Abstract
Abnormalities of cortisol and the hypothalmic–pituitary–adrenal (HPA) axis have been extensively documented in patients with depressive disorders. These abnormalities include increased 24 h cortisol levels, elevated urinary cortisol levels, failure of suppression of cortisol by dexamethasone, increased levels of corticotropin-releasing hormone (CRH) and alterations in the cortisol response to CRH [1].
In recent years there has been increasing interest in the impact of childhood or developmental experiences on later HPA axis activity in both humans and animals. In humans, childhood sexual abuse, perinatal stress and preteen adversity have all been shown to impact on later HPA axis activity [2–4].
In this paper we examined whether childhood variables, including quality of perceived parenting, physical and sexual abuse influences plasma cortisol levels in adults suffering from major depression. We also examined whether clinical features of depression, such as melancholia, influenced cortisol levels, and whether the childhood variables would still impact on cortisol levels when clinical features were controlled for.
Methods
The depressed patients were recruited for a treatment study [5–7], which had been approved by the Canterbury Ethics Committee, and written informed consent was obtained. Inclusion criteria were principal current diagnosis of major depressive disorder; antidepressant medication as appropriate treatment; age ≥18 years; free of any physical illnesses that may affect neuroendocrine assessment (e.g. diabetes); and free of prescribed psychotropic drugs for a minimum of 2 weeks. Patients were excluded if current moderate or severe alcohol or drug dependence was considered to be the principal current diagnosis. Patients were also excluded if they had a history of mania, schizophrenia or severe antisocial personality disorder.
After giving consent, patients were scheduled for a detailed clinical and neurobiological assessment. Patients were assessed using the Structured Clinical Interview for DSM (SCID), the Montgomery–Asberg Depression Rating Scale (MADRS), the Hamilton Depression Rating Scale (HDRS), and the Mental State Examination for Melancholia (MSE) [8], which assesses observable retardation and agitation. During this assessment patients also completed the Parental Bonding Instrument (PBI) [9]. The PBI consists of 25 questions about how they were treated by their mother and 25 questions about their father, during the first 16 years of their life. Two scores, care and protection, are obtained for each parent. Categories of care and protection were created by trichotomizing the scores. Affectionless control is the combination of low care with high protection. Severe affectionless control required that the patient was in the lowest tertile for both low care and high protection. A research nurse independently interviewed each patient about recent and childhood (prior to 16 years) physical abuse and sexual abuse [10, 11]. Based on this interview both childhood physical and sexual abuse were coded as absent (0), mild (1) or severe (2).
For their neurobiological assessment, patients reported to the Clinical Research Unit at approximately 13.00 hours, after a 3 h fast. While reclining, an indwelling i.v. catheter was inserted. From 14.00 hours to 15.00 hours, five blood samples were drawn at 15 min intervals for the measurement of cortisol. Blood samples were centrifuged, and plasma was frozen and stored for later cortisol measurement by an enzyme-linked immunosorbent assay [12]. The mean afternoon cortisol level was obtained by averaging the five cortisol levels.
Mean afternoon cortisol levels were not normally distributed. The results were made to approximate a normal distribution by a logarithmic transformation. For clarity cortisol levels are presented as untransformed geometric means. Relationships of cortisol levels with clinical and childhood variables were initially assessed in a univariate analysis of variance (ANOVA), and significant variables were then entered into a multiple linear regression. Continuous measures such as depression severity, maternal and paternal care and protection, were trichotomized, thus creating categories such as low/medium/high, before statistical analyses.
Results
Of the 195 depressed patients recruited for this treatment study, there was complete neuroendocrine data on 192. Table 1 shows the baseline characteristics of the sample.
Clinical and demographic characteristics of the depressed sample
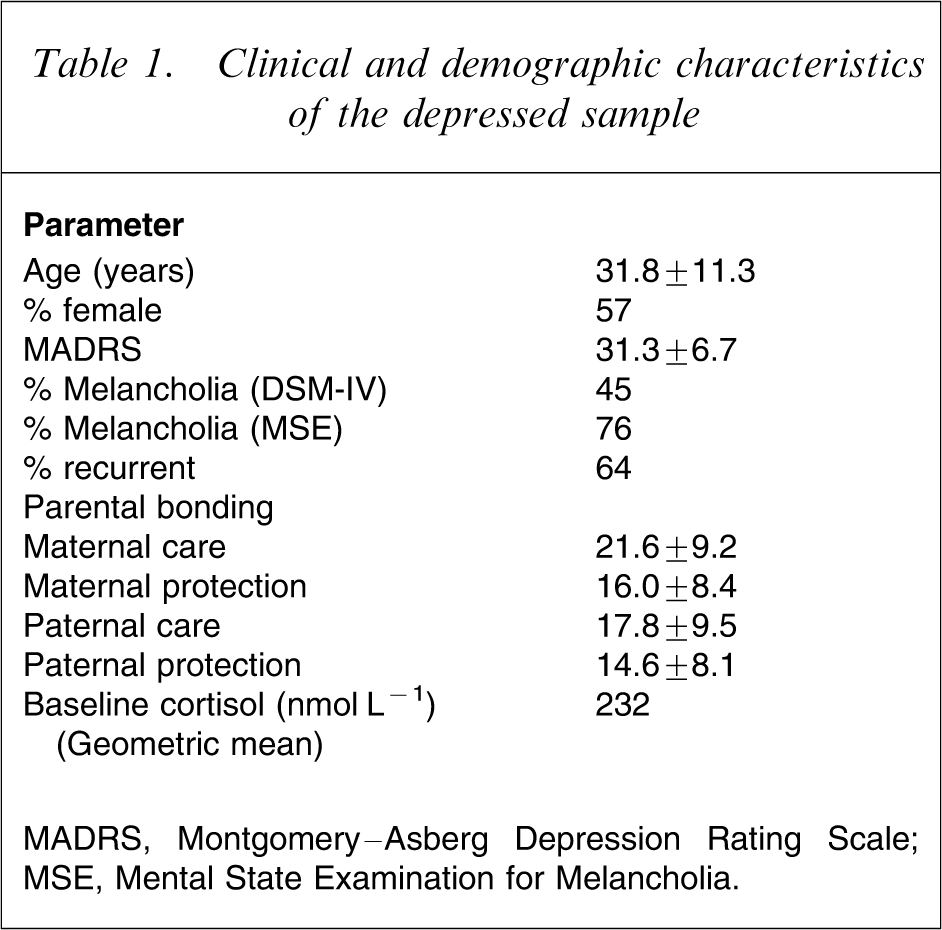
MADRS, Montgomery–Asberg Depression Rating Scale; MSE, Mental State Examination for Melancholia.
The 84 men had higher mean afternoon cortisol levels than the 108 women (255 nmol L−1 vs 215 nmol L−1, F = 6.50, p = 0.012), but cortisol levels were not related to age, depression severity (as measured by MADRS or HDRS), or a DSM-IV diagnosis of major depression with melancholia. However, if melancholia is defined using the Mental State Examination, which assesses observed retardation and agitation, then melancholic depressives have higher cortisol levels than non-melancholic depressives (234 nmol L−1 vs 211 nmol L−1, F = 6.57, p = 0.01).
Table 2 shows that the depressed patients who reported low maternal care, high maternal protection, or maternal affectionless control (i.e. low care and high protection) had lower cortisol levels. Both childhood sexual and physical abuse were also associated with cortisol levels. By contrast paternal care, protection or affectionless control were not related to cortisol levels.
Relationships between cortisol levels and childhood experiences
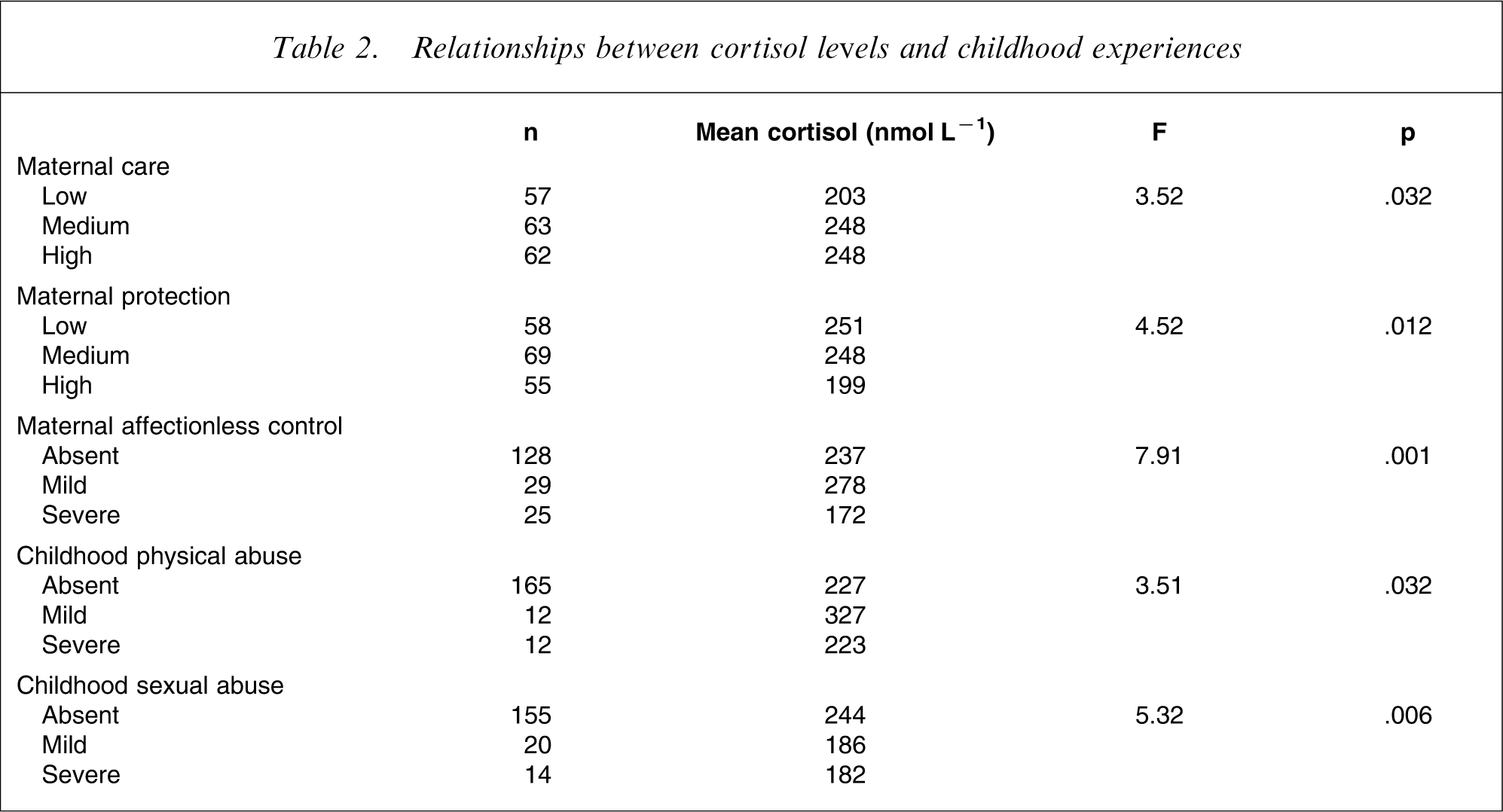
Because maternal affectionless control was derived from low care plus high protection, we entered maternal affectionless control, along with childhood physical abuse, childhood sexual abuse, melancholia (as defined by MSE) and gender into a multiple linear regression to predict afternoon cortisol levels. Together, these variables explain 22% of the variance in the cortisol levels. The strongest predictor was maternal affectionless control (F = 8.39, df = 2, p < 0.001), followed by physical abuse (F = 4.13, df = 2, p = 0.018), sexual abuse (F = 3.92, df = 2, p = 0.022), and melancholia (F = 3.13, df = 1, p = 0.079). Gender (F = 1.60) was not significant in the multiple regression.
Discussion
In this sample of 192 depressed outpatients recruited for a treatment trial, approximately 20% of the variance in their mean afternoon cortisol levels can be explained by a combination of adverse childhood experiences; a small amount of the variance is explained by current melancholic symptoms while depressive severity was unrelated. These results are consistent with the growing data in both animals and humans that childhood experiences have a long-term impact upon regulation of the HPA axis. Of the childhood experiences, maternal affectionless control, which involves the combination of low care with high protection, had the greatest impact. Both maternal affectionless control and childhood sexual abuse were associated with lower cortisol levels; findings consistent with the lower cortisol levels reported in post-traumatic stress disorder [13].
The persisting neuroendocrine effects of maternal behaviour is compatible with animal research that suggests that offspring of low-care rat mothers have alterations in the epigenome at a glucocorticoid receptor gene promoter in the hippocampus. This effect emerges over the early weeks of life and is related to differences in DNA methylation. Interestingly, this early behavioural programming with persistent HPA axis effects can be reversed by methyl supplementation [14–16].
All the subjects in the present study suffered from current major depression therefore conclusions cannot be drawn regarding the relationship between childhood experiences, HPA axis function and susceptibility to later depression. In this sample of outpatients with depression it is of note that childhood experiences had a greater effect on cortisol levels than severity of depression or presence of melancholia. This might not be the case in subjects with, for example, psychotic depression in whom HPA axis abnormalities are more prominent.
This animal research, plus our findings that variations in maternal care can have a major lasting impact on adult HPA axis activity, support the growing literature linking childhood experiences to adult psychopathology, and provide a plausible neurobiological mechanism for this association [2–4].
Footnotes
Acknowledgements
This research was funded by grants from the Health Research Council of New Zealand.
