Abstract
Study design
Retrospective Cohort Study
Objectives
To describe surgeries and treatment outcomes of metastatic pheochromocytomas and paragangliomas (PPGLs) on the spine.
Methods
We reviewed a cohort of 18 patients with spinal PPGLs who were consecutively treated in our spinal center. Their clinical data was reviewed. The survival period and its relevant factors was then analyzed.
Results
The cohort included ten cases of pheochromocytomas and eight paragangliomas. The local pain and neurological deficits were the two most common symptoms. One third of the spinal PPGLs were diagnosed as functional tumors, arousing secondary hypertension. The imaging features were consistent with those of osteolytic lesions. The surgical strategies for the cohort included percutaneous vertebroplasty, neurological decompression and partial tumor resection, and total en-bloc resection. The postoperative courses were uneventful except 1 patient developed heart failure. The adjuvant therapies were implemented in 6 patients with 131I-MIBG, five with radiotherapy, two with chemotherapy, and 1 with target therapy. The median survival period was 39 months, and the overall survival rate of 1 year was 77.8% (14/18). The patients’ Karnofsky performance scores were positively correlated with the survival period (P < .05).
Conclusion
Surgery is indicated for intractable local pain and neurological impairment in the patients with spinal PPGLs. Palliative surgical strategies, including neurological decompression and partial tumor resection, could bring fair outcomes, especially for the patients in poor physical conditions.
Introduction
Pheochromocytoma and paragangliomas (PPGLs) are rare endocrine tumors arising from chromaffin cells, which are derived from the neural crest.1,2 According to the 2017 World Health Organization (WHO) classification system, 3 Pheochromocytoma (PHEO) is an adrenal tumor, and paragangliomas (PGL) is an extra-adrenal tumor. Since the two tumor types cannot be differentiated on the basis of histologic findings, anatomical location is used to distinguish between them. PHEO develops from the adrenal medulla, while PGL arises in extra-adrenal sites, including the abdomen, thorax, head, and neck. PPGLs has a low incidence, which is estimated at 2 to 8 cases per million per year, in which PHEOs are more common than PGLs.1,4 These neoplasms are mostly seen in the age of fourth to sixth decade, and do not show significant gender disparity. PPGLs have the capacity to synthesize, store, and secrete a variety of neurotransmitters, especially catecholamine, which predispose the patients to symptoms such as hypertension, palpitations, throbbing headaches, and diaphoresis.1,5 However, their clinical manifestations are extremely variable, depending on locoregional and distant involvement, tumor size, the secretion of catecholamines and function of altitude, and the malignancy potential.1,2,4,6
All PPGLs have the potential to metastasis, according to the latest WHO classification in 2017. 3 The prevalence of metastasis to non-chromaffin organs and structures is estimated 10% in PHEOs and 35% to 40% in PGLs, relatively.1,4,6,7 The common metastatic sites include lymph nodes, liver, lung, urinary system, and bone.4,6 Spinal involvement is rare and was sporadically reported.8–17 Currently, there is no mature guideline on the treatment of metastatic spinal PPGLs. Surgical resection is the treatment of choice for the spinal lesions, and a complete resection remains the goal of surgery, which, however, is hard to achieve due to the complex anatomic structures, adjacent to vital organs and heavy bleeding due to hypervascularity.4,7,16,18 In the present study, we retrospectively reviewed the patients who were consecutively treated in our spinal center and described their clinical features and surgical outcomes. Furthermore, we searched medical databases for reports, case series, and researches on metastatic spinal PPGLs, in order to present a deep and precise appraisal of current therapeutic modalities.
Materials and Methods
Patients Inclusion
The clinical data of 18 patients with metastatic spinal PPGLs, who were consecutively treated in our spinal center from May 2007 to August 2019, was reviewed. The inclusion criteria were as the following: (1) having a history of PHEOs or PGLs, and the pathological specimens of spinal lesions were confirmed by the department of pathology in our center; (2) receiving surgical treatment in our center; (3) being regularly followed-up; and (4) the clinical data were fully accessible. This study was approved by our institutional ethics committee, and the written informed consent was obtained from all the patients.
Data Collection
In this study, we mainly focused on the review and analysis of clinical features, treatment information, and follow-up data regarding spinal metastatic lesions. Specifically, the neurological status was rated according to ASIA scaling system. The life capacity of the patients was assessed with the Karnofsky performance scale (KPS). 19 The Spinal Instability Neoplastic Score (SINS) system was used to assess spinal stability, 20 and the Tomita and Tokuhashi scoring systems were used to assess the prognosis of patients and assist in the scheming of surgical plans.21,22 The advice from our institutional multidisciplinary team on the spinal tumors was also incorporated into the plans. The surgical strategies were categorized as the following: total resection (TR), partial resection (PR, including separation surgeries), and percutaneous vertebroplasty procedures (PVP). Besides, neurological decompression was undertaken in case of symptoms and signs of neurological compression. Surgical data, including blood loss, operation duration, and perioperative complications, was reviewed and analyzed. The surgical outcomes were appraised mainly using overall survival (OS) after the indexed operation.
Statistical Analysis and Data Presentation
Data analysis was performed using IBM SPSS statistics for Windows Version 20 (IBM Corp., Armonk, NY, USA). Lilliefors test, which is an adaptation of the Kolmogorov–Smirnov test, was used to examine whether the data was normally distributed. Data presentation was provided in the forms of percentages, mean ± standard deviation, or median (range). Two-tailed unpaired Student’s t test and two-tailed Pearson’s χ2 test (or Fisher’s exact test) were utilized to make a comparison between different groups. Spearman’s rank correlation test was used for correlation analysis between the two parameters. Statistical significance was set at .05.
Results
Demographic Data and Medical History
Clinical Data and Medical History of the Included Cohort.
aThis item refers to the metastasis to other organs and structures except for the spine.
Symptoms and Imaging Findings
Clinical Features, Treatment, and Outcomes of the Metastatic Spinal PPGLs.
aInstrumented fixation was performed in all cases but case #1 and #2. PPGL stands for pheochromocytoma and paraganlioma; SINS, spinal instability neoplastic score; ND, neurological deficits; PR, partial resection of the tumor; TR, total resection of the tumor; PVP, percutaneous vertebroplasty; MIBG, metaiiodo-benzylguanidine.
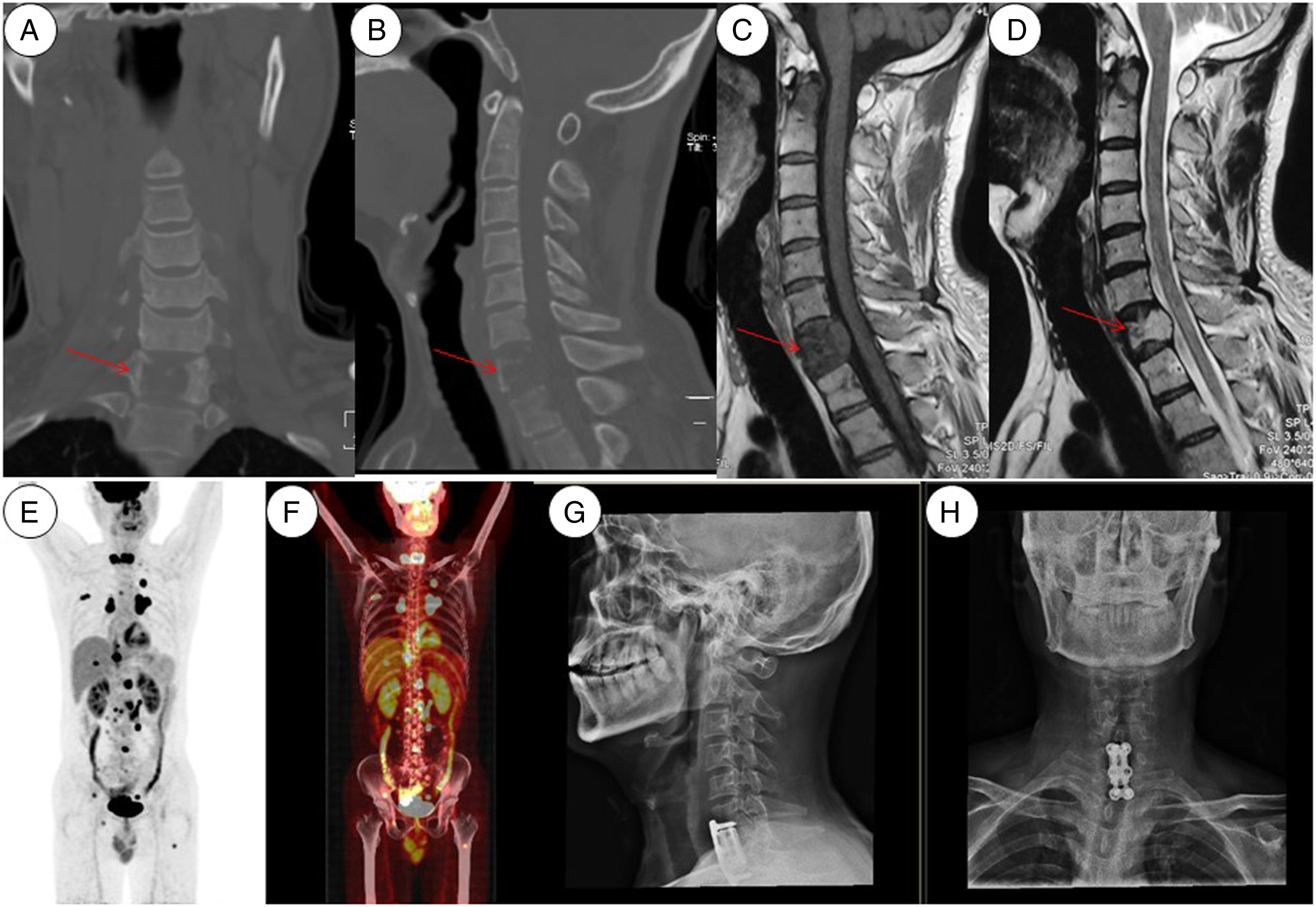
Imaging presentation of case #15. CT reconstruction films revealed an osteolytic lesion inside the C7 vertebral body (arrows in a and b). The lesion showed hypo-intense signal on T1-weighted MRI sequences and hyper-intense signal on T2-weighted sequences (arrows in c and d). PET/CT revealed elevated uptake of 18F-FDG in multiple foci, including systemic lymph nodes, lung, liver, pelvis, and spine (e and f). The patient received C7 corpectomy and spinal reconstruction with 3D-printing prosthetic vertebral body (g and h).
Surgical Strategies and Adjuvant Therapies
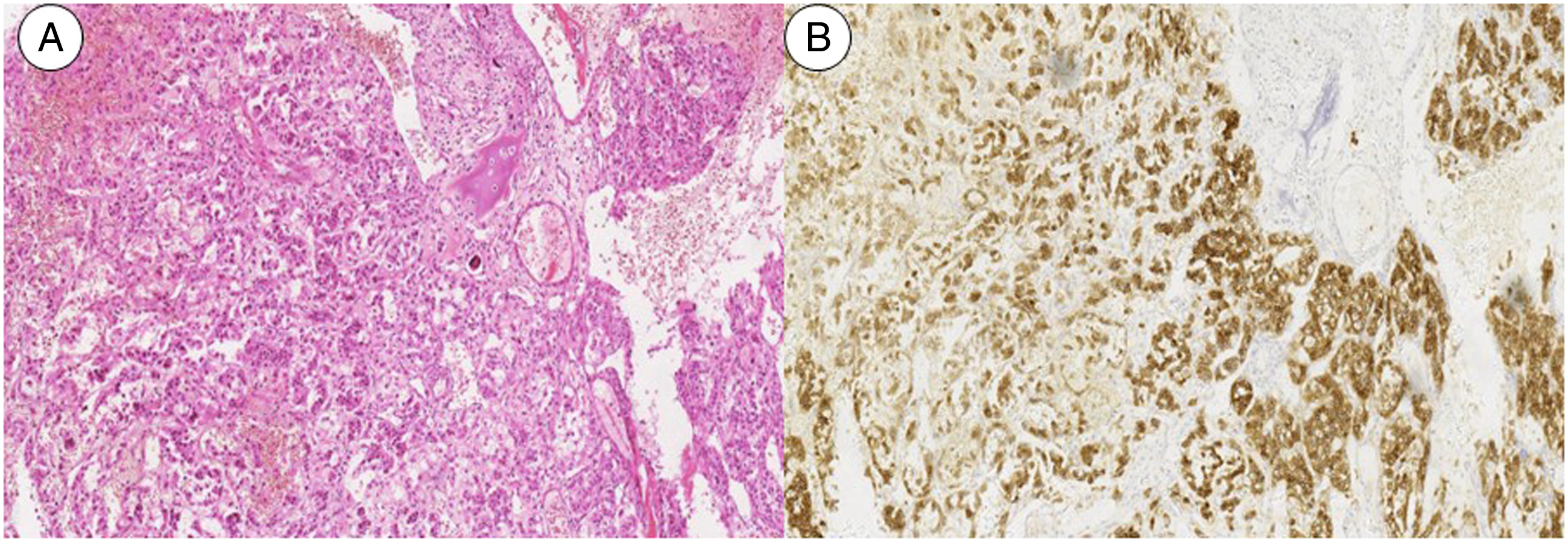
Surgical options for the cohort included PVP in one case (case #1), en-bloc tumor resection in one case (case #3, and Figure 2), and partial tumor resection in the other 16 cases, among whom 13 cases received instrumented fixation and neurological decompression procedures in 12 cases. The mean bleeding volume for open surgeries in 17 cases was 1105.9 ml. Histological and immunostaining results of the removed specimens were consistent with PPGLs (Figure 3). Preoperative embolization was performed in three patients to mitigate the bleeding during the operation. The postoperative course was uneventful in 17 patients, but 1 patient (case #10) developed heart failure (Table 2). Presentation of total tumor resection (case #3). Preoperative imaging work-up revealed an osteolytic lesion inside C2 vertebral body (a, b, and c). The patient underwent CT-guided biopsy (d) and the result was consistent with paraganglioma. Total tumor resection was obtained via one-staged posterior and anterior surgical approach. The reconstruction of anterior spinal column was made with a customized 3D-printing vertebral body (e and f). Histological result of spinal lesion. Photomicrograph showing characteristic nests of tumor cells separated by vascular septa (Zellballen) with cells showing significant nuclear pleomorphism with prominent nucleoli (H and E, original magnification x20) .Synaptophysin immunostaining shows strong, diffuse cytoplasmic staining in the tumor cells. (immunostaining, original magnification x20).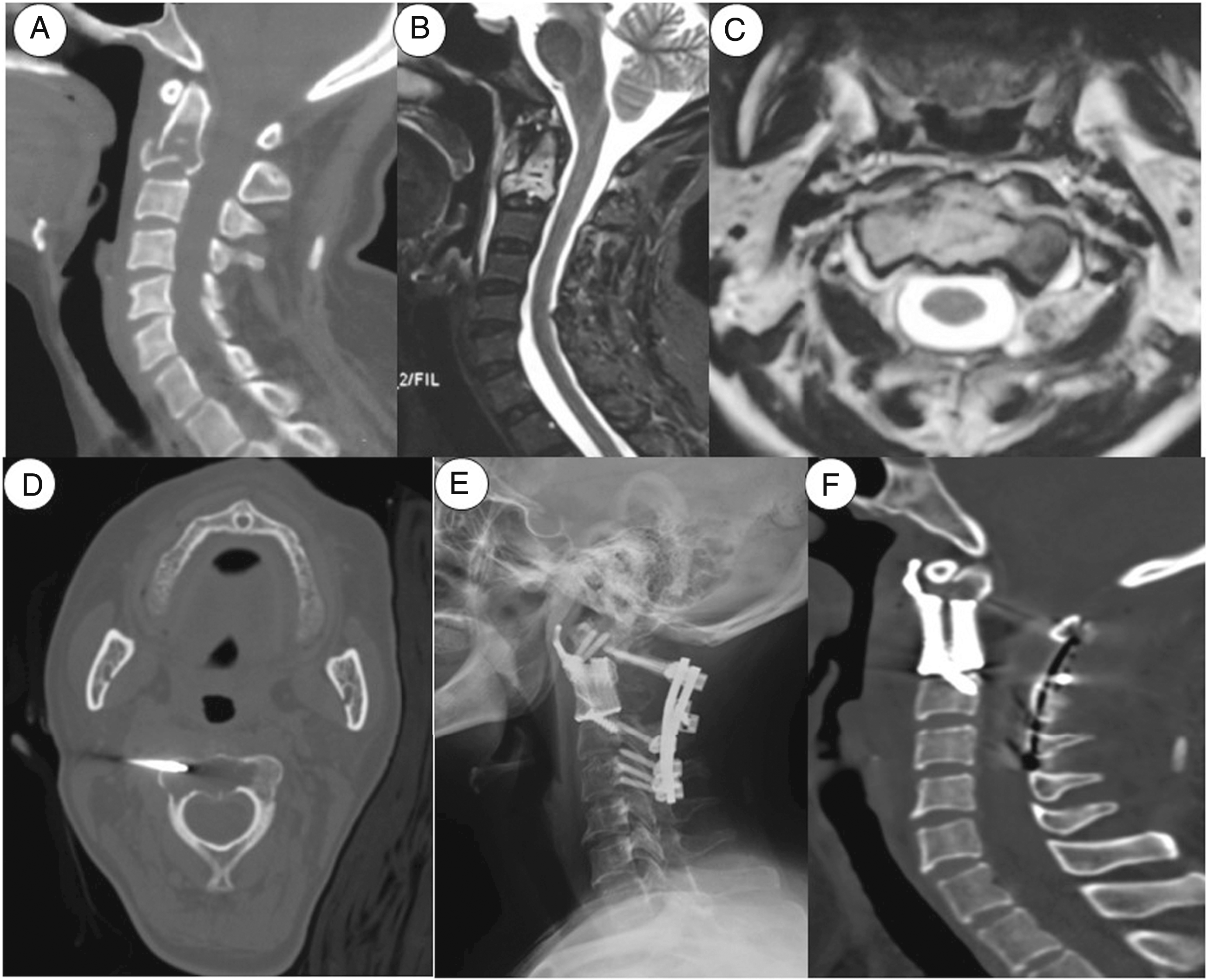
Adjuvant therapies were offered according to the advice from our institutional multidisciplinary treatment (MDT) team on spinal tumors. Five patients were treated with radiotherapy, two patients treated with chemotherapy, 6 patients treated with iodine(131)-metaiodobenzylguanidine (131I-MIBG), and 1 patient treated with targeted therapy. The other four patients did not receive adjuvant therapies (Table 2).
Follow-Up Results
Up to the last follow-up, eight patients died due to tumors or tumor-related events from five to 39 months after the operation, and 1 patient (case #6) died of pulmonary infection and thoracic hemorrhage 4 months after the operation. The 1-year OS rate was 77.8% and the median survival period was 39.0 months (Figure 4). We found that survival period was related with numbers of spinal metastases (solitary versus multiple, P < .05, two-tailed student t test) and significantly correlated with KPS (rho = .537, P < .05, Spearman’s correlation analysis). Kaplan-Meier survival curve of the cohort.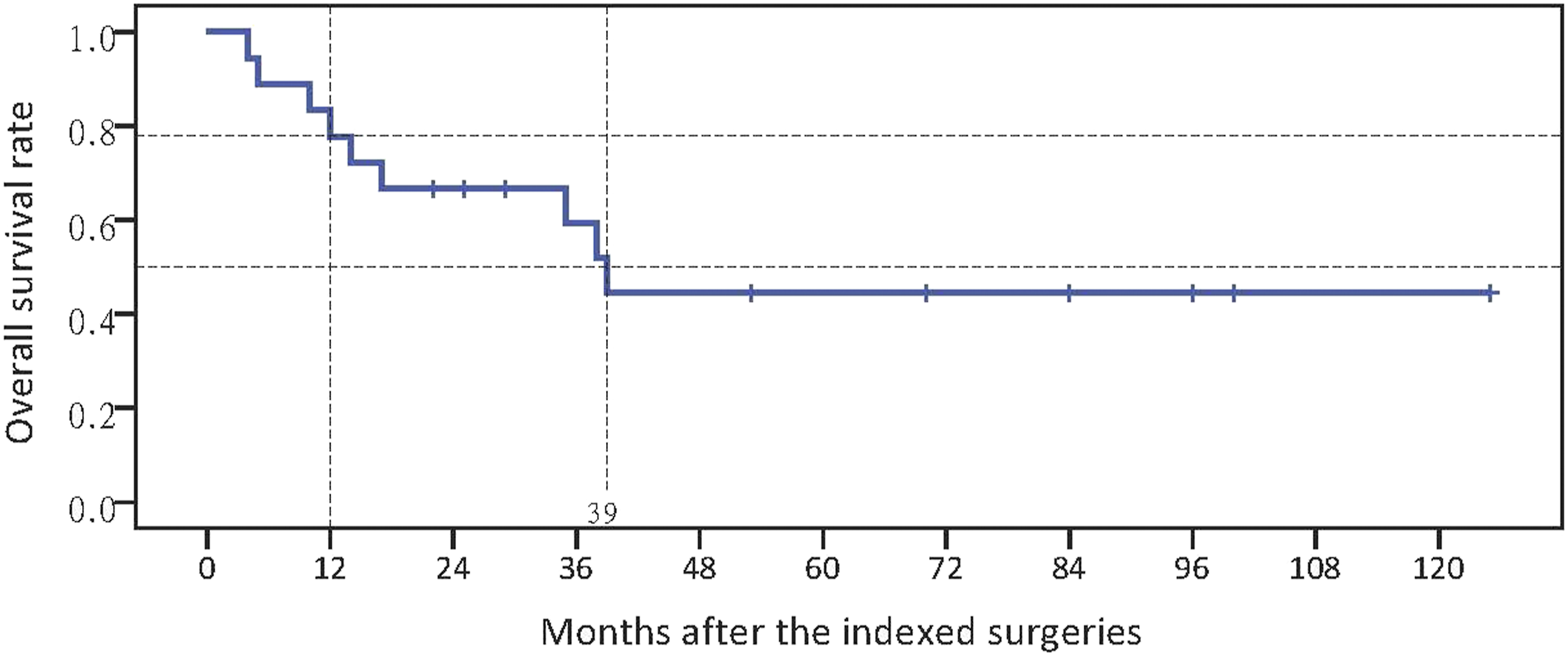
Discussion
PHEOs and PGLs have the same cellular origin. They are generally slow-growing but all have potentials of recurrence and metastasis, according to the WHO classification (2017 version) on endocrine tumors.1,3 Symptomatically, the patients with PPGLs are usually latent until huge massing effect and/or metastasis has emerged. Previous researches identified some factors associated with risk of metastasis, including the primary tumor site and size, age at diagnosis and some specific genotypes.1,3–7,23 The bone is one of the most common metastatic sites (72%), and spinal involvement is usually a dreadful situation.24,25 Metastatic spinal PPGLs can lead to some devastating skeletal-related events, such as pathological fracture and severe local pain and paraplegia, and significantly impair patients’ activity of daily living and quality of life. 24 Literature regarding this entity is lacking, so few references are availed about the clinical features and treatment outcomes of the spinal metastasis. In this report, we described and analyzed a cohort of 18 patients with metastatic spinal PPGLs, hoping to provide references for the future clinical practice in this field.
This study found that the spinal PPGL was mostly seen in mid-aged patients (Table 1), and that 94.4% (17/18) cases were younger than 60 years. The study noted gender variation with a predominance of males (2.6/1). The time between the prior surgery of primary site and the spinal metastasis varied in a wide range, from 1 to 27 years. The clinical symptoms of the cohort were not pathognomonic. Local pain and neurological dysfunction were related to bony erosion and pathological fractures. As the primary lesions of PPGLs, metastatic lesions can also be functional and capable to secrete catecholamines (Table 2). Typical catecholamine-arousing symptoms include headache, palpitation, fatigue, flushing, sweating and/or paroxysmal hypertension.1,4,24 In our case series, there were 6 patients (33.3%, 6/18) having secondary hypertension, which were presumably related to the functional metastatic lesions. Malignant hypertension may cause a series of hemodynamic disorders and vascular events.1,25 Therefore, we prepared these patients with α-adrenergic receptor antagonists before the operation, in avoidance of hypertension crisis and lethal vascular events.1,4,6,25
In this study, all cases had different extents of pathological fractures, from cortical destruction to severe vertebral collapse. The thoracic spine was the most susceptible metastatic site, with a percentage of 55.6%. The imaging manifestations of metastatic PPGLs were consistent with osteolytic lesions. CT and MRI scans are the modalities of choice to identify the involved vertebral segments. On CT films, the lesions are usually a homogenously osteolytic mass within the vertebral body, with paraspinal soft tissue mass in some cases. Different from the inflections and metabolic bone diseases, the surrounding cancellous bone is normal and no existence of sclerotic girdle. On MRI scans, the lesion is a mass with hypo-intense signal on T1-weighted images and hyper-intense signal on T2-weighted images (Figure 1). Sometimes, we could find the signs of vascular flow void on T2 sequences, due to the heavy vascularity in some cases.16,18
The treatment strategies of the case series were schemed by our institutional MDT team in a concerted way. As for spinal metastases, a holistic set of treatment modalities are emphasized, including surgery, radiotherapy, chemotherapy, and/or other anti-tumor therapies.4,7,11,16,25 Preoperative evaluation was carried out by our MDT team. Especially, the patients’ physical condition (KPS), the spinal instability neoplastic score (SINS), Tokuhashi and Tomita staging systems were evaluated, to facilitate the scheming of surgeries. Surgical resection is the most effective way to reduce the tumor burden, and it is recognized as the treatment of choice for spinal PPGLs, especially for those with intractable local pain and neurological impairment.4,16,25–28 Therefore, surgery was considered in our center for the patients: 1. having symptoms and signs of spinal instability; 2. progressive neurological dysfunction; 3. severe and refractory local pain. Preoperative hemodynamic reconditioning is essential to the safety of the surgeries. Preoperatively, adrenergic blockade is typically accomplished with either a nonselective or a selective α-adrenergic receptor antagonist (e.g., phenoxybenzamine, and doxazosin), usually started at least 1 week before surgery.7,15,27 A β-adrenergic antagonist should be administered to control tachycardia after α-adrenergic blockade has been effective in normalizing blood pressure. Another factor needs to be taken into account in the surgical planning of metastatic spinal PPGLs: the vascularity of the tumor. This spinal metastasis tends to be hypervascular, which can lead to significant blood loss intraoperatively. Our case series had an over 1100 ml of blood loss averagely. It has been suggested that preoperative embolization of the supplying arteries can minimize blood loss 24h–48h before surgery. 18
Palliative surgeries, including separation surgeries, neurological decompression, and vertebral augmentation procedures, are the mainstay of treatment choice, for the patients with multiple metastases, recurrent spinal lesions, acute neurological deterioration, and intractable local pain. In our case series, 17 patients underwent palliative surgeries, and the main surgical goals were to restore spinal stability with or without decompressing the neurological elements. In recent years, separation surgery has gained wide acceptance for metastatic spinal tumors. In our series, the surgeries of 13 patients were performed under the principles of separation surgeries. Through the separation surgery, dual goals were achieved: providing sufficient neurological decompression and sparing a safe distance for radiotherapy at the same time. Percutaneous vertebral augmentation procedure was proven to be an effective method to control the local pain for the patients with no symptoms of neurological impairment (case #1).
For the patients with solitary spinal metastasis, total en-bloc resection shall be attempted. PPGLs have a high recurrent rate when partially resected. Richter et al. 13 reported that total en-bloc spondylectomy complemented with radiotherapy led to tumor-free survival at the 10-year follow-up in their patient. Yin et al. 16 shared a series of 18 patients with spinal PGLs and 15 of them received total tumor resection. The surgical outcomes were encouraging, with a relapse-free period over 40 months. However, given the intricate growth pattern and encasement of neural tissue by spinal metastases, intrusion to the spinal cord and paraspinal vital structures, and heavy bleeding during the surgery, total resection is not always feasible. Therefore, we should make a reasonable choice, and balance the safety and efficacy of tumor resection for each surgical option. In recent years, the advances in the surgical tools and instruments, such as 3D-printing prosthetic vertebra and ultrasonic bone scalpel, facilitate the challenging procedure of tumor resection.29,30 Even in the procedures of PR, maximal tumor mass resection should be attempted, to reduce the tumor load and spare more space in avoidance of relapse of neurological compression.
Locoregional recurrence or progression is a big concern for patients with spinal tumors. When removal of the entire tumor is not possible, radiation treatment is recommended for the remaining mass after the operation, to provide a better local control.1,16,24 In our cohort, five out of 13 patients who underwent separation surgeries received radiotherapy after the operation, among whom 1 patient died 5 months after the therapy and the other four patients manifested a satisfying local control after more than 2 years of follow-up. The doses delivered to our cases ranged from 35Gy to 45Gy. The dose was referred to that of giant cell tumors and chordomas. For the past several decades, systemic therapies for PPGLs have been mainly referred to 131I-MIBG. Current knowledge of systemic therapies relies on the results of a few retrospective studies. 25 Up to 70% of metastatic PPGLs do not respond to the treatment significantly.31,32 Overall, 6 patients in our study were treated with 131I-MIBG, while three patients died ten to 14 months after the therapy, one died 38 months later, and the other two patients enjoyed a long survival. The outcomes suggest that the use and efficacy of 131I-MIBG therapy in spinal PPGLs need further exploration and verification.
The use of chemotherapy is controversial. The single or combined use of chemotherapeutic drugs is often empirically decided rather than based on high-level medical evidence given the rarity of the diseases. CVD regimen (cyclophosphamide, vincristine, and dacarbazine) is recommended as the best protocol. Ayala-Ramirez et al. 24 reported that 5-year OS rate of PPGLs patients who received CVD regimen was 51%. A meta-analysis found that only 37% of malignant PPGLs showed response to the regimen. 33 U-King-Im et al. 32 reported successful therapeutic use of octreotide in a patient with carotid body PGL that metastasized to multiple vertebrae. Mertens et al. 11 demonstrated the successful regression and palliation of metastases of PGL with chemotherapy, particularly with the CVD regimen. However, the application and efficacy of chemotherapy after the surgeries were not well verified and in need of evidence from high-quality, large-scale clinical trials. In our case series, we found that the application of adjuvant therapies did not correlate with OS, and these patients had a 1-year OS rate similar to non-adjuvant therapies (21.4% versus 25.0%, respectively; P = 1.000, Fisher’s exact test).
The present study has several limitations. Firstly, it is a retrospective study with a small sample size. Thus, the results of this study need further examination and references with caution. Secondly, the primary tumors were operated many years ago in other hospitals, so access to information related to surgical margin status, the size of the primary tumor, and treatment after primary tumor resection was limited. Thirdly, it was difficult to obtain the continuity, regularity, periodicity, and dose of radiotherapy and chemotherapy after the indexed surgeries for spinal PPGLs because some of adjuvant therapies were not implemented in our hospital.
In conclusion, this study described the clinical features, surgical strategies, and outcomes of metastatic spinal PPGLs. The spinal metastases were generally latent and did not be recognized until local pain and the occurrence of neurological dysfunction. Functional spinal PPGL is a big concern during the surgical preparation and hemodynamic reconditioning with α-adrenergic receptor antagonists is recommended to avoid severe vascular events. PPGLs are heavy vascular tumors, so preoperative embolization of supplying vessels is recommended to mitigate the blood loss during the operation. The median survival period was 39 months and 1-year OS rate was 77.8%. Surgery is the treatment of choice for spinal PPGLs, and palliative surgery combined with 131I-MIBG and/or radiotherapy was proven a feasible and safe therapeutic strategy, with fair outcomes.
Footnotes
Acknowledgments
We thank our institutional multidisciplinary treatment team on spinal tumors including colleagues from departments of anesthesiology, pathology, oncology, intensive care unit and radiotherapy for their participation in the treatment scheming and implementation and in the preparation of this article.
Author Contributions
S. Du and P. Hu designed the study, reviewed the patients, collected and process the clinical data, and drafted the manuscript. S. Yang and H. Zhuang reviewed the patients and collected the data. F. Wei designed the study, selected the patients, processed the data and supervised the study. X. Liu and Z. Liu supervised the data collection and processing. All authors reviewed this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the institutional research fund (Peking University Third Hospital; grant number: Y73504-03; recipient: F. Wei). The fund provider has no influence on the design of the study and data collection, analysis, and interpretation of data and in the preparation of the manuscript.
Ethics Statement and Approval
The study was approved by the institutional ethics committee of Peking University Third Hospital (IRB approval number: M2021085), and performed in accordance with the ethical standards of the Declaration of Helsinki (1964) and its subsequent amendments.
