Abstract
Study Design
Retrospective study.
Objective
We reviewed cases of surgically treated cervical spondylotic myelopathy (CSM) or chronic, degenerative myelopathy of the subaxial cervical spine to study the incidence of inadequate surgical decompression.
Methods
We included all persons treated at our institution after a first surgical decompression for CSM over a 3-year period. Inadequate original surgical decompression was defined as neurologic decline within 12 months postoperatively and ongoing impingement of the spinal cord with <1-mm change in anteroposterior canal dimension from pre- to postoperative magnetic resonance imaging (MRI) leading to revision decompressive surgery. Revisions for other reasons were not counted as inadequate.
Results
Of 50 patients, 5 (10%) required revision decompression for neurologic decline and inadequate change in space available for the cord on postoperative imaging; 4 patients declined within the first 6 months and 1 patient at 8 months postoperatively. None of the 5 declined further after posterior revision, but none recovered from the interval loss. All 5 had undergone anterior approaches, for an anterior inadequacy rate of 23% (5 of 22). None of the 28 patients having posterior or combined approach declined at 2 years or had <1-mm change on postoperative MRI. The difference between anterior and posterior approaches was statistically significant (p = 0.018).
Conclusions
The rate of inadequate surgical decompression for CSM was greater than expected in this series and directly associated with an anterior approach. No cases of inadequacy occurred for posterior or combined approaches. Postoperative neuroradiographic imaging such as MRI should be entertained routinely for this entity or at least for anterior-only approaches.
Keywords
Introduction
The natural history of cervical myelopathy from spinal cord compression has been described as a progressive process, with intermittent exacerbations of symptoms often followed by periods of static, or only slightly worsening, function. 1 Patients with cervical myelopathy may complain of gait abnormalities, numbness or weakness in the upper extremities, diminished fine motor control, and changes in bowel or bladder function with the most severe end stage being wheelchair-bound tetraplegia or death. 2 Myelopathic symptoms are largely irreversible, and the limited degree of recovery depends on the severity of the myelopathy at the time of intervention in addition to other factors such as the duration of symptoms and patient age. 3 The literature suggests that surgical decompression may alter the natural history of this disease most typically by halting progression. 4 , 5 , 6
There is a paucity of published data on the incidence of patients with myelopathy who have undergone a surgical decompression that was subsequently found to be inadequate. This lack of data may be because postoperative magnetic resonance imaging (MRI) or equivalent neuroimaging of the decompression is not commonly obtained unless, or until, a subsequent neurologic decline is noted, most often without report in the medical literature. We performed a retrospective review of all surgically treated chronic, degenerative myelopathy cases transferred to our rehabilitation center to determine the rate of inadequate surgical decompression at the intended level(s) leading to revision surgery due to neurologic decline.
Methods
Database query and retrospective review of medical records was performed for all patients who were referred to our rehabilitation center from 2009 to 2012 and who were identified as first-time postoperative for a diagnosis of cervical spondylotic myelopathy (CSM) or chronic, degenerative spinal cord myelopathy in the subaxial cervical spine that required surgical decompression of an offending degenerative, nonneoplastic, noninfectious mass as part of the surgical intervention. The indication for all index surgeries was clinically significant myelopathy as assessed by each individual treating spinal surgeon. The interval selected represents the time at which the senior author began consulting at this institution up to the writing of this report to allow minimum 2-year follow-up for all. Patients were excluded for reducible instability not requiring decompression, tumor, infection, and thoracic or occiput–C2-level disease.
Fifty consecutive patients met the inclusion criteria and underwent postoperative MRI to compare with the preoperative imaging, on average 2 months or more after surgery to avoid acute postoperative imaging changes influencing interpretation and measurements. Cases of inadequacy were identified due to worsening neurologic signs or symptoms occurring within 12 months of the index surgery if, and only if, a clear inadequacy of decompression was noted at a treated level when comparing the preoperative to postoperative neuroimaging. To qualify, a “near-zero” change in the anteroposterior (AP) dimension of the spinal canal was required such that less than a 1-mm change in this dimension could be resolved on MRI in association with ongoing cord compression. No cases had medial-lateral compressive lesions as compared with AP. The neurologic assessments were made using the American Spinal Injury Association (ASIA) examination, and patients were graded on the ASIA impairment scale and assigned a Nurick score for myelopathy. Second surgeries were excluded from the label of inadequacy if the revision was performed for nonunion, adjacent-level degeneration, new surgical lesion not present at the index surgery, or instrumentation failure/misplacement. One patient with CSM was excluded for new, severe neurologic deficit sustained intraoperatively (motor complete).
The statistical analysis was performed using IBM SPSS Statistics (IBM Corp., Armonk, New York, United States). The rate of inadequate surgical decompression was determined from the number of identified cases divided by the whole. The rates of inadequacy were determined for the entire group and for individual approaches, anterior, posterior, or combined. Body mass index, age, and the number of actually occurring medical comorbidities (i.e., hypertension, diabetes, coronary artery disease, liver disease, chronic kidney disease, chronic obstructive pulmonary disease, major psychiatric illness, rheumatoid arthritis, stroke, thyroid disease, and drug or alcohol abuse) were compared between the groups.
Referred cases came from multiple university, private practice, county hospital, and HMO settings representing a range of experience, practice variety, and orthopedic and neurologic surgeons throughout Los Angeles County, thereby providing a broad representative sample of spine surgeons at large in the region.
Results
In total, 50 patients were identified as first-time postoperative for a diagnosis of chronic, compressive, subaxial cervical myelopathy over the 3-year period (Table 1); 5 of the 50 underwent revision surgery due to worsening neurologic signs or symptoms attributable to persistent cord compression (i.e., less than 1-mm AP change in canal dimension on pre- to postoperative neuroimaging measured on axial slices) for an overall inadequacy rate of 10% (Table 2). All 5 cases of inadequacy were anterior in approach, so the rate of inadequacy for the anterior approach was 23% (5/22). Of the remaining 45 patients, all had a greater than 1-mm change in canal diameter and none declined during 2-year follow-up, so there were no other near-zero cases that remained neurologically stable. In addition, none of the posterior or combined approach cases were revised for inadequacy of decompression and none of these declined neurologically. The inadequacy rate for anterior decompression was statistically significantly greater than posterior (p = 0.018). Patients revised for inadequacy were not different from all others for body mass index (mean of 28 for revisions versus 27, p = 0.72), number of comorbidities (mean of 2 for revisions versus 2.11, p = 0.86), or age (mean of 63 for cases versus 60, p = 0.44). There were 3 male and 2 female patients in the inadequate group and 33 male and 12 female patients in the remainder.
Index surgery for cervical spondylotic myelopathy
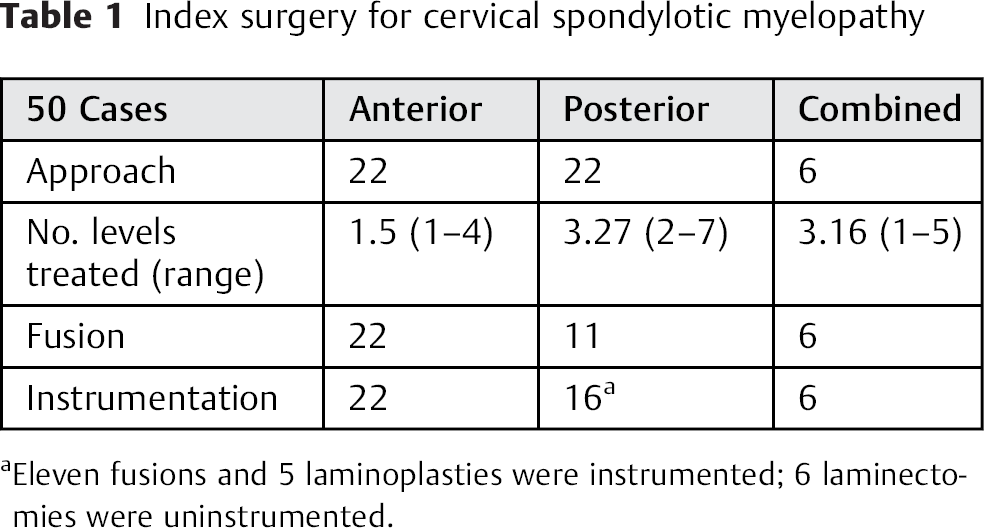
Eleven fusions and 5 laminoplasties were instrumented; 6 laminectomies were uninstrumented.
Cases of inadequate surgical decompression
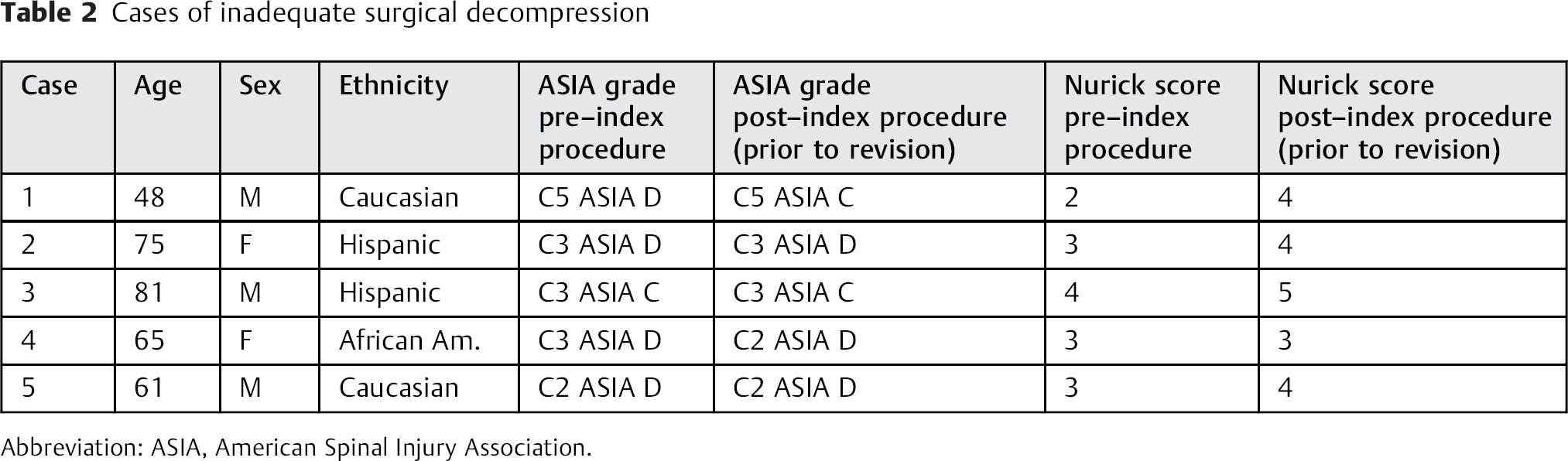
Abbreviation: ASIA, American Spinal Injury Association.
The neurologic changes prompting the revisions in conjunction with a near-zero change are listed in Table 3; 4 of the 5 patients declined within 6 months of the index surgery and 1 patient at 8 months. None of the 5 declined further after posterior revision decompression, but neither was there any improvement above the new decline. A second postoperative MRI after these revisions revealed adequate decompression with restoration of cerebrospinal fluid flow anterior and posterior to the spinal cord; none of the patients experienced operative complication. The operating microscope was used surgically for each of the 5 cases.
Interval neurologic changes in cases of inadequate surgical decompression
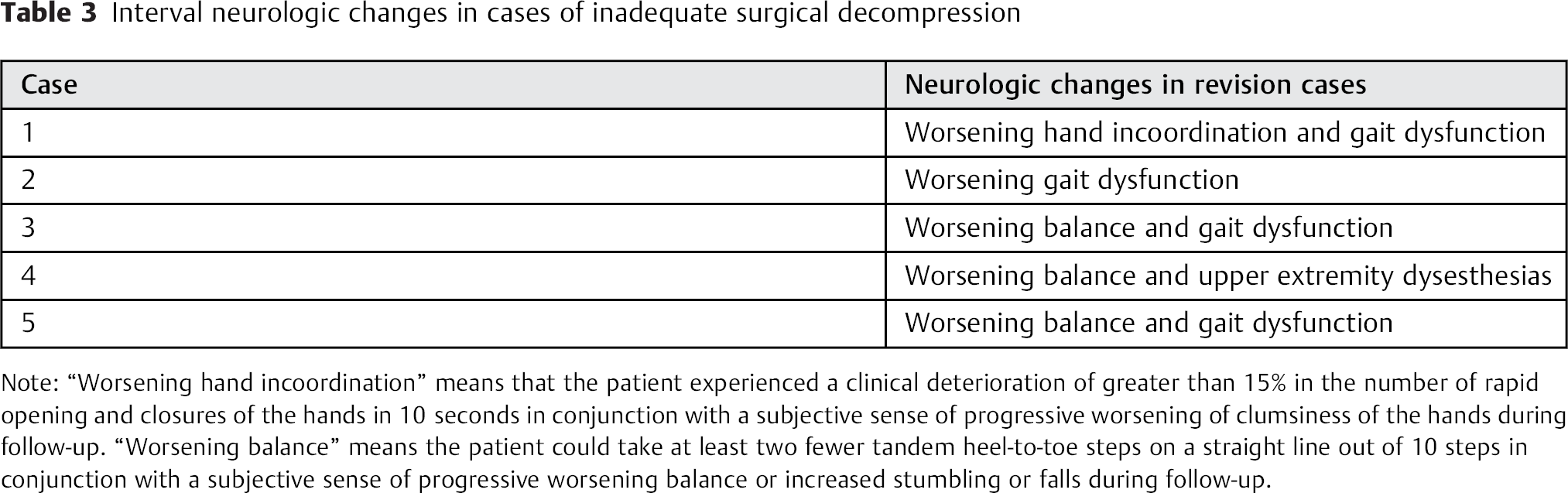
Note: “Worsening hand incoordination” means that the patient experienced a clinical deterioration of greater than 15% in the number of rapid opening and closures of the hands in 10 seconds in conjunction with a subjective sense of progressive worsening of clumsiness of the hands during follow-up. “Worsening balance” means the patient could take at least two fewer tandem heel-to-toe steps on a straight line out of 10 steps in conjunction with a subjective sense of progressive worsening balance or increased stumbling or falls during follow-up.
The mean Nurick score for all 50 patients was 3.36. 7 For the 45 patients in the nonrevision group, the mean preoperative Nurick score was 3.4 and remained stable or slightly improved during 2-year follow-up. For revision cases, the mean index preoperative Nurick score was 3.0, slightly better initial function than the nonrevision group, which fell to a mean of 4.0 postoperatively prior to revision (Table 2). No statistical differences were found for age, gender, or disease severity at presentation between the two groups. Figs. 1 and 2 present illustrative pre- and postoperative MRIs of a revision case versus a nonrevision case.
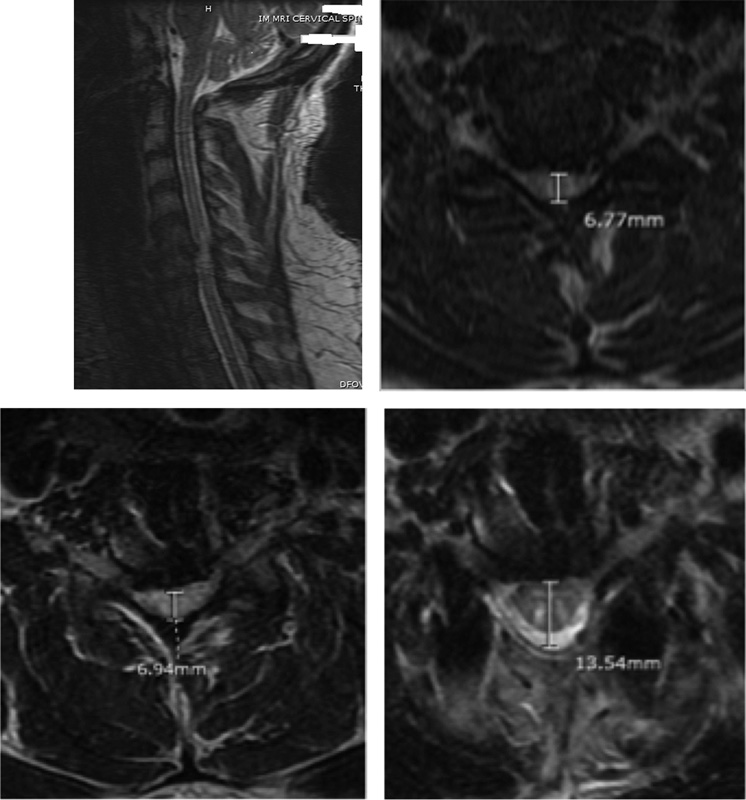
Upper left: Sagittal preoperative T2-weighted magnetic resonance imaging (MRI) of a revision case. Upper right: Preoperative T2-weighted axial MRI with canal measurement of 6.77 mm. Lower left: Postoperative T2-weighted axial MRI with canal measurement of 6.94 mm and “near-zero,” less than 1-mm, change. Lower right: Second postoperative T2-weighted axial MRI after posterior revision with canal measurement of 13.54 mm.
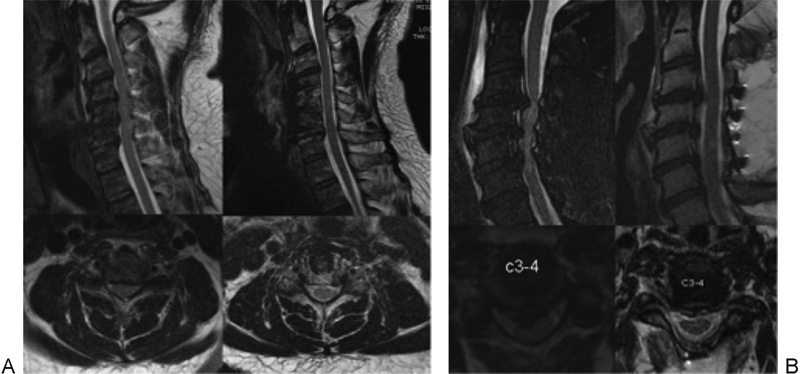
(A) Magnetic resonance imaging (MRI) of an adequately decompressed two-level anterior cervical decompression and fusion. Preoperative T2-weighted sagittal MRI and representative C4–C5 axial (left column) versus postoperative (right column). (B) Adequately decompressed four-level posterior cervical laminoplasty. Preoperative T2-weighted sagittal MRI and representative C3–C4 axial (left column) versus postoperative (right column).
One major intraoperative event (ASIA B motor complete with changes noted on intraoperative neuromonitoring) occurred from an anterior approach and was excluded from analysis for inadequate decompression. Therefore, the overall major intraoperative event occurrence rate was 1 in 51 or 1.9%. None of the 50 included cases had an intra- or perioperative event or neuromonitoring changes. The documentation for all anteriorly approached cases included removal of the posterior longitudinal ligament (PLL) with visualization of the thecal sac. There was visualization and expansion of the thecal sac after laminectomy for all 5 posterior laminectomy and fusion revisions.
Discussion
We identified 5 of 50 cases of inadequate surgical decompression performed for chronic, degenerative, first-time postoperative myelopathy, for an inadequacy rate of 10%. All cases of inadequacy were approached anteriorly so that the rate of inadequacy for the anterior (alone) approach was 23% (5/22). These numbers likely represent a minimum rate of inadequacy given our strict inclusion criteria of near-zero surgical change (i.e., less than 1-mm change in AP dimension from pre- to postoperative MRI) in conjunction with worsening neurologic status within 12 months of the attempted decompression. There was also one new major intraoperative neurologic event (motor complete), which was also approached anteriorly, excluded from this analysis, yielding a 1.9% occurrence rate.
It appears that the anterior cervical approach is highly susceptible to decompressive inadequacy as regards degenerative myelopathic compression. We surmise this result may be due to the smaller working portal through the disk space with this approach versus the posterior one. Though surgical probing can be performed to assess decompression beyond the viewing window, this maneuver may not always be fully reliable to surgeons. We wondered whether an older paradigm for fusion without removal of the entire posterior annulus and PLL might also be responsible, but the extracted operative dictations for all five cases documented removal of the posterior annulus and PLL with visualization of the thecal sac in each. MRI findings revealed failure to remove the PLL in these cases, suggesting there may be inherent difficulty in deciding or identifying the PLL or dura mater or both in some cases, and intraoperative assessment may not be reliable for all cases.
Another possible explanation for the decline in the revision cases is preexisting irreversible cystic necrosis of the spinal cord as seen with severe cord signal change on MRI. Reports of decline despite adequate decompression have been issued in this setting. 8 , 9 Only 1 of the 5 revision cases had significant preoperative cord signal change (Fig. 1), and because the remaining 45 nonrevision cases had slightly worse levels of cord signal change preoperatively and none declined neurologically nor had less than 1-mm change in AP canal diameter, persistent compression seems more likely.
As spine surgeons conduct routine postoperative radiographic surveillance of instrumented cases to assess union and position, given the considerable overall 10% rate of failed decompression leading to further irreversible neurologic decline suggested by this study, we recommend consideration of routine postoperative MRI or equivalent neuroradiographic imaging for surgical cases involving myelopathy. Although one might adopt a watch-and-wait approach pending resolution of symptoms to save time and money spent on additional MRI in decompression for radiculopathies, such an approach seems unsound for the more dangerous and largely irreversible myelopathy. That is to say, a watch-and-wait approach carries a real risk of neurologic decline, which may not be regained upon subsequent realization and revision, as evident in this study. It is significant to note that the posterior or combined approach in this small series had no occurrence of inadequacy and the anterior alone approach contained all the inadequacies. As such, postoperative MRI should be considered at least for the anterior approach, particularly given that intraoperative assessment does not appear consistently reliable given this data. Furthermore, although this study suggests that the posterior or combined approach may be superior with regard to avoidance of inadequate decompression, it makes no statement as to whether one approach is better than another when the decompression is adequate and there may be other reasons why the anterior approach is preferable such as recovery, amount of muscular dissection, alignment, complications, and so on. Nonetheless, we contend that the most important consideration for patients with myelopathy is an adequate decompression given that the surgical goal is to arrest the neurologic decline in the first place, and so, ceteris paribus, the posterior approach may be preferable for myelopathy, which is a potentially catastrophic, irreversible entity.
An advantage to this study is that the sample presented reflects cases performed by a broad cross section of spinal surgeons practicing throughout the Los Angeles County basin and neighboring regions, one of the largest in the United States. The patients were referred from public and private university and academic centers and private and HMO-based institutions. Thus, the treating surgeons are likely to represent a full-spectrum sample of actual practicing orthopedic and neurosurgical spine surgeons with variable practices, experience, and skill levels versus an isolated series from a high-volume or single-surgeon ultraspecialist. Nonetheless, the sample is small and the rate reported may only represent the rate for patients with CSM referred to our institution.
One weakness of this study is that it is a retrospective review. Future study might include a prospective analysis as well as larger numbers of patients with longer follow-up to clarify subtle differences in neurologic recovery or stability relative to varying amounts of decompression achieved against the severity of initial compression and clinical status, and we are presently engaged in this endeavor. As regards longer follow-up, our study is likely to underreport at only the minimal rate of inadequacy because cases may be slower to appear with marginal, but not near-zero, decompression and may not have surfaced yet. Though our inclusion criterion for inpatient rehabilitative care may bias our patient population, it allowed us to accurately identify and reimage all cases of postoperative myelopathy because clear records and consults exist for all transfers. Also, all 50 patients were transferred for simple rehabilitative optimization with an average Nurick score of 3.36 and were at least minimally ambulatory.
In sum, the rate of inadequate surgical decompression for spinal cord myelopathy is significant, and intraoperative assessment of decompression does not appear to be consistently accurate, at least for the anterior approach. When inadequacy occurs, the disease is permitted to progress until neurologic decline makes it known and revision decompression does not appear to afford improvement above the interval loss. Given these results, routine postoperative neuroradiographic assessment of surgical decompression should be entertained for patients with myelopathy treated with the anterior approach, possibly all approaches, given the high stakes, unless or until routine intraoperative use of MRI or its equivalent becomes a reality.
Disclosures
Amandeep Bhalla: none
Kevin W. Rolfe: none
