Abstract
The aim of this study was to measure the pharmacokinetics and pharmacodynamics of subcutaneously injected 40 IU/ml porcine lente insulin preparation (Caninsulin, Intervet BV, The Netherlands) in diabetic cats. The pharmacological properties of the insulin in poorly controlled or untreated cats were compared with those after several weeks of treatment, to determine if improved diabetic stability altered the pharmacology of this insulin. In addition, the pharmacological properties of intravenously injected 100 IU/ml regular porcine insulin (Actrapid MC, NovoNordisk, Denmark) were measured.
Serial plasma samples were collected after subcutaneous injection of porcine lente insulin from 25 diabetic cats in the first week of admission to a 12-month diabetic treatment trial. Samples were also collected after 4 or 8 weeks of treatment, in those cats which had not achieved diabetic remission by this time. At this time, serial plasma samples were also collected from these cats after intravenous injection of porcine regular insulin. Plasma samples were assayed for glucose, anti-insulin antibodies were extracted using a PEG technique, and samples were assayed for insulin using an RIA kit with low sensitivity for endogenous feline insulin, but high sensitivity for exogenous porcine insulin in feline plasma.
Caninsulin injected subcutaneously in diabetic cats led to a peak insulin concentration in plasma after 1.7 ± 0.1 h, and a nadir of blood glucose after 4.1 ± 0.3 h. Insulin and glucose concentrations returned to baseline within 12 h. There was no significant change in the onset or duration of Caninsulin action between the first week of treatment and 5 or 9 weeks of treatment. Actrapid MC injected intravenously had a peak insulin at 0.36 ± 0.03 h, and a nadir of blood glucose at 1.9 ± 0.3 h. Insulin and glucose returned to baseline within 6 h. It was concluded that Caninsulin injected subcutaneously has suitable pharmacological properties for the twice-daily treatment of diabetes mellitus in cats. In addition, Actrapid MC insulin injected intravenously has suitable pharmacological properties for injection every 4–6 h in diabetic cats.
Porcine lente insulin 40 IU/ml (Caninsulin, Intervet BV, The Netherlands) is an insulin-zinc suspension with a retarded onset and duration of action through crystallisation (Hallas-Møller et al 1954). The insulins termed ‘lente’ are a mixture of three parts of the amorphous semilente (short-acting) forms and seven parts of the crystalline ultralente (long-acting) forms, and have an intermediate duration of action (Hallas-Møller et al 1954, Hildebrandt 1991). Insulin duration of action is also influenced by the effect that the amino acid sequence of the insulin has on its association state, with the effect that a porcine insulin has a slightly longer duration of action than an equivalent formulation of human insulin (Brange et al 1990, Hildebrandt 1991). In normal humans, the duration of action of subcutaneously administered porcine lente insulin is similar to that of human ultralente insulin, although the maximum reduction in blood glucose of porcine lente insulin is greater (Frier et al 1984). In humans, it has been found that there can be variation in the duration of action of an insulin in diabetic vs normal patients, with some diabetics demonstrating longer duration of action of the insulin than normal patients, perhaps due to hyperglycaemia (Gardner et al 1986, Hildebrandt 1991).
Studies have been reported describing the clinical use of intermediate- and long-acting insulins in diabetic cats, including the use of NPH, lente, ultralente and PZI insulins (Moise & Riemers 1983, McMillan & Feldman 1986, Nelson et al 1992, Bertoy et al 1995). Intravenous regular insulin is mainly used in cats for the intensive treatment of diabetic ketoacidosis (Greco et al 1995). Pharmacokinetic studies have been reported on the use of beef-pork regular, NPH and PZI insulins in normal cats (Wallace et al 1990). Pharmacokinetic and pharmacodynamic studies have been reported with porcine and bovine ultralente, and PZI, also in normal cats (Broussard & Peterson 1994). To date, the only study reporting insulin pharmacokinetics in diabetic cats involved the use of NPH and PZI insulin in a very small number of cats (Moise & Riemers 1983). The pharmacological properties of Caninsulin have been reported in diabetic dogs (Graham et al 1997), but there have been no previous reports of the pharmacology of Caninsulin or other porcine lente insulins in normal or diabetic cats.
This aim of this study was to measure and compare the pharmacological properties of Caninsulin in diabetic cats between cats at initial treatment and after several weeks of treatment, and to measure some of the pharmacological properties of 100 IU/ml regular porcine insulin (Actrapid MC, NovoNordisk, Denmark) in diabetic cats.
Materials and methods
Animals and diabetes treatment regime
A total of 25 feline diabetics (17 male, eight female, median age at admission 12 years, range 6–15 years; 12 domestic short hair, six Burmese, two Burmese cross, three domestic long hair, one Abyssinian, one Tonkinese) were recruited from cats referred to The University of Queensland Veterinary Teaching Hospital for treatment. The over-representation of males and Burmese cats is in accordance with previous epidemiological studies on diabetes mellitus in cats (Panciera et al 1990, Rand et al 1997). Cats were treated with Caninsulin (40 IU/ml porcine lente insulin, Intervet BV, The Netherlands).
The initial dose rate for Caninsulin was based on baseline blood glucose samples. Cats with baseline blood glucose up to 19 mmol/l were given Caninsulin at 0.25 IU/kg bodyweight, and cats with baseline blood glucose greater than 19 mmol/l were given Caninsulin at 0.5 IU/kg bodyweight. These doses were used up to the third day of treatment, when the first plasma collection for insulin pharmacology was performed. In the first 10 cats, insulin was given once daily for the first 3 days; all subsequent cats were treated with Caninsulin twice daily from the first day. Subsequent to the third day of treatment, dosage was determined for each cat on the basis of serial blood glucose measurements. Blood glucose measurements were made every 2 h for 12 or 24 h in each cat, at intervals daily for the first week of treatment, every 1–2 weeks for the next 4 weeks, and subsequently every 4 weeks. For the study on Actrapid MC, all cats were given 0.3 IU/kg bodyweight.
At least 2 days prior to blood collection, cats were admitted, and jugular catheters were placed (Martin & Rand 1999b).
Study design
Samples for the study on subcutaneous porcine lente insulin were collected at −0.5, 0, 0.5, 1, 1.5, 2, 4, 6, 8, 10 and 12 h after injection. In all cats, samples were collected for the first study on the third day of treatment (Table 1). The second study was performed 4.5 weeks later (after 5 weeks of therapy) in cats 1–10 (Table 2), and at 8.5 weeks later (after 9 weeks of therapy) in cats 11–25 (Table 2). The second study was omitted in cats which had attained diabetic remission by the time at which this study was scheduled (Table 2).
Details of insulin doses and pharmacokinetic parameters—Caninsulin given subcutaneously at week 1

This cat was started on a higher dose rate of Caninsulin as it had been treated with once-daily Caninsulin for several months previously.
Not calculated as differing dose rates were used for each cat.
Details of insulin doses and pharmacokinetic parameters—Caninsulin given subcutaneously and Actrapid MC given intravenously at week 5 or 9
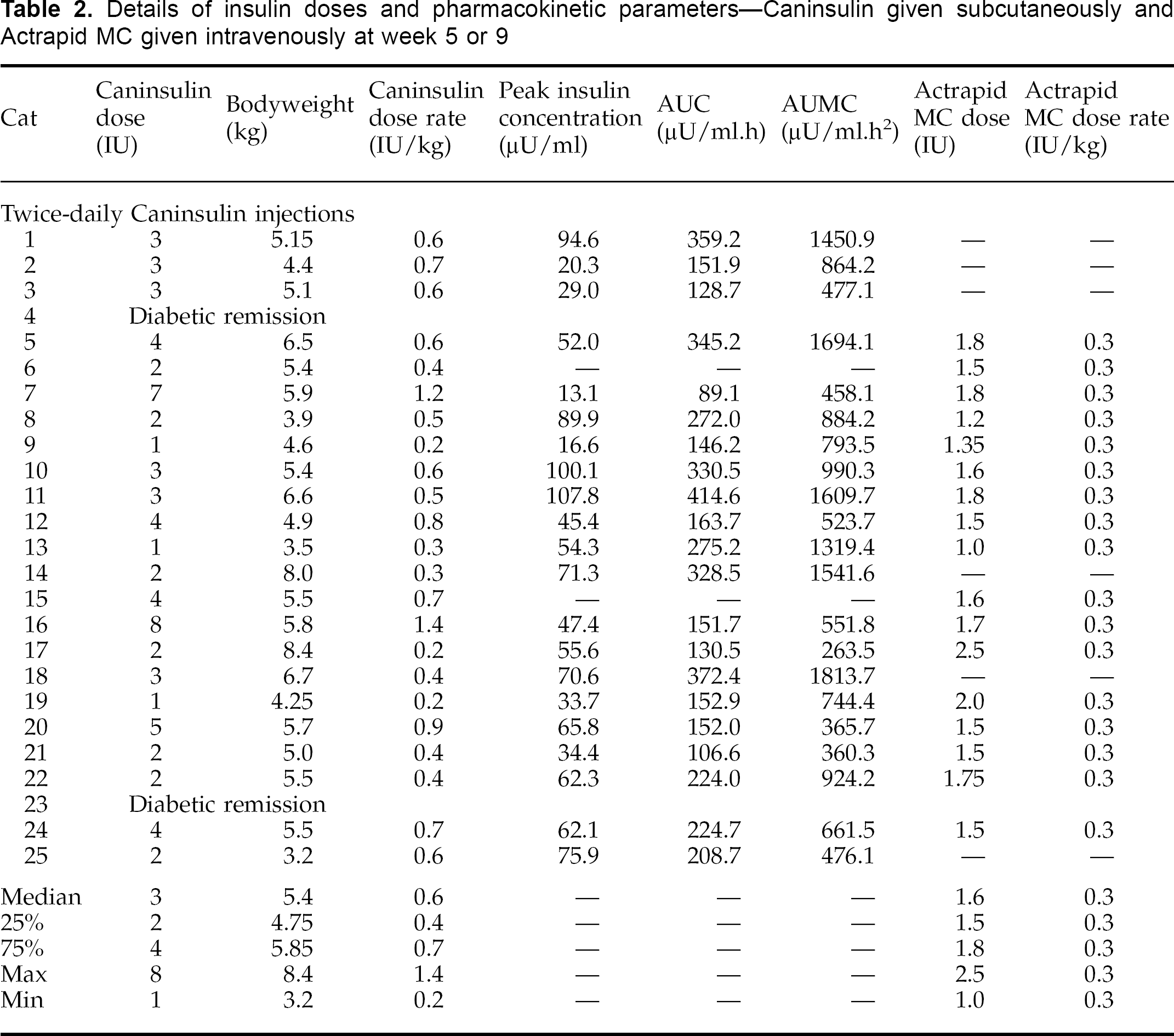
Samples for the study on intravenous porcine soluble insulin were collected at −0.5, 0, 0.25, 0.5, 0.75, 1, 1.5, 2, 3, 4, 5 and 6 h after injection (Wallace et al 1990). Cats were given the same dose (0.3 IU/kg) of Actrapid MC intravenously as a bolus. This study was performed on the day following the second study on subcutaneous porcine lente insulin, and was not performed in all cats (Table 2). This study was performed primarily to allow calculation of the bioavailability of subcutaneously injected Caninsulin in diabetic cats (Wallace et al 1990).
Data were not available for Caninsulin in week 1 from cats 1 and 2. In weeks 5 or 9, cats 4 and 23 were in remission, so no measurements were performed in these cats. In week 1, plasma insulin concentrations were barely above baseline after Caninsulin injection in cats 15 and 16, so data from these cats were excluded. Likewise, after Caninsulin injection at weeks 5 or 9, plasma insulin concentrations were barely above baseline in cats 6 and 15, so data from these cats were excluded.
Sample collection and handling
Blood samples were collected into tubes containing K3EDTA (1 ml Vacuettes, Greiner Labotechnik GmbH; Kremsmuenster, Austria) and 0.05 ml (500 KIU) of the protease inhibitor aprotinin (Trasylol; Bayer Corporation, Germany), kept on ice. Plasma was separated by centrifugation at 4°C within 60 min. Plasma was aspirated into paired aliquots and stored at −70°C until antibody removal was performed up to several months later. Once antibody removal had been performed, samples were stored at −70°C and assayed within 60 days.
Assays and analysis
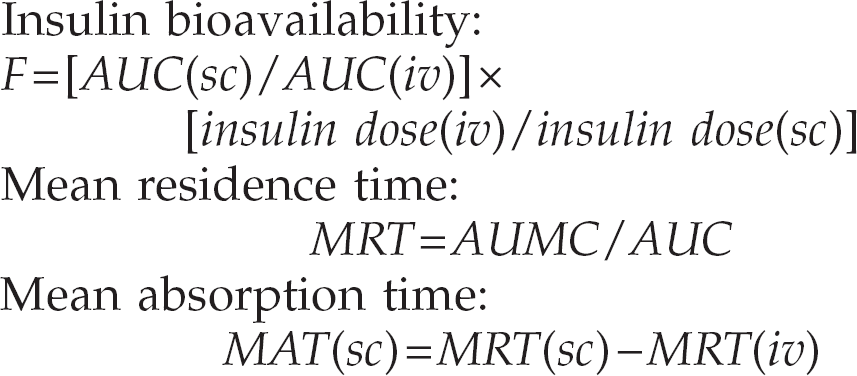
In addition, insulin clearance from plasma (CL) and insulin volume of distribution (VD) was calculated for regular porcine insulin using the equations:

Insulin concentration was regarded as having returned to baseline concentration when it was less than the minimum concentration which could be reliably assayed by the insulin RIA kit (11 μU/l).
Data were analysed using Microsoft Excel (Microsoft Corporation, Seattle, WA, USA) and SigmaStat (SPSS, Inc, Chicago, IL, USA). Normally distributed data was described and analysed parametrically and non-normally distributed data was described and analysed non-parametrically
Results
At week 1, doses ranged from 0.2 to 0.9 IU/kg bodyweight, with a median dose of 0.4 IU/kg bodyweight. At week 5 or 9, doses ranged from 0.2 to 1.4 IU/kg bodyweight, with a median dose of 0.6 IU/kg bodyweight (Tables 1 and 2). However, there was no statistically significant increase in median dose rate (P>0.05, Mann-Whitney rank sum test) between week 1 and week 5 or 9. An example of typical changes in plasma glucose and insulin concentrations following subcutaneous injection of Caninsulin in a diabetic cat is shown in Fig 1.

Typical plasma insulin and glucose concentrations in a diabetic cat (cat 1, week 1) after subcutaneous injection of Caninsulin at time=0.
Insulin concentration in plasma peaked at 0.36 ± 0.03 after injection of intravenous Actrapid MC, and plasma insulin and glucose concentrations returned to baseline within 6 h of injection (Table 3).
Pharmacokinetic and pharmacodynamic properties of subcutaneous Caninsulin and intravenous Actrapid MC in diabetic cats

Not calculated for Caninsulin as differing dose rates were used for each cat.
There was no statistically significant correlation between the total insulin dose given or the insulin dose rate, and the time to the insulin peak concentration, the time for insulin concentrations to return to baseline, the time to the nadir of blood glucose, and the time for the blood glucose levels to return to baseline (P > 0.05, Pearson product moment correlation).
There was no significant difference in the time to peak plasma insulin concentration or time to the nadir of blood glucose concentration (Table 3) after Caninsulin injection between week 1 and week 5 or 9 (P > 0.05, paired t-test). There was no significant difference in the time for plasma insulin or glucose to return to baseline (Table 3) after Caninsulin injection between week 1 and week 5 or 9 (p > 0.05, Wilcoxon signed rank test).
Discussion
This study showed that Caninsulin has a shorter duration of action in diabetic cats than it does in diabetic dogs (Graham et al 1997). It is typical of an insulin in cats that it has shorter onset and duration than in other species (Greco et al 1995). In some dogs, Caninsulin has a sufficient duration of action that is able to be given once daily (Graham et al 1997), but the results of this study indicate that Caninsulin should be given twice daily to diabetic cats. All cats in this study required twice daily Caninsulin injections (Martin & Rand 1999a). In contrast, intravenously injected porcine insulin in diabetic cats in this study had a more prolonged mean residence time (MRT) than has been previously reported in diabetic dogs (diabetic cats MRT=0.9 h, vs diabetic dogs MRT=0.35 h) (Nelson et al 1990). The longer MRT for intravenous insulin in cats as compared with dogs, but the shorter MRT of subcutaneously injected insulins in cats compared with dogs, suggests that extravascularly injected insulins are more rapidly absorbed from the injection site in diabetic cats than in diabetic dogs.
Based on the results of this study, Caninsulin has similar pharmacological properties to beef-pork NPH insulin, and has a shorter onset and duration of action compared with human and beef-pork ultralente insulin, and beef-pork PZI insulin (Moise & Riemers 1983, Wallace et al 1990, Broussard & Peterson 1994). The pharmacology of Caninsulin implies it should be administered twice daily in diabetic cats, as is the case with 100 IU/ml beef-pork lente insulin (Bertoy et al 1995).
There was no significant difference between the onset or duration of Caninsulin appearance in plasma or of Caninsulin action between the cats during the first week of treatment and after 5 or 9 weeks of treatment. This suggests that any changes which occur in insulin dose responsiveness over time in diabetic cats are due to factors other than insulin kinetics, such as an increase in insulin sensitivity in response to treatment (Garvey et al 1985), which was not measured in this study.
Caninsulin had a similar bioavailability (45 ± 4%) to beef-pork NPH insulin in normal cats (45.4 ± 6.7%) (Wallace et al 1990). In humans, insulins generally have a higher bioavailability in diabetic than in non-diabetic individuals (Ferrannini & Cobelli 1987). Whether the bio-availability of Caninsulin is lower in nondiabetic cats compared with the diabetic cats studied, was not determined.
As insulin distributes into multiple compartments in the body, the pharmacokinetics of insulin is complex. In humans and in dogs, insulin follows a two- or three-compartment model (Binder et al 1984, Brown et al 1987), but this has not been determined in the cat. Additionally, the absorption model of insulin from a subcutaneous site is not clearly defined (de Meijer et al 1989, Mosekilde et al 1989, Brange et al 1990). Due to the lack of an appropriate model for insulin kinetics in the cat, analysis was performed in this study using non-compartmental methods (Rowland & Tozer 1995). Insulin half-life was not calculated in this study, as insulin elimination does not follow a first-order process. The conceptually similar parameter MRT was therefore calculated from the non-compartmental data.
The insulin RIA kits used in this study were re-tested for sensitivity to feline insulin in normal cats following the method previously described by Wallace and Peterson (Wallace et al 1990), and were shown to have little or no sensitivity to endogenous feline insulin (Link 2000). Diabetic cats are likely to have reduced or absent endogenous insulin secretion due to glucose toxicity from prolonged hyperglycaemia (Link & Rand 1996). Injection of exogenous insulin suppresses endogenous insulin secretion (Wallace et al 1990). Therefore, although there may have been some endogenous insulin secretion present in the diabetic cats studied, taken in conjunction with the low sensitivity of the assay used, potentially measurable levels of endogenous feline insulin were regarded as having minimal significance to the assayed plasma insulin levels.
In four of the samples (cats 15 and 16 in week 1, cat 6 in week 5, and cat 15 in week 9), insulin levels assayed in plasma were barely above baseline levels throughout. As the PEG technique precipitates out antibody-bound insulin, leaving only unbound insulin (or free insulin) (Nagakawa et al 1973), absorbed insulin may have been sequestered by circulating insulin-binding antibodies. These cats (6, 15 and 16) were all antibody positive, with cat 15 having the highest antibody levels of the cats assayed (Hoenig M, unpublished data). However, it is also possible that poor or delayed absorption from the injection site led to the low plasma insulin levels in these cats (Nelson 1995).
Spuriously high levels of insulin in plasma samples assayed by insulin RIA are suspicious of anti-insulin antibodies in the samples (Kurtz & Nabarro 1980, Ihle & Nelson 1991). When plasma samples from the cats in this study were assayed with the insulin RIA kit without PEG antibody extraction, spuriously high insulin levels were seen in some cats. Pooled plasma samples from individual cats collected over the first 6 months of the treatment trial were assayed for anti-insulin antibodies, with several of the cats being positive for anti-insulin antibodies (Hoenig M, unpublished data). An attempt was made to extract anti-insulin antibodies from the plasma using the acid-ethanol technique (Heding 1972), however, spuriously high insulin levels persisted in some samples. Following anti-insulin antibody extraction using the PEG technique described, assayed plasma insulin levels were reduced to expected levels.
Ideally, insulin clearance is measured using steady-state insulin infusion; however, bolus doses of insulin may be used (Ferrannini & Cobelli 1987). The median clearance of porcine soluble insulin measured in diabetic cats in this study (1.6 × 103 l/h/kg) was similar to the insulin clearance measured in humans (Ferrannini & Cobelli 1987). However, insulin clearance is much lower in diabetic dogs (Nelson et al 1990). In addition, the volume of distribution of porcine regular insulin in the diabetic cats in this study was lower than in human diabetics, similar to that of non-diabetic humans (Ferrannini & Cobelli 1987), and significantly higher that of dogs (Nelson et al 1990).
In conclusion, the pharmacology of Caninsulin (a 40 IU/ml porcine lente insulin preparation) given subcutaneously, and Actrapid MC (a 100 IU/ml regular porcine insulin) given intravenously, were described in diabetic cats. The pharmacological properties of Caninsulin imply it is suitable for twice-daily treatment of diabetes mellitus in cats. The pharmacological properties of Actrapid MC given intravenously imply it is suitable for administration every 4–6 h in cats with diabetes.
Footnotes
Acknowledgements
The authors would like to thank Intervet International BV, Netherlands, for funding the study, and also Mararethe Hoenig of the University of Georgia for performing the insulin antibody assays. The authors wish to thank Lynn Knott for performing the insulin assays, and the staff of the University of Queensland Veterinary Teaching Hospital for their assistance with patient care.
