Abstract
This study aims to evaluate the effects of plasma beta endorphin level and exercise on migrainous headache. Forty patients with migrainous headache without aura (MWOA), firstly exercised on a treadmill and afterwards were asked to continue an aerobic home exercise program for six weeks. On evaluation, basal plasma beta endorphin level was negatively correlated with total duration of attacks in the last month period (P = 0.01). Exercise was found to have beneficial effects on all migraine parameters (P < 0.0001) and to increase beta endorphin levels after the treadmill practice (P < 0.0001) and after the home exercise program except four patients not regularly attended to the program (P < 0.0001). Any of the changes in headache parameters was not correlated with the change in beta endorphin level. However, pre-exercise beta endorphin level was found to be negatively correlated with the changes in the number of attacks (P < 0.05) and total duration of attacks (P = 0.01) and also with the change in beta endorphin level due to exercise (P < 0.0001). As a result, this study emphasizes the beneficial effect of exercise on migrainous headache, especially in patients with lower basal beta endorphin level.
Introduction
In recent years, exercise is recommended in a multidisciplinary regimen as a part of the multimodal wellness treatment program (1–3). However, in the literature the relationship between exercise and migraine is not so clear yet. There are conflicting results about the subject (1, 2).
Some authors promote aerobic exercise as a method of migraine management (4–9). The pathways by which exercise may alter migraines have been theoretically explored. It is assumed to be related to some factors, one of which is the large amounts of encephalins and endorphins secreted during exercise (1). In one study, cerebrospinal beta endorphin levels were found to be closely correlated with the severity of disease (10).
This study aims to evaluate the role of plasma beta endorphin level in migrainous headache and to assess the effect of exercise on migrainous headache and whether this effect was due to the mediation of beta endorphin level and to find an indication for better treatment of the disease.
Methods
Forty patients with migrainous headache without aura (MWOA) who did not have any clinical affective disorder were enrolled into the study. The patients were attending the Headache Centre of Erciyes University Medicine Faculty. Their diagnosis were made according to the criteria of IHS. Six of the patients were male (15%) and 34 were female (85%); mean age was 32 ± 7 years (range 19–42 years). The patients were mostly using simple analgesics during their migraine attacks but they were not using any other drug, including those which could modify beta endorphin levels like oral contraceptives.
A detailed migraine history and information about their analgesic intake were obtained. All patients gave informed consent, indicating that they understood the study process and could stop the testing and participation at any time. The study was approved by Erciyes University Ethical Committee.
The drugs used for the prophylaxis of migraine were stopped 6 weeks before the procedure. During this period, the patients were instructed to fill a headache diary to monitor their headache activity (frequency, intensity and duration). The intensity of headache was described according to a scale with four degrees for evaluation (10); (degree 4: total disability, even with medication; degree 3: partial disability with medication; degree 2: no disability after medication, degree 1: no medication required). At first, patients exercised on the treadmill at submaximal capacity (at 80% of maximal heart rate) at the Center of Sports Medicine in Erciyes University. Female patients practised on the day at the end of their menstruation period (i.e. in follicular phase of the menstrual cycle), not coinciding with beta endorphin peak at the mid-cycle. Submaximal exercise level was obtained gradually and was continued for three minutes and thereafter gradually decreased. At the end of a total 10-min period, the exercise was stopped gradually. During exercise, heart rates and blood pressures were measured. No adverse event was observed. At least 5 ml blood was taken for each of two examinations of beta endorphin level performed prior to and within one minute after completion of the exercise.
The same patients were instructed in an aerobic exercise program at 60% of maximal heart rate lasting 40 min (10 min warming up, 20 min active exercise, 10 min resting period). They were asked to continue this exercise program at their homes three times per week for 6 weeks during their headache free periods. The patients were again instructed to complete a headache diary during their home exercise period. The patients did not change analgesic use, continuing in the same way as before the exercise program.
Blood samples were drawn for examination of beta endorphin level from all patients at the end of their exercise program. But four female patients reported that they did not practise the program regularly.
For the detection of beta endorphin concentration, venous blood samples was collected by plastic heparinized syringes, drawn into ice-cold vacutainer tubes containing a mixture of EDTA and aprotinin, and centrifuged under refrigeration, afterwards frozen on dry ice, and stored at −70°C. Plasma samples were analysed by radioimmunoassay method (Nichols Institute Diagnostics, San Juan Capistrano, USA). The lower limit for quantification was about 10 pg/ml. According to the manufacturer, there was no cross-reactivity with beta endorphin, beta melanocyte stimulating hormone or beta lipotropin at concentrations less than 100 ng/ml. The intra-assay variation coefficients were 7.1% and 12.2% at concentrations of 260 and 1000 pg/ml, respectively. The reference range was quoted as <126 pg/ml, with a mean of 29 pg/ml.
The number and total duration of attacks in the last one month period and the average intensity of pain were considered as headache parameters in the statistical assessments. Pre- and post-exercise values of headache parameters and beta endorphin levels were evaluated using the Student t-test for dependent groups or Wilcoxon rank analysis. For correlation analysis, Pearson and Spearman tests were used appopriately.
Results
On evaluation of 36 patients who practised their aerobic exercise program regularly, effects on all headache parameters by exercise were beneficial and statistically important (Table 1).
Headache parameters and beta endorphin concentrations of the patients before and after aerobic home exercise program
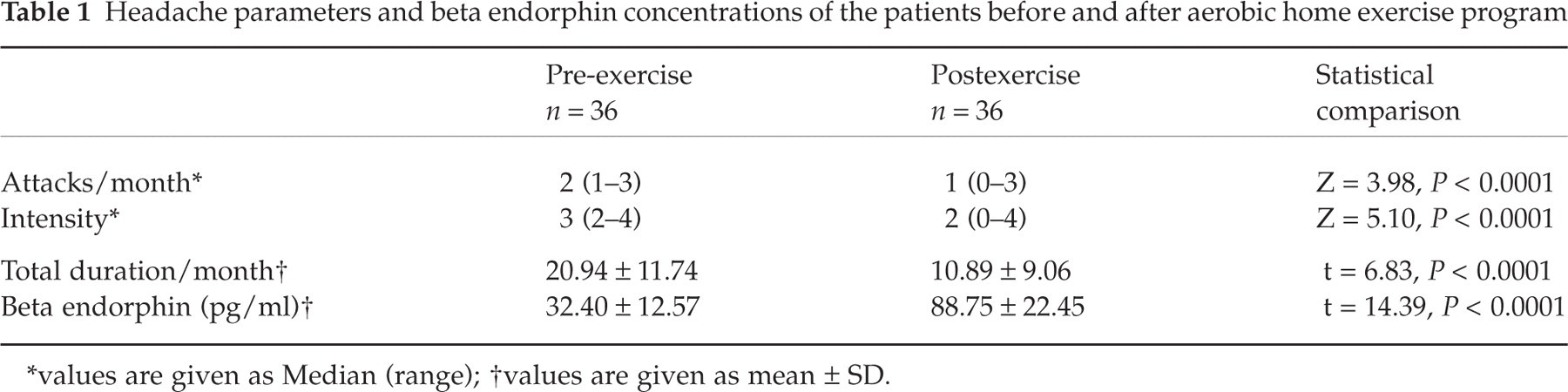
∗values are given as Median (range);
†values are given as mean ± SD.
As a result of treadmill exercise, beta endorphin levels increased in all patients. Mean beta endorphin level following the exercise was statistically higher than the level prior to the exercise (Means ± SD: 84.61 ± 38.30, 32.30 ± 11.94; t= 9.85, P < 0.0001). The level after the home exercise program also increased except in the 4 patients who had not practised the program regularly (Table 1). The increase ratios (after/before exercise trials) were shown in Table 2. There was no difference in the tendencies of the described changes between male and female patients. The levels of beta endorphin after treadmill exercise and home exercise program were found to be correlated (Pearson r = 0.47, P < 0.005).
Post/pre beta endorphin level ratios of treadmill and home, exercise trials

The ratio of increase in beta endorphin level was found to be negatively correlated to the level before home exercise program (Pearson r = 0.69, P < 0.0001). A likely negative correlation was also found between the ratio of increasement in beta endorphin level after treadmill exercise to the level before but not in a significant degree.
On correlation with the migrainous headache parameters, pre-exercise beta endorphin level was found to be negatively correlated with the total duration of attacks in the last one month period (Pearson r = 0.43, P= 0.01). Nevertheless, there was not any similar significant relation with the level following the home exercise program.
The beta endorphin increasement ratio after the home exercise program (postexercise/pre-exercise beta endorphin) and changes in migrainous headache parameters did not not correlate with each other. However pre-exercise beta endorphin level was found to be correlated with the changes in the number of attacks (Spearman r = 0.35, P < 0.05) and negatively correlated with the changes in total duration of attacks (post/pre-exercise durations) (Pearson r =–0.43, P= 0.01). Any similar relation was not found between postexercise beta endorphin level and changes in the headache parameters.
Discussion
In this study, we tried to evaluate the role of plasma beta endorphin level in migrainous headache and to assess the effect of exercise on migrainous headache and if this effect was with the mediation of beta endorphin level. As a result, basal plasma beta endorphin level appeared to be negatively correlated with the duration of attacks. Exercise was found to have beneficial effects on all migraine parameters (frequency, duration and intensity of attacks) and to increase beta endorphin level in both acute (treadmill) and chronic trial (six weeks program). Although the change in beta endorphin level with exercise (postexercise/pre-exercise beta endorphin level) was not correlated with changes in headache parameters, we obtained some data making us think that the beneficial effect of exercise on migrainous headache was with the mediation of beta endorphin level. We noticed that positive changes in headache parameters due to exercise was negatively correlated with pre-exercise beta endorphin level, which was found to be in negative correlation with change in beta endorphin level.
All the findings together support the presence of beneficial effect of exercise in migraine and emphasize the role of beta endorphin in migrainous headache. It is also interesting to see that the patients with lower basal (pre-exercise) beta endorphin level benefit more from exercise. Basal plasma beta endorphin level appeared to be a determinator of exercise induced increasement in the endorphin level and beneficial effects on headache parameters.
Beta endorphin is an endogenous opioid peptide primarily synthesized by adenohypophysis. It is derived from a prohormone, pro-opiomelenocortin (POMC) by a post-translational processing and released together with ACTH, which is also produced from the same prohormone (11, 12). Iranmanesh et al. (11) found the half life of endogenously generated beta endorphin as 22 ± 1.7 min. This is a sufficiently high biological half life unlike other endogenous opioid peptides (13). There are many studies reporting the presence of dysfunction of the endogenous opioid system in migraine. Beta endorphin levels in plasma and cerebrospinal fluid were found to decrease in migrainous subjects during headache free periods and/or during attacks (10, 14–19). In one study, cerebrospinal beta endorphin level was found to be closely correlated with the severity of disease. It was in the lowest level in the state of continuous migraine with interparoxysmal headache (10). In our study, beta endorphin level was found to be negatively correlated with total duration of attacks in one month period. This meaned the patients with lower beta endorphin level had longer duration of headache.
As a method of migraine management, exercise has been frequently discussed, but an examination of the literature provides conflicting results (1). Some authors reported exercise induced migraines or prodrome symptoms (20–22) and some called our attention to sports migraine (23–26). However, advocates of exercise in migraine management asserted that some special accompanying circumstances such as strenous exercise (27) and lack of proper warm up before exercise (28) caused these phenomena. In our study, submaximal aerobic exercise and warm up period before exercise were applied to the patients to circumvent the probable detrimental factors. In contrast to the reports of exercise precipitated migraine attacks, there are many studies in the literature reporting the beneficial effect of both single sessions (4, 6, 29) and prolonged programs of exercise (5, 7–9). However, in some studies, the beneficial effects of aerobic exercise programs on migrainous headache did not reach a statistically significant level (5). In our study, we found the effects highly significant on all parameters of migraine (frequency, duration, intensity). We performed our study on patients with MWOA despite others, on patients with migrainous headache with aura (MWA). The difference between the results in concern to their statistical significance might be due to the difference between study populations. This result brings a question in minds such as if exercise is more useful in patients with MWOA.
The relationship between the benefits of exercise and the treatment of migraine is complex (1). Exercise is known to increase plasma beta endorphin level in healthy individuals (30–35). We also observed a similar effect in patients with MWOA both after a single session (treadmill exercise) and prolonged program. We detected that the increase in beta endorphin levels after the two trials of exercise were well correlated with each other. So, the beneficial effect of exercise on our patients was most probably due to increasement in beta endorphin level. This is in concordance with the findings of decreased levels of endorphine in migraine patients (10, 14–19) and of endorphin mediated increasement in pain threshold induced by exercise (36–38). Also, we found that positive changes in headache parameters after exercise was negatively correlated with pre-exercise beta endorphin level, which was in negative correlation with changes in the beta endorphin level.
It is unlikely that a single factor is involved in exercise-induced changes in migraine (1). Besides neurochemical factors such as endorphins, increase in cardiovascular, cerebrovascular fitness and psychological states, such as relief from depression, stress and anxiety, were hypothesized as exercise-induced changes (1, 39). Additionally, exercise-induced changes to depression, anxiety and stress reactivity may also be mediated by alterations to neurotransmitters including endorphins (1). We have not made such a multifactorial evaluation. We have just tried to increase our knowledge of endogenous opiates in exercising migrainous patients and its effects.
All patients were asked to continue the exercise program for the same duration to standardize the method of the study. This could be a drawback in female patients because of the possible changes in beta endorphin level during different stages of the menstrual cycle. In literature, plasma beta endorphin levels was shown to peak at mid–cycle, with no obvious difference between follicular and the luteal phases (40, 41). We took the first blood sample from female patients at the end of their menstruation period (i.e. in follicular phase) not to coincide with the peak level. So, the second examination most probably came during the luteal phase but this is not certain, since hormonal examination for an exact timing of the menstrual cycle was not performed because of the difficulties in standardization of method. To our knowledge, in most of the studies of exercise and beta endorphin, this is a common deficiency, general trend is to describe in subjective terms the participants’ menstrual characteristics.
This study emphasizes the beneficial effect of exercise on migrainous headache, especially in patients with lower basal beta endorphin level. It has been performed on patients with MWOA. We think that a study population including also patients with MWA will provide a comparison between these two subtypes of migraine and probably will improve our understanding on the subject.
