Abstract
Cognitive control deficits and associated abnormalities of cortical activation, particularly involving the prefrontal cortex (PFC), are some of the most consistent findings in established forms of schizophrenia [1–4]. These findings have been linked to a range of phenomenological features of this disorder, particularly disorganization symptoms and prominent working memory deficits [5–7]. Recently studies suggest that such deficits exist at the first episode (FE) of illness [6, 8, 9], although they may be less marked than in chronic patients [10]. One explanation for this might be that PFC dysfunction is an enduring characteristic present early in the course of schizophrenia that becomes progressively worse with ongoing disease processes [10]. Another possibility is that PFC dysfunction may be a dynamic reactive disturbance in response to the onset and unstable nature of psychiatric symptoms, including the stress and emotional trauma associated with the more acute stages of the illness. However, with remission, this dysfunction quickly improves early in the course of the disorder, although it again becomes progressively dysfunctional with continued episodes of relapse.
To date there has only been one functional neuroimaging study that has tracked such changes through the early course of the illness in a longitudinal manner [11]. In that study the authors reported hypoactivation of the dorsolateral PFC (dlPFC), and the anterior cingulate cortex (ACC) at baseline in medication-naïve FE subjects. Following 4 weeks of antipsychotic (AP) treatment, ACC activity normalized together with improvements in symptoms (predominantly in positive symptomatology). However, the dlPFC remained significantly hypoactive despite the remission of acute psychosis. While these findings support a model emphasizing dlPFC dysfunction as an enduring characteristic in schizophrenia, it is possible that 4 weeks of treatment in that study was not sufficient for the effects of medication or clinical improvement to lead to the associated changes in cortical activity.
In the present study we evaluated AP-naïve FE patients within the first week of the onset of the illness and matched healthy controls to perform a well-validated task of prefrontal function and cognitive control while undergoing positron emission tomography (PET) to assess regional cerebral blood flow (rCBF). Patients and controls were then followed up after 8 weeks (patients treated with low-dose APs), at which time the PET paradigm was repeated. We predicted that compared with baseline, FE patients would show increased PFC rCBF and improved Stroop task performance at follow up when they are less symptomatic or distressed and are medicated.
Methods
Subjects
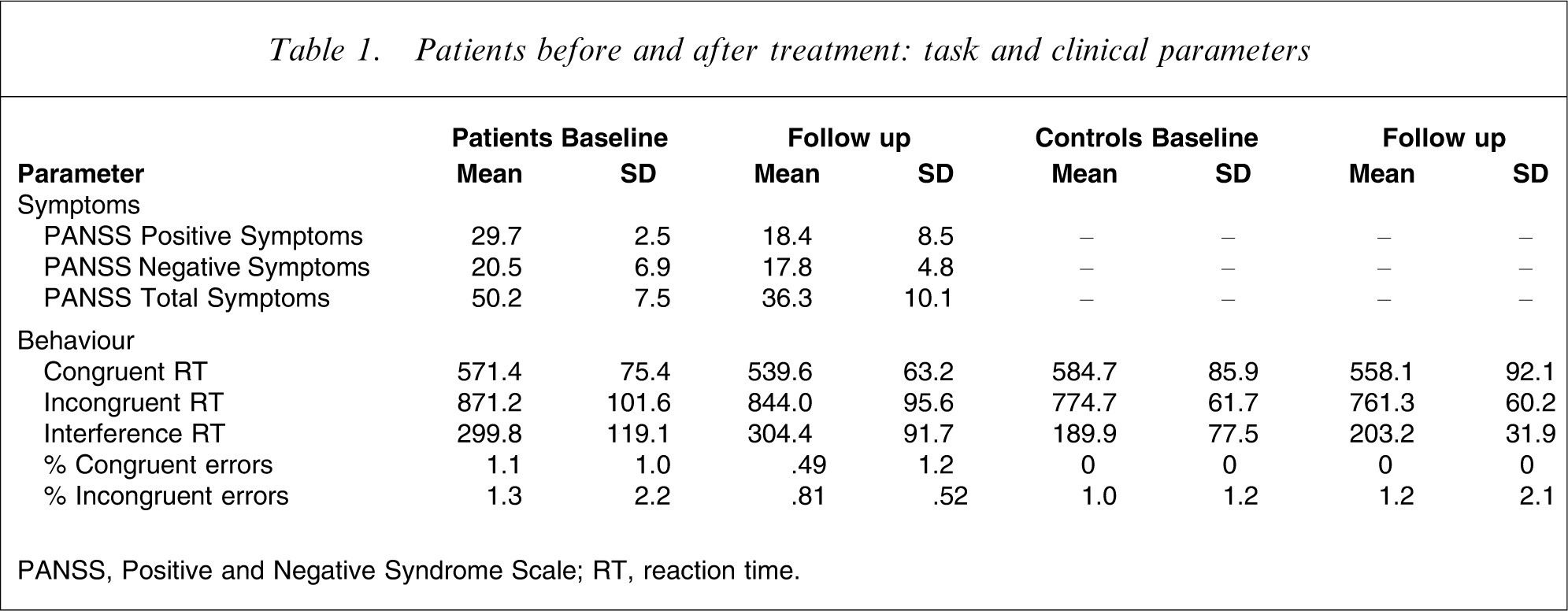
Eight right-handed male patients with first-episode psychosis (FEP; mean age 21.2±3.0 years; one inpatient and seven outpatients) at baseline and seven at follow up were recruited and scanned within 3 days of contact with ORYGEN Youth Health, Melbourne. The diagnosis of schizophrenia was based on the Structured Clinical Interview for DSM-IV [12] administered 6 months after their participation in this study. Clinical admission criteria have previously been published [13]. Patients underwent PET scanning while performing the Stroop interference task [14]. The age of onset of symptoms was between 16 and 30 years and the presence of active psychosis as reflected by at least one of the following: (i) delusions; (ii) hallucinations; (iii) disorder of thinking/speech, other than simple acceleration or retardation; and (iv) disorganized, bizarre, or markedly inappropriate behaviour. Patient scores on the Positive and Negative Syndrome Scale [15] (PANSS [16]) were: total = 50.3±7.5; positive symptoms = 29.7±2.5; and negative symptoms = 20.5±6.9 (Table 1). Exclusion criteria included a significant current history of alcohol or illicit drug dependence and recent history of psychoactive medication use, including steroids, or any other contraindication to PET scanning. Baseline findings have previously been reported [17].
Patients before and after treatment: task and clinical parameters
PANSS, Positive and Negative Syndrome Scale; RT, reaction time.
Seven of the aforementioned patients completed the same functional imaging paradigm after 8 weeks of low-dose AP treatment. Four patients received fixed doses of risperidone (2 mg day−1) and three patients haloperidol (2 mg day−1). Patients underwent a second PET scan approximately 60 days, or 8.4 weeks, after initiating treatment with the EPPIC program (mean 58.8±6.7 days; range 47–64 days). Patient clinical symptomatology (i.e. PANSS ratings) at the time of follow-up scanning indicated a stable-to-mild level of illness, total = 36.3±10.1; positive symptoms = 16.00±6.2; and negative symptoms = 17.66±5.2 (Table 1).
Eight control subjects (seven male; one female) were also recruited by approaching ancillary hospital staff and their families, or via local advertisements (mean age 22.6±2 years). Control subjects were matched with the patient group for age (t(14) = 0.95, p < 0.36) and estimated premorbid IQ, (t(1,15) = 1,16, p < 0.12; patients 101.1±7.1; controls 108.5±9.9) with the National Adult Reading Test (NART; [18]). All participants entering the study were screened for co-morbid medical and psychiatric conditions by clinical assessment, physical and neurological examination. All participants spoke English as a first language and presented with adequate visual and auditory functioning. Five subjects in each group were smokers and three were non-smokers. All participants gave written, informed consent to participation in the present study, which was approved by the Behavioural Research and Ethics Committees for the North Western Mental Health Care Network, Melbourne and the Austin Hospital Human Research Ethics Committee.
Task, data acquisition and analysis
Subjects completed the Stroop colour–word paradigm, identical to that used in our previous studies [9, 19]. The two Stroop conditions (congruent, incongruent) were performed eight times (baseline and follow up) in an ABABABAB design with 36 stimuli per block.
Image acquisition and preprocessing was identical to those previously published [9, 19, 20]. PET scans were acquired using a Siemens/CT1 951R ECAT PET scanner (Siemens Medical Solutions, USA), which generates 31 transaxial slices across an axial field of view of 10.8 cm. PET images were reconstructed resulting in data volumes with 128×128×31 voxels (each of 2.43×2.43×3.375 mm3). High-resolution T1-weighted magnetic resonance imaging (MRI) was also acquired for each subject (GE Sigma 1.5 T scanner, voxel size 0.9×0.9×1.4 mm3; Genesis Medical Imaging, Inc., USA). Spatial realignment of the individual PET images was performed in SPM2. Data were smoothed with a 12 mm full width at half maximum (FWHM) Gaussian filter. Normalization to standard space was performed using FSL (http://www.fmrib.ox.ac.uk/fsl/index.html).
Functional analysis
We performed voxel-wise within- and between-groups univariate (fixed-effects) analyses in SPM2. Global differences in rCBF were removed using subject-specific ANCOVA within each group, and the grand mean of each group was scaled to 50. The factor of interest was the change in experimenter-specified contrast (follow up > baseline) of the study conditions (incongruent–congruent blocks) to isolate variance specific to the task effect of Stroop interference over time. Activation clusters are reported as significant if exceeding a peak probability of Puncorrected=0.001 and >20 contiguous voxels.
Results
Behavioural
Clinically, patients demonstrated significant improvement in total PANSS scores after AP treatment (F(1,14 = 9.39, p < 0.009). This change was due to an improvement of positive symptomatology (F(1,14) = 32.4, p<.001), while there was minimal change in PANSS ratings of negative symptoms (F(1,14)=.69, p < 0.42; Figure 1).
Patients before and after treatment: task and clinical parameters. PANSS, Positive and Negative Syndrome Scale.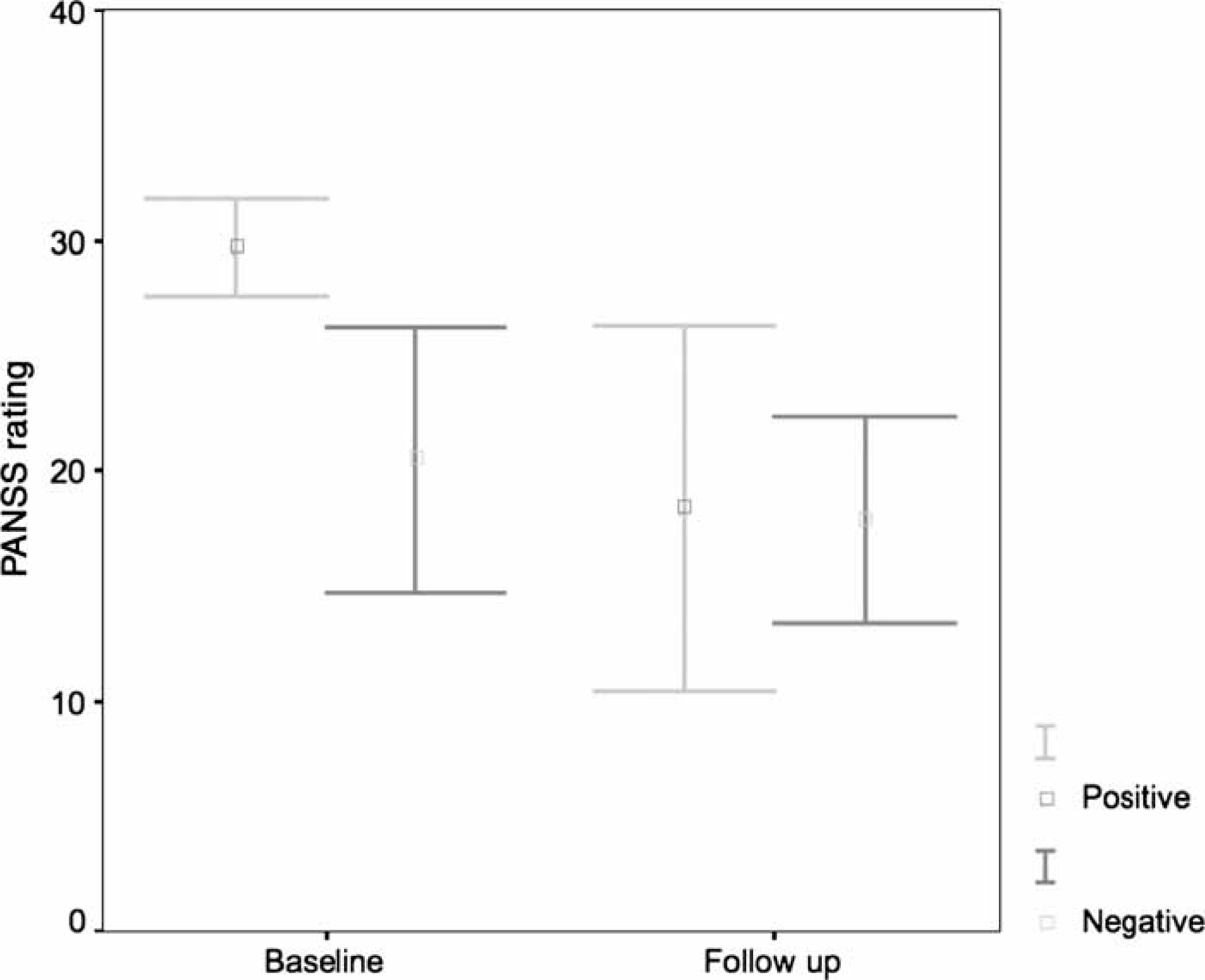
There was no significant task×group interaction between patient reaction time (RT) scores (before and after AP treatment F(1,6) = 0.04, p < 0.95), or main effect of time (F(1,6) = 2.5, p < 0.16). However, there was a main effect of task, signifying an overall interference effect between the two task conditions (F(1,6) = 5.6, p < 0.001; Figure 2). There was no significant trial×group interaction for patient error scores before and after AP treatment (F(1,5) = 0.08, p < 0.78) or main effect of time (F(1,5) = 1.8, p < 0.23), although there was a trend towards main effect of task (F(1,5) = 4.3, p < 0.09), indicating that patients committed less overall errors on the incongruent versus congruent trials after treatment (Table 1). These results indicate that there were no meaningful behavioural changes in task performance in the patient group, before and after AP treatment.
Patients at baseline and follow up: Stroop task reaction times.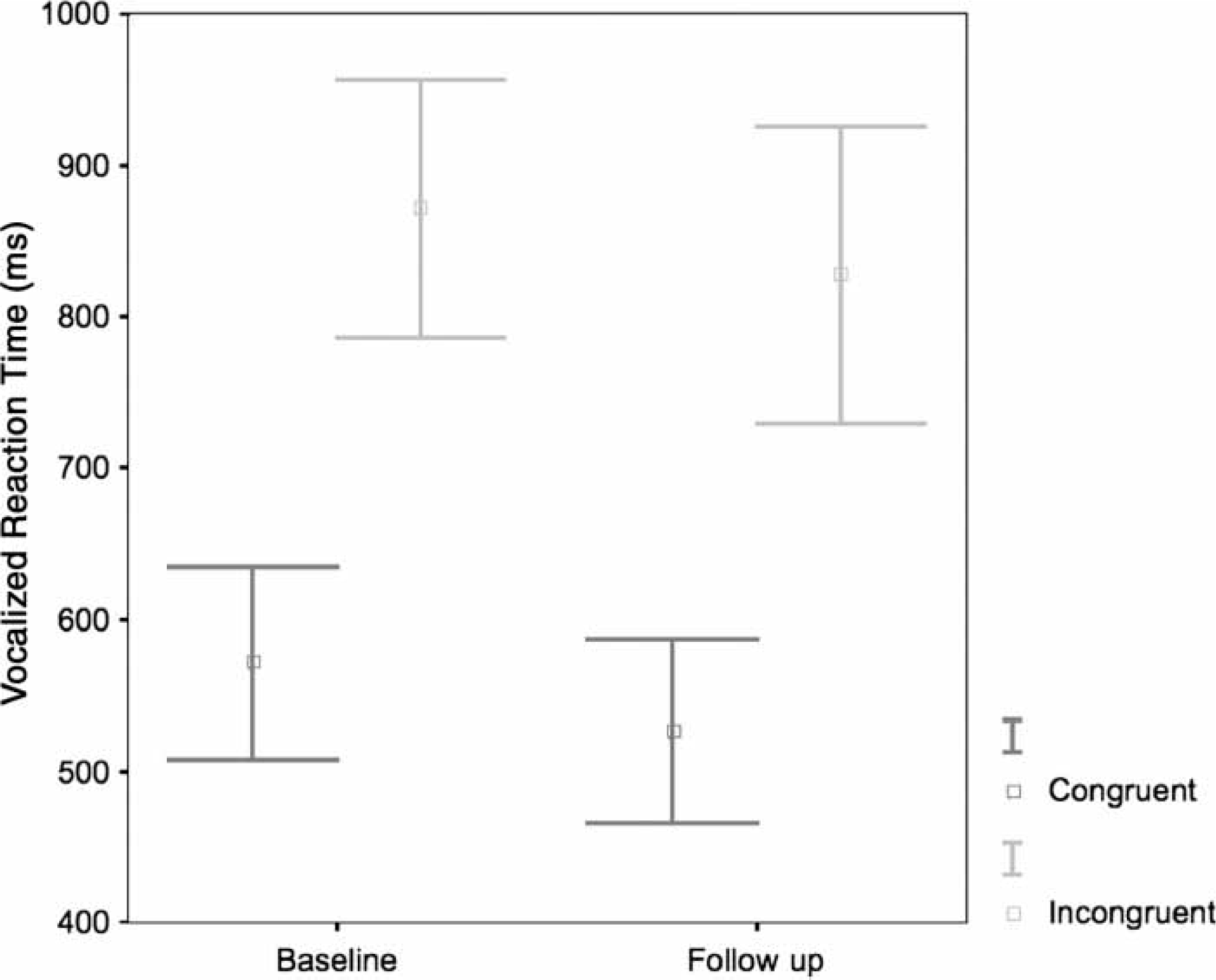
The pattern of results between patients after treatment and control subjects was also relatively unchanged. Again, there was a significant trial×group interaction between patients and control subjects (F(1,12) = 5.5, p < 0.03), signifying greater RT interference in the patient group. Analysis of main effects showed that after treatment patients continued to show significantly slowed RT latencies during incongruent colour naming trials (F(1,13) = 5.36, p < 0.03), but comparable performance during the congruent naming trials (F(1,13) = 1.3, p < 0.27). There was no trial×group interaction between patients and controls for errors on the Stroop task (F(1,12) = 2.8, p < 0.11).
Functional
Controls
Figure 3b illustrates areas of significant activation at follow up > baseline. Suprathreshold clusters (Z-score range = 4.67–3.41, cluster range = 280–29 voxels, p < 0.0001) were characterized bilaterally in the superior parietal cortex (x,y,z=30, −64, 44; and 16, −82, 50; and −24, −76, 42); right supramarginal gyrus (54, −50, 38); left precuneus (−10, −56, 60); and pre-central gyrus (−2, −32, 30; −34, −48, 50); left post-central gyrus (−48, −20, 62); right extrastriate cortex (22, −78, −2); left posterior cingulate gyrus (−6, −46, 60); right superior temporal gyrus (36, 10, −14); left inferior frontal gyrus (−54, 20, 8); left orbital gyrus (−8, 24, −14); right superior frontal gyrus (18, 2, 68); and brainstem (substantia nigra 14, −10, −6). These results indicate that controls engaged largely posterior parietal and motor cortices to a greater extent at follow up than initial testing.
Cerebral blood flow changes (follow up > baseline) in (a) patients and (b) healthy controls.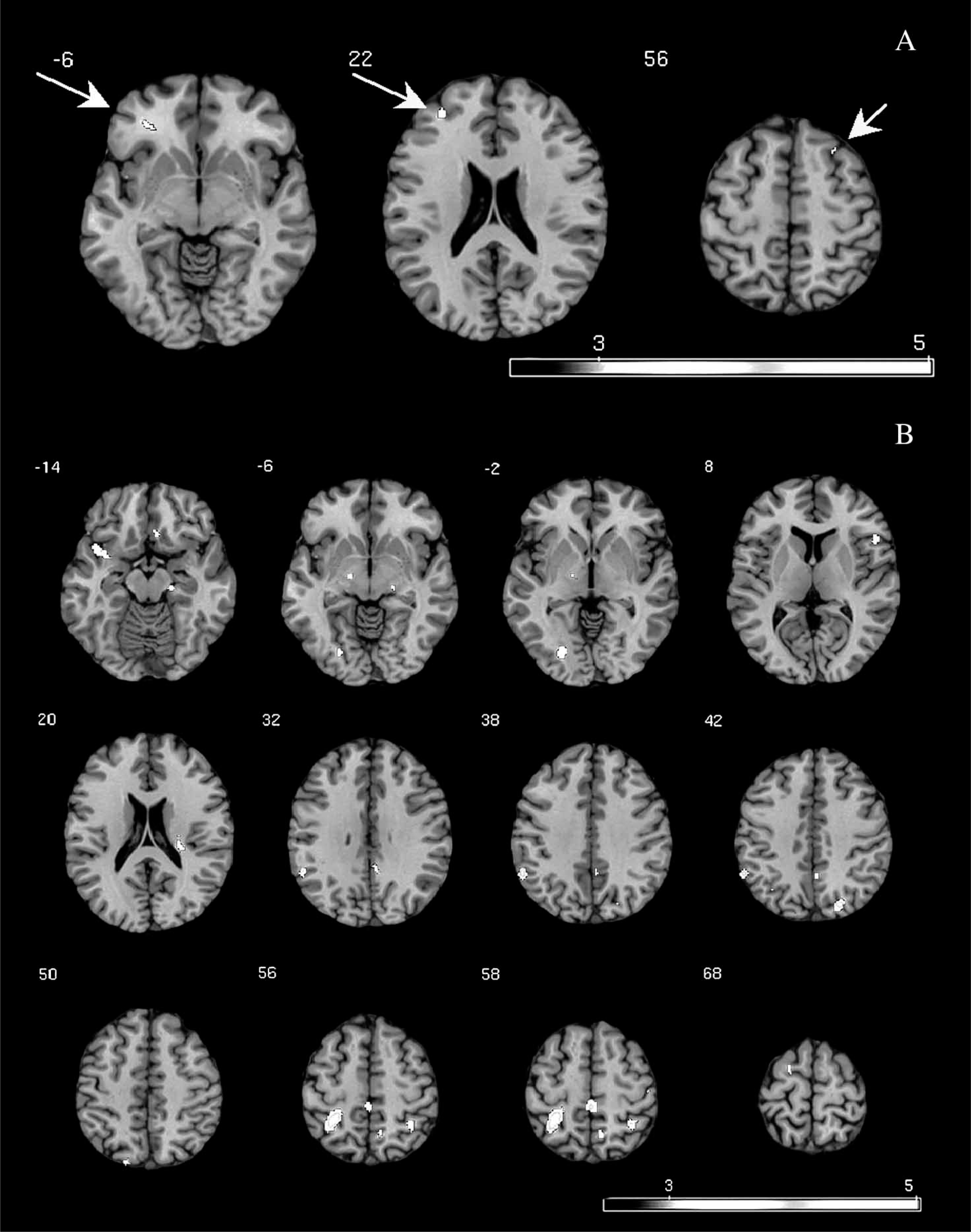
Patients
Figure 3a illustrates areas of significant activation at follow up > baseline. Suprathreshold clusters (Z-score range = 3.72–3.40, cluster range = 59–21 voxels, p < 0.0001) were characterized in the right medial and lateral frontal regions (x,y,z=30, 46, 22; and 30, 40, −6). These results indicate that patients engaged dorsal and inferior regions of the lateral PFC (indicated by arrows) to a greater extent at follow up than initial testing.
Discussion
This is the first longitudinal functional neuroimaging study of FE schizophrenia to investigate PFC activity after an 8 week period of treatment with APs. We used a well-validated task to explore patterns of regional cerebral blood flow in AP-naïve patients within 3 days of the onset of the illness and following treatment with low doses of APs. There were no changes in behavioral performance in both groups, despite improvement in positive symptoms in the clinical group. For the control group, task performance was associated with greater recruitment of posterior brain regions at follow up compared to baseline, while for the patients, this involved greater recruitment frontal regions. The findings support our prediction that following significant remission of the acute symptoms of psychosis, short-term intervention with AP treatment had an observable effect on activity of the lateral PFC. However, it is still unclear whether this increase in frontal activity is due to symptom stabilization per se, the effects of medication, or a lack of the normal neurophysiological learning that occurs with experience/practice.
To our knowledge there has been only one functional neuroimaging study of schizophrenia examining the nature of PFC function in patients at the FE stage of illness, before and after receiving AP treatment [11]. In that study hypofunctional activity in dlPFC regions found at baseline did not improve over time. However, as the authors themselves suggest, 4 weeks may not have allowed adequate time for measurable improvement [21]. Indeed, we found increased lateral PFC activation 8 weeks after illness onset. However, in contrast to the findings of Snitz et al.[11], we did not observe increases in ACC activity over time following symptom stabilization. Given the specific association between ACC activity and symptoms of poverty and disorganization that have previously been reported [11, 22], our failure to find ACC-related changes in activity might reflect the fact that there was little change in these symptoms, particularly negative symptoms in the present patient sample between baseline and follow-up assessment.
Our findings of limited behavioural change at follow up relative to baseline are consistent with that of Snitz et al.[11]. It may be that these processes (i.e. blood flow and RT measures) are not necessarily related. For example, the patients’ behavioural performance did not improve at follow up despite the increased PFC activity at the same stage. Alternatively, it may be that improvements in behavioural measures emerge with longer duration of treatment, or are found with larger numbers. While previous studies have documented beneficial effects on cognition after short-term treatment, it has been suggested that domains such as the more complex executive functions may be modulated over longer periods [23, 24]. In line with these studies, our findings suggest that elements of executive control are not immediately responsive to improvements in the neural systems that mediate these functions. Thus, the overall functional changes in the frontal regions of the clinical group in the absence of associated executive performance improvement could be interpreted as the initial stages of a longer process of functional normalization. It is more likely in these early phases of illness onset that the increased prefrontal activity reflects ongoing inefficiency in maintenance of task performance. That is, while healthy comparison subjects are using more posterior, and presumably more automated processes to complete the task at follow up, patients are still using frontal-executive, and presumably more effortful processes to achieve a comparable level of behavioural performance. However, the present numbers are small and this issue requires further exploration with more sophisticated designs (e.g. parametric design experimentally manipulating the cognitive load) and larger samples.
No behavioral changes were found in the control subjects at follow up compared to baseline performance. Controls demonstrated significant activation changes after 8 weeks. Their recruitment of more posterior brain regions including the superior parietal cortex might reflect either a greater demand on these regions that are thought to be a part of the inhibitory control network [25], or more likely, practice-related automatization of task performance [9, 26]. A third possibility is that APs impeded the recruitment of these more posterior brain regions. To this end, it could also be agued that the differential treatment of patients in the present study with risperidone (i.e. atypical AP) and haloperidol (i.e. typical AP) may have influenced the current findings. However, there is emerging evidence that treatment with either class of neuroleptics may have comparable effects on cognition in schizophrenia when compared at low doses. For example, in a 2 year randomized trial, Green et al. reported no specific advantage with risperidone treatment compared to low-dose haloperidol, where both showed some effect in improving basic attentional performance [27]. This has since been replicated with other atypical (e.g. olanzapine) and typical compounds (e.g. zuclopenthixol) in patients with FE schizophrenia [23, 28].
Taken together, the present findings suggest that disturbances of the PFC often seen in FEP are at least partially associated with acute symptom expression. However, it is still unclear whether the observed increase in frontal activity is due to symptom stabilization per se, the effects of medication, or differences in neural/cognitive learning. Furthermore, it is unclear whether this increase in PFC activity is reflective of a healthy or unhealthy pathway. In the context of the control findings, we interpret the patient findings as reflecting continued inefficiency in the performance of tasks requiring executive ability. However, these findings are preliminary and therefore should be taken in the context of study limitations that include a small number of FE patients in the analyses, mixed treatments, and the lack of a placebo group.
Footnotes
Acknowledgements
Associate Professor Brewer was supported by the National Health and Medical Research Council (NHMRC): Schizophrenia Research Unit and an NHMRC of Australia Program Grant (ID 350241); Janssen-Cilag also provided support along with the Colonial Foundation. Dr Yücel is supported by an NHMRC of Australia Program Grant (ID 350241) and the Colonial Foundation. Dr Harrison is supported by an NHMRC Training Award (ID 400420). Neuroimaging analysis was facilitated by the Neuropsychiatry Imaging Laboratory managed by Ms Bridget Soulsby at the Melbourne Neuropsychiatry Centre and supported by Neurosciences Victoria.
