Abstract
Objectives
Primary objectives were outcomes comparison of instrumented surgery used for de-novo spinal infections in terms of infection recurrence, reoperations, primary failure, mortality, and length of stay relative to non-instrumented surgery. Secondary objectives were outcomes for surgical and non-surgical treatment of de-novo spinal infections regarding recurrence of infection, mortality, quality of life, and length-of-stay.
Methods
A systematic literature review was performed using the PubMed database. Studies comparing outcome variables of patients with de-novo spinal infections (DNSI) treated with and without instrumentation and surgical versus non-surgical treatment were included. Studies primarily focusing on epidural abscesses or non-de-novo infections were excluded. A meta-analysis was performed for infection recurrence, reoperation, primary treatment failure, mortality, and quality-of-life parameters.
Results
A total of 17 retrospective studies with 2.069 patients met the inclusion criteria. 1.378 patients received surgical treatment with or without instrumentation; 676 patients were treated non-surgically. For the comparison of instrumented to non-instrumented surgery Odds-Ratios were .98 (P = .95) for infection recurrence, .83 (P = .92) for primary failure, .53 (P = .02) for mortality and .32 (P = .05) for reoperation. For the comparison of non-surgical to surgical treatment, Odds-Ratios were .98 (P = .95) for infection recurrence, and 1.05 (P = .89) for mortality.
Conclusion
Available data support that instrumented surgery can be performed safely without higher rates of infection recurrence or primary failure and lower reoperation and mortality rates compared to nonsurgical treatment for DNSI. Furthermore, spine surgical treatment may generally be performed without higher risk of infection recurrence and mortality and better quality-of-life outcomes compared to generic non-surgical treatment.
Keywords
Introduction
Spontaneous spinal infections such as discitis with or without vertebral osteomyelitis, in the following referred to as ‘de-novo spinal infections’ (DNSI), are an increasing medical problem. The incidence of spinal infections has nearly doubled in developed countries from 2010 to 2019. 1 Due to profound differences regarding the disease origin and the clinical course, it appears sensible to differentiate non-pyogenic diseases and implant-associated perioperative infections as well as purely epidural abscesses from DNSI which historically predominantly managed non-surgically with intravenous antibiotics and immobilization.2-5 The basic management tenants of DNSI have remained unchanged starting with identification of the infectious pathogen, providing targeted antibiotic therapy, immobilizing the affected area of the spinal column, to providing pain control, and emphasizing nutritional support.2,3 Surgical treatment for DNSI, unlike the management of implant-associated perioperative infections, has remained reserved for more severe cases presenting such as new or progressive neurological impairment, development of unacceptable secondary spinal deformity, manifest spinal instability, insufficient response to antibiotics, and severe and intractable pain.3,6 In contrast, principles of infection treatment for implant-associated perioperative infections routinely consist of surgical debridement, hardware and bone graft management, and antibiotic treatment.
Preservation of spinal stability is an important aspect of spinal infection treatment and can be predictably achieved by instrumented spine surgery. In contrast to this, the use of instrumented surgery for de-novo infections remains disputed among practitioners of different specialties. Such concerns about instrumented spine surgery usually arise out of more generalized assumptions extrapolated from perioperative implant-associated infections, such as infected arthroplasties and trauma surgery as well as cases of revision spine surgery, even though these are entirely different disease entities.2,6-8 Such specific concerns fear higher rates of recurrent infection or emergence of chronic infections that could be incurred through biofilm formation on implant surfaces as a foundation for bacterial colonization.7-9 Proponents of spinal instrumentation in DNSI identify the potential to provide affected patients with more physiologic spinal column alignment and stability of the impaired spinal segments, protect neural element passage space integrity and allow for earlier and more aggressive mobilization with fewer pain.10,11
Methods
A protocol was registered with PROSPERO (CRD42022337612) and our Systematic Review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 12
Inclusion and Exclusion Criteria
We included studies that compared outcome variables of patients with DNSI treated with non-surgical and operative therapy and studies that compared surgical treatment with and without instrumentation in the management of DNSI. Our lower cohort threshold for inclusion was a minimum of 20 patients. Included studies were limited to German and English language publications. Those studies reporting only epidural abscesses, postoperative surgical site infections, revision surgeries, spinal tuberculosis (Pott’s disease), pediatric cases, spinal fungal infections, studies that reported on specific subgroups, and studies solely including data for surgical, non-surgical, instrumented or non-instrumented treatment without an internal comparison cohort for the treatment modality were excluded from our SR. Case reports and studies with populations that mainly consisted of spinal infections other than DNSI, such as perioperative infections, were also excluded from this study. We created cohorts based on the general therapeutic approach used and based on key outcomes. If the study cohorts had similar but non-substantial differences for comorbidities and an existing internal comparison cohort for the treatment modality (surgical versus non-surgical or instrumented versus non-instrumented surgery), meta-analyses were performed.
Instrumented surgery was defined as surgery that involved the use of medical devices, including cages, screws, rods, or hooks, while non-instrumented surgery was defined as a surgical procedure in the OR including surgical decompression, debridement with or without irrigation, discectomies, or surgical bone debridement. Interventional procedures like puncturing of the infective process or biopsy procedures were not defined as surgical procedures.
Database and Search Items
A Pubmed database search with the search item “(Spondylodiscitis [title/abstract] OR Spondylodiskitis OR “vertebral osteomyelitis” [title/abstract] OR osteodiscitis [title/abstract] OR osteodiskitis [title/abstract] OR discitis [title/abstract] OR diskitis [title/abstract]) AND (“treatment failure” [title/abstract] OR “treatment” [title/abstract] OR “antibiotics” [title/abstract] OR surgery [title/abstract] OR instrumentation [title/abstract]) NOT (tuberculosis [title/abstract])” was performed. A total of 17 publications reached the inclusion criteria.
Data Collection
All available demographic data, clinical data, and relevant outcomes data were extracted into an Excel database. Demographic and clinical data included age, sex, number of patients, comorbidities, and location of infection. Outcomes data included mortality, infection recurrence, reoperation, primary failure, pain, quality of life using patient-reported outcomes (PRO’s), and length-of-stay (LOS). Each included study was assessed for the risk of bias using the corresponding SIGN methodology checklist.
Statistical Analysis
Means were built based on the number of patients pooled for each variable. When available, odds ratios were calculated based on the pooled data with ReviewManager (RevMan V. 5.4.). In addition, RevMan was used to build forest plots for the variables of ‘infection recurrence’, ‘primary treatment failure’, ‘mortality’, ‘physical activity, and ‘pain’. Odds ratios for individual studies and the total of all included studies were calculated using the Mantel-Haenszel test. Study heterogeneity was tested with the chi-square and I2 test. Significant heterogeneity was considered when the I2 value exceeded 50% or the chi-square value was within a 10% level of significance (P < .10).
Results
Study Selection
Our formal search produced a total of 2.373 publications. Each headline was screened by two investigators for suitability to our inclusion criteria. Subsequently, a total of 120 abstracts were reviewed for inclusion. This resulted in 28 articles that underwent a full-text article review. 13 publications met our stated criteria for inclusion. A citation search of the 13 included publications was performed, and four additional studies were included in the review. No automation tools were used. The study’s selection process is shown in Figure 1. Study selection process using PubMed database and citation search.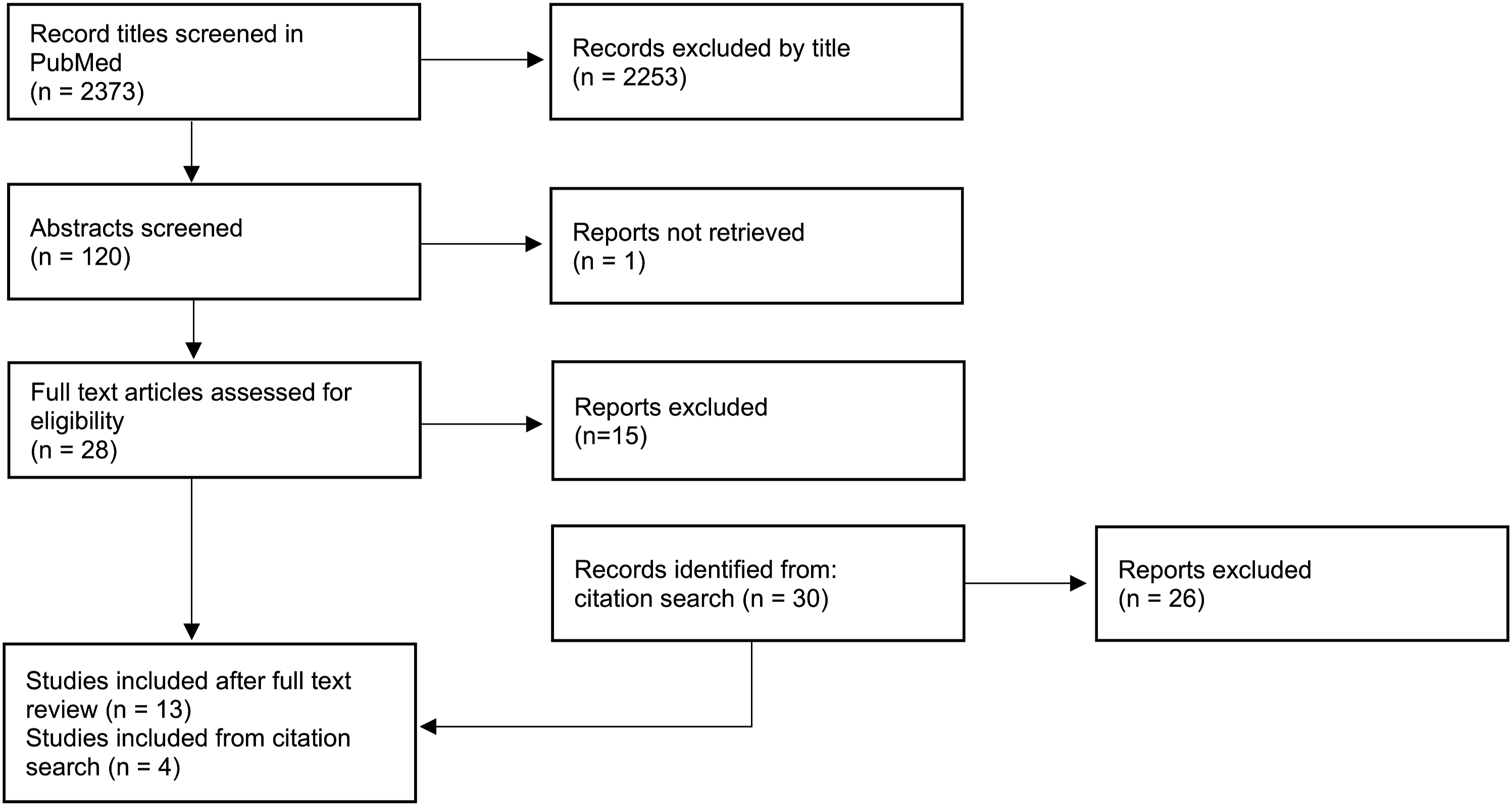
Studies like that of Pojskic et al. 2021: “Neurosurgical Management and Outcome Parameters in 237 Patients with Spondylodiscitis” or Dietz et al. 2019: “Outcomes of decompression and fusion for treatment of spinal infection” were excluded as they included a substantial proportion of patients with postoperative surgical site infections.13,14
Demographic and Clinical Data
Overview of Included Studies Comparing Surgical Treatment to Non-Surgical Treatment for Spinal Infections.
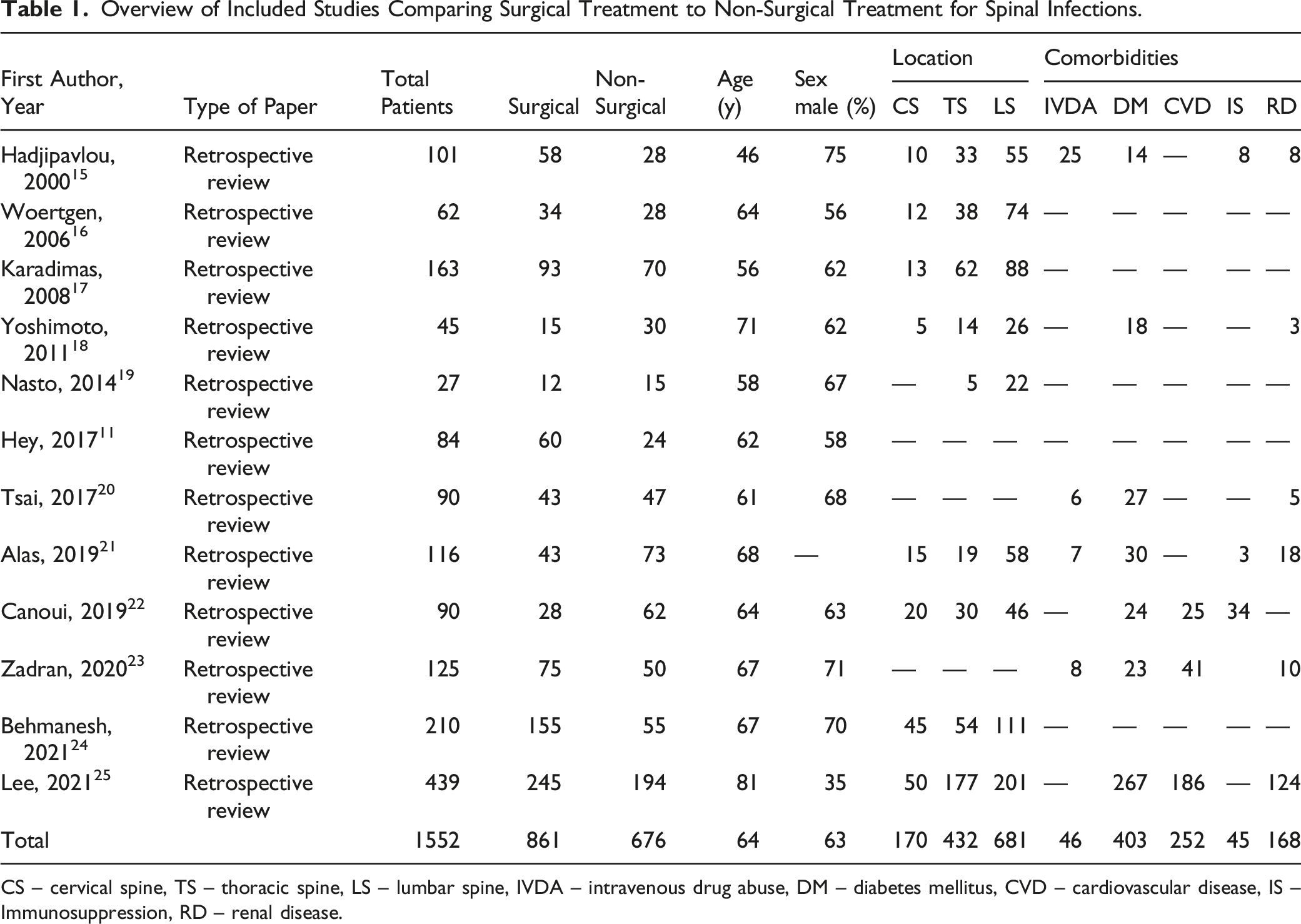
CS – cervical spine, TS – thoracic spine, LS – lumbar spine, IVDA – intravenous drug abuse, DM – diabetes mellitus, CVD – cardiovascular disease, IS – Immunosuppression, RD – renal disease.
Overview of Included Studies Comparing Instrumented Surgery to Non-Instrumented Surgery for Spinal Infections.
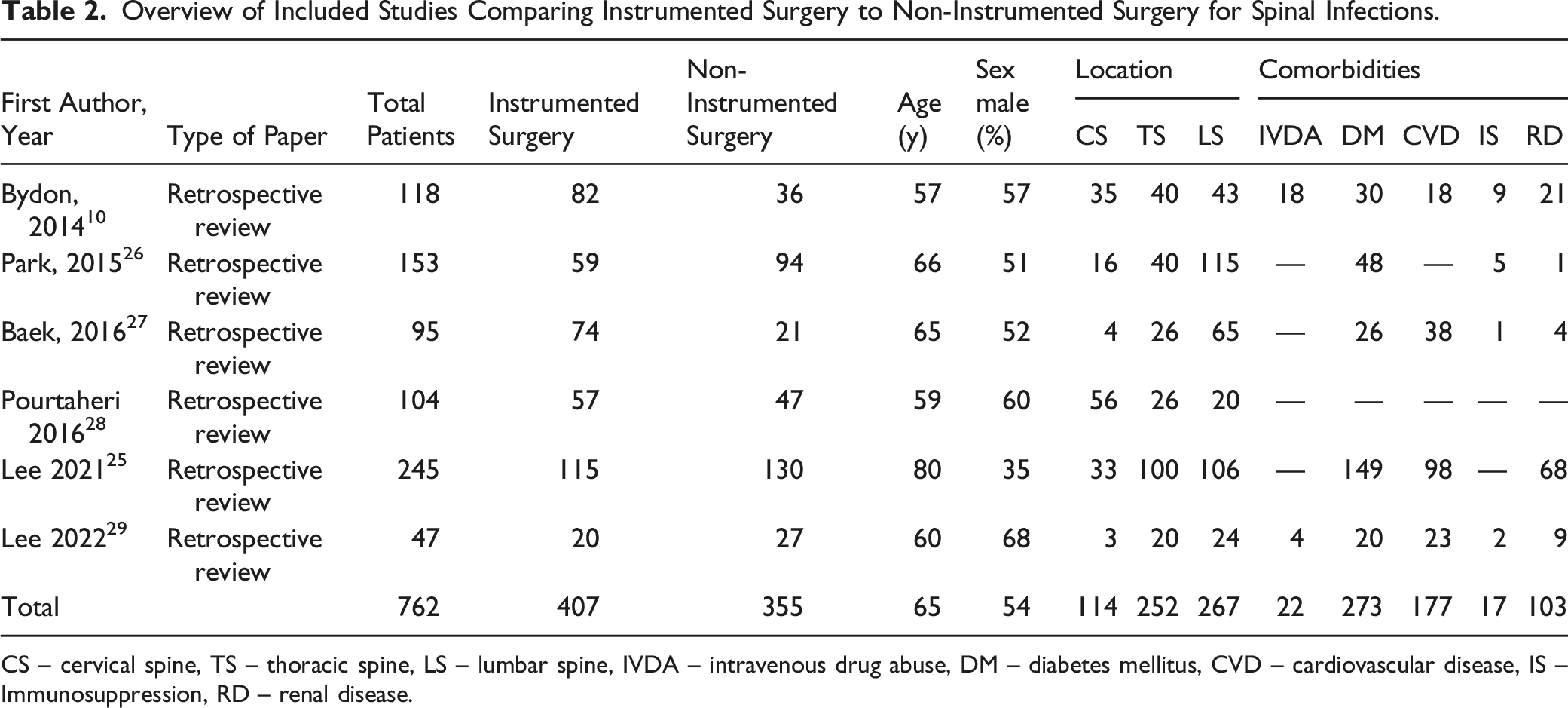
CS – cervical spine, TS – thoracic spine, LS – lumbar spine, IVDA – intravenous drug abuse, DM – diabetes mellitus, CVD – cardiovascular disease, IS – Immunosuppression, RD – renal disease.
Overall, 16% of the spinal infections were in the cervical spine (284/1.815), 32% were thoracic spine infections (584/1.815), and 52% were in the lumbar spine (948/1.815). The most common comorbidity identified was diabetes mellitus (36%, 527/1.419), followed by cardiovascular diseases (36%, 331/914), renal disease (15%, 203/1.329), intravenous drug abuse IVDA (11%, 68/597) and immunosuppressive disorders (9%, 62/720).
Successful Infection Management: Recurrence of Spinal Infection, Reoperation and Primary Failure
Six of the included studies reported recurrence of infectious processes.11,22,25-27 Four studies reported reoperation rates10,11,17,29 and two reported primary failure.26,27
Overall, none of the included investigations showed a significantly higher rate of infection recurrence when instrumented surgery was used (OR .91 (.50-1.64) P = .74 (Figure 2)).10,21,26,27 In addition, Hey et al. in a comparison of medical management to surgical debridement and surgical debridement with spinal instrumentation found that there was no higher rate of infection recurrence in the group treated with spinal instrumentation surgery (OR 1.00 vs. .88-1.31 vs. .82-1.20, P = .46 and .95).
11
Furthermore, no difference was seen for infection recurrence (5% vs. 4% P = 1.00)
22
when comparing medical treatment to surgical treatment for DNSI. The same findings were reported by Alas et al., who showed no significant difference in recurrence of spondylodiscitis for patients treated operatively versus non-surgically (11.6% vs. 16.4%, P = .59).
21
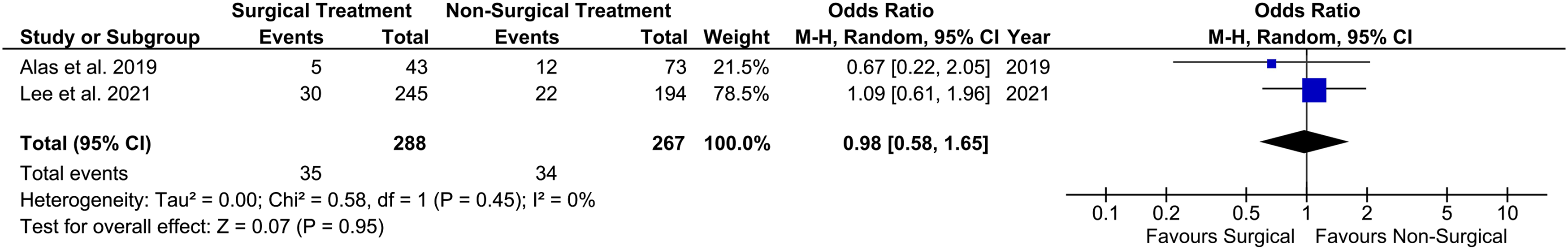
Cumulative results are demonstrated in forest plots in Figure 2 and Figure 3. Recurrence of infection for instrumented vs. non-instrumented surgery – the forest plot showed no difference in overall Odds-Ratios for infection recurrence for instrumented surgery compared to noninstrumented surgery. Recurrence of infection for surgery vs. non-surgical treatment - Forest plot showed no significant difference for the overall Odds-Ratios for infection recurrence.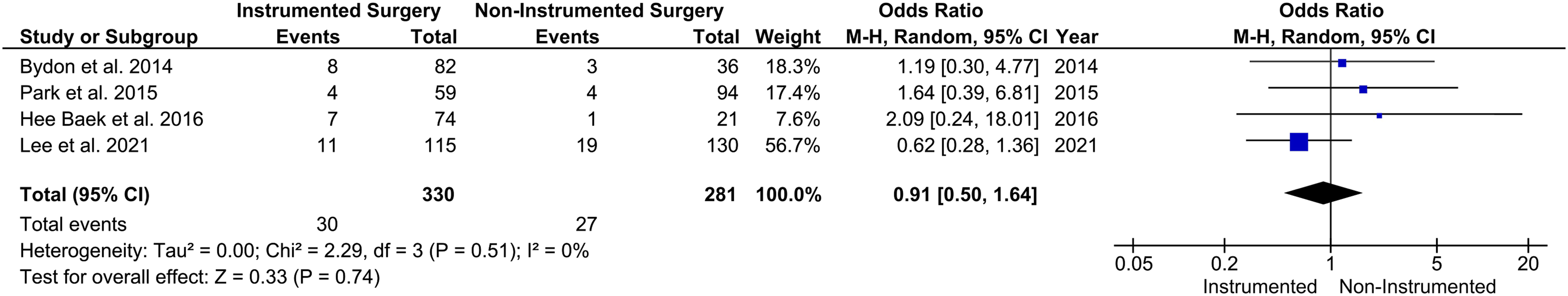
In addition, Hee Baek et al. and Park et al. showed no significant differences in primary failure for non-instrumented compared to instrumented spine surgery26,27 (Figure 4). In both studies, primary failure was defined as patients who needed surgical debridement after surgical intervention before completion of the antibiotic treatment due to uncontrolled infection.26,27 Primary failure - Forest plot demonstrated no significant difference for overall Odds-Ratios for instrumented surgery compared to non-instrumented surgery.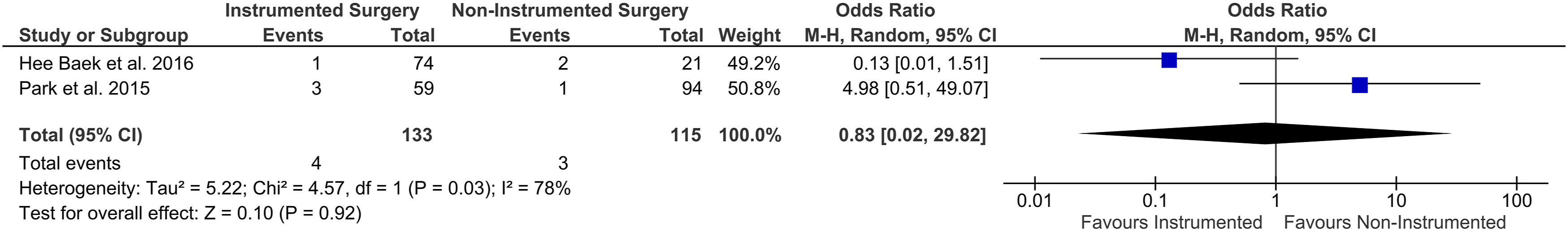
Bydon et al. reported that patients without instrumentation had similar rates of ongoing discitis/osteomyelitis and similar overall reoperation rates compared to instrumented surgery (8.3% vs. 9.8 P = .80; 19.44 vs.17.07 P = .76).
10
Furthermore, Hey et al. showed no significant difference in reoperation rates between surgical treatment with debridement or debridement with instrumentation and non-surgical treated patients (OR .93 (.84-1.02) P = .013 and OR .91 (.83-1.01) P = .07).
11
In fact, Bydon et al., Lee et al., and Karadimas et al. found lower reoperation rates for instrumented to non-instrumented surgery (OR .32 (.10-1.00) P = 0,05)11,17,29 (Figure 5). Reoperation - Forest plot showing significantly lower overall Odds-Ratios for reoperation for instrumented surgery compared to non-instrumented surgery for the included studies.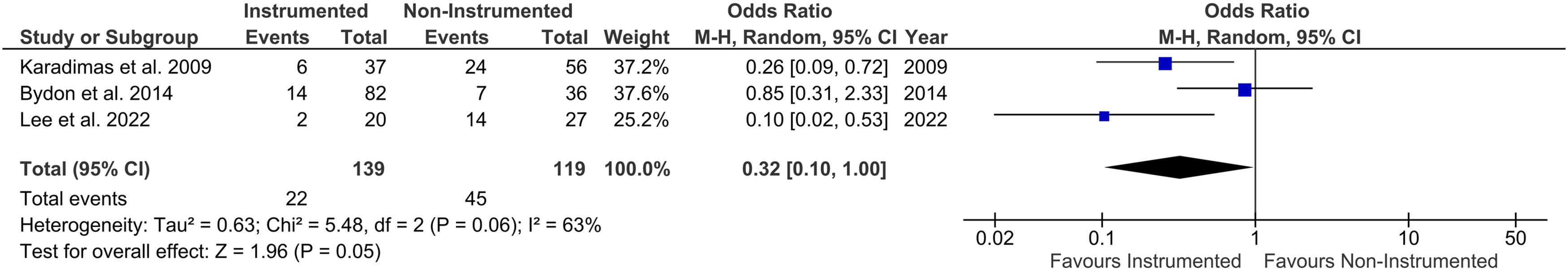
Mortality
Eight studies compared the mortality rates of nonsurgical management and surgical treatment with instrumented to non-instrumented spine surgery.11,21–23,25–28 The overall Odds-ratios for the included studies showed no significant difference in mortality rates for surgical compared to non-surgical treatment (OR 1.05 (.50-2.24) P = .89),21,22,25 whereas mortality was significantly lower in the instrumented compared to the non-instrumented group (OR .53 (.32-.89) P = .02)25-28 (Figure 6 and 7). Mortality rates for surgical compared to non-surgical therapy - Forest plot showed no significant difference for overall Odds-Ratios for mortality for surgery compared to conservative treatment. Mortality rates for instrumented compared to non-instrumented surgery - Forest plot represents a significantly lower overall Odds-Ratios for instrumented surgery compared to non-instrumented spine surgery.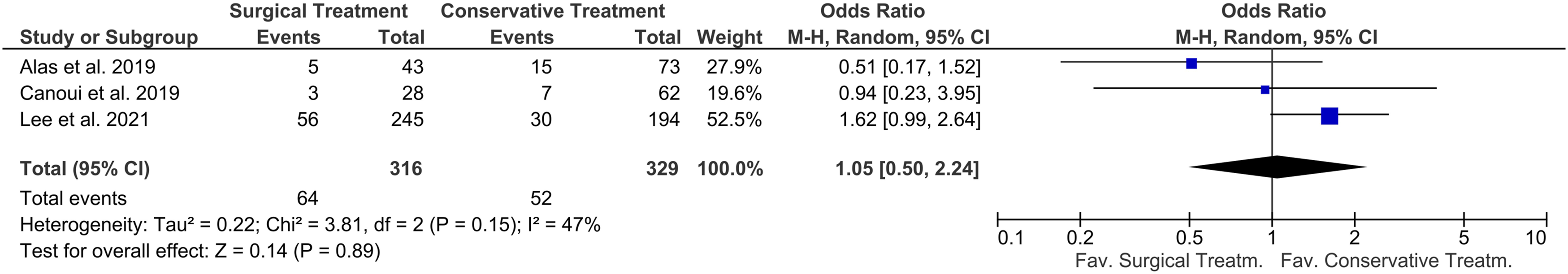
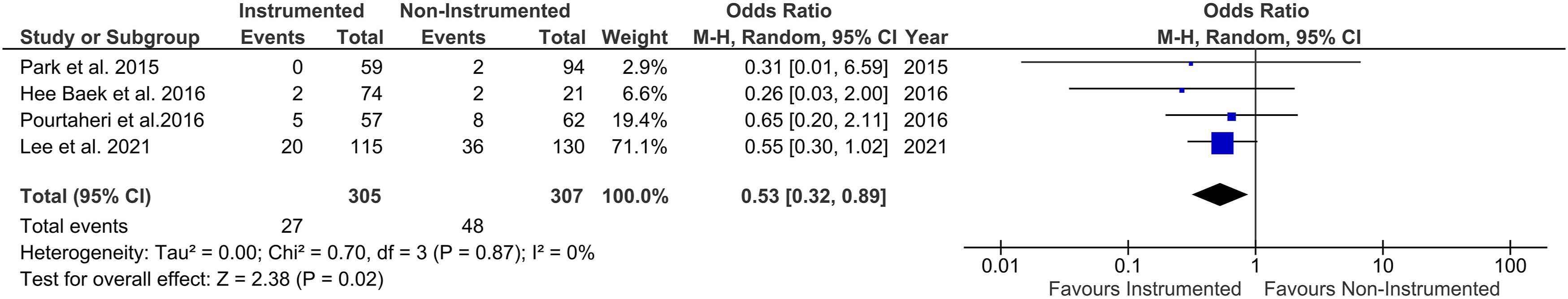
For non-instrumented spinal surgery, higher mortality rates were found in the study by Lee et al. compared to the nonsurgical treatment group for patients over 75 years.
25
In a different study by Hey et al. significantly lower mortality rates were reported for a cohort with a mean age of 62 for instrumented and non-instrumented surgery compared to conservative treatment (OR .82 and .80, P = .02 and P = .01).
11
SF-36 Physical activity - Forest plot presented significantly lower overall mean SF-36 for physical activity component for surgery compared to conservative treatment.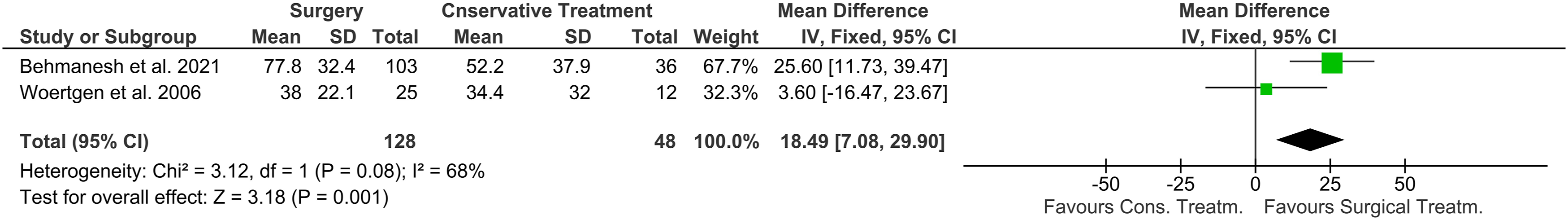
Quality of Life
Three studies reported on quality-of-life patient-reported outcomes (PRO’s). Two utilized the SF-36 questionnaire, whereas one used the SF-12 questionnaire.16,19,24
The categories of the SF 36 physical activity and bodily pain were compared using forest plots shown in Figures 8 and 9.16,24 Besides substantial heterogeneity (I2 = 68%), mean scores of the SF-36 for physical activity were significantly higher in the surgery group compared to the patients treated conservatively (OR 18.49 (7.08-29.90) P < .01).16,24 Nasto et al. also showed higher scores for the mental and physical components of the SF-12 1 and 3 months after surgery. However, after nine months, mental and physical scores showed no difference.
19
VAS for surgical vs. non-surgical therapy - Forest plot demonstrate significantly lower overall VAS scores for surgery compared to conservative treatment.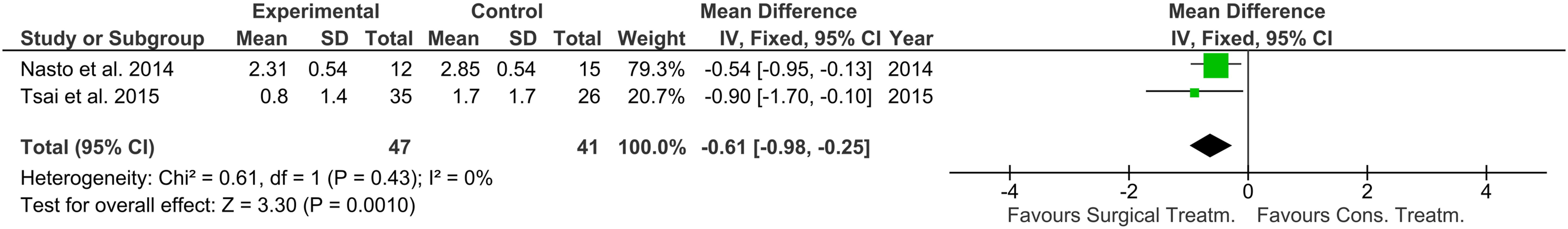
Pain
Four studies reported on pain as an outcome variable. Two of these studies used the visual analog scale (VAS),19,20 while the other two used the bodily pain component of the SF-36.16,24 The included studies show a lower pain VAS for surgery compared to the mean VAS for conservative treatment (-.61 (-.98—.25) P < .01, I2 = 0%, P = .43)19,20 Figure 9 and higher scores for the SF-36 component for bodily pain Figure 10.16,24 SF-36 Bodily Pain - Forest plot showed significantly higher overall mean SF-36 bodily pain component for surgery compared to nonsurgical management.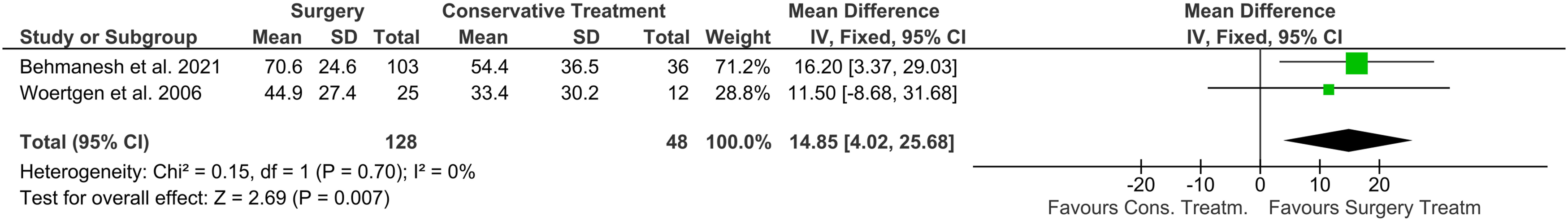
Length of Stay
Five studies reported on the length of hospital stay (LOS). The length of hospital stay was shorter for patients who had surgical treatment than those with nonsurgical management (31.0 ± 16.9 vs. 46.0 ± 21.7 P < .01). 20 The comparison of instrumented to non-instrumented surgery yielded inconsistent results. Pourtaheri et al. showed a significantly shorter hospital stay for non-instrumented spine surgery (13 (2-63) vs. 19 (3-75), P = .02), 28 while Lee et al. 2022 showed no significant difference in length of stay for decompression compared to decompression and fusion (13.5 vs. 16,7 days, P = .13). 29 Lee et al. 2021 also found no significant difference between non-instrumented surgery compared to instrumented spine surgery (52 (47-59) vs. 54 (47-63) P = .084). 25 In contrast, Bydon et al. showed significantly shorter hospital stays for instrumented surgery compared to decompression alone (14.36 ± 8.44 vs. 24.93 ± 23.15, P < .01). 10
Discussion
Seen as whole, spinal infections present as a heterogeneous clinical problem group. In general, they can be differentiated based on their pathoorganism, their anatomic location and local propagation into discitis, spondylodiscitis, epidural space, retroperitoneal space, and facet joints as well as their causal relationship to previous spine surgery. 2 Although still subject of debate, isolated epidural abscesses can be interpreted as a separate entity. While an epidural abscess can develop secondary to spondylodiscitis, primary epidural abscesses are relatively rare and commonly are treated surgically.4,5 To address the unresolved issue of the safety and outcomes of using spinal instrumentation in presence of a DNSI we undertook this formal Systematic Review. In order to answer our study question, we excluded studies primarily dealing with the treatment of epidural abscesses and also implant-associated spinal infections. Over the recent past, several studies have addressed the role of surgical spinal hardware in peri- or postoperative surgical site infections.7,8,30 In contrast, there has been a paucity of studies concentrating on DNSI.6,31 Unfortunately, important differences in clinical presentation and outcomes for DNSI and perioperative hardware-associated infections have been insufficiently elucidated. 31 The available data suffices, however, in the recommendation that DNSI should be seen as a different “disease” compared to spine implant-associated postoperative infections. While spinal implant infections are usually treated with a combination of surgery and antibiotics, DNSI have historically usually been managed nonsurgically.2,7,8,30 However, outcomes did show different results when comparing these two disease entities. Tschugg et al. reported a significantly higher mortality rate for DNSI compared to spondylodiscitis following spinal surgery. Also, they described a clinically more complicated course for de-novo spondylodiscitis compared to postsurgical infection patients. 31 The question, therefore, arises if lessons learned from perioperative spine infection management may be extrapolated to the management of DNSI patient, where surgically indicated.
Surgeons are, however, commonly confronted with questions of the ultimate fate of newly placed stabilizing spinal implants used in the management of active spinal infections and if such use ultimately could lead to failure of infection treatment along with higher rates of reinfections and the need for more unplanned reoperations, as well as a higher probability of other unfavorable outcomes, even death. Such concerns have resulted in a general consensus that has placed surgical intervention for DNSI into the role of a second-line therapy instead of a primary treatment modality equal or perhaps preferable to nonsurgical management.
Our study, which we performed under the assumption of potential biases and an existing inherent heterogeneity of the included studies in our meta-analysis, showed no significant differences in terms of recurrence of infection for instrumented surgery compared to non-instrumented spine surgery (OR .91 (.50-1.64) P = .74)10,25-27 or surgery compared to nonsurgical management (OR .98 (.58-1.65) P = .95).21,25 Furthermore, mortality rates showed no significant difference between surgical and non-surgical treatment (OR 1.05 (.50-2.24) P = .89)21,22,25 and were significantly lower for instrumented compared to non-instrumented spine surgery (OR .53 (.32-.89) P = .02).25-28
We could only exclude potential selection biases to a certain extent for our mortality comparison for instrumented and non-instrumented surgical treatment. None of the included studies demonstrated significantly different comorbidities within their treatment groups of instrumented and non-instrumented surgery. Only two of the included studies reported the Charlson comorbidity index.25,28 Pourtaheri et al. showed no difference for the Charlson comorbidity index for instrumented compared to non-instrumented patients with an overall lower mortality rate for patients treated with instrumentation. 28 Even though mortality rates for instrumented surgery were also lower in the study by Lee et al. focusing on patients over 75 years, the shown Charlson comorbidity index was significantly lower for the instrumented group. 25 However, all included studies tended to lower mortality rates when comparing instrumented to non-instrumented surgery without reaching the level of significance.25-28 Besides these existing limitations, this could imply that the potential benefits of spine surgery, especially where indicated, may eclipse the seemingly baseless appearing fear arising out of the potential for ineffective local control of the infectious disease processes. Furthermore, we found that reoperation rates for DNSI were lower for instrumented compared to non-instrumented spine surgery (OR .32 (.10-1.00) P = 0,05) I2 = 63%, P = .06). Even though none of the included studies for our reoperation analysis (Figure 5) stated the indication or the specific degree of instability triggering the use of instrumented surgical treatment versus non-instrumented surgery, this could underscore the importance of spinal stability as a relevant component for infection control, while also allowing for more definitive debridement. It could further indicate that the risk for reoperation may rise when spinal instrumentation is avoided out of concern for an active infectious process.
From a more general viewpoint considerations of the “safety” of spinal infection has historically been reduced to achieving infection control instead of allowing for a more multidimensional outcomes assessment, which would include preservation of spinal stability and alignment, effective neural element decompression, pain management, return to function in form of physical activities of daily life beyond the core goal of successful infection management.
For instance, untreated and under-treated pain can be expected to result in increased adverse outcomes due to reduced mobility. It can result in chronic pain disorder associated with loss of strength, immunologic impairment, dependence on opiates, and other adverse effects.32,33 Our meta-analysis showed that spine surgical intervention in DNSI seemed effective in pain management and that surgery seemed to result in greater pain reduction than nonsurgical management. This could also explain our finding of better return to physical activity and improved of overall quality of life (IV 18.49 (7.08-29.90) P < .01).16,19,20,24
The measurement of hospital length of stay has become an important expression of cost containment for stressed healthcare systems globally. It can serve as a key measure of healthcare efficacy but can also be an indicator of patients’ quality of life. 34 A shorter LOS for instrumented spinal surgery compared to non-instrumented surgery was shown by Bydon et al. (14.36 ± 8.44 vs. 24.93 ± 23.15, P < .01). 10 While LOS is a multifactorial variable and was not significantly shorter for instrumented spine surgery in all included studies, the overall shorter LOS for surgical treatment compared to nonsurgical management (31.0 ± 16.9 vs. 46.0 ± 21.7 P < .01) 20 could indirectly underline the positive quality of life effect achievable through surgical treatment.
Limitations of our SR include the relatively small number of included studies. Furthermore, we only included German and English language studies, all of which were retrospective. This may have introduced a number of selection biases. Beyond our strict adherence to our listed in- and exclusion criteria, it is possible that not every study was purely addressing DNSI since some studies did not specify their inclusion and exclusion criteria in detail. Furthermore, our meta-analyses showed substantial heterogeneity within the included studies, which might have been caused by data from investigations of different countries with different socioeconomic standards and distinct clinical treatment strategies. The presented pooled data may have been further influenced by several different biases including selection, treatment indication, or affiliation-caused influences. This could have resulted in inhomogeneous cohorts. However, we only included studies with de-novo spinal infections and existing internal comparison cohorts for the included treatment modalities to minimize the risk of bias-related statistical inaccuracy between purely non-surgical or surgical studies and purely instrumented to non-instrumented surgical studies. We also did not include non-pyogenic and fungal diseases, which likely follow very different immunologic and outcomes pathways and are further subject to different sets of outcomes expectations. Finally, interesting surgical technique questions, such as timing of surgical intervention relative to symptom onset, minimally invasive versus traditional surgery, and multi-versus short-segment surgeries could all not be addressed due to a lack of specific analytically useful data.
This systematic review presents a comprehensive outcome variable analysis for surgical treatment of de-novo spinal infections (DNSI), with a special focus on instrumented spine surgery. Aside from the question of accomplishing control of the infectious disease process we also addressed further outcome-relevant variables such as quality of life, pain, and mortality. Future investigations, especially well-designed prospective studies which include improved risk profiling of patients and disease states, generally underrepresented in the field of surgery, would likely significantly add to our current knowledge base and could lead to a change in management strategies. Specific surgical techniques including aspects of surgical approaches, timing, staging, and fusion options are currently insufficiently represented in our clinical literature all relevant for more dedicated clinical comparative effectiveness research. Based on our current findings, however, we conclude that spinal instrumentation, where indicated, does not pose an additional risk in the management of DNSI patients and may be associated with more favorable outcomes.
Conclusion
Spine surgery in de-novo spine infections (DNSI) may safely be performed without additional risk of higher reinfection rates or mortality rates compared to non-surgical management. In addition, instrumented surgery, compared to non-instrumented spine surgery, showed lower reoperation and mortality rates in the treatment of patients with DNSI. Based on current literature, improved outcomes in terms of quality-of-life and pain reduction were associated with the surgical intervention of DNSI compared to non-surgical management.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
