Abstract
Schizophrenia has been associated with impaired emotion processing that may present itself phenomenologically as reduced expression and experience of affect, as well as excessive emotionality [1–3]. Overall, male patients tend to be more withdrawn and exhibit blunted affect whereas female patients display more irritability and inappropriate affect [4, 5]. In recent years studies have used functional neuroimaging methods to investigate neural substrates of impaired emotion processing in schizophrenia but were unable to uncover any potential sex differences; most likely due to the fact that tested groups of patients consisted exclusively or predominantly of male subjects. For example, Schneider et al. observed diminished amygdala activation in patients (13 men) relative to controls during sad mood induction [6]. In a different study, Crespo-Facorro et al. have found that relative to control participants, patients (16 men, two women) exhibited diminished activation in limbic and paralimbic cortex, including insula, ventral striatum, and parahippocampal gyrus, during experience of unpleasant odors, and recruited a compensatory set of frontal cortical regions instead [7]. Paradiso et al. have shown that despite matched performance with healthy volunteers, patients (16 men, two women) did not activate the amygdala during conscious evaluation of aversive images [8]. Similarly, Gur et al. used the task of facial emotion discrimination among happiness, sadness, anger, fear, and disgust, and observed that schizophrenia patients (10 men, four women) were characterized by reduced activation in the amygdala and hippocampus relative to control subjects [9]. Takahashi et al. have also observed diminished activation of several regions including the amygdala, hippocampus, thalamus, basal ganglia, midbrain, medial prefrontal cortex and cerebellum, in patients (10 men, five women) while viewing unpleasant pictures [10]. Other studies however, revealed exaggerated limbic and corticolimbic activations in schizophrenia patients relative to healthy controls during experience and/or exposure to affective stimuli [11–13]. These inconsistencies can be partly explained by a heterogeneous symptomatology of tested patients because some studies have linked anomalous cerebral activations during emotion processing with specific symptoms of schizophrenia [12–16].
We have suggested recently that the sex of tested patients may also play a significant role in the final results of functional neuroimaging studies [17], especially because there is rich body of data supporting the existence of sex differences in the cerebral function during processing of emotions in the general population [18–20]. For example, in a functional magnetic resonance imaging (fMRI) study investigating neural correlates of happiness and sadness, Wrase et al. observed stronger activations in men exposed to positive visual stimuli in the inferior and medial prefrontal cortex, while women exhibited stronger activations for affectively negative pictures in the anterior and medial cingulate gyrus [21]. Lee et al. found that male and female subjects used a different pattern of cerebral activations when processing emotional faces, particularly when they were portraying sadness [22, 23]. Thus, men exhibited bilateral frontal, right temporal and right lentiform activation, whereas women showed left parietal, left lentiform and right occipital activation. A meta-analysis of functional neuroimaging studies of emotion processing concluded that women more frequently activate midline limbic structures, including anterior cingulate, thalamus, midbrain, and cerebellum, whereas men exhibit more activation in the left inferior frontal and posterior cortex [24].
In our previous report we found a dissimilar pattern of differences between male and female schizophrenia patients to what has been observed in the general population. Specifically, we have found that relative to female patients, male patients exhibited significantly greater activations in the anterior and middle portion of the cingulate gyrus, the medial and bilateral dorsal prefrontal cortex, as well as the right inferior parietal cortex, during passive viewing of sad versus neutral film excerpts. In addition, men activated bilateral parahippocampal cortex, right amygdala and right superior temporal gyrus, whereas women did not exhibit any significant activations despite equivalent ratings of the experienced emotions [17].
The aim of the present investigation was to extend these results in a more carefully designed experimental paradigm and find out whether the effect we have observed is specific to sadness or whether it can be generalized over other emotions such as fear and disgust. Based on our previous report and the existing structural neuroanatomical literature suggesting reversed sexual dimorphism in schizophrenia patients in some corticolimbic structures [25–29], we hypothesized that exposure to aversive stimuli will result in significantly greater cerebral activations in schizophrenia men than in women in the areas implicated in the processing of emotions.
Method
Participants
Fifteen men and ten women diagnosed with schizophrenia according to DSM-IV independently by at least two psychiatrists were recruited to participate in the study. All patients were assessed with the Positive and Negative Symptoms Scale (PANSS) [30]. Patients were clinically stable with a mean duration of illness 5 ± 3 years. All patients were receiving treatment with at least one antipsychotic medication (four patients received one, eight received two, one received three, and one received four antipsychotics: haloperidol, n = 4, mean dosage 7.5±2.9 mg; zuclopentixol, n = 4, mean dosage 137.5 mg ±12.5; risperidone, n = 12, mean dosage 4.0±2.4 mg; olanzapine, n = 8, mean dosage 20.0±4.6 mg; and/or quetiapine, n = 12, mean dosage 470.8±182.7 mg). The groups of men and women were equivalent in terms of age (26.4±5.8 vs 28.4±9.1 years), years of education (10±3.4 vs 11±2.9 years), age at illness onset (22.9±2.7 vs 22.6±5.2 years), symptomatology (total, positive, negative, and general PANSS scales), medication status (chlorpromazine equivalence), and all were right-handed. Patients were clinically stable with no concomitant axis I or axis II disorder, and no medical or neurological disease. All subjects were judged by the attending physicians to be competent to give informed consent and capable of cooperating during the imaging procedure. They gave full, informed, written consent to participate in the study approved by the local ethics and scientific committees. Although it is somewhat surprising that there were no significant differences either in the illness onset or in the expressed symptomatology between men and women, this might have been due to a biased sample, that is, relatively well functioning patients who were willing to undergo testing with fMRI.
Neuroimaging procedure and analysis
Blood-oxygen-level-dependent (BOLD), echo-planar images (EPI; 28 5 mm thick slices, TR = 3 s, TE = 44 ms, flip = 90°, field of view =215 mm, 64×64 matrix) were acquired on a 1.5 T system (Magnetom Vision, Siemens Electric, Erlangen, Germany) during two experimental conditions: negative and neutral. Negative condition consisted of 44 emotionally laden negative pictures (e.g. plane crash, angry face, electric chair), whereas neutral consisted of 44 emotionally neutral pictures (e.g. boat, neutral face, chair). The two categories of pictures were selected from the International Affective Picture System (IAPS), were matched in terms of visual complexity, but differed in affective valence (5.74±1.47 vs 2.66±1.58) and in level of arousal (6.11±2.14 vs 2.97±2.08). Four blocks of negative and four blocks of neutral pictures, separated by periods of rest, were presented to the subjects in an alternating manner. Each picture was presented for a period of 2.88 s and each block, which lasted 31.68 s, consisted of 11 pictures. Blocks were separated by resting periods of 14.4 s, during which subjects viewed a blank cyan screen. Subjects were instructed to look carefully at the pictures and assess their subjective responses to the stimuli at the end of each run on the scale ranging from 0 (absence of any emotional reaction) to 8 (strongest emotional reaction ever felt in one's lifetime).
Data were realigned, spatially normalized, smoothed and statistically analysed using Statistical Parametric Mapping software (SPM99, Wellcome Department of Cognitive Neurology, London, UK). Following fixed-effects analyses of individual data, the single image for each participant was created based on the aversive minus neutral contrast and entered into random-effects analyses, which estimates the error variance for each condition across subjects, thus allowing one to make inferences on the population from which participants are deemed representative [31]. This procedure also alleviates the problem related to the unequal sample sizes of the groups. During this stage, any potential differences between men and women in the negative versus neutral condition were explored using a two-sample t-test, whereas within-group differences were assessed with one-sample t-tests for each group separately. All analyses were performed for entire brain volume at the corrected cluster level (p ≤ 0.05, thresholded at p ≤ 0.001) and the uncorrected voxel level (p ≤ 0.001) as implemented in SPM99 [32].
Results
There were no significant differences between men and women in the intensity of subjective responses towards aversive pictures (3.67±2.8 vs 4.9±3.3; p > 0.01). Random-effects within-group analyses demonstrated that during passive viewing of aversive versus neutral pictures, women activated right inferior frontal, left lingual, and left middle occipital cortices, as well as bilateral cerebellum. In comparison, men activated bilateral lingual and right inferior frontal cortex, bilateral hippocampal and left middle temporal gyrus, bilateral precuneus and cerebellum (Table 1). The between-group analysis (sex × condition interaction) revealed that men exhibited greater cerebral activations than women in bilateral thalamus, cerebellum, temporal gyrus, right occipital gyrus, left cuneus and posterior cingulate, while women showed greater activation than men in the left middle frontal gyrus (Table 2).
Areas of significant cerebral activation in schizophrenia during passive viewing of negative versus neutral pictures†
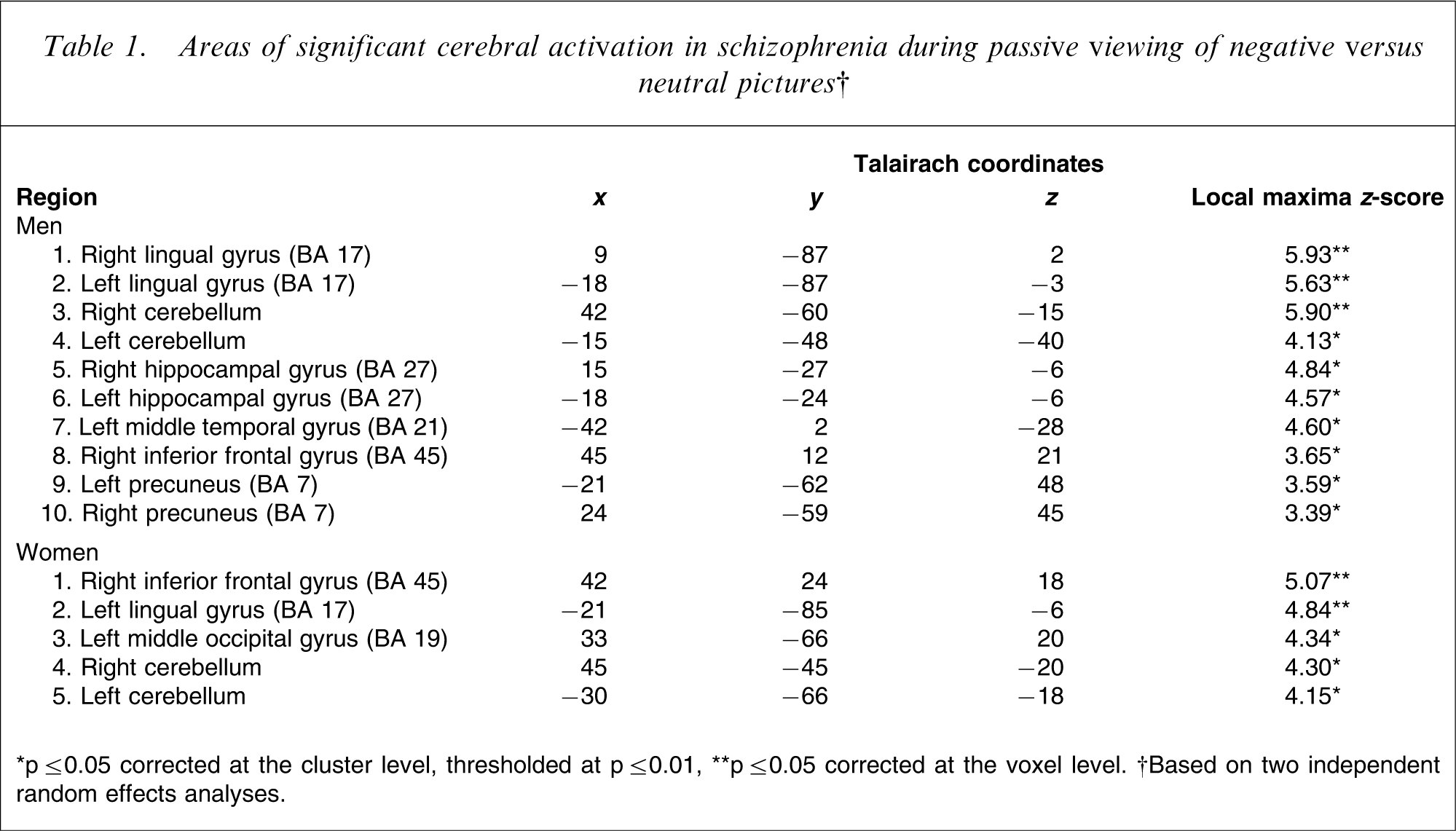
∗p ≤ 0.05 corrected at the cluster level, thresholded at p ≤ 0.01, ∗∗p ≤ 0.05 corrected at the voxel level. †Based on two independent random effects analyses.
Summary of the areas of significant differences in cerebral activation between schizophrenia men and women during passive viewing of negative versus neutral pictures†
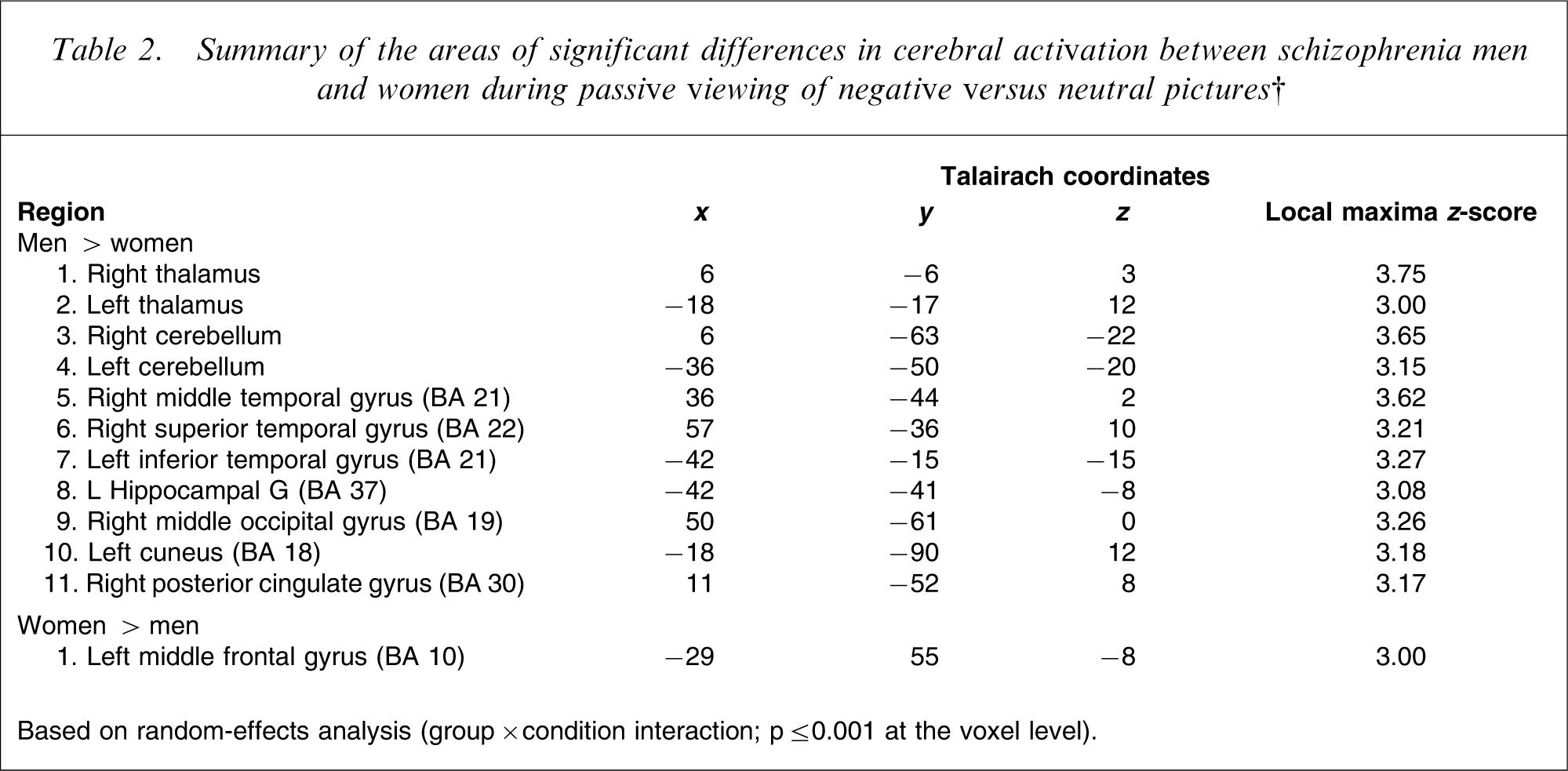
Based on random-effects analysis (group×condition interaction; p ≤ 0.001 at the voxel level).
Discussion
The data analyses revealed that despite no differences in the subjective rating of emotions evoked by the presented pictures, schizophrenia men exhibited more widespread and more intense activations than women (Figure 1). Thus, while both groups activated bilaterally cerebellum as well as occipital, parietal and prefrontal cortex, men exhibited additional activations in bilateral hippocampal and middle temporal gyrus. The direct comparison between the sexes indicated augmented activations in men relative to women in the thalamus, cerebellum, temporal and occipital cortex, as well as the posterior cingulate gyrus. In comparison, women showed significantly greater activations than men only in a circumscribed region of the left middle frontal gyrus.
Illustration of cerebral activations rendered on horizontal slices of the brain at different levels (three columns) in schizophrenia men (top panel) and women (bottom panel) during passive viewing of the negative versus neutral pictures.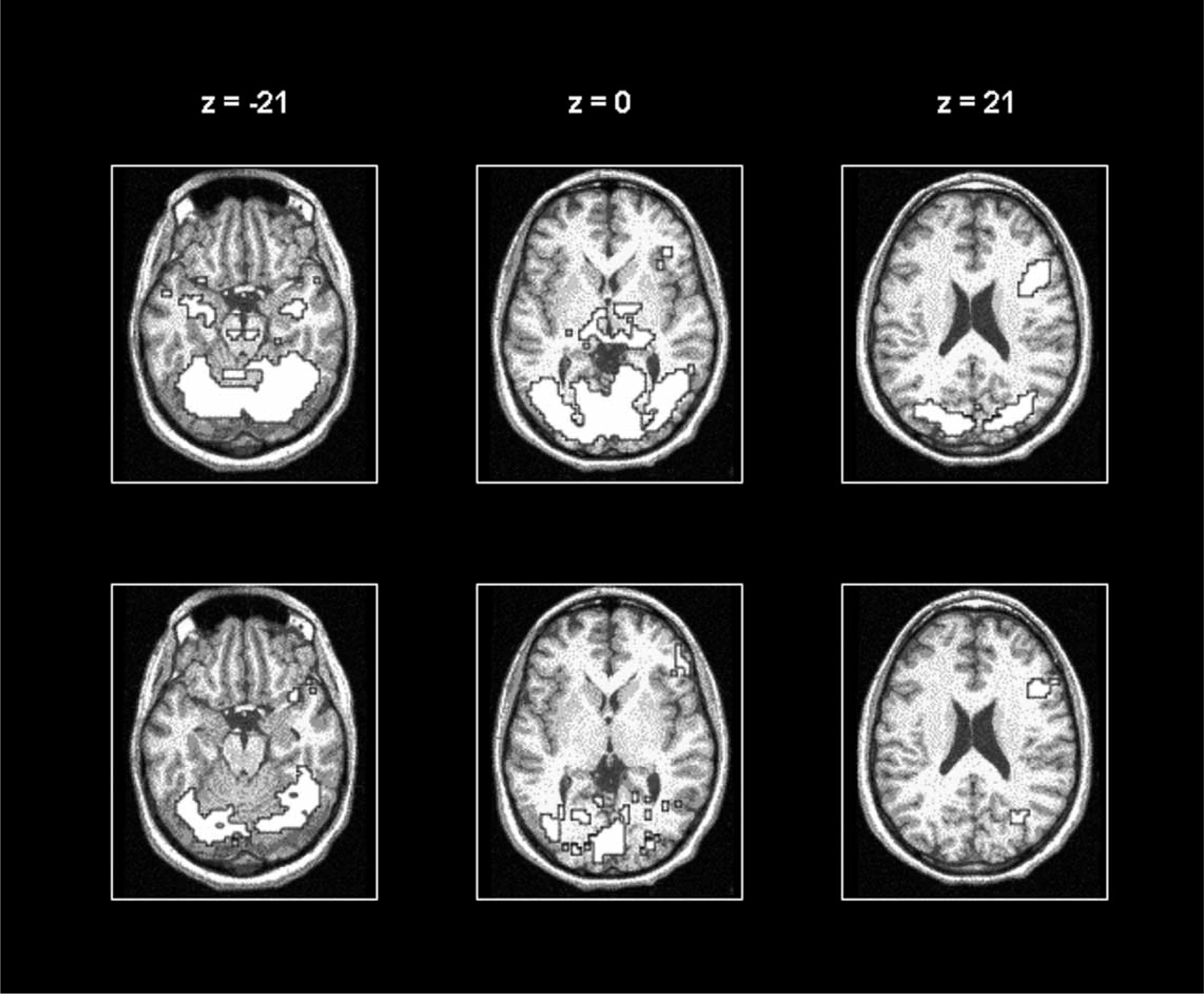
These results cannot be accounted for by differences between the groups in any measured demographic variable, symptomatology, medication, or subjective experience of negative affect, and imply that the difference stems from the cerebral function associated with the way in which men and women with schizophrenia process emotions. For example, in the general population women are more susceptible to emotion contagion [33] and exhibit greater psychophysiological responses to affective stimuli [34] than men. A meta-analysis of functional neuroimaging studies of emotion processing concluded that whereas women activate more frequently midline limbic structures, including anterior cingulate, thalamus, midbrain, and cerebellum, men exhibit more activation in the posterior cortex and the left inferior frontal region [24]. This difference has been attributed to the possibility that while women perceive and experience emotions more directly and viscerally, men take a more analytic and evaluative approach when faced with affective stimuli [35]. Present results in schizophrenia patients represent almost a total reversal of the typical pattern of cerebral activation during exposure and experience of negative affect in healthy individuals [24]. Thus it is possible that in schizophrenia it is women who are more rational and men who are more emotional (at least internally because they did not rate their reactions differently), although this remains highly speculative.
The present findings are consistent with our previous report of the overall greater activation in schizophrenia men during experience of sadness, although the regions of significant differences included alternative circuitry (i.e. portions of prefrontal, parietal and temporal cortex, as well as anterior cingulate and amygdala) [17]. This difference arouse most likely due to experimental paradigm, which evoked different affective states in patients.
Although comparisons between structural and functional neuroanatomical studies are not easily interpretable, it is interesting to note that the present findings are somewhat analogous to reports of abnormal sexual dimorphism in schizophrenia in brain regions implicated in the processing of emotions. For example, while in healthy volunteers the grey matter volume of anterior cingulate is greater in women than in men [36], there is no apparent difference in anterior cingulate between men with and without the diagnosis of schizophrenia and a significant reduction in women with schizophrenia relative to healthy women [25]. The findings were confirmed in schizophrenia patients [28] and in individuals with schizotypal personality disorder [29]. In addition, while in healthy volunteers the orbitofrontal cortex: amygdala ratio is higher in women than in men [26], men with schizophrenia have a higher ratio than healthy men, and women with schizophrenia have a lower ratio than healthy women, suggesting feminization of men and masculinization of women with schizophrenia [27]. Although the latter supposition is intriguing, we cannot support it with the present data because the study focused specifically on differences between men and women with the diagnosis of schizophrenia and we did not test any healthy volunteers.
Conclusion
Together with our previous fMRI report [17], present results indicate the existence of sex differences in the cerebral function associated with the processing of negative affect by schizophrenia patients. Whether this difference deviates significantly from the normal sexual dimorphism present in the general population remains to be verified in future studies.
Footnotes
Acknowledgements
We would like to thank all the patients for their participation in this study and Hospital Louis-H. Lafontaine Research Foundation for their support.
