Abstract
Introduction
Intervertebral (IVD) disc degeneration is characterized by a cascade of structural, chemical, and cellular changes. The current standard for surgical treatment of degenerated discs involves total disc extraction followed by fusion or implantation of a motion-preserving disc prosthesis. However, complications arising from such procedures have stimulated interest in tissue-engineered- (TE-)IVDs as a potential biological treatment for degenerated discs. TE-IVDs have the potential to mimic the physiological properties of native discs better than conventional implants.1-6 Previous approaches to disc engineering have focused on creating AF or NP analogues. In the presented study, we evaluated the viability of tissue engineered IVD composites in vivo.
Materials and Methods
This study aimed to evaluate the viability of our TE-IVD implants in vivo according to radiological, histological, biochemical, and biomechanical parameters. TE-IVDs were implanted into 20 rats at C3/4 following discectomy. Twelve rats were sacrificed after 6 months for biochemical and biomechanical analysis. MR and X-ray imaging was performed on those rats at 6 weeks and 6 months. Eight rats were sacrificed after 8 months for histological fixation of the TE-IVD segment. MR and X-ray imaging was performed at 1, 5, and 8 months on those rats. Control group underwent only discectomy and were sacrificed at 8 months for histological analysis. The proximally adjacent disc was also studied as a healthy control.
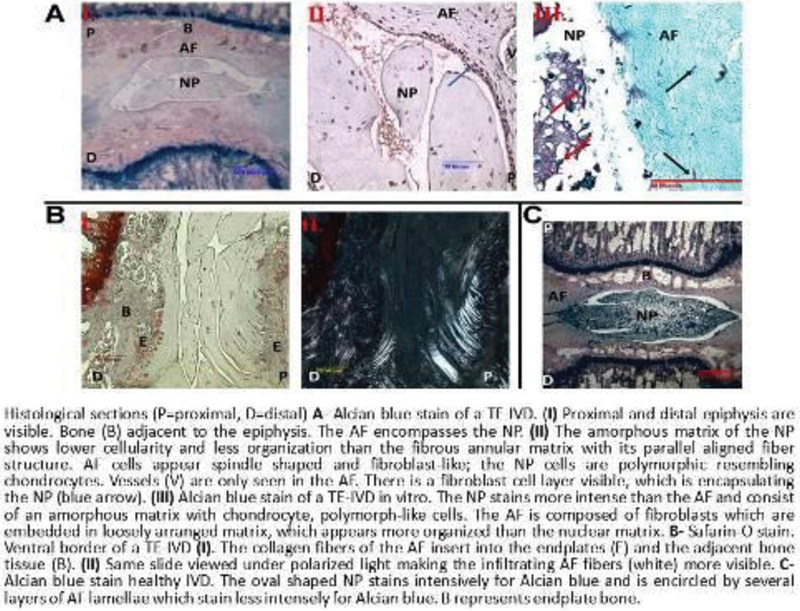
The nucleus pulposus (NP) and annulus fibrosus (AF) cells of our TE-IVDs were harvested from sheep IVDs and embedded in alginate (NP) and collagen (AF).2 Biomechanical tests performed at 6 months, including stress-relaxation tests and frequency sweep tests, measured equilibrium modulus and hydraulic permeability. Biochemical tests determined collagen and proteoglycan content. MR and X-ray imaging were used to study disc height and morphology of implanted TE-IVDs. NP size was assessed as NP voxel count according to the number of MRI voxels that composed it. MR T2-relaxation time (RT) measurements were used to assess water content and volume of the NP, and T1ρ-RT to measure proteoglycan content. Eight-month histological sections were stained with Alcian Blue and Safranin O for glycosaminoglycans and proteoglycans.
Results
Biochemical analysis showed that at 6 months collagen and proteoglycan content of TE-IVDs was similar to that of the native AF and NP. Collagen type I was found throughout the disc, while collagen type II was concentrated in the NP; similarly, proteoglycan content was greater in the NP than the AF. Mechanical tests revealed hydraulic permeability, equilibrium modulus, ability to pressurize, and load-bearing ability comparable to intact native discs. Disc height of TE-IVDs remained a constant 68 to 74% of that of healthy control discs over 8 months. MR imaging showed that TE-IVDs maintained native disc-like morphology over that same time period; the NP midsection was thinner but remained hyperintense, and was surrounded by slightly thickened hypointense AF tissue. These qualitative measurements were supported by mean voxel count measurements. T1ρ-RT measurements found that NP water content was similar to healthy discs, but NP size reduced. T2-RT measurements indicated proteoglycan synthesis in TE-IVDs. Histological sections showed AF tissue consisting of an organized fibrous structure encapsulating the NP. The NP matrix appeared amorphous, stained for proteoglycans, and contained chondrocyte-like cells. The AF fibers infiltrated the bony endplates almost to the epiphysis without any signs of immune reaction. The discectomy-only control group showed complete disc collapse and no signs of viable disc tissue on MRI and histology.
Conclusion
In vivo TE-IVDs demonstrated similar properties to native discs—including ability to remain in place, withstand mechanical loads, and maintain satisfactory disc height and chemical composition—and remained viable over 8 months. Discs showed evidence of dynamic adaptation to the host environment, with extracellular matrix production and cell proliferation. TE-IVDs should be studied further in larger animals to move the construct closer to clinical application.
None declared
Bowles R, Gebhard H, Dyke J, et al. Transplantation of Composite Tissue Engineered Intervertebral Discs to Restore Function to the Rat Spine. New Orleans, LA: Orthopaedic Research Society; 2010
Bowles RD, Gebhard HH, Härtl R, Bonassar LJ. Tissue-engineered intervertebral discs produce new matrix, maintain disc height, and restore biomechanical function to the rodent spine. Proc Natl Acad Sci U S A 2011;108(32):13106–13111
Bowles RD, Williams RM, Zipfel WR, Bonassar LJ. Self-assembly of aligned tissue-engineered annulus fibrosus and intervertebral disc composite via collagen gel contraction. Tissue Eng Part A 2010;16(4):1339–1348
Gebhard H, Bowles R, Dyke J, et al. Total disc replacement using a tissue-engineered intervertebral disc in vivo: new animal model and initial results. Evid Based Spine Care J 2010;1(2):62–66
Gebhard H, James AR, Bowles RD, et al. Biological intervertebral disc replacement: an in vivo model and comparison of two surgical techniques to approach the rat caudal disc. Evid Based Spine Care J 2011;2(1):29–35
Mizuno H, Roy AK, Vacanti CA, Kojima K, Ueda M, Bonassar LJ. Tissue-engineered composites of anulus fibrosus and nucleus pulposus for intervertebral disc replacement. Spine 2004;29(12):1290-1297, discussion 1297–1298
