Abstract
Schizophrenia is a disorder characterised by disturbances in perception, thought, volition, socialisation, psychomotor behaviour and the sense of self. While the frontal lobes have received a great deal of attention in schizophrenia research, several lines of investigation have implicated dysfunction or pathology in other areas with which the frontal lobes connect, such as the medial temporal lobe, basal ganglia and thalamus [01–04]. Hippocampal disturbance in schizophrenia has been suggested by clinical observations of the relationship between temporal lobe epilepsy and schizophrenia as well as neuroimaging, neuropsychological and neuropathological research. Despite broad agreement that schizophrenia is associated with a subtle disorder of brain structure [5], the nature, timing and course of the associated neurobiological changes have remained elusive [6]. Kraepelin's notion of dementia praecox, which was based on clinical and neuropathological observations, implied a premature and progressive degeneration of the brain, similar to Alzheimer's disease [7]. The prevailing current view is that schizophrenia is a neurodevelopmental disorder in which structural brain changes, caused by an early antenatal or perinatal insult, confer a predisposition to the development of schizophrenia, but are not progressive beyond the onset of symptoms [08–10]. These investigators draw on various sources of evidence, such as the higher incidence of obstetric complications, winter births, minor physical abnormalities and soft neurological signs in schizophrenia to support their arguments. Furthermore, the motor, cognitive, social and emotional changes that have been described in children who go on to develop schizophrenia may be subtle, early manifestations of such a lesion [11],[12]. According to the neurodevelopmental model, the lesion produced by an early insult will interact with normal postpubertal brain maturation to produce the clinical symptoms of schizophrenia.
The hippocampus is seen as an important structure in such models because of the role it plays in cognitive and emotional functioning, its vulnerability to early insults and its connections to other brain structures, in particular the prefrontal cortex. The findings of hippocampal pyramidal cell disarray and reduction in pyramidal cell numbers seen in neuropathological studies of schizophrenia are consistent with an early, nonprogressive insult that interferes with cell proliferation and migration [9]. One important factor may be the late myelination of major hippocampal efferent fibres [13]. Alternatively, an early hippocampal lesion may lead to anomalous synaptic reorganisation in hippocampal projection sites, particularly those to the prefrontal cortex [14]. Adrenal and gonadal stimulating hormones released during adolescence act as neurotrophic factors for axonal sprouting and synaptic expansion [14]. Animal studies have shown that early perinatal stress can produce hippocampal volume reductions and postpubertal behavioural disturbances [15]. The emergence of these postpubertal behavioural changes can be prevented by the administration of haloperidol, a drug used in the treatment of schizophrenia [16]. These studies suggest that an interaction between early brain insult and maturational processes involving the hippocampus may have aetiological significance for the development of schizophrenia.
Modern neuropathological studies, while not conclusive or consistent, have favoured a neurodevelopmental process, but are limited by their cross-sectional nature, small numbers and methodological issues specific to post-mortem studies (discussed further by Lipska et al. [6]). In vivo neuroimaging techniques, especially magnetic resonance imaging (MRI), are well placed to address the question of brain changes in schizophrenia, particularly the issue of longitudinal progression. Recent reviews of the area [17–19] have identified that the most consistent cross-sectional study findings are of enlarged ventricles, smaller medial temporal and frontal structures and subcortical changes. These reviews have identified the need for further studies at different stages of illness and the importance of longitudinal studies that span the period before and after the initial onset of psychosis. The aim of the present paper is to examine whether the currently available cross-sectional and longitudinal neuroimaging studies can shed light on the issue of progressive brain changes, particularly within medial temporal structures, in schizophrenia and to discuss the way forward for research in the field.
The hippocampus and its relevance to schizophrenia
Papez conceptualised the limbic system as critical to the expression and experience of emotion [20]. It has been proposed that the limbic system is critical to memory, including working memory, and motivation [21],[22] and that the role of the hippocampus is to assign meaningfulness to events in order to make them memorable [23]. The meaningfulness of an event is, in part, determined by emotion and motivation [24]. Deficits in memory, motivation and emotion are recognised features of schizophrenia. The hippocampus also has close connections with other areas that are considered to be important in schizophrenia, in particular with prefrontal cortical regions such as the dorsolateral prefrontal cortex and the anterior cingulate [25–27]. The behavioural implications of these connections and their relevance to schizophrenia have been recently elaborated by several investigators [3],[28],[29]. One influential model suggests that the consistent findings of frontal lobe deficits in schizophrenia may result from disruption of the circuitry connecting prefrontal cortex with limbic structures, particularly the hippocampus [3],[26]. These investigators provide evidence, derived from their structural and functional neuroimaging studies, to suggest that structural changes in the hippocampus explain the functional deficits related to dorsolateral prefrontal cortex.
Cross-sectional evidence for hippocampal abnormality in schizophrenia
Clinical evidence
The strongest clinical evidence for a link between brain structure and schizophrenia comes from the observation that temporal lobe epilepsy is associated with an increased incidence of psychotic symptoms [30]. Patients with epilepsy who present with first rank Schneiderian psychotic symptoms are more likely to exhibit medial temporal limbic pathology [31]. Left temporal abnormalities have been identified in patients with epilepsy and psychosis on MRI compared to epilepsy patients without psychosis [32]. Further, in a postmortem study of epilepsy patients with and without psychosis, Bruton and colleagues reported that, while mesial temporal sclerosis was apparent in both groups, the psychosis groups had larger cerebral ventricles, excess periventricular gliosis, and more focal cerebral damage compared with nonpsychotic epileptic patients [33], all of which have been reported in schizophrenia. Patients with epilepsy who develop psychoses are more likely to have had epilepsy since late childhood or early adolescence [34] and about 10%% of children with mixed psychomotor and generalised epilepsy who progress to adolescent epilepsy develop schizophreniform psychoses [14]. Further, it is those patients with developmental lesions such as dysplasias and hamartomas, particularly those affecting the left side, who are most likely to have ictal psychoses [34]. These findings are generally supportive of the hypothesis that an early, developmental brain lesion is responsible for the symptoms of schizophrenia.
A major argument against a nonprogressive process in schizophrenia has been the variable course that schizophrenia may take. Thus, a proportion of patients have one episode and recover, some patients have an initial course of relapse and remission followed by a plateauing of symptoms, while up to 50%% of patients have a severe illness with a relatively poor outcome, with about 5–10%% having progressive, treatment-resistant illness. (For further discussion see references [35],[36].) This wide disparity in illness course is particularly manifest in the instability of the presentation of schizophrenia, which may be due to aetiological heterogeneity or, alternatively, a variable pathophysiological process. However, there is nothing to preclude an early, neurodevelopmental lesion and a later, progressive degenerative process as features of the disorder, possibly interacting with the heterogeneity suggested earlier.
Neuropsychological evidence
In their review of neuropsychological studies in schizophrenha, Gold and Harvey identified impairments in attention, memory and problem solving and suggested that these findings implicated multisystem dysfunction involving frontal, temporal, thalamic and basal ganglia areas [4]. Recently, researchers have sought to link the range of phenomena observed in schizophrenia to the neurocognitive deficits by utilising distributed neuronal network models [26],[37]. According to this approach, the control of any cognitive function is distributed across a number of interconnected nuclei through the brain. Disruption to any of these nuclei or their interconnections results in changes in cognitive function. As noted earlier, there are two models that have sought to explain the symptoms of schizophrenia in this way.
Weinberger and colleagues have proposed a model to explain deficits in executive function in schizophrenia [3]. These investigators have drawn on the animal literature for working memory to suggest the neuropsychological dysfunction in schizophrenia results from disruption of the circuitry connecting the hippocampus with the dorsolateral prefrontal cortex [22]. An alternative model argues that the positive symptoms of schizophrenia are due to a failure of an internal monitoring system that depends upon links between the hippocampus and the prefrontal cortex [38]. Frith and Done regard the hippocampus as an internal monitor that compares sensory information with an internally generated model of the world [38]. Positive symptoms occur because there is a failure to monitor self-generated intentions. Subsequent actions are then perceived as unintended and delusionally attributed to psychotic phenomena. While these models differ conceptually, both emphasise the importance of the hippocampus and its prefrontal projections in explaining the range of clinical phenomena in schizophrenia.
Neuropsychological evidence for a nonprogressive lesion in schizophrenia comes from both crosssectional and the limited number of longitudinal studies [39],[40]. Although there are a number of confounding variables in these studies, such as medication, reliability and validity of diagnoses, dropout rate and practice effects, clear trends have emerged. Specifically, the main conclusion from both crosssectional [4],[41],[42] and follow-up studies [39],[40],[43],[44] is that, after the onset of the disorder, cognitive deficits are stable, although there are reports of improvements in performance with remission of symptoms or with certain treatments [45–47]. The fact that the cognitive impairment is generalised can also be attributed to a neurodevelopmental cause, with additional features arising during acute psychotic episodes [40]. To date, no studies have reported neuropsychological data from the period either side of the transition to psychosis. As this is the period of maximal behavioural change, it is reasonable to consider this as a time of neuropsychological change also.
Neuropathological evidence
Microscopic descriptions of schizophrenia have frequently concentrated on whether gliosis is a core feature of the disorder. As gliosis is a sign of past inflammation, its presence or absence is of importance in understanding the underlying aetiology of the illness. Initial studies, such as those by Stevens, which found gliosis in roughly 70%% of cases [48], have been questioned by findings that gliosis may be related to the large number of incidental findings in patients with schizophrenia rather than being specific to the illness. Studies of neurones have tended to indicate decreased neuropil and neuronal size, rather than neuronal loss, in the hippocampus [49] and dorsolateral prefrontal cortex [50]. In addition there does appear to be decreased neuronal density in the dorsal thalamus [51]. Other findings, which are much less robust, include synaptic changes that may support the hypothesis of excessive synaptic pruning in the disorder [52]. Disturbances in hippocampal pyramidal cell orientation, reductions in pyramidal cell density and cell size have been reported in postmortem brains of patients with schizophrenia [6]. Thus, the neuropathological evidence provides support for a neurodevelopmental rather than neurodegenerative pathological process in the hippocampus. However, this conclusion has been challenged for a number of reasons. First, the suggestion that the lack of gliosis in neuropathological studies implies a very early insult may be contested, because it is by no means confirmed that the glial response can only occur after the second trimester. Second, the glial response may be regionally variable [6]. Gliotic change is not always present even after postnatal injury with clearcut neuronal loss [53], so the subtle kinds of neuropathology that are a feature of schizophrenia may not be sufficient to cause gliosis no matter when they occurred. Third, a neurodevelopmental lesion may leave the brain vulnerable to further changes, particularly at the time of illness onset or with acute relapses.
It is also the case that the neurodevelopmental model generally limits the timeframe for the pathogenetic process to fetal or perinatal life [5]. However, it is now clear that a number of developmental processes, such as apoptosis, neurite proliferation, axonal myelination and neurite pruning continue into postnatal life, either as a normal [54] or abnormal [55] feature. Indeed, it appears that even neurogenesis [56] or structural brain changes [57] can occur late in life as a normal part of development. This means that any aberrant development that occurs as a feature of schizophrenia may not be restricted to the early period of life. A finding of progressive deterioration would have profound implications, with the possibility of arresting or reversing that progression by therapeutic means [7].
Imaging studies of the hippocampus in schizophrenia
Two key predictions of the neurodevelopmental model are that structural lesions will be present before and at the onset of a schizophrenic illness and that the lesion will be nonprogressive. In order to examine whether structural changes are progressive during the course of the illness, two strategies are useful. First, a cross-sectional design that includes subjects at all stages of the illness would provide indirect evidence of the effects of illness duration. However, as discussed by Kraemer and colleagues, these can provide misleading information depending on a number of factors, such as the nature of the progressive process [58]. A second, more powerful technique is to use a prospective longitudinal design that follows patients from or before the onset of illness. The imaging literature relating to these two methodologies is discussed in detail later.
Cross-sectional studies
The most consistent finding to emerge from a large body of computed tomography (CT) scan research in schizophrenia was the finding of lateral ventricular enlargement and cortical atrophy [59]. Computed tomography scans cannot provide the spatial resolution needed to estimate hippocampal size, other than through indirect means such as enlargement of the temporal horn of the lateral ventricles [60]. The advent of MRI has allowed the delineation of smaller structures such as the hippocampus.
Early MRI research largely failed to identify hippocampal abnormalities. Hippocampal volume reductions have been more consistently found with the advent of improved technology, providing thinner brain slices and improved image resolution. There is now broad agreement that reduced medial temporal lobe volume is a feature of schizophrenia and is present from illness onset [17–19]. However, the specificity of structural brain changes to schizophrenia remains unresolved because structural abnormalities such as increased ventricle to brain ratio (VBR) have been described in other psychoses [59],[61],[62]. One MRI study that compared patients with schizophrenia, bipolar disorder and normal control subjects on 10 mm MRI slices failed to find any difference in hippocampal volumes between any of the groups [62]. Hippocampal reduction was not identified in a study of depressed patients [63], though shortened T1 relaxation time in the hippocampus of depressed patients has been reported [64]. In our own published study, we identified bilaterally smaller hippocampi in patients with chronic schizophrenia, while first-episode patients with schizophrenia or schizophreniform psychosis and affective psychoses had significantly smaller left hippocampi [65],[66]. We have now measured the hippocampal volumes of more than 400 subjects at various stages of illness, from prodrome to chronic schizophrenia, including a large number of healthy controls. This more recent data indicates that, when controlled for intracranial volume, only chronically unwell subjects (n = 92) have bilaterally reduced hippocampi compared to controls (n = 96). Patients in their first episode of psychosis (n = 161) have reduced left hippocampal volumes only (D. Velakoulis, unpubl. obs., 1999). This finding implies that either patients with a chronic course exhibit more brain abnormalities in the first episode, or that there is some progression of hippocampal volume loss with continuing illness. As either case would have major implications for prognosis and treatment, we have further investigated this using an automated technique (the Structural Magnetic Resonance Tool kit, or SMaRT), which has been developed by collaborators at the Institute of Psychiatry (London, UK) [67]. This technique classifies voxels as grey matter, white matter, cerebrospinal fluid or dura on the basis of their signal intensities on proton density and T2-weighted images. This segmentation is a probabilistic one, taking account of partial volume effects and allows a comparison to be made between groups, or to correlate brain volume with some other variable such as duration of illness. In keeping with the findings mentioned earlier, reduced right medial temporal lobe volume was associated with increased duration of illness, with no other brain area demonstrating such an association (D. Velakoulis et al. unpubl. obs., 1999). Only one other study has found a relationship between illness duration and the severity of structural change [68]. While no hippocampal volume reduction was identified in either the first episode or chronic schizophrenia groups in that study, a significant association was found between reduced left temporal lobe volume and illness duration.
The failure of cross-sectional imaging studies to find evidence of ongoing structural change in the brain does not, in itself, speak against neurodegeneration in schizophrenia. This is because it is unknown at what stage during the course of illness the observed volume loss occurred. If it occurred mostly during the prodrome (i.e. the period before the diagnosis of psychosis but after the onset of behavioural changes), then further progression might only be seen rarely in the context of clinical deterioration [7],[69].
In order to examine this we have studied high risk individuals who were being treated in a specialist clinic, the Personal Assessment and Crisis Evaluation Clinic (PACE) [70], to whom young (16–30 years old) people are referred if they are thought to be at risk of developing psychotic disorders. Individuals were included in the study sample if they met one or more of three sets of operationalised criteria. They had either to display frequent attenuated subthreshold psychotic symptoms (a number of which are considered under DSM-IV schizotypal disorder) or transient psychotic symptoms (lasting less than one week), or have both a first-degree relative with a history of psychosis or schizotypal disorder, plus a loss of 30 points or more on the Global Assessment of Functioning (GAF) scale for at least one month.
This group represents a different population to the traditional high-risk studies, which have tended to examine relatives of patients with schizophrenia. These ‘genetic’ high-risk studies have identified differences in brain structure, such as enlarged ventricles [71] and smaller medial temporal lobes [72]. However, because the number of high-risk subjects who become psychotic is low and transition occurs over a long period, this type of study is expensive and logistically difficult. In contrast, the advantage of our ‘close-in’ strategy is that it yields a much higher rate of transition to psychosis than family history alone and does so within a relatively short follow-up period (41%% transition rate to psychosis within a 12-month period [73]). We have termed this group ‘ultra-high risk’ to distinguish it from previous studies, which used family history alone. By investigating the period of maximal disturbance of behaviour and cognitive function, namely, the first emergence of psychosis, we hypothesised that this would also be the period when maximal neuroanatomical change is likely to occur [74].
Our initial baseline study of the hippocampi of 63 ultra-high risk subjects indicated that there was a significant reduction in volume on the left side only [75]. This is in line with data previously published by the Edinburgh High Risk Group [72]. However, when our baseline data was divided into those ultrahigh risk subjects who later developed a psychotic illness and those who did not, what we found was counter-intuitive. Contrary to our expectations, those subjects who developed a psychotic illness within 12 months (UHR-psychotic) did not have smaller hippocampal volumes compared to controls. Instead, it was the group who did not become unwell (UHR-nonpsychotic) whose left hippocampi were significantly smaller than a normal control group [75]. Further, survival analysis for hippocampal and whole brain volume found that larger left hippocampi were associated with a higher risk of psychosis [75]. Because the UHR-psychotic group have left hippocampal volumes at baseline that are larger than those of a first-episode group, one possibility is that there is an active process occurring during the evolution of psychosis.
Longitudinal studies
Structural magnetic resonance imaging follow-up studies in schizophrenia
The earliest longitudinal CT studies generally considered ventricular size to be stable over time but they suffered greatly from methodological problems such as small numbers, no comparison group or variable follow-up interval [101]. Despite improvements in technology, recent MRI studies have suffered from similar methodological constraints, and the confusion over the issue of progression remains. One study of a unique population of adolescents with childhood-onset schizophrenia has shown widespread changes in cortical and subcortical regions over a period of 3–5 years [89],[93],[96],[97]. However, it is not clear how comparable this population is to adult-onset patients, given that there is likely to be extensive interaction between the disease process and normal brain development.
The only published study that has longitudinally examined hippocampal volume in patients with adultonset schizophrenia failed to find any hippocampal volume change in the 4 years following the firstepisode of psychosis [88]. Our cross-sectional data suggests that the critical period for hippocampal volume change may be before the time period examined in this latter study; that is, during the period of evolution of psychosis. In order to test this, we utilised the SMaRT technique described earlier, in an initial pilot study, to investigate longitudinal change in two UHR groups; seven who became psychotic within a year and nine who did not. The two groups were clinically similar at intake with regard to symptom scores, premorbid IQ and gender, although the UHR-psychotic group were significantly younger at both time points. The comparison between baseline and follow-up scans for the UHR-nonpsychotic group indicated no difference between the two time points in any brain region, which is concordant with the lack of clinical progression in those subjects. However, in the UHR-psychotic group, two regions of the left hemisphere reduced in volume over the follow-up period. These were a left inferior frontal region, and a left medial temporal region that included the hippocampus, surrounding parahippocampal gyrus and the fusiform gyrus [102].
This finding, if confirmed by further investigations, implies that there is change occurring in the brains of patients developing schizophrenia, something that could perhaps be prevented or ameliorated by early intervention. However, the SMaRT technique cannot distinguish between volume loss as a result of neuronal loss, glial cell loss or other pathological processes. Magnetic resonance technology that allows us to further evaluate this issue is MR spectroscopy. In our recent review of the available studies utilising this technique in schizophrenia, we concluded that there was a reasonably consistent finding of reduced N-acetylaspartate (NAA) in the hippocampi of patients when compared to controls [103]. NAA is a biochemical that is found almost entirely in neurones and, as such, can be considered a marker of neuronal loss or dysfunction. Hence, the spectroscopic findings correspond well with the structural findings in established schizophrenia and first-episode psychosis. However, to date there have been no studies using MR spectroscopy in highrisk populations (either ‘genetic’ or UHR groups), and no longitudinal studies of such groups. Clearly these studies will be important in developing our understanding of the role the hippocampus plays in schizophrenia.
Summary
Neuroimaging has been a highly useful tool for the investigation and understanding of structural abnormalities in schizophrenia. Evidence from a number of sources points to a neurodevelopmental origin for the illness, but the data outlined earlier suggest that structural changes may be also occurring later in life, particularly in the period around the first onset of illness. We concur with McCarley and colleagues [104] in postulating an additional neural insult that occurs during the evolution of psychosis. Our data suggests a model in which an early neurodevelopmental lesion renders the hippocampus vulnerable to a later lesion, which is possibly mediated through the effects of stress [105], cannabis or some other as yet unidentified pathogen at the time of the first episode of illness. The result of this is to cause an already vulnerable left hippocampus to lose volume. Following this reduction in volume early in the course of psychosis, we suspect that there might be a slower reduction in right hippocampal volume with continued psychotic illness. This could be due to neuroleptic medication, or other agents similar to those suggested previously. The available data therefore supports a possible three-hit model, with an early insult during neurodevelopment being followed by further insults during the transition to illness and with progression to chronic persistent illness (Fig. 1). We are presently attempting to confirm our findings in further work with ultra-high risk and first-episode patients, both through structural and spectroscopic MR.
Three-hit model for hippocampal damage in schizophrenia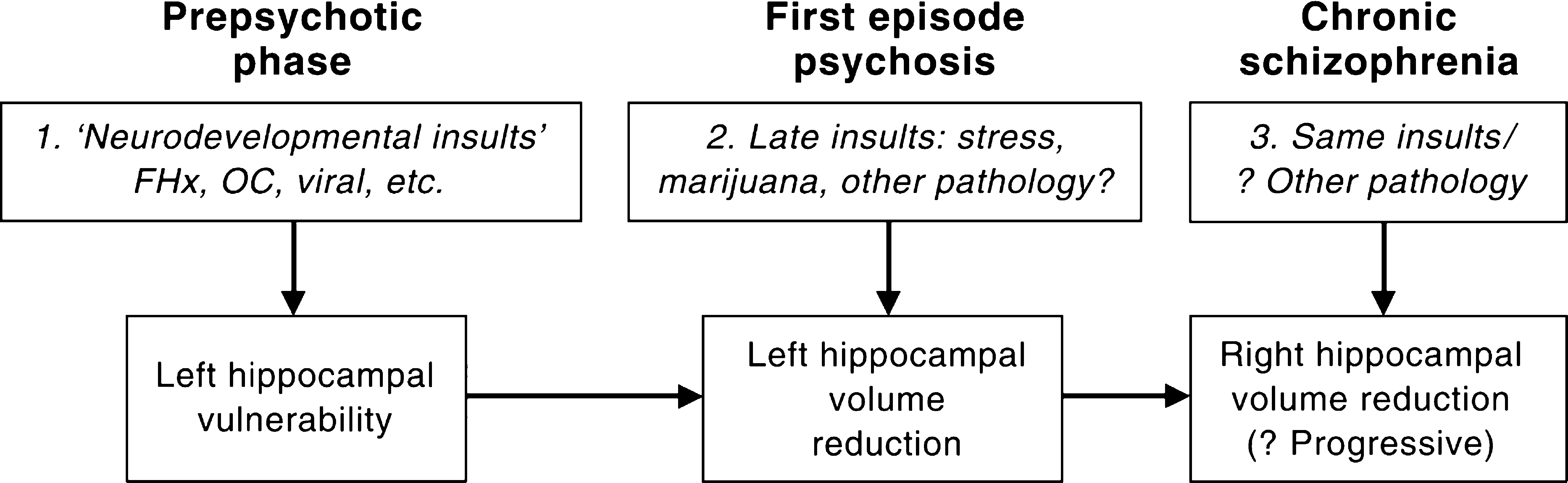
Footnotes
Acknowledgements
This work was supported by project grants from the National Health & Medical Research Council (NH&MRC; grant ID nos. 970598, 970391, 98112), Victorian Health Promotion Foundation, the Stanley Foundation, Ian Potter Foundation, Woods Family Trust, Jack Brockhoff Foundation, the L. E. W. Carty Trust and the Percy Baxter Charitable Trust. Professor McGorry is supported by a NARSAD (National Alliance for Research on Schizophrenia and Depression) Distinguished Investigator Award. Dr Wood is an NHMRC Research Officer, supported by an NHMRC grant (no. 981112).
