Abstract
Introduction
Although there are different causes of low back pain, intervertebral disc (IVD) degeneration is one of them. In healthy discs, nociceptive nerve fibers and mechanoreceptors penetrate up to the outer annulus fibrosus (AF). In contrast, the endplates are heavily innervated. However, neither the inner AF nor the nucleus pulposus (NP) are innervated. Discogenic pain can occur due to degenerated discs acquiring cracks and fissures (annulogenic pain) or endplate damage (vertebrogenic pain) resulting in increased nerve fibers that penetrate the inner AF and NP. The increased expression of neurotrophins nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF) has been identified in human and animal models of degenerating IVDs.1 Proinflammatory cytokines interleukin (IL)-1β and tumor necrosis factor-α (TNF-α) have been shown to trigger the expression of NGF, BDNF, and substance P in human NP and AF cells. In related studies, bone morphogenetic protein (BMP) was shown to suppress innervation while overexpression of the BMP inhibitor noggin resulted in a significant increase in nerve fibers.2 However, the use of growth factors in clinical practice is limited by their high cost. Using synthetic peptides, such as link N, which stimulate BMP signaling, can circumvent this cost. The purpose of the present study was to evaluate the effect of link N on neurotrophins and substance P by human IVD cells stimulated with proinflammatory cytokines as well as in injured bovine IVDs.
Materials and Methods
Lumbar IVDs were obtained through organ donations within 6 hours after death. The procedure was approved by the local Research Ethics Board. Cells were isolated from NP and AF regions of the discs by sequential digestion with pronase followed by collagenase IA digestion for NP and collagenase II digestion for AF, respectively. They were cultured in monolayers and stimulated with TNF-α (100 ng/mL) and IL-1β (10 ng/mL) in the presence or absence of link N (1 μg/mL) for 48 hours. Total RNA was isolated and gene expression was measured using reverse-transcriptase polymerase chain reaction. The release of substance P into the culture media was measured after cells were stimulated by TNF-α (100-ng/mL) and IL-1β (10 ng/mL) in the presence or absence of link N (1 μg/mL) at different time points (1, 2, 4,and 24 hours). Coccygeal IVDs from the tails of adult bovine steers (20-25 months) were used for disc isolation. Four discs with cartilage end plates were isolated and were treated (control, capsaicin [1.5 μg/mL], punctured with a 16G needle, link N [10 μg/mL] treated) after preconditioning for 24 hours in complete DMEM. Disc culture media was collected at different time points for analysis. Substance P in the media was concentrated by solid phase extraction and was assayed by ELISA.
Results
Link N is known to induce proteoglycan synthesis by isolated disc cells and in degenerate rabbit and human discs, and to enhance chondrogenesis of MSCs in vitro. It is, however, not known if link N can suppress inflammatory mediators and neurotransmitters in disc degeneration. Without intervention or with link N supplementation alone only trace amounts of NGF gene expression in human lumbar disc cells from AF regions was observed. When human lumbar disc cells from AF regions were exposed to TNF-α for 48 hours, NGF gene expression was observed to increase significantly (p < 0.05). However, supplementing 1.0 µg/mL link N to this media led to a significant downregulation of NGF gene expression. Further, link N significantly suppressed substance P release from punctured bovine discs after 24 hours of treatment as compared with the untreated punctured disc (p < 0.05) (Fig. 1). We also showed that link N can suppress TNF-α and IL-1β-induced messenger RNA levels of brain-derived neurotrophic factor (BDNF) and TAC 1 in AF cells. Thus, link N appears to reduce inflammatory mediators and neurotransmitters in human AF cells from normal and degenerated discs, as well as in injured bovine IVDs.
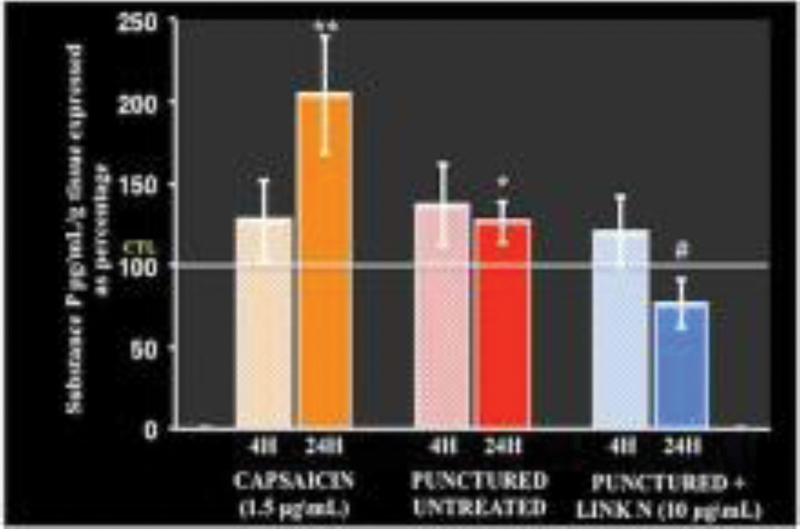
Link N treatment significantly reduced substance P release into culture medium from punctured IVDs isolated from adult bovine tails.
Conclusion
Link N is a promising agent for biological repair of degenerated human discs but an affective treatment for discogenic pain will also depend on stopping inflammation. Previous studies have shown that BMPs can suppress peripheral innervation in the skin.2 Because BMPs and link N both share the Smad 1/5 signaling pathway, it was not unreasonable to assume that link N could also suppress inflammatory mediators associated with discogenic pain. The present study indicates that link N can also suppress NGF, BDNF, and TAC 1 in human disc cells as well as substance P release in injured bovine discs. This suggests that link N has the potential to inhibit pain induced by neuronal innervation caused by disc degeneration.
Disc degeneration is often associated with low back pain; link N represents a potential economical growth factor with beneficial effects on disc repair. It would be of clinical significance to see if link N has any potential in reducing the pain caused by neuronal invasion during disc degeneration.
None declared
García-Cosamalón J, del Valle ME, Calavia MG, et al. Intervertebral disc, sensory nerves and neurotrophins: who is who in discogenic pain? J Anat 2010;217(1):1–15
Guha U, Gomes WA, Samanta J, Gupta M, Rice FL, Kessler JA. Target-derived BMP signaling limits sensory neuron number and the extent of peripheral innervation in vivo. Development 2004;131(5):1175–1186
