Abstract
Study Design
Case report.
Objective
Temporary external ventricular drainage for refractory thoracolumbar cerebrospinal fluid (CSF) leak is not reported in the literature. We describe a recent case that utilized this technique.
Methods
Retrospective review of the patient's case notes was performed and the literature on this subject reviewed.
Results
The patient underwent multiple complex spinal surgeries for resection of innumerable metastatic ependymoma lesions. A case of significant refractory CSF leak developed and as a last resort a right frontal external ventricular drain was placed. The CSF leak ceased, and the patient was eventually discharged home without further complication.
Conclusion
External ventricular drainage can be a viable option for temporary proximal CSF diversion to treat refractory thoracolumbar CSF leaks.
Introduction
Temporary external ventricular drainage for refractory thoracolumbar cerebrospinal fluid (CSF) leak is not reported in the literature. We present a young man who had multiple complex spinal surgeries for resection of innumerable metastatic ependymoma lesions. During his last hospital course, he developed a significant refractory CSF leak. As a last resort, we decided to place an external ventricular catheter for proximal controlled CSF drainage with great success.
Case Report
The 32-year-old man had a history of spinal myxopapillary ependymoma World Health Organization (WHO) grade I with local metastatic spinal disease. The patient first presented in 2005 with a 1-year history of severe low back pain. He was found to have an intradural lesion, and subsequently had a T12–L3 laminectomy for resection of the tumor. Small amounts of the tumor adherent to the nerve roots were purposefully left behind. Each time an attempt was made to remove these residual small pieces of the tumor, significant amounts of neuromonitoring activity was noted. Hence, the residual tumor was left behind so as to not cause a neurologic deficit. Approximately 10 days after surgery, the patient fell at home and noticed significant amounts of a clear fluid consistent with CSF leaking from his incision. He was taken to the operating room at that time and a dural defect was identified and closed primarily, then covered with Gelfoam (Baxter Healthcare, Deerfield, IL, United States) and Tisseel (Baxter Healthcare). The patient again noted another CSF leak from a different area of his incision 5 days later, and he was taken back to the operating room for another primary closure, as well as placement of a fat graft and more Tisseel. Adjuvant radiation treatment was undertaken after he recovered from the initial surgeries. In 2008, the patient presented again with signs and symptoms from thoracic spinal cord compression. He was found to have a dorsally located intradural lesion at the T8–T9 region, which was resected via a T7–T10 laminectomy. In 2010, the patient presented with progressive low back pain and radicular symptoms. He was found to have growing intradural lesions at L4 and L5. At that time, L4–S1 laminectomies for the resection of three separate tumors was undertaken. One year later, our patient presented yet again with multiple thoracolumbar intradural lesions (Fig. 1). He underwent redo decompressive laminectomies from T8–S1 with a total of 13 intradural tumors being excised (Fig. 2). The dural closure proved to be quite complex, as there were several areas that were macerated and very difficult to reapproximate. Fat grafts, Duraform (DePuy Synthes, West Chester, PA, United States), and Tisseel were all used on top of the attempted primary dural closure.
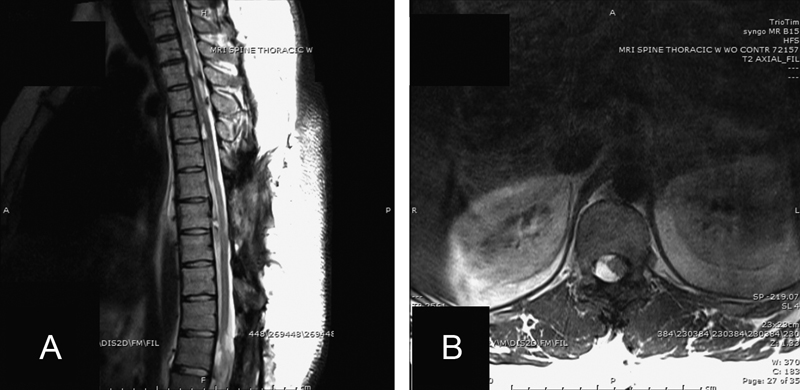
Preoperative sagittal T2-weighted magnetic resonance imaging (MRI) (A) of the thoracic spine revealing multiple intradural tumors as well as an axial T2-weighted MRI (B) revealing significant compression of the spinal cord.
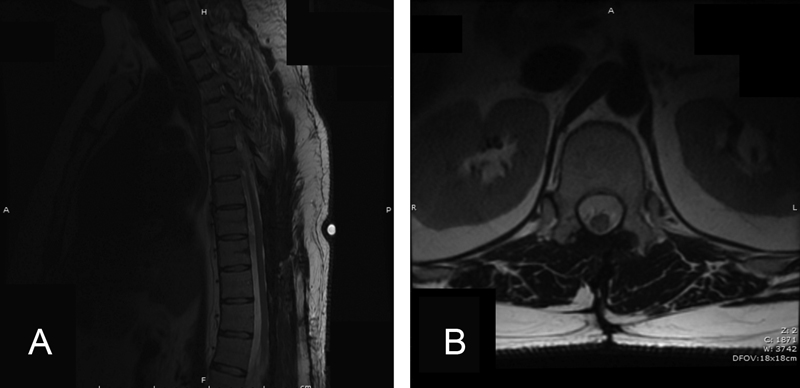
Postoperative T2-weighted sagittal (A) and axial (B) magnetic resonance imaging revealing gross total resection of multiple intradural tumors. Radiographic evidence of a refractory thoracolumbar cerebrospinal fluid leak was not readily evident on the immediate postoperative films.
During his hospital course, the patient developed a spontaneous CSF leak from his wound. It was initially treated by oversewing the skin, but the leak persisted over the next day. We decided to bring the patient back to the operating room for an exploration and an attempt at repairing the dural defects. Multiple dural defects were noted. As a result of the significant dural maceration, another primary closure was not attempted. We placed more pieces of Duraform, fat graft, and Tisseel over the areas of CSF leakage, and we again performed a tight multilayer closure of the muscle, fascia, fat, and subcutaneous tissue. Despite our best efforts to stop the CSF leak, the leak continued through the incisions from multiple areas. The patient then began showing clinical signs of meningitis. As a last resort, we decided to place a right frontal external ventricular drain (EVD; Integra Life Sciences, Plainsboro, NJ, United States) after obtaining a preprocedure computed tomography scan of the head, to ensure the ventricular size could accommodate the catheter (Fig. 3A). The EVD was successfully placed (Fig. 3B) and kept for 11 days. The patient's hospital course was further complicated by pneumonia and reintubation. He was placed on antibiotics for both the pneumonia as well as the CSF leak and subsequent meningitis. On the day 8 of the EVD in place, we continued to see cessation of the CSF leak and decided to perform a 3-day clamping trial. No further CSF leak was noted from the incision during the clamping trial. The EVD was removed, and the patient was eventually discharged home without further complications.
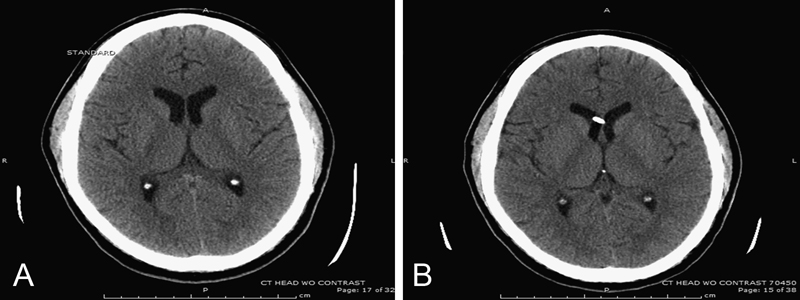
Preprocedure computed tomography scan of the head to ensure the ventricular size could accommodate an external ventricular drain (A). Postprocedure scan showing successful placement of a right frontal external ventricular drain (B).
Discussion
There are no specific guidelines regarding the treatment of postoperative spinal CSF leaks. A watertight dural closure is essential in reducing the risk of CSF leakage in spinal surgery. Eismont et al recommended the meticulous closure of the dura at the time of surgery and reoperation and repair of dural tears upon the discovery of a persistent CSF leak. 1 Conservative management may be initially attempted with bed rest for 3 days with subsequent slow advancement to ambulation. Eighty-six of eighty-eight patients with dural tears studied by Wang et al had resolution of CSF leaks with primary dural closure at the time of surgery followed by bed rest. 2 Others have confirmed the efficacy of treating such leaks via CSF diversion in the lumbar region. 3
A review of the literature revealed scarce reports of CSF leakage in more complex patients such as the patient in this case. This complexity arose from the multiple surgeries that were performed due to the recurrence of this intradural disease. Myxopapillary ependymomas predominantly occur in the lower conus and filum. They are benign, slow-growing, WHO grade I tumors that are amenable to surgical resection with optimal outcomes when gross total resection is achieved. Although pathologically they may be “low grade,” they can be metastatic locally within the spine via the CSF route. Recurrence of the disease is usually in the same region and is substantiated by subtotal or piecemeal resection. 4
Spine surgeons occasionally encounter lumbar CSF leak. Routine iatrogenic CSF leaks can usually be repaired primarily by suturing the dural defect, with the subsequent placement of a liquefied dural sealant such as Tisseel. Our case, however, had a refractory CSF leak. Our patient's dura was manipulated on multiple occasions over the span of half a decade. With each surgery to remove more metastatic lesions, the dura became more macerated. This damaged dura created multiple avenues for CSF to leak, which was essentially refractory to all of our attempts at a repair.
A case report by Yeager demonstrated the use of an EVD with subsequent conversion to a shunt for a persistent cervical CSF leak after a schwannoma resection. 5 However, temporary external ventricular drainage for refractory lumbar spinal fluid leaks is not reported in the literature. Lumbar drains have been utilized in the case of persistent cervical CSF leaks. 6 We feel that a lumbar drain in our particular case would not have been best suited for the patient we presented. We placed an EVD in this situation as opposed to a lumbar drain because maximal CSF diversion from the macerated dura would take place with a proximal avenue for controlled CSF drainage, which would essentially decrease the pressure gradient of CSF in an attempt to traverse the dural defects more distally. Although the placement of external ventricular drains comes with associated risks, if they are placed safely after a preprocedure computed tomography scan is done, their usage can be a viable option for temporary proximal CSF diversion for refractory thoracolumbar CSF leaks.
Disclosures
Michael A. Galgano, none
Ali Hazama, none
Eric M. Deshaies, none
