Abstract
Study Design
Systematic review.
Objective
We assessed the current state of spine registries by collecting spine trauma data and assessing their compliance to defined registry standards of being clinical quality. We ascertained if these registries collected spinal cord injury data alone or with spine column trauma data.
Methods
A systematic review was performed using MEDLINE and Embase databases for articles describing dedicated spinal cord and spine column databases published between January 1990 and April 2011. Correspondence with these registries was performed via e-mail or post. When no correspondence was possible, the registries were analyzed with best information available.
Results
Three hundred eight full-text articles were reviewed. Of 41 registries identified, 20 registries fulfilled the criteria of being clinical quality. The main reason for failure to attain clinical quality designation was due to the unavailability of patient outcomes. Eight registries collected both spine column and spinal cord injury data with 33 collecting only traumatic spinal cord injury data.
Conclusion
There is currently a paucity of clinical quality spine trauma registries. Clinical quality registries are important tools for demonstrating trends and outcomes, monitoring care quality, and resolving controversies in the management of spine trauma. An international spine trauma data set (containing both spinal cord and spine column injury data) and standardized approach to recording and analysis are needed to allow international multicenter collaboration and benchmarking.
High-quality dedicated spine trauma registries benefit the community by being powerful tools for treatment planning and the organization and delivery of services. They are extremely valuable research assets as they allow assessment of trauma management and outcomes. Although there are several of these registries, it is known that the majority of them collect only spinal cord injury data. 1 One main reason is the significant morbidity and the extremely high lifetime cost associated with spinal cord injuries when compared with spine column trauma alone.2 To be most useful, registries or databases (referred collectively as registries) should meet the five standards that define clinical quality, namely: (1) having mergeable data, (2) having a standardized data set, (3) having rules for data collection, (4) having knowledge of patient outcomes, and (5) having a queriable database. 3 Clinical quality registries (CQRs) typically focus on conditions and procedures where outcomes vary and where improvements in management have potential to improve outcomes and reduce cost. We therefore conducted a systematic review to identify and describe all existing dedicated spine registries that collect spine trauma data and to evaluate whether they attain the designation of being clinical quality.
Materials and Methods
Data Sources and Searches
We searched Medline (1950 to April 2011) and EMBASE (1980 to April 2011) using keywords and Medical Subject Headings (MeSH) based on the following terms: registry/database/audit/surveillance, fracture/wound/injury/damage/contusion/laceration/transection, ischemia and spine/spinal/spinal cord.
Study Selection
Each potentially eligible study was independently assessed by two reviewers (J.W.T. and P.C.H.C.). Titles retrieved from searching and their reference lists were screened to identify potentially relevant publications. For any potentially relevant abstracts, full-text articles were obtained and reviewed. Studies describing dedicated spine registries collecting spine trauma data were identified for data extraction.
Data Extraction
Dedicated spine trauma registries were subsequently investigated using Internet resources and contact with key personnel. The registries’ Internet home pages or relevant Web pages were visited to acquire contact details of their governing bodies. They were then contacted by e-mail or post to confirm the existence and operational status of the registries and to ascertain whether the registries meet clinical quality requirements through the following questions: (1) Is your data mergeable (stored in a format that allows researchers to query the database)? (2) Do you have a standardized data set (same data collected for all patients in the registry)? (3) Do you have rules for data collection (inclusion and exclusion criteria)? (4) Are you able to observe a patient over time (a patient is recorded as a single continuous record for the duration of the registry, prospectively)? and (5) Do you have knowledge of patient outcomes (follow-up)?
Two further attempts were made to contact nonrespondents. If unsuccessful, these registries were analyzed according to data available from Internet resources and scientific publications. Two of the authors (J.W.T. and P.C.H.C.) independently completed a standardized extraction checklist for each study. Disagreements on data extraction and classification of study results were resolved by consensus.
Results
In all, 3,756 articles were identified using the EMBASE and Medline electronic databases; 3,437 articles were excluded following title and abstract review. Full-text review was performed on 317 articles with 308 articles selected for data extraction. These articles are listed in the online Supplementary Appendix 1.
Forty-nine potential dedicated spine trauma registries were identified. Completed questionnaires were received from 39 (80%). Of the 49, 8 were established for specific research projects and were not functioning as ongoing registries. Of the remaining 41, 31 (75%) were currently collecting and reporting data, and 5 had ceased to operate. We were not able to ascertain the operational status of the remaining 5 due to lack of information.
Twenty registries attained the designation of being clinical quality; 10 of the 20 CQRs contributed information to their national registry (National Spinal Cord Injury Statistical Centre in the United States). Of the 20 CQRs, 15 collected only spinal cord injury data, and 5 collected both spinal column and spinal cord injury data. Of the 21 non-CQRs, 18 collected only spinal cord injury data, and 3 collected both spinal column and spinal cord injury data.
Twenty-one registries failed to attain the designation of being clinical quality. Four of them contributed information to their national registry (Australian Spinal Cord Injury Register). Twenty registries did not have follow-up data, 14 did not have a single continuous record for storage of longitudinal data, 2 did not have a queriable database, and 1 did not have a systematic or prospective method of data collection. However, all 21 had a standardized data set (Table 1). None of the registries exclusively collected spinal column injury data.
Dedicated spine registries collecting spine trauma data
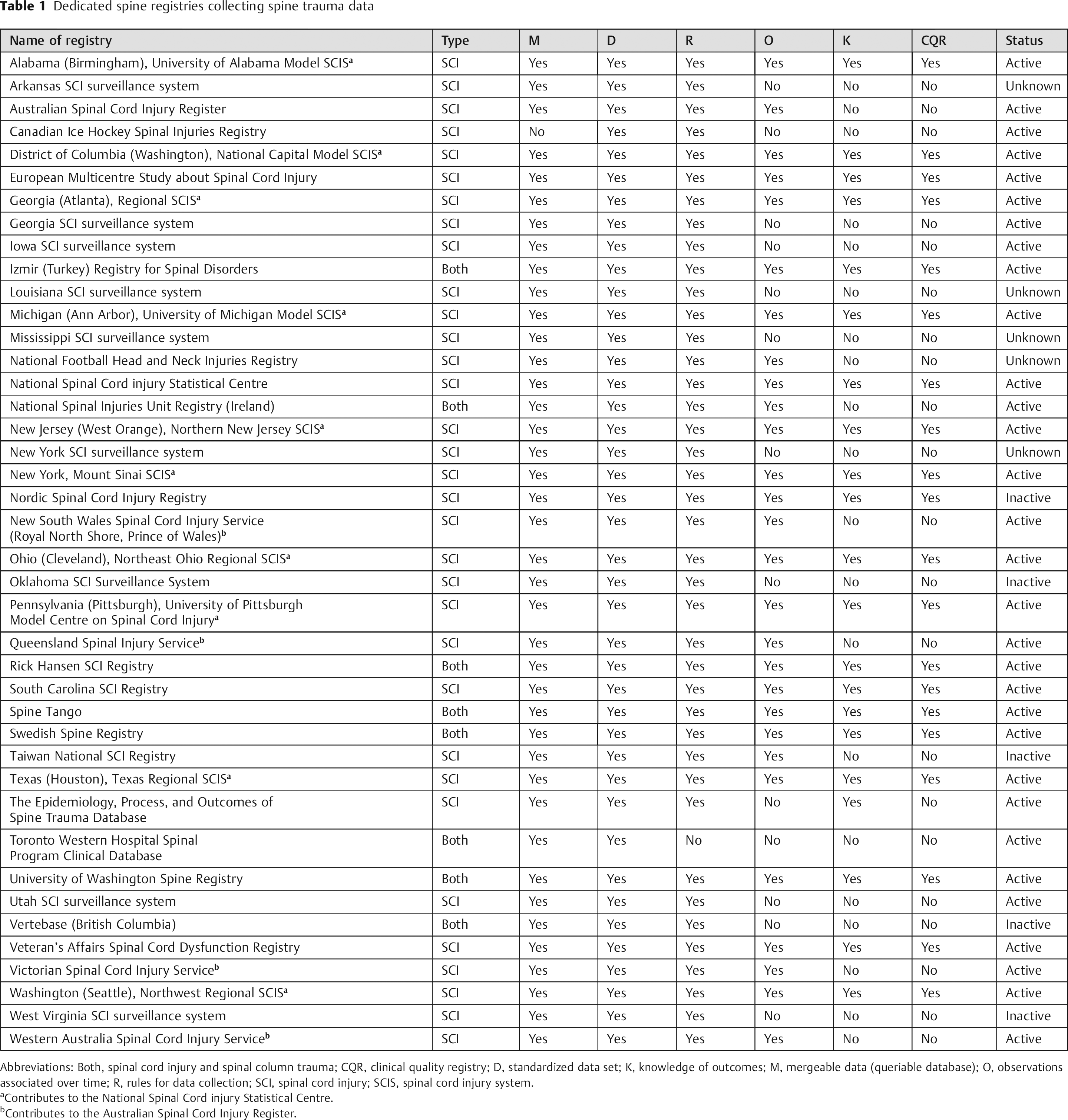
Abbreviations: Both, spinal cord injury and spinal column trauma; CQR, clinical quality registry; D, standardized data set; K, knowledge of outcomes; M, mergeable data (queriable database); O, observations associated over time; R, rules for data collection; SCI, spinal cord injury; SCIS, spinal cord injury system.
Contributes to the National Spinal Cord injury Statistical Centre.
Contributes to the Australian Spinal Cord Injury Register.
Discussion
Registries have many functions. Among them are the elucidation of demographics and injury trends, monitoring of level of care via outcome assessment and patient advocacy, planning of resource allocation and delivery of services, collaboration and outreach, benchmarking, and last, an invaluable research tool. As a result, its data are the target of interest of both academics and more recently clinicians. The majority of dedicated spine registries do not collect data on vertebroligamentous injuries despite being very common. We argue that, without this information, spine trauma clinicians may lack the complete appreciation of spine trauma pathology on which to guide management and to report outcomes. Setting up a clinical quality spine trauma registry collecting both spinal cord and spine column trauma data enables the establishment and use of the same language and data elements to evaluate spine trauma in terms of research and formulation of algorithmic spine trauma classification and treatment paradigms.
There is significant variation of practice in the management of spine trauma, especially in patients with polytrauma. Spine trauma CQRs enable the understanding of natural history and allow comparison of different treatment regimens. An example is the case of a patient with traumatic central cord syndrome without evidence of mechanical instability. The Subaxial Cervical Spine Injury Classification System (SLIC) does not recommend any treatment option but leaves it to the discretion of the treating clinician. 4 The collection of both spinal cord and spine column trauma data enables improvement and modification of current spine trauma treatment algorithms such as the SLIC and TLICS (Thoracolumbar Injury Classification System) algorithms to be more relevant as the severely injured trauma patient with spine trauma is perhaps the most complex patient to treat. 4 , 5 With this, standardized outcome data in the form of both physician-reported (radiology outcomes and clinic reviews) and patient-reported (health scores or scales) outcomes are crucial in determining which management strategy, if any, leads to better outcomes and the nuances that inevitably exist.
Key Elements of the Spine Trauma Data Set
The cornerstone of every spine trauma registry is the minimum data set. Choosing the right data items is a balance between comprehensiveness and practicality. It must contain the data needed to be useful, but not so much that it is impractical to collect routinely. An example of a clinical quality spine trauma minimum data set was recently published. 6 A spine trauma minimum data set can be divided into five broad categories: patient history, clinical examination, spine injury characteristics, patient management, and patient outcome. Paramount to a spine trauma CQR is the collection of both spinal cord and spine column trauma data. Spinal cord injury data such as the American Spinal Injury Association impairment score and scale are crucial elements in a spine trauma data set. However, a spinal cord injury registry would not necessarily place importance on spine column trauma data such as injury morphology (differentiate between compression, burst, translational, rotational, and distraction injuries) and soft tissue integrity. This is particularly important as diagnosis, treatment, and outcome can then be correlated to ensure financial accountability and patient advocacy. An example of this is a lumbar spine burst fracture with minimal angulation in a neurologically intact patient without lower back pain. The TLICS recommends conservative management for this injury type. However, it is not uncommon for this injury type to be instrumented with or without fusion. Further studies and registry data assessing long-term patient- and physician-reported outcomes is invaluable in providing further evidence in this matter.
In our review, spinal cord injury data were collected more frequently than spine column trauma data. Only five CQRs collected both spine column and spinal cord trauma data. This situation may be due to the preponderance of research on spinal cord injuries. 1 Although a single case of spinal cord injury is associated with significant morbidity and an extremely high lifetime cost, the incidence of spinal column trauma is much greater than that of spinal cord injuries.2,7, 8 , 9 This results in a substantial economic burden by virtue of the sheer number of injuries. 10 A significant proportion of these patients are high functioning but suffer from chronic pain or mild disability and are unable to return to work. It is also uncommon for spinal cord injury secondary to trauma to occur without spinal column injury.
Patient management data items are divided into three subgroups: (1) conservative management, including orthotic device utilization; (2) operative management, including description of the performed procedure; and (3) spinal devices, including instrumentation, cages, and bone substitutes. Spine trauma care is multidisciplinary with clear role delineations. The same should apply to registry data collection. Data entry about operations and spinal devices should be entered into the database by members of the treating surgical unit. Similarly, the orthotist should enter details of orthotic devices used, and clinical nurse specialists or other dedicated staff members ensure the completion of data entry into the registry database.
A sound methodology for data collection is crucial for CQRs. The gold standard is a systematic, prospective, and most importantly simple data collection protocol. Also, having a dedicated data collection team of spine trauma clinicians and spine trauma clinical nurse specialists is crucial to ensuring registry protocol integrity. This is especially so as data entry for a patient occurs continuously and is not a one-off event. The database should be able to merge different entries pertaining to the same patient into an individual aggregate data set.
The interpretation and data capture of spine injury characteristics data items may be more complicated. The Spine Trauma Study Group noted moderate intraobserver and interobserver agreement regarding the radiologic classification of spine fractures. 11 This could lead to inaccuracy with spine column trauma data entry and skew study results. We have proposed the use of synoptic reporting to circumvent this issue. 12 This method of reporting has been used successfully in oncologic histopathology and has led to improved patient outcomes. 13 , 14 It comprises a method template reporting and structured data capture utilizing standard nomenclature and a set of universally required findings, facilitating a consistent report structure. Using a multidisciplinary modified Delphi approach, a synoptic reporting template was created (online Supplementary Appendix 2). 15 This is likely to improve observer reliability and accuracy of spine injury characteristics data item interpretation, especially if consultant radiologists were to perform the reporting.
Outcome Measures
Our study showed that the failure to attain CQR standard in dedicated spine trauma registries was most often due to not having the infrastructure enabling collection and storage of patient outcome data. Imaging results and clinical examination are far inferior to objective validated measures reflecting outcomes that are important to patients, clinicians, and funders. 16 The majority of spine trauma patients are independent in terms of daily function, but a proportion of them may have significant morbidity. Patient outcome data items are divided into two subgroups. Physician-reported outcomes (follow-up imaging, clinical examination) assessed during outpatient clinic review should be entered into the registry at that time. Spine trauma CQRs have the ability to capture patient-reported outcomes using pain scores, the Short Form-12 or -36 Health Survey, Functional Independence Measure, and Glasgow Outcome Scale. 17 Although these outcomes can be ascertained by telephone interview, trained assessors are needed. 18 The evaluation of the cost-effectiveness of a treatment may be met with instruments that are designed for cost-utility analyses and for comparisons of therapeutic effects across different diseases. The EuroQol-5D, which measures generic health-related quality of life, is one such measure. 19 These outcome measure data should be entered into the database on completion of assessment. In our experience, clinical nurse specialists play an important and efficient role as gatekeepers to ensure data quality and completeness.
Constraints and Limitations
We believe that the development and maintenance of a CQR underpins spine trauma clinical research and treatment. However, it may not be possible or appropriate for every institution. Setting up of a spine trauma CQR in a region without preexisting robust data collection systems and infrastructure is particularly difficult in a resource-poor setting with a shortage of skilled personnel, funding, information technology support, or existing registry infrastructure. However, such institutions can collect data that can be submitted to a regional, national, or international spine trauma CQR. The major challenge for a CQR is whether the data collection can be sustained indefinitely. Often, the longevity and success of a registry depends on the involvement, enthusiasm, and technical ability of participating clinicians. In the middle of 2011, an international spinal cord injury registry ceased its operations. From our correspondence with its governing body, it ceased its operations due to funding and participation issues. It is difficult to predict the success for any CQR; however, elements crucial to its survival are prevention of registration fatigue (by having a concise yet complete minimum data set), sound data collection methodology and infrastructure, financial backing, and most importantly dedicated staff.
By being a registry that meets clinical quality standards, it is inferred that the registry has at least met basic quality standards. This is because registry quality assessment is often difficult and controversial as empirical evidence for evaluating parameters purported to indicate quality may not meet international standards. Moreover, these data are heavily guarded. An indirect simplistic manner of doing this is by assessing scientific articles based on registry data. Again, this is fraught with bias, as the quantity and quality of the publications and presentations are often the result of researches and may be a poor reflection of registry quality.
Future Directions
Synoptic reporting for spine trauma is another technology for spine clinicians to embrace. Because this method of reporting incorporates discrete and scientifically validated data elements, the synoptic data used in spine trauma reporting can be exported to research databases collecting data on spine trauma. The development and maintenance of a successful spine trauma CQR is challenging. With it we will be able to compare results and benchmark against other databases and health systems to facilitate improvement in patient care.
Conclusion
Clinical quality spine trauma registries are important tools for demonstrating trends and outcomes, monitoring care quality, and resolving controversies in the management of spine trauma. There is currently a paucity of dedicated spine trauma clinical quality registries. We encourage the establishment of clinical quality spine trauma registries collecting both spinal column and spinal cord injury data. These CQRs should use common definitions and coding to allow comparative or multicenter research. An international spine trauma data set (collecting both spinal cord and spine column injury data) and standardized approach to recording and analysis are needed.
Disclosures
Jin W. Tee, None
Patrick C. H. Chan, None
Jeffrey V. Rosenfeld, None
Russell L. Gruen, None
