review [11] may have been substantially inflated. For example, the mean pre–post effect sizes for medication in that review of 1.13 (95% CI = 0.82–1.25) was 2.5 times higher than the overall effect size of 0.46 (95% CI = 0.37–0.55) reported in a recent meta-analysis of randomized controlled trails of psychopharmacology for pediatric OCD [15]. While O'Kearney et al.[16] recently reviewed randomized controlled trials of CBT for pediatric OCD and found two studies that examined CBT efficacy against a non-active control, because of substantial differences between these studies and their exclusion of pre–post designs it remains unclear if pre–post estimates for CBT are also inflated. In addition, none of the attempts at summarizing the evidence for CBT for pediatric OCD so far have considered study quality in regard to potential risk of bias nor discussed study heterogeneity in considering pooled effect size estimates.
Inferences about treatment effectiveness from single group designs are also more vulnerable to error because of the impact of testing or re-test effects [17, 18]. These effects refer to the consistent observations of significant decreases in negative states in groups that have not received treatment. In particular, Arrindell [17] reported effect sizes of 0.33 and 0.38 for a group of 95 adult OCD patients for decreases in self-report measures of OCD symptom severity over time without intervention. These effect sizes represent increased improvement rates due to a testing effect of between 16 and 18%. While data is available from several studies to estimate this effect for OCD measures in children and youth, none of the reviews or meta-analyses of CBT for pediatric OCD have included a correction for this effect in estimates of effect sizes from one-group studies.
Consideration concerning study design, risk of bias and study comparability are particularly important from a clinical perspective when examining the relative benefits of CBT against other available and effective treatments. Evidence for the efficacy of pharmacological treatment of OCD in children and adolescents, particularly for the SSRIs fluoxetine and sertraline, appears conclusive from good quality controlled trials [3, 10, 15, 19]. Nevertheless, many clinicians are understandably reluctant to prescribe psychotropic medication to children and adolescents. Consequently, clinicians often recommend CBT, when available, as the first-line treatment. While there are many other factors besides relative efficacy that influence a clinician's treatment recommendation for an individual patient, it is important that evidence about relative efficacy not be misinterpreted by advocates of either medication or CBT. For example, in her summary of the relative benefits of CBT and medication Turner [13] implies that CBT is superior because it ‘produces consistently larger effect sizes and greater rates of clinically significant improvement in comparison to psychopharmacology’ (p. 18). Comparing effect sizes between studies is inherently problematic [20] because of the impact of between study differences. When combined with inflated treatment effects for CBT because of the use of pre–post estimation, such comparisons risk providing a conclusion inconsistent with the evidence. In their review, O'Kearney et al.[16] located only two studies comparing CBT and medication for pediatric OCD, both of which reported equivalent results for the two treatments. In view of the controversy over the potential for self-harm with SSRIs, despite evidence of their benefits for children and adolescents with OCD, it is extremely important that researchers and practitioners provide non-partisan readings of the evidence.
It is also important for clinicians to have reliable and realistic estimates of the relative benefits of CBT in order to set reasonable benchmarks for treatment progress for their patients. Regardless of the recent advances in treatment, OCD remains a difficult disorder to treat with the adult research indicating that OCD symptoms rarely remit and few patients remain symptom-free. In addition, the ongoing process of improving treatment options and developing new treatments requires accurate estimates of the benefits of existing ones in order for researchers and clinicians to advocate for treatments that may produce benefits beyond these levels.
The present study is a critical appraisal of the evidence about the benefits of CBT for pediatric OCD. It presents the first comprehensive consideration of the quality, comparability and statistical rigor of the evidence from two types of studies: controlled trials and single-group pre to post designs. It provides a critical summary of the findings and their implications for clinical practice, research and policy-making. Overall, the study asks how beneficial CBT is for children and adolescents with OCD and what its relative benefits are compared to medication. It substantially extends recent reviews [12, 16] by contrasting estimates of the benefits of CBT from controlled and one-group designs taking into account study quality. The paper also aims to demonstrate the importance for clinicians, policy-makers and clinical researchers of considering the type and quality of evidence in their recommendations and advocacy of particular treatments for individuals and services.
Method
Inclusion criteria
Studies were selected if they were judged to be randomized controlled trials, quasi-randomized trials or one-group ‘open’ trials. Case studies were excluded but case series where estimates of group pre–post treatment effects were provided were included.
Eligible studies included participants who were 18 years of age or younger at the time of treatment or who were considered ‘children and adolescent’ as defined by the study. Participants had a diagnosis of OCD, established by clinical assessment or standardized diagnostic interview. Studies were included if they have used any of the main CBT techniques (exposure with response prevention, psycho-education, cognitive training, anxiety management training and parental or family involvement), either alone or in combination. Trials were included regardless of ‘treatment’ status of comparison, and the non-CBT control included active drug, placebo or wait list. Studies of medication efficacy that used CBT as a comparison group were included as well as studies that combined CBT and medications.
Measures
Primary outcomes
For the comparison designs, the primary continuous outcome examined was between-group differences in endpoint (post-treatment) measures on OCD symptoms’ severity. For the one-group design, the primary outcome examined was prior to post-treatment differences in OCD severity. Continuous outcome measures included the frequency, duration and degree of distress of obsessions and compulsions measured by clinician-rated or self-monitoring standard measures (e.g. Children's Yale-Brown Obsessive Compulsive Scale [CY-BOCS]). Binary primary outcomes examined were post-treatment improved/not-improved status or diagnosis remitted/non-remitted as defined by the studies. Where individual's CY-BOCS scores were available and binary outcomes were not reported, we classified endpoint scores of >10 as non-remitted.
Search strategy for identification of studies
Electronic searches were conducted on Medline and PsychINFO using the following search strategy: (a) RCT (phase 1), 1 RANDOMIZED CONTROLLED TRIAL.pt, 2 CONTROLLED CLINICAL TRIAL.pt. 3 RANDOMIZED CONTROLLED TRIALS.sh. 4 RANDOM ALLOCATION.sh., 5 DOUBLE BLIND METHOD.sh., 6 SINGLE BLIND METHOD.sh., 7 or/1 6, 8 ANIMALS.sh. not HUMAN.sh., 9 7 not 8, Clinical Trial (phase 2), 10 CLINICAL TRIAL.pt.,11 exp CLINICAL TRIALS/, 12 (clin$ adj25 trial$).ti, ab.,13 ((singl$ or doubl$ or trebl$ or tripl$) adj25 (blind$ or mask$)).ti, ab.,14 PLACEBOS.sh. 15 placebo$.ti, ab.,16 random$.ti, ab., 17 RESEARCH DESIGN.sh.,18 or/10 17, 19 18 not 8, 20 19 not 9, Comparative/Evaluation Studies (phase 3), 21 COMPARATIVE STUDY.sh. 22 exp EVALUATION STUDIES/23 FOLLOW UP STUDIES.sh.,24 PROSPECTIVE STUDIES.sh.25 (control$ or prospective$ or volunteer$).ti, ab.,26 or/2125 27 26 not 8, 28 27 not (9 or 20), Combine all 3 phases, 29 9 or 20 or 28,(b) Health Condition, 30 exp Obsessive Compulsive Disorder/Combine (a) & (b)), and EMBASE (using the following search strategy: (a) RCT (phase 1), #1 RANDOMIZED-CONTROLLED-TRIAL: It #2 CONTROLLED-CLINICAL-TRIAL: It #3 RANDOMIZED-CONTROLLED-TRIALS #4 RANDOM-ALLOCATION#5 DOUBLE-BLIND-METHOD #6 SINGLE-BLIND-METHOD #7 #1 or #2 or #3 or #4 or #5 or #6 #8 HUMANS/lim#9 7 and 8 Clinical Trial (phase 2) #10 CLINICAL-TRIAL: It #11 CLINICAL-TRIALS/exp #12 (clin∗ ∗25 trial∗):ta, ab. #13 ((singl∗ or doubl∗ or trebl∗ or tripl∗) ∗25 (blind∗ or mask∗)):ta, ab. #14 PLACEBOS #15 placebo∗:ta, ab. #16 random∗:ta, ab. #17 RESEARCH-DESIGN #18 #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 #19 #18 and #8 #20 #19 not #9 Comparative/Evaluation Studies (phase 3) #21 COMPARATIVE-STUDY #22 EVALUATION-STUDIES/exp #23 FOLLOW-UP-STUDIES #24 PROSPECTIVE-STUDIES #25 (control∗ or prospective∗ or volunteer∗):ta, ab. #26 #21 or #22 or #23 or #24 or #25 #27 #26 and #8 #28 #27 not (#9 or #20)Combine all 3 phases#29 #9 or #20 or #28, (b) Health Condition #30 Obsessive–compulsive-Disorder/exp Combine (a) & (b)).
The Cochrane Collaboration Depression Anxiety and Neurosis Controlled Trials Registry was also searched, using search terms Diagnosis = Obsessive–compulsive, and Age Group = Children or Adolescent.
In addition, the reference lists of all selected studies were inspected for more published reports and citations of unpublished research. Other relevant papers and major textbooks, which cover anxiety disorders, were checked. Any journals or conference proceedings specifically relating to behavioural treatment of OCD were searched.
Where required, the contact author of the included studies was contacted for clarification or additional information or data.
Data analysis
Effect sizes are reported as standardized mean differences (SMD; Hedge's g) for the comparison studies. Where the same measurement scale (e.g. CY-BOCS) was used across studies for a comparison and the studies have low to moderate heterogeneity, the weighted mean difference (WMD) was used to pool mean post-treatment outcomes with the weight given to each study determined by the precision of its estimate of effect. For the one-group designs, effect sizes (Cohen d) were estimated adjusting for sample size using the criteria described by Morris and DeSohn [21].
For dichotomous outcomes (improved/not-improved), relative risks together with the 95% confidence intervals were calculated at post-treatment for the comparison studies and absolute risk at post-treatment for the one-group designs.
Results
After reviewing the titles, abstracts and full texts, when necessary, of the 153 relevant titles initially identified by our search, 30 studies providing data on CBT for OCD in children and adolescents were located. Nine studies were excluded; four [22–25] were case studies, two [26, 27] had samples of only three participants, three [28–30] failed to report data separately for the participants who were less than 18, while one [31] looked at outcomes for a group of adolescents with various anxiety disorders and did not report separate outcomes for OCD. One of the comparison studies [32] was a report on the design and methods of another study [36], leaving five [33–37] comparison studies and 14 [38–51] one-group designs with data suitable for extraction.
Participants
Table 1 presents the characteristics of the patient samples for each of the included studies grouped by study type. The comparison studies had a total of 262 children and adolescents ranging in age from 7 years to 18 years 2 months. Gender distribution was about even for all studies. The ethnicity of the participants was Dutch [33], Australian [35], American [34, 36] and Brazilian [37]. Participants were diagnosed with OCD by clinical interview [33, 34, 37] or using a well-established diagnostic semi-structured interview for anxiety disorders (Anxiety Disorders Interview Schedule [ADIS]; Barrett et al.[35] used the parent as respondent [ADIS-P]; the POTS Team [36] used the child as respondent [ADIS-C]). Of those assessed for eligibility, 28% [33] and 27% [36] were excluded, while the numbers excluded in Neziroglu et al.[34], Barrett et al.[35] and Asbahr et al.[37] were not reported.
Descriptive characteristics of patients in the CBT studies and of the CBT delivered by study type
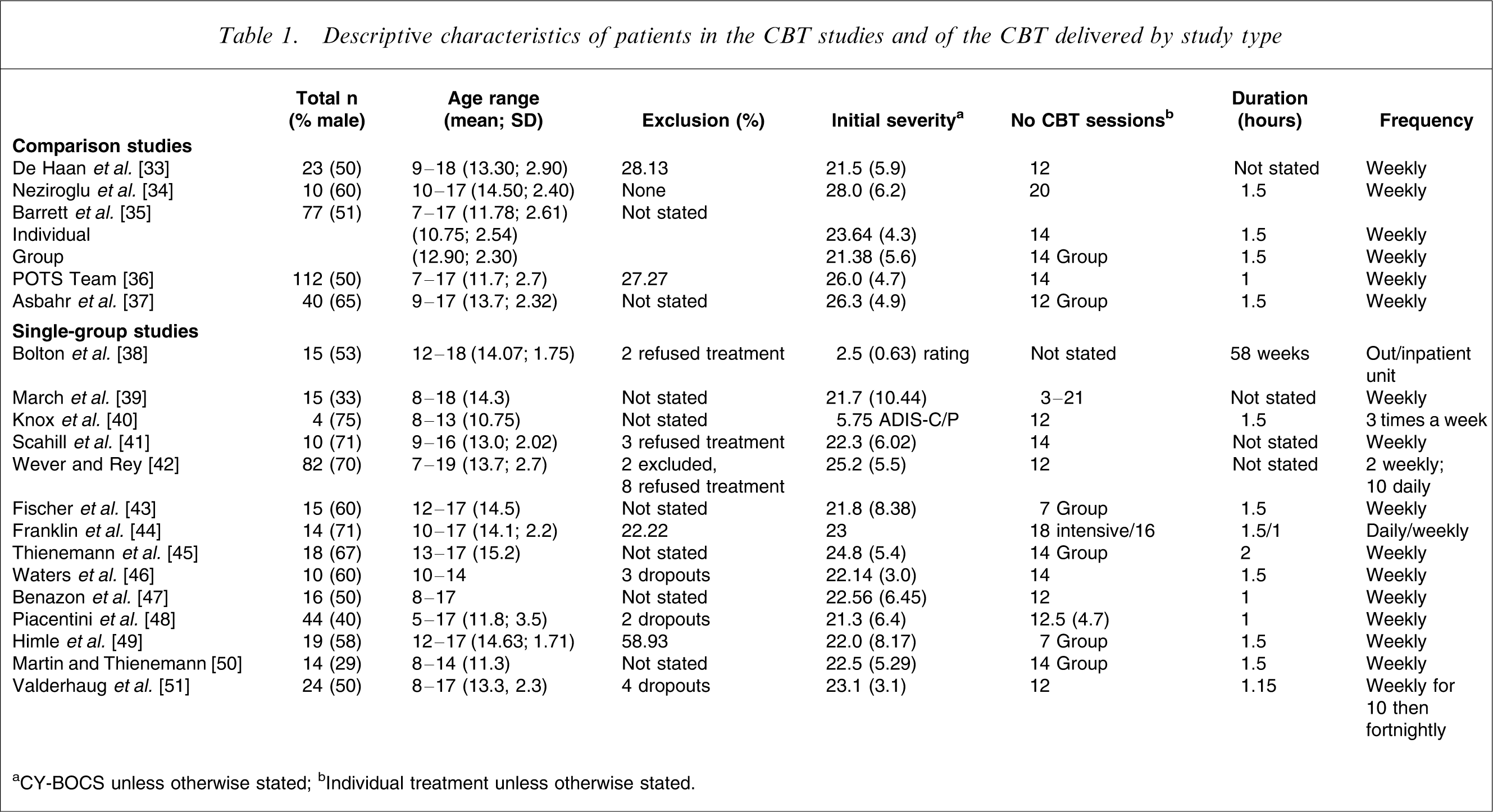
aCY-BOCS unless otherwise stated; bIndividual treatment unless otherwise stated.
The one-group studies also had a total of 300 children and adolescents ranging in age from 5 years to 18 years. The percentage of males in each group ranged from 29 to 75. The ethnicity of the majority of participants was American with the exception of one British study [38], two Australian studies [42, 46], and one Norwegian study [51]. Participants were diagnosed with OCD mainly with the use of an unstructured clinical interview, except for two studies that used the ADIS-C [48, 46], and three studies that used the Schedule for Affective Disorders and Schizophrenia for School-Age Children [47, 49, 51]. Of those participants assessed for eligibility, 22% [44], 59% [49], and 2% [42] were excluded, while the numbers excluded in the remaining studies were not reported.
Cognitive–behavior therapy protocols
The comparison studies delivered CBT interventions of between 12 to 20, 1 or 1.5-hour weekly sessions with total hours of treatment of 30 hours for Neziroglu et al.[34], 21 for Barrett et al.[35], 18 for Asbahr et al.[37], 14 for the POTS Team [36] and 12 hours for de Haan et al.[33]. In Neziroglu et al.[34], therapy focused exclusively on exposure with response prevention. For the other studies [33, 35–37], therapy was multi-modal, manualized and had equivalent components including psycho-education, cognitive therapy and ERP. These studies were based on a similar protocol [2, 52] and included parent involvement (all sessions for Barrett et al.[35], at least three sessions for POTS [36] with additional sessions determined by the symptom picture and developmental stage of the child, two sessions in Asbahr et al.[37] and not specified for de Haan et al.[33],). Barrett et al.[35] also included sibling involvement and compared CBT delivered in an individual or group format to a waitlist condition. Except for Neziroglu et al.[34] and Asbahr et al.[37], which did not report on fidelity checks, fidelity to the treatment protocol was protected by regular supervision of therapists in all studies. Barrett et al.[35] reported strong concordance between actual sessions and the manual using videotapes of sessions.
The one-group studies described between seven and 18 sessions of 1, 1.15, 1.5 or 2 hours. Most sessions were provided weekly except for: Knox et al.[40], which had three sessions per week; Wever and Rey [42], which had 10 daily and two weekly sessions; and Franklin et al.[44], which had two CBT conditions, an intensive condition with 18 daily sessions and a group condition with 16 weekly sessions. Bolton et al.[38] did not stipulate the number of sessions but gave the number of months of inpatient and outpatient treatment for each participant. Treatment in the one-group studies was mostly administered as individual sessions except for five studies that ran group treatment [43–45, 49, 50]. Both individual and group treatment focused on exposure and response prevention with varying levels of psycho-education, parent training, and pharmacology included. Nine one-group studies [39, 41, 43–47, 49, 50] based their treatment on the protocol developed by March [2] or were similar to methods of March et al.[39]. Four other studies used different manualized treatments [40, 42, 48, 51]. Bolton et al.[38] described their treatment as primarily behavioural (response prevention) and also included family therapy, medication, psychotherapy, and ‘milieu’ therapy. The amount of parent involvement varied between studies from an optional parent session [49], to weekly 30-minute parenting skills sessions [46], to weekly 60-minute parent session and 30-minute combined child and parent session [50]. The number of participants concurrently on medication in the one-group studies ranged from 21% [51] to 100% [42]. Fidelity to the treatment protocol was protected by regular supervision of therapists in the individual treatment studies, except for Bolton et al.[38], March et al.[39], Wever and Rey [42], and Martin and Thienemann [50], which did not report their fidelity checks.
Non-CBT comparison group
Four of the comparison studies used a medication comparison group (clomipramine [33], fluvoxamine [34], sertraline [36, 37]), one [36] had a pill placebo control and one [35] had a waitlist control. In the POTS Team [36] CBT was combined with medication in one group. Asbahr et al.[37] compared group CBT with medication. The medication administration was standardized and well-described. All control groups were assessed at post-treatment except for Barrett et al.[35], where it was assessed after 4 to 6 weeks wait because of ethical concerns.
Risk of bias
The methodological criteria required to lower risk of bias were based on the various aspects of study design, implementation, analysis and reporting that are specified for controlled trails [53], and also on consideration of the additional threats to internal validity for one-group designs. Because different aspects of study design are important to risk of bias for the two types of studies, only the relevant design and implementation aspects was used in assessing quality. Two raters independently judged each study on the criteria described below, allocating points from 0 to 2 according to the degree there was evidence that the study met the criteria. The total score was used to classify the studies as low, low to moderate, moderate to high, or high risk of bias. Any disagreements between the raters in assignment of a study to a risk category were resolved through discussion.
Risk of bias: comparison studies
Selection bias
The POTS Team [36] reported on the method of randomization and on allocation concealment that were adequately done. De Haan [33] clarified the method of randomization and reported on allocation concealment, which appear adequate. Neziroglu et al.[34], Barrett et al.[35] and Asbahr et al.[37] did not report on the method of randomization or on allocation concealment. In Barrett et al.[35], the process of block randomization described suggested quasi-randomization. De Haan et al.[33], the POTS Team [36] and Asbahr et al.[37] checked for baseline comparability on all outcomes, demographics and co-morbidity. Barrett et al.[35] checked for comparability on age and co-morbidity but not on outcomes and made appropriate adjustment in the analysis for the between-group age difference. Neziroglu et al.[34] did not check for baseline comparability.
It was not possible to blind participants and therapists in the CBT conditions. For the pills-only conditions in the POTS Team [36] (sertraline, placebo), both participants and psychiatrists were blinded to condition. There was no blinding in the CBT-combined-with-sertraline condition in the POTS Team [36]. De Haan et al.[33] and Neziroglu et al.[34] do not report on blinding for their medication condition. De Haan et al.[33], Barrett et al.[35] and the POTS Team [36] described processes to enhance the fidelity of the delivered intervention with the manual by using supervision by experienced clinicians. Barrett et al.[35] formally assessed protocol adherence by the therapists and found it to be good. Neziroglu et al.[34] and Asbahr et al.[37] do not describe any fidelity or quality assurance processes.
Detection bias
Blinding of outcome detection was not possible for participant self-report measures. For the CY-BOCS and other clinical-rated instruments, assessors were clearly blinded in the POTS Team [36] and Asbahr et al.[37]. De Haan et al.[33] and Neziroglu et al.[34] do not report on the status of assessors. Barrett et al.[35] described the assessors at the initial assessment as blind to study hypotheses but it is not clear if assessors at post-treatment for CY-BOCS were also blinded. Barrett et al.[35] compared endpoint status on all measures assessed 4 to 6 weeks after initial assessment for the waitlist condition and after 12 weeks for CBT conditions.
Attrition bias
The POTS Team [36] described flow of participants and reasons for attrition from all conditions and performed intention to treat analyses using last observation carried forward.
Barrett et al.[35], de Haan et al.[33] and Asbahr et al.[37] reported loss to follow-up for the CY-BOCS of two, one and two case(s), respectively, but did not use intention-to-treat analyses. There was no attrition at post-treatment in Neziroglu et al.[34].
Summary
The POTS Team [36] study overall had a low risk of bias, while de Haan et al.[33] and Neziroglu et al.[34] were considered as low to moderate risk of bias because they failed to specify method of allocation concealment, whether assessors were blinded, and did not use intention-to-treat analyses. Barrett et al.[35] was judged as moderate risk of bias arising from the method of randomization, between-group differences in timing of endpoint assessments, non-blinded post assessment and lack of intention-to-treat analyses. Asbahr et al.[37] was also considered moderate risk of bias because of lack of specification of method of randomization and incomplete reporting of recruitment, flow-though study and post-test data.
Risk of bias: one-group studies
Selection bias
Stability of symptoms at pre-treatment is important for meeting the threat to internal validity of one-group studies due to maturation effects or symptom variability. Children and adolescents who are on improving trajectories before commencement of treatment are likely to bias the strength of prior to post-treatment change. None of the studies use multiple pre-treatment assessments to establish a stable baseline. Participants recruited from treatment settings are more likely to have a stable symptom pattern than those recruited from community settings or via media announcements. Bolton et al.[38], Wever and Rey [42], and Valderhaug et al.[51] recruited participants from community outpatient and inpatient mental health settings and approached consecutively referred children and adolescents to take part. Martin and Theinemann [50] and Thienemann et al.[45] recruited from a specialist clinic and reported long durations of illness for all participants. March et al.[39] and Waters et al.[46] included recruitment via advertisement as well as referrals to their clinics, while the other studies do not report details of referral and recruitment procedures.
Detection bias
Only Himle et al.[49] clearly identifies outcome assessors as blinded, although blinding may be inferred in Thienemann et al.[45], Waters et al.[46] and Martin and Theinemann [50]. The other studies either do not use blind assessors for measurement of outcomes [41, 42, 51] or do not report of the status of assessors [38–40, 43, 47, 48].
Attrition bias
All the studies report dropout and/or refusal rates if relevant. Wever and Rey [42] report that eight children refused treatment but do not provide reasons. There were generally very small numbers of dropouts from the studies. Only Bolton et al.[38], March et al.[39], and Scahill et al.[41] accounted for dropouts and included them in their analysis. None of the studies corrected effect sizes for testing effects.
Summary
In addition to the lack of a control group, the main sources of differential bias in one-group studies come from the selection process and non-blinding of outcomes assessors. While none of the studies could be clearly considered low risk of bias, Bolton et al.[38], Thienemann et al.[45], Himle at al.[49] and Martin and Theinemann [50] had overall a low to moderate risk of bias. Knox et al.[40], Scahill et al.[41] and Waters et al.[46] were judged to have moderate to high risk of bias because of an additional threat to validity due of very small sample size. The other studies had a moderate risk of bias arising from recruitment issues and non-blinding of assessors.
Outcomes
OCD severity
All of the comparison studies reported data on the clinician-rated CY-BOCS that has strong reliability and validity [54] and is considered the gold standard of outcome assessment for OCD. It assesses the frequency and intensity of obsessions and compulsions, as well as the amount of interference and degree of distress they produce. Neziroglu et al.[34], Barrett et al.[35] and Asbahr et al.[37] also reported data about OCD severity using the clinician-rated single-item National Institute of Mental Health Global Obsessive Compulsive Scale (NIMH-GOCS) and also on the Clinical Global Impressions Severity and Improvement Scales (CGIS: CGII). All the one-group studies also used the CY-BOCS except for Bolton et al.[38], which used a three-point clinician-rated OCD severity and Knox et al.[40], which used the ADIS-C eight-point clinician rating of severity. Martin and Thienemann [50] and Valderhaug et al.[51] also used the Child Obsessive Compulsive Impact Scale (COIS) [55] that assesses functional impairment at school and home and in social relationships due to the OCD, using child and parent reports. All studies provide data that allowed comparisons of rates of dichotomized outcomes (non-remission from OCD) at post-treatment or provide some index of improved/not-improved status, with the exception of Fischer et al.[43].
How effective is CBT for pediatric OCD?
Table 2 presents the effects sizes for severity of OCD symptoms and the relative or absolute risk of remaining disordered at post-treatment grouped by type of study and nature of comparison. Three comparisons (two studies [35, 36]) tested the efficacy of CBT against a non-active control group, and 14 one-group studies looked at pre–post change with CBT. All of the studies reported statistically significant effect sizes showing superiority or efficacy of CBT.
Effect sizes (95% CI) favouring CBT group for severity of OCD symptoms and for risk of not improved status by study type
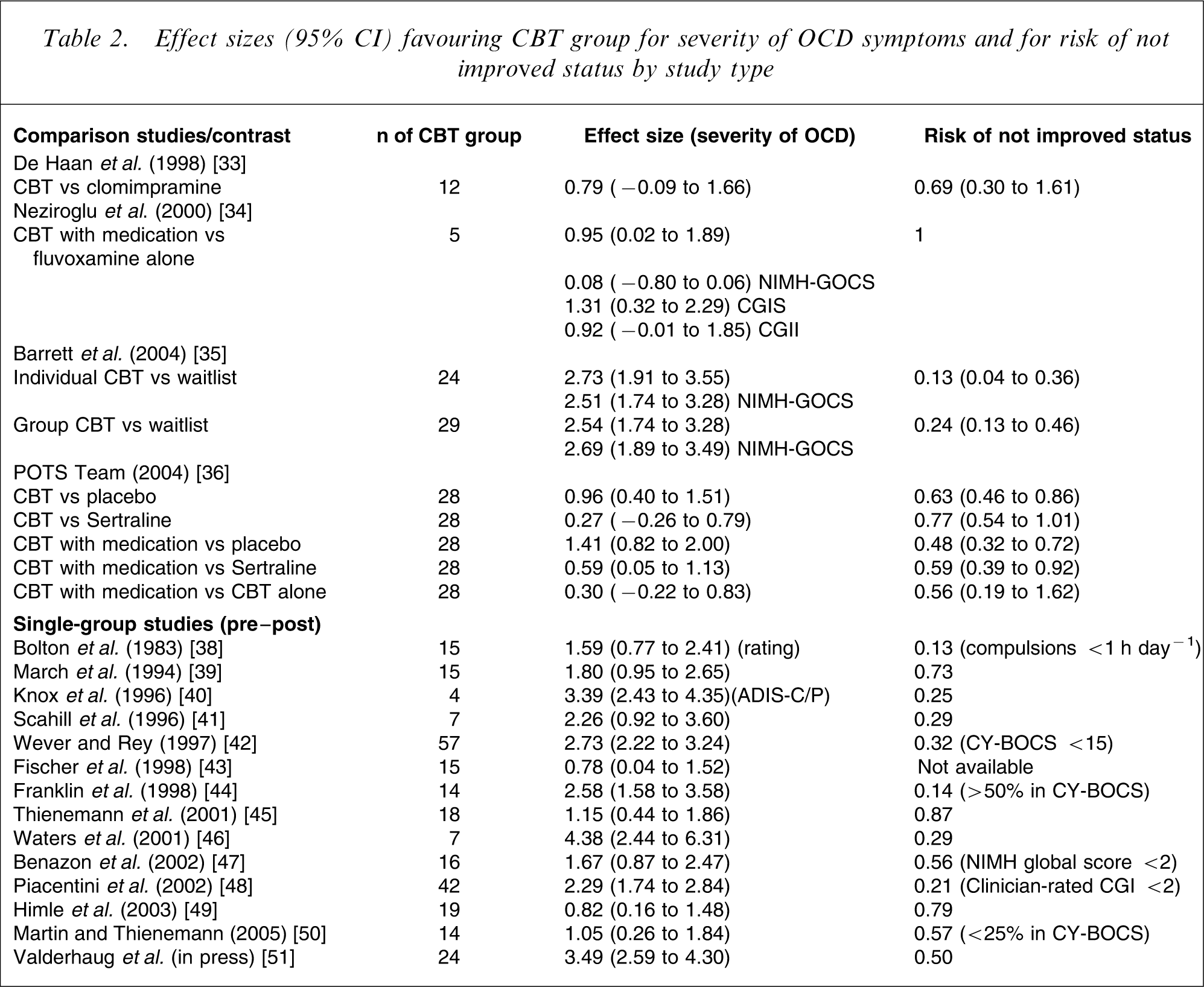
In regard to the controlled trials, because of marked statistical heterogeneity (I2=89.3%) between Barrett et al.[35] and the POTS Team [36] pooling of estimates was not undertaken. The POTS Team [36] with low risk of bias showed an effect size of 0.96 (95% CI = 0.40–1.51) in favour of the CBT group. Barrett et al.[33] reported substantially larger effects sizes of 2.73 (95% CI = 1.91–3.55) in favour of individual CBT and of 2.54 (95% CI = 1.74–3.28) in favour of group CBT. Outcomes from the one-group studies ranged from a mean effect change in CY-BOCS scores favoring the CBT group of 0.78 (95% CI = 0.04–1.52) [43] and 0.82 (95% CI° = 0.16–1.48) [49] to mean effect sizes of 2.58 (95% CI = 1.58–3.58) [44], 2.73 (95% CI = 2.22–3.24) [42] and 3.49 (95% CI = 2.59–4.30) [51].
The POTS Team [36] used a cut off of > 10 on the CY-BOCS to classify participants as still having OCD. The relative risk of participants continuing to have OCD at post treatment was 0.63 that of the risk in the placebo group (95% CI = 0.46–0.86). Barrett et al.[35] used the ADIS-P diagnosis and found the relative risk of participants continuing to have OCD at post-treatment was 0.13 (95% CI = 0.4–0.36) the risk in the waitlist group for the individual-CBT group and 0.24 (95% CI = 0.13–0.46) the risk in the waitlist group for the group-CBT group. Risk of not-improved status at post-treatment in the one-group studies ranged from highs of 0.87 [45], 0.79 [49] and 0.73 [39] to lows of 0.29 [46], 0.21 [48] and 0.13 [38]. It needs to be noted that the criteria for not improving varied considerably between studies.
Figure 1 depicts each study's effect size estimates for OCD severity with their 95% confidence intervals grouped by type of study. With the exception of Bolton et al.[38], all of the studies have been carried out since 1994. The figure demonstrates that the range of estimates for improvement in symptom severity with CBT overall is very wide (0.82 to 4.38), with no indication of a narrowing of this variability over time. The two relevant randomized controlled trials produced considerably different estimates, although the effect size of the study with the lowest risk of bias [36] (ES = 0.96) was the third lowest of all the studies. Ten of the single-group studies produced point estimates of the CBT effect outside of the upper 95% confidence interval for the POTS Team study.
Effect sizes (95% CI) for OCD severity for each study by type of study.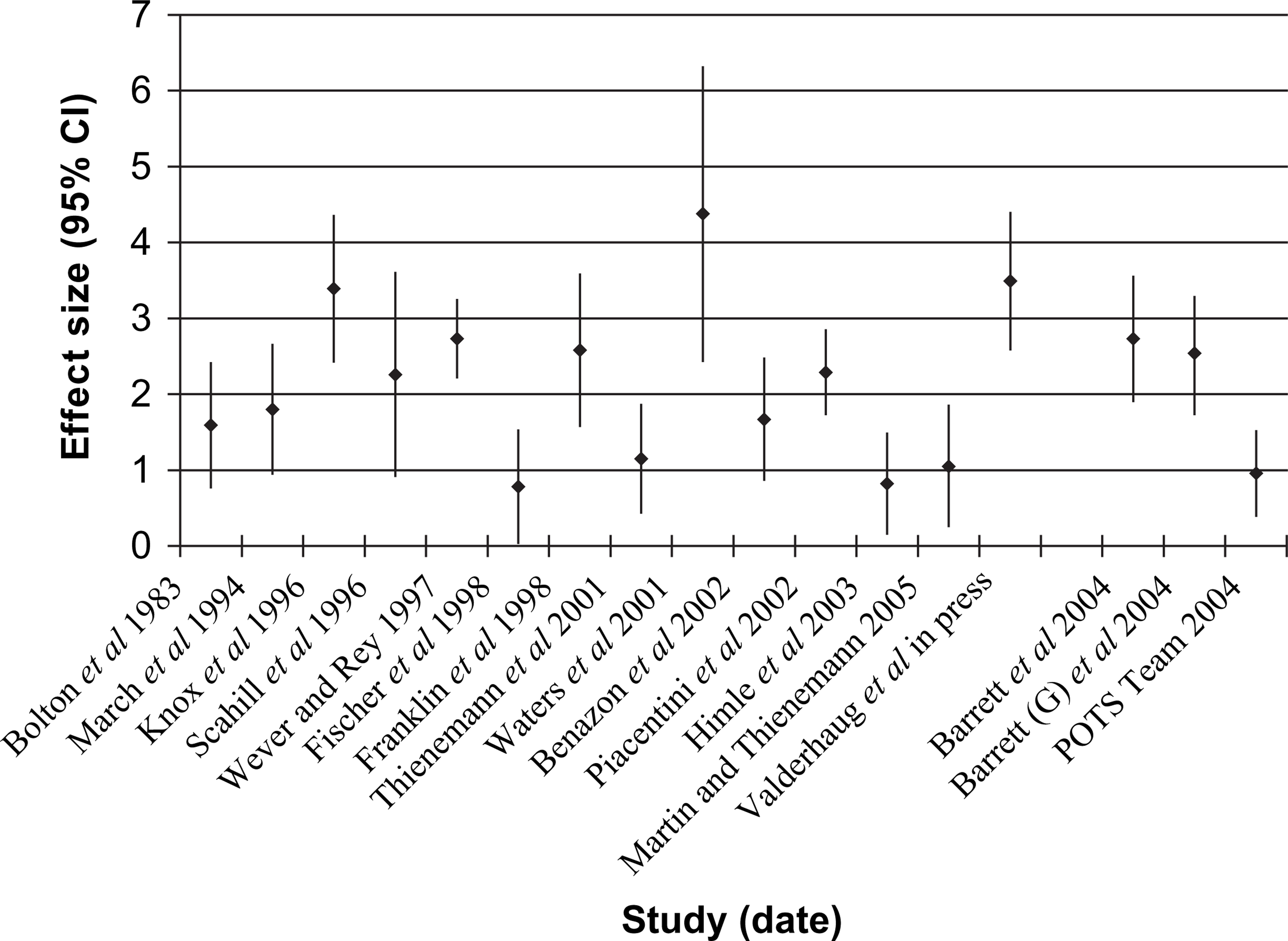
Does quality matter?
Effect sizes for the single-group studies were adjusted by subtracting the testing effect. This effect for the CY-BOCS was estimated as the average standardized change (dc=(Xpost–Xpre)/SDpre) in non-treated groups in three studies [35, 36, 54]. Barrett et al.[35] reported a standardized mean increase in CY-BOCS total scores for the waitlist group of 0.19, the POTS Team [36] reported a standardized mean decease for the placebo-control group of 1.12, and Storch et al.[54] reported a standardized mean decrease for their standardization sample in estimating test–retest reliability of 0.56. The average standardized change from these three studies equals −0.49. This is similar to the average effect size for the drug-placebo groups in controlled trails of SSRIs for pediatric OCD of −0.48 [11]. Figure 2 depicts the adjusted effect sizes studies grouped in terms of categories of risk of bias. The figure clearly shows that lower-risk-of-bias studies produce lower effect sizes. Only one study (20%) in the low- and low-to-moderate-risk-of-bias categories report adjusted effect sizes above 1, while 11 (92%) in the moderate- and moderate-to-high-risk-of-bias category report effects about 1, and seven (58%) report effects above 2.
Effect sizes adjusted for testing effect for each study by risk category (∗RCT).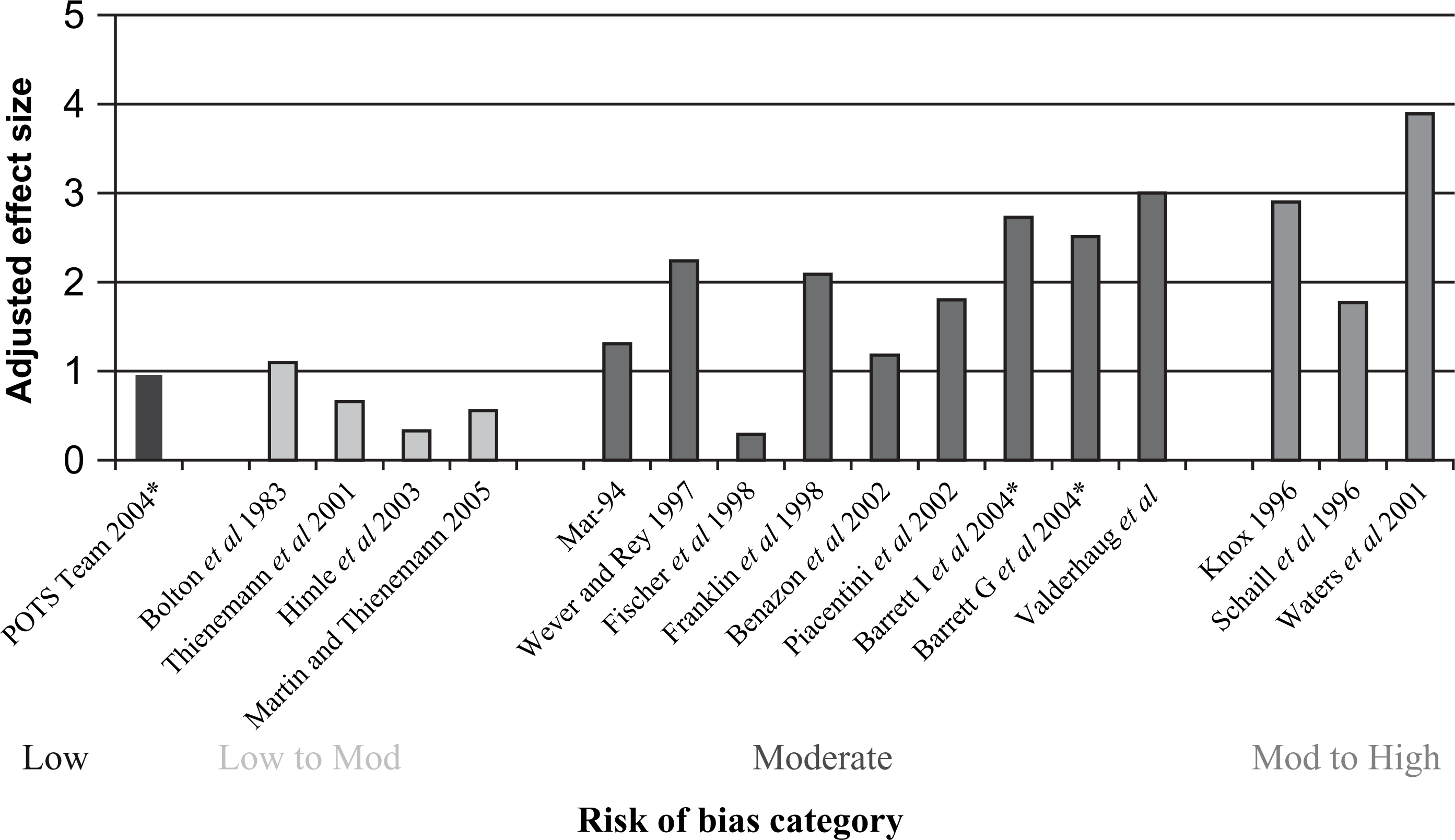
CBT versus medication
Three comparisons [33, 36, 37] tested the relative efficacy of CBT alone against medication (clomipramine, sertraline). The pooled weighted mean difference from two of these studies [33, 36] showed equivalence of the two treatments (WMD = − 3.87, 95% CI = − 8.15–0.41). Neither study found a significant difference in post-treatment CY-BOCS between the treatments (CBT versus clomipramine WMD = − 8.50, 95% CI = 17.44–0.44 [33]; CBT versus sertraline WMD = − 2.50, 95% CI = − 7.37–2.37 [36]). Asbahr et al.[37] does not provide endpoint scores on total CY-BOCS and there are inconsistencies between outcomes presented in tables and those depicted in figures in the study write-up. While they conclude that the two treatments (group CBT; sertraline) are equally beneficial, the standardized mean difference could not be computed. Asbahr et al.[37] have not, to date, responded to request for clarification of their data.
To allow comparability for binary outcomes, we classified de Haan et al.[33] participants into those continuing to have OCD at post-treatment using the POTS Team criteria (CY-BOCS >10) [36]. The pooled data indicated that CBT and medication had an equivalent proportion of participants continuing to have OCD at post-treatment (RR = 0.75, 95% 0.54 to 1.05). In de Haan et al.[33], participants in the CBT group (5/12) and in the medication group (6/10) were equally likely to continue to have OCD at post-treatment (RR = 0.69, 95% CI = 0.30–1.61). In the POTS Team [36], participants in the CBT group (17/28) and medication (22/28) were also equally likely to continue to have OCD at post-treatment (RR = 0.77, 95% CI = 0.54–1.10). Asbahr et al.[37] do not provide numbers for those with not-improved status at post-treatment. They do report marked discontinuation effects in the sertraline group by 4 weeks post treatment.
CBT combined with medication versus other groups
The POTS Team [36] post-treatment data showed a superior effect of CBT combined with medication relative to placebo (SMD = − 10.30, 95% CI = − 14.06– − 6.54). Neziroglu et al.[34] and the POTS Team [36] compared CBT combined with medication relative to medication alone. Outcomes on the CY-BOCS at the two post-treatment points (43 and 52 weeks) were averaged in the Neziroglu et al. study [34]. The pooled weighted mean difference in favour of the combined treatment was −4.55 (95% CI = − 7.40– − 1.70), with both studies reporting a significant superior effect for CBT combined with medication relative to medication alone. The POTS Team [36] also compared CBT combined with sertraline with CBT alone. Examination of unadjusted CY-BOCS scores post treatment showed that the two treatments were not significantly different (SMD = − 2.80, 95% CI = − 7.55–1.95).
Neziroglu et al.[34] participants were classified into those continuing to have OCD at post-treatment using the POTS Team criteria (CY-BOCS >10) [36]. As all participants in both groups scored above 10, the relative risk could not be estimated. In the POTS Team [36], participants in the CBT combined with medication group (13/28) were significantly less likely to continue to have OCD at post-treatment compared to the medication alone group (22/28) (RR = 0.59, 95% CI = 0.38–0.92) and the placebo group (27/28) (RR = 0.48, 95% CI = 0.32–0.72). The relative risk of participants continuing to have OCD in the CBT combined with medication group (13/28) was not significantly different from that of the CBT alone group (17/28) (RR = 0.76, 95% CI = 0.47–1.26).
Other primary outcomes
Barrett et al.[35] compared the group and individual CBT to their waitlist condition on the NIMH-GOCS measure. Both CBT groups were superior to the waitlist group with a mean difference of 5.50 (95% CI = − 6.74– − 4.28) in favour of individual CBT and of 5.69 (95% CI = − 6.87– − 4.51) in favour of group CBT. Bolton et al.[38] reported an effect size for prior to post change for the CBT group on a clinician OCD severity rating of 1.59 (95% CI = 0.77–2.71), while Knox et al.[40], using the ADIS-C/P severity rating, found an effect size for prior to post change of 3.39 (95% CI = 2.43–4.35).
In terms of life-functioning, both studies [50, 51] that used the COIS reported higher effect sizes for parent's report of improvement (SMD = − 0.90 [50]; SMD = − 0.96 [51]) than for the child's self-report of functioning (SMD = − 0.22 [50]; SMD = − 0.64 [51]). These effect sizes for improvement in function were less notable than the effect size for symptom severity in the respective studies.
Discussion
This systematic re-examination of the evidence clarifies the benefits of CBT for pediatric OCD. It found that in terms of post-treatment levels of symptom severity and interference due to symptoms using the gold standard outcome (CY-BOCS) that CBT is an empirically supported treatment. There remains, however, considerable variability in estimates of the strength of CBT's efficacy. It is difficult in view of the elevated risk of bias in many of the studies and the association between low risk of bias and lower effect size estimates to confidently attribute the larger effect sizes to intervention differences. This review suggests that the best current estimate of CBT efficacy relative to no treatment is about 1 standardized mean difference equivalent to a treatment effect of about 8 points on the CY-BOCS. The highest confidence is in the results of the POTS Team study (SMD = 7.5; 95% CI = 3.45–11.55 [36]). This effect represents a reduction in the risk of continuing to have OCD of about 37% (95% CI = 14–54%). Treatment of about three pediatric OCD patients with CBT (95% CI = 2–7) would be needed to facilitate remission of OCD in one extra child or adolescent.
It is interesting to compare these conclusions with the pooled pre–post effect size of 1.98 (95% CI = 1.40–2.56) for eight uncontrolled and two controlled studies of CBT reviewed by Abramowitz et al.[12]. The present review demonstrates that effect size estimates for CBT's efficacy for pediatric OCD are significantly moderated by study design and by the risk of bias in the studies. In addition, testing effects in pre–post designs play a substantial role in elevating effect sizes estimates. Taken together, these considerations question the usefulness of the results of uncontrolled trials in meta-analytic thinking about the benefits of a treatment. While they have an important function in initial assessments of the potential of new treatments, their overrepresentation in the efficacy evaluations of CBT for pediatric OCD (14 of 16; 88%) highlights the fact that the paucity of high-quality studies places ongoing questions on the accuracy and precision of conclusions about the degree of benefit associated with CBT. This is further illustrated by the differences in precision of the best estimate of CBT (Hedge's g = 0.96; 95% CI = 0.40–1.51 [36]) when compared to the effect size estimates for OCD-specific medications (Hedge's g = 0.46; 95% CI = 0.37–0.55 [9]). One implication of the current study for clinical research is that a well-conducted replication of POTS [36] would be important for enhancing confidence about the magnitude of the benefits of CBT for pediatric OCD.
The evidence reviewed here places CBT as an equally effective first-line treatment with SSRIs in the treatment of OCD in children and youth. The two treatments could not be distinguished in terms of reductions in symptom severity and interference, or in post-treatment remission rates in three head-to-head studies. Rather, the evidence from the current review provides some support for CBT combined with medication as the ‘treatment of choice’. This advice is clear in regard to a non-active treatment with a strong relative effect for CBT combined with medication (Hedge's g = 1.14, 95% CI = 0.82–2.00) but also relative to medication alone (pooled effect size of 0.68, 95% CI = 0.21–1.15). But the utility of the combined treatment as treatment of choice relative to beginning with CBT alone is less compelling (Hedge's g = 0.30, 95% CI = − 0.22–0.83). While favoring the combined treatment over CBT alone, the confidence interval around this estimate from one randomized controlled trial does not exclude the possibility of a superiority of CBT alone. Clearly, decisions to begin treatment for pediatric OCD with CBT alone or with CBT and medication rather than medication alone are warranted on the basis of the reviewed evidence. Of course, a number of other important issues such as patient preference, likelihood of adverse effects of medication, the availability of skilled CBT practitioners, cost to patients and the patient's treatment history, need to be included in decision-making in individual cases. The small number of quality studies also dampers the confidence with which decisions made on the basis of the current evidence can be made.
In addition to the caveats to conclusions due to the quantity and quality of studies, there are a number of other limitations to the current evidence that need to be considered. Most studies used the CY-BOCS as the main outcome indictor. While it is regarded as the ‘gold standard’ measure for child and adolescent OCD and has good reliability and validity [54], there are some recent indications that the CY-BOCS total score may underestimate symptom severity in many pediatric patients [56, 57]. In addition, there are no normative data on the CY-BOCS for typical children and youth so it is not possible to assess the clinical importance of treatment-related reductions in terms of the likelihood of end-state functioning being more typical than disordered. The cut-offs used to categorize responders or remitters in the studies reviewed are rules of thumb or based on adult data. There is some suggestion [58] that it may be more normative for younger children to spend more time on behavioural and cognitive rituals making it difficult to define obsessions and compulsions as disordered or problematic by using measures that rely on frequency and duration. In addition to better normative data across the developmental span for the CY-BOCS, it would be useful in supplement assessment of the clinical importance of treatment effects with sound measures of the impact of the OCD on the child's or youth's life-functioning. The Child Obsessive Compulsive Impact Scale [55] is a promising measure in this regard and has been shown to be sensitive to treatment effects in medication trails. It is also beginning to be applied in CBT outcome evaluations [50, 51]. The results of these two studies suggest that the degree of symptomatic improvement may not be reflected in functional improvement particularly from the child or adolescent's point of view. While there are best bets for the estimates of the effects of CBT on pediatric OCD it is still not clear what the clinical and functional importance of these effects are.
Besides the implications for practice coming out of this review, there are a number of general conclusions about the direction of research into the benefits of CBT for pediatric OCD that can be made from its observations. One is the need to improve the quality of the design, implementation and reporting of evaluations of CBT. The need for further randomized controlled trials carried out with low risk of bias is clear, with the direction of findings from the reviewed studies suggesting further comparisons of CBT with medication and CBT combined with medication are a priority. Given other important questions such as age-related response to both CBT and medication, and whether sequencing between CBT and medication may optimize benefits, such studies should attempt to both strengthen and extend the current evidence. Recently, Valderhaug et al.[51] tested the effectiveness of CBT for OCD in children and youth in a community-based clinical setting in an ‘open’ trial. Because availability of competent CBT practitioners for pediatric OCD is often an obstacle to treatment choice, it is important to carry out further controlled evaluations in general child and adolescent clinical settings with clinicians trained and supervised in the delivery of CBT protocols to OCD patients. As well, trials of alternative modes of delivery of CBT for pediatric OCD, such as group and self-directed approaches, are warranted in order to increase access to empirically-validated treatments.
Overall, the current review reaffirms CBT as a promising treatment for OCD in children and youth. On the basis of the reviewed evidence, CBT should be regarded as an equivalent first-line treatment to anti-OCD medication with the potential to lead to better outcomes when combined with medication than medication alone can provide. Nevertheless, there are significant problems with the quantity and quality of CBT trials that obscure the degree and nature of the benefits associated with CBT. Further open trails are unlikely to clarify these benefits. Rather, the direction for clarification should be towards well-conducted controlled trials similar to the POTS Team [36] study using a range of outcomes including measures of life-functioning. In the interim, clinical researchers and policy-makers need to attend to ways in which CBT might be made widely available as a treatment option for children and youth who suffer from OCD.
Footnotes
Acknowledgements
Joanne Speyer and Kaarin Anstey undertook the risk-of-bias ratings for the one-group and controlled studies, respectively.
