Abstract
Introduction
Multiple therapies for treating intervertebral disc (IVD) degeneration are under development and range from injectable biologics to implantable devices. Treatments that show promise in small animal and cell culture systems often fail in human trials. There is a lack of models that are comparable to human IVDs, and this is a major obstacle preventing effective translation of basic science findings to human treatment. Human IVDs remodel and accumulate damage over years of life and disease, and anatomical features of vertebrae and IVDs are likely to influence clinical outcomes. Organ culture of IVDs from human autopsy provides a model that represents the spectrum of IVD degeneration. Prior studies demonstrated that whole human IVD organ culture is possible, but required organ donation and described removal of vertebral endplates which can lead to swelling and matrix loss.1 The objectives of this study are to determine the feasibility of whole human IVD organ culture using human autopsy specimens and to highlight important anatomical features using a range of tinctorial stains.
Materials and Methods
Bioreactor Design
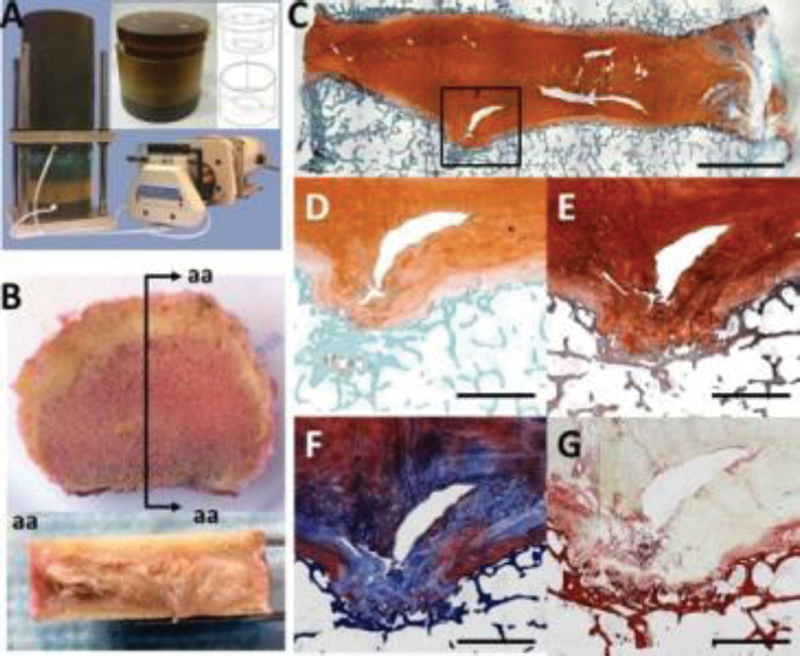
An organ culture chamber for use with human IVDs was designed, based on a previously described bovine IVD system.2 IVDs from human autopsy are isolated, placed between two permeable platens of the chamber, sealed and loaded to prevent swelling (Fig. A).
Human Autopsy IVD Isolation
Six human spines were collected from autopsy services with approval and X-rayed to assess degenerative level and establish feasibility for organ culture. IVDs that retained the vertebral and cartilaginous endplates were harvested and vertebral endplates were cleaned as previously described.2
Culture Conditions
IVDs from 3 different autopsy donors (average age 36 ± 24) were loaded for 7, 14, and 21 days in organ culture at static (0.2MPa) or diurnal (0.1-0.2MPa) conditions. All cultures occurred in 37°C and 5% CO2 using DMEM (high glucose, 10%FBS, 1% Pen/Strep, 0.2% ascorbic acid, and primocin) with media changes every 3-4 days. To assess transport through the endplates, 500 µM Texas Red labeled dextran (MW=3 kDa) was added to the media. Following culture, tissue was taken for cell viability (cryosections stained with MTT for live cells and DAPI for nuclei) and histology.
Tissue Histology
The 21 day organ culture specimens and additional human autopsy IVDs (n = 6) were isolated for tinctorial staining. Tissue was fixed, embedded in plastic (PMA), and 5µm sagittal sections were used for histology (Fig. B). Multiple stains were compared: Safranin-O, Toluidine Blue, Picrosirius Red & Alcian Blue,3 modified Gomori's Tri-chrome, and XFAST.4 Annulus fibrosus (AF), nucleus pulposus (NP), endplate, Schmorl's node, and transitional regions were compared.
Results
Human IVDs from autopsy were successfully cultured for up to 21 days. At the culture period completion, evidence of dextran transport was observed at the interface of the cartilaginous endplate and AF. Effective transport was confirmed with the high cell viability that was maintained in both NP and AF through day 21. Tinctorial staining after 21 day organ culture highlighted many structural elements of human IVDs (Fig. C). A distinctive Schmorl's node region stained with XFAST revealed a protrusion disrupting the organization of chondrocytes and matrix in the cartilaginous endplate and intercalated blue and orange staining in the matrix surrounding cell clusters (Fig. D). Localized variations in proteoglycan staining were apparent from both XFAST and Safranin-O (Fig. E) staining including diminished staining in cartilage endplates. Color gradients were more obvious with XFAST than Safranin-O. The tri-chrome stain (Fig. F) highlighted the disrupted structure at the endplate to NP interface and surrounding the entire Schmorl's node, however it was difficult to discern proteoglycan rich and collagen rich regions. Picrosirius red and Alcian blue staining (Fig. G) highlighted the collagenous structure of the cartilaginous endplate and in pericellular regions around cell clusters, although proteoglycan staining was not well differentiated in this region with this stain.
Conclusion
Key structural features in human IVDs are important in replicating clinical conditions; therefore the development of a relevant human organ culture model serves as an important tool to screen therapeutics and repair strategies to treat IVD degeneration. The presence of dextran at the interface of the endplate and AF, and high cell viability in the AF and NP after 21 days in culture demonstrated the feasibility of human organ culture using IVDs from autopsy. Tinctorial stain comparisons of sagittal IVD sections highlighted the complex structure of human IVDs, which are difficult to replicate in animal model systems. The use of IVDs from autopsy provides a viable source for ex vivo experiments to assess mechanisms of pathophysiology and to evaluate therapies for IVD degeneration in a highly clinically relevant model system.
None declared
Gawri R, Mwale F, Ouellet J, et al. Development of an organ culture system for long-term survival of the intact human intervertebral disc. Spine 2011;36(22):1835–1842
Illien-Jünger S, Gantenbein-Ritter B, Grad S, et al. The combined effects of limited nutrition and high-frequency loading on intervertebral discs with endplates. Spine 2010;35(19):1744–1752
Gruber HE, Ingram J, Hanley EN Jr. An improved staining method for intervertebral disc tissue. Biotech Histochem 2002;77(2):81–83
Leung VY, Chan WC, Hung SC, Cheung KM, Chan D. Matrix remodeling during intervertebral disc growth and degeneration detected by multichromatic FAST staining. J Histochem Cytochem 2009;57(3):249–256
