Abstract
Stress is the leading precipitating factor for migraine attacks but the underlying mechanism is currently unknown. Nitric oxide (NO) has been implicated in migraine pathogenesis based on the ability of NO donors to induce migraine attacks. In the present study, we investigated in Wistar rats the effect of air stress on nitric oxide synthase (NOS) mRNA and protein expression in dura and pia mater using real-time polymerase chain reaction and Western blotting, respectively. Endothelial (e)NOS protein expression was significantly increased in dura and pia mater after air stress. Significantly augmented neuronal (n)NOS protein expression was detected in pia mater after air stress but not in dura mater. Inducible NOS mRNA and protein expression levels in dura and pia mater were unaffected by stress. The increased expression of eNOS in dura mater and eNOS and nNOS in pia mater seen after stress could not be antagonized by treatment with the migraine drug sumatriptan. These findings point towards the involvement of increased NO concentrations in dura and pia mater in response to air stress. However, the role of these findings in relation to migraine pathophysiology remains unclear.
Introduction
Despite stress being one of the leading precipitating factors for migraine attacks, the physiological mechanisms and biochemical pathways initiating the headaches are largely unknown (1, 2). Prospective studies, where migraineurs kept a diary, showed that 1–4 days preceding a migraine attack appear more stressful to the migraineur than other days (3–6). The free radical gas nitric oxide (NO) is believed to play a key role in the pathogenesis of migraine (7). Endogenous NO is formed from the amino acid L-arginine by nitric oxide synthases (NOS), namely endothelial NOS (eNOS), neuronal NOS (nNOS) and inducible NOS (iNOS). eNOS and nNOS are constitutively expressed, but their expression is the object of regulation at the transcriptional, translational and post-translational levels (8). Although the expression of iNOS is detected primarily during inflammatory reactions, low basal levels of constitutive iNOS mRNA and protein have been found in human tissues (9, 10).
Migraine patients have an increased sensitivity towards the exogenous NO donor glyceryl trinitrate (GTN) when compared with non-migraineurs. During intravenous infusion of low doses of GTN, more migraine patients develop headache than healthy controls and at high doses the headache experienced by migraine patients is more severe (11). Furthermore, NO is able to act as an experimental migraine trigger, as infusion of GTN leads to the development of a delayed migraine attack in eight out of 10 migraine patients at an average of 5.5 h following termination of the infusion (12).
Vessels within the meninges are among the few intracranial structures that are able to generate nociceptive signals. When large cerebral vessels and vessels in the dura and pia mater are stimulated experimentally during brain surgery, patients experience a throbbing unilateral migraine-like pain (13), indicating that these structures are involved in migraine pain. In rats, eNOS protein expression has been demonstrated in the craniovascular system, including the endothelium of dural middle meningeal artery and pial vessels (14, 15). nNOS immunoreactivity has been detected within the endothelium of rat pial vessels and in rat pial perivascular nerve fibres (15–17). In the cerebral circulation of the rat, nNOS is primarily found in nerves belonging to the parasympathetic nervous system and, to a minor degree, in trigeminal nerves (18). Under physiologically normal circumstances, iNOS is rarely detected within rat CNS (19).
In relation to stress, immobilizing rats for 6 h increases the protein expression and enzyme activity of iNOS in rat brain cortex (20). Likewise, immobilization stress (6 h) causes up-regulation of nNOS mRNA expression and enzyme activity in the pituitary and hypothalamus (21). Furthermore, rats stressed for 1 h every day for 4, 9 or 14 days have a significantly increased NOS activity and an accumulation of NO metabolites in their hippocampus after 4 and 9 days of stress (22).
The aim of the present study was to investigate the hypothesis that stress increases the formation of NO in the pain-sensitive tissues dura and pia mater by increasing the expression of NOS. We examined the expression of eNOS, nNOS and iNOS in rat dura and pia mater after non-traumatic air stress at the mRNA and protein levels using reverse transcription real-time polymerase chain reaction (PCR) and Western blotting.
Methods
Animals
Animal experiments were performed on 114 male Wistar rats weighing 200–300 g, which were purchased from Taconic M&B, Denmark. The rats had a 12-h light/dark cycle with lights on at 06.00 h and had free access to food and drinking water. The rats were allowed to acclimatize for at least 7 days prior to the experiments. The experimental protocol was approved by the Danish committee for experiments on animals.
Air stress protocol
The rats were stressed between 09.00 h and 13.00 h in a laboratory adjacent to the animal housing room and afterwards they were returned to the animal housing facility. The control rats remained in the animal housing room during the procedure. The rats were air-stressed by placing them in a box of 10 × 25 × 10 cm and exposing them to an air flow through two tubes attached to holes in the box. The rats were stressed for 1 h/day for three successive days. On the third day the animals were sacrificed during an overdose of pentobarbital 30 min (only for mRNA analysis), 1, 2, 3 and 4 h after the air stress exposure and perfused transcardially with 100 ml of a cold 0.9% NaCl solution. The dura and pia mater were dissected out and frozen in 2-methylbutane at − 40°C. The samples were placed on dry ice and later homogenized.
Air stress combined with sumatriptan treatment
Rats stressed for 1 h per day for three consecutive days received an i.p. injection of 0.5 ml containing either the migraine drug sumatriptan (300 µg/kg Imigran®; GlaxoSmithKline, Uxbridge, UK) or vehicle (0.9% NaCl) immediately after the end of air stress on the last day. All animals were killed during an overdose of pentobarbital 2 h after the end of air stress and were transcardially perfused with 100 ml of ice-cold saline. The dura and pia mater were dissected out and frozen in 2-methylbutane at − 40°C. The samples were placed on dry ice and later homogenized.
Homogenization
For real-time PCR analysis, the frozen tissue samples were homogenized by sonication in 1 ml of TRIzol (Invitrogen, Carlsbad, CA, USA) using a Vibra Cell ultrasonic processor (Sonics and Material Inc., Newtown, CT, USA) and stored at − 70°C until further use. For Western blotting, the frozen tissue samples were sonicated in 250 µl of homogenization buffer containing 10 m
Western blot analysis
The protein content of the samples was determined using a Bio-Rad DC protein assay (Bio-Rad, Hercules, CA, USA). The protein samples were diluted 1 : 1 with loading buffer and heated to 95°C for 5 min. Sample protein (12.5 µg) was loaded in each lane. Subsequently, the proteins were separated by electrophoresis on a 4–12% gradient polyacrylamide Tris-glycine gel (BioWhittaker Molecular Applications, Walkersville, ME, USA), which had a separation range from 25 to 250 kDa. Positive controls were a human endothelial cell lysate (eNOS) (BD Transduction Laboratories, San Jose, CA, USA), purified rat nNOS (Alexis Biochemicals, Lausen, Switzerland) and a liver sample from a lipopolysaccharide (LPS) (5 mg/kg i.p.; Sigma-Aldrich)-treated rat (iNOS). The separated proteins were transferred to nitrocellulose membranes (Pierce, Rockford, IL, USA) during wet transfer using a Novex XCell II blotting apparatus (Invitrogen). The blots were blocked for 1 h with a 5% non-fat dry milk solution in Tris-buffered saline with 0.05% Tween-20 (TBS–T). Blots were cut in half and the upper half was incubated overnight in TBS–T containing 3% skim milk and either a monoclonal mouse anti-eNOS antibody (1 : 1000, 610297; BD Transduction Laboratories), a monoclonal mouse anti-nNOS antibody (1 : 1000, 610309; BD Transduction Laboratories) or a polyclonal rabbit anti-iNOS antibody (1 : 1000, sc-650; Santa Cruz Biotechnology, Santa Cruz, CA, USA). The lower halves of the blots were incubated with a loading control polyclonal rabbit antibody against β-actin (1 : 1000, A5060; Sigma-Aldrich). The following day, the eNOS, nNOS and β-actin blots were incubated for 1 h with goat anti-mouse or anti-rabbit secondary antibody linked to alkaline phosphatase (1 : 15 000; Amersham Biosciences, Little Chalfont, UK). The iNOS signal was amplified using an antirabbit Vectastain® ABC alkaline phosphatase kit (avidin–biotin kit) according to the manufacturer's instructions (Vector Laboratories, Burlingame, CA, USA). An enhanced chemifluorescence system (Amersham Biosciences) was used for visualization of the protein bands, while the membranes were scanned on a Storm scanner (Amersham Biosciences). Optical density measurements were performed using the ImageQuaNT software (Amersham Biosciences). To compensate for any differences in the amount of loaded protein, the intensity of the NOS bands were divided by the intensity of the β-actin bands for each sample. The iNOS antibody was specific, as preabsorption with the immunizing peptide eliminated the iNOS band. The antibodies against eNOS, nNOS, iNOS and β-actin gave rise to bands of the expected size (Fig. 1).
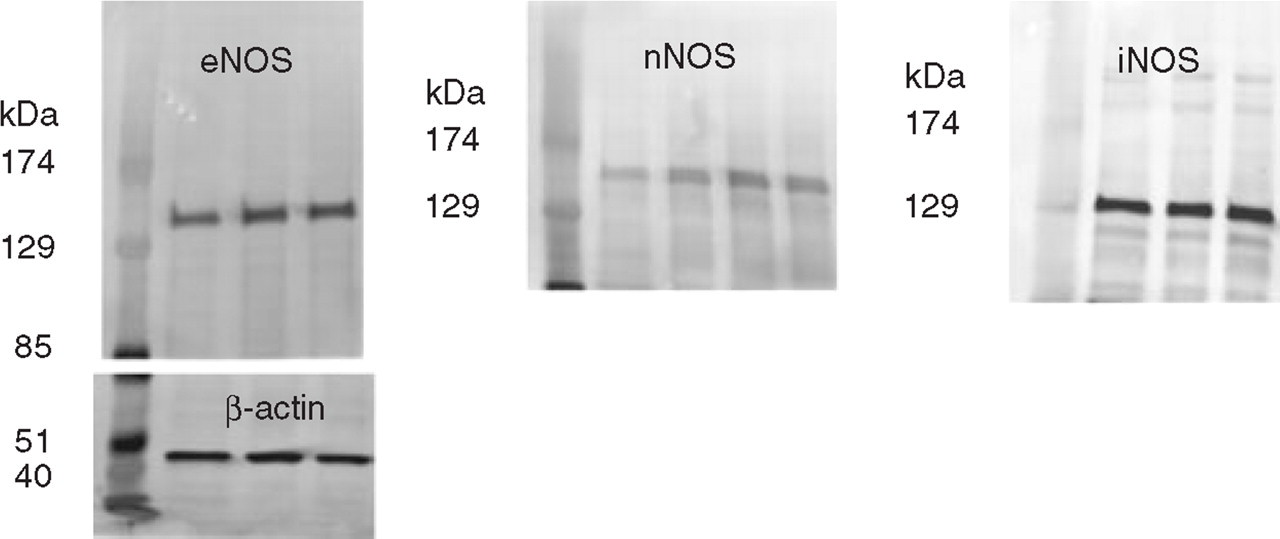
Examples of the obtained endothelial nitric oxide synthase (eNOS), neuronal (n)NOS, inducible (i)NOS and β-actin Western blots. Besides a protein marker, the depicted eNOS blot shows from left to right control, 1-h and 2-h rat dura mater samples with the β-actin blot shown below. On the nNOS blot are shown the results from the analysis of control 1, control 2, 1-h and 2-h dura mater samples, while the iNOS blot depicts pial control, 1-h and 2-h samples. The NOS monomers display sizes corresponding to approximately 135 kDa, 160 kDa and 130 kDa for eNOS, nNOS and iNOS, respectively, while β-actin has a size of approximately 45 kDa. Note that the iNOS bands appear amplified.
cDNA synthesis
Total cellular RNA was extracted using the TRIzol (Invitrogen) extraction method following the manufacturer's instructions. The resulting RNA pellet was redissolved in 20 µl diethyl-pyrocarbonate treated water and the RNA concentration and purity was measured spectrophotometrically (GeneQuant; Amersham Biosciences). The total RNA was DNase-treated with DNase I (Sigma-Aldrich) according to the manufacturer's instructions and repurified using a RNeasy Mini Kit (Qiagen Inc., Valencia, CA, USA). cDNA was synthesized from 100 ng of DNase-treated RNA in a 20-µl reaction volume using reagents from Applied Biosystems (Foster City, CA, USA). The RNA was incubated at 70°C for 10 min with oligo-dT primers (16 bases; MWG Biotech, Ebersberg, Germany) and then placed on ice. Subsequently, the reaction mixture was added and the reaction was incubated at room temperature for 10 min with a final concentration of 1 unit RNase inhibitor, 0.5 m
Real-time PCR
Real-time PCR analysis of the relative eNOS and nNOS expression in the samples from the air stress experiments was performed using a GeneAmp 5700 Sequence Detection system (PerkinElmer, Wellesley, MA, USA). The growth of DNA product in the samples was monitored by adding GeneAmp SYBR® Green to the reactions (Applied Biosystems). SYBR® Green is a fluorescent dye that binds to double-stranded DNA and the GeneAmp 5700 Sequence Detection System thereby monitors the growth of DNA in real time with an optic and imaging system. Specific primers for rat eNOS, nNOS and the housekeeping gene glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were designed as shown in Table 1. A mastermix of the following reaction components was prepared to the indicated end concentration: 15 µl H2O, 3 µl forward primer (0.6 µ

Crossing point (CP) vs. log pia mater cDNA concentration for the neuronal nitric oxide synthase (nNOS), endothelial (e)NOS and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) primer pairs performed on the GeneAmp 5700 Detection system (n = 2). The corresponding real-time PCR efficiencies were calculated according to the equation: E = 10( −1/slope).
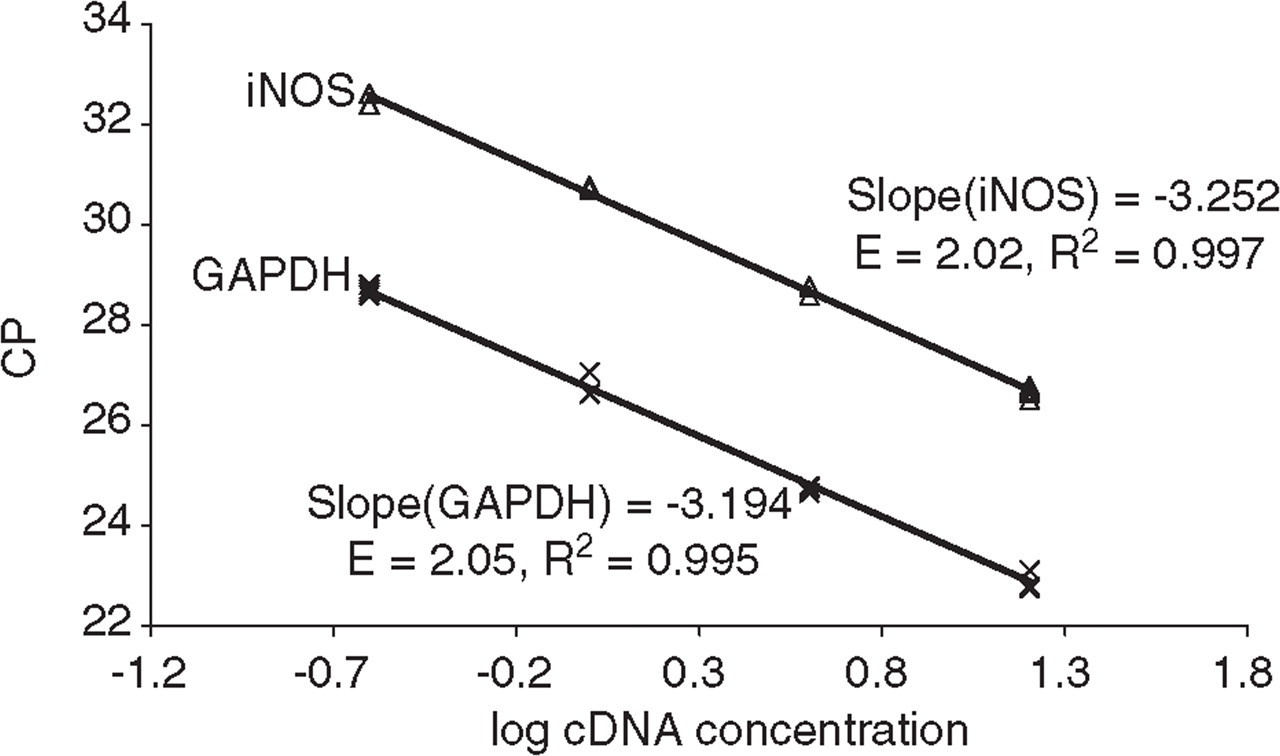
Crossing point (CP) vs. log cDNA concentration for the inducible nitric oxide synthase (iNOS) and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) primer pairs performed on the LightCycler Detection system (n = 4). The corresponding real-time PCR efficiencies were calculated according to the equation: E = 10( −1/slope).
Primers designed for detection of mRNA expression for endothelial nitric oxide synthase (eNOS), neuronal (n)NOS, inducible (i)NOS and glyceraldehyde-3-phosphate dehydrogenase (GAPDH)

Each set of forward and reverse primers were designed from GeneBank extracted sequences corresponding to the shown accession numbers (Acc. No.). The primer sequences, the amplified part of the GeneBank sequence (Seq. No.) and the size of the amplified products are shown.
Corticosterone radioimmunoassay
Twenty-four rats were randomly assigned to one of four groups: control, 1 day, 2 days and three consecutive days with 1 h air stress per day. All rats were accustomed to handling prior to the experiment. To avoid circadian variability, all blood samples were collected between 09.00 h and 10.15 h. Control rats were brought from the animal room and killed by decapitation in less than 40 s. All air stress experiments were performed in a room adjacent to the animal housing facility. After air stress exposure, the rats were decapitated immediately and trunk blood was collected in tubes containing EDTA (1 mg/ml blood) and the protease inhibitor aprotinin (Sigma-Aldrich) (0.55 TIU/ml blood). The collected blood was cold-centrifuged and the plasma samples were stored at − 70 °C until the day of analysis. Plasma corticosterone concentrations were measured in triplicates using a 125I-corticosterone radioimmunoassay kit (DPC, Los Angeles, CA, USA).
Data analysis
In order to limit the number of animals, the control and stress/vehicle rats from the sumatriptan study were added to the 2-h group in the time profiling Western blot study. Statistical analysis of the Western blot data was performed on optical density raw data using the SAS system for Windows V8 (SAS Institute, Cary, NC, USA). The comparisons between groups were performed using one-way
Results
NOS expression in dura mater after air stress
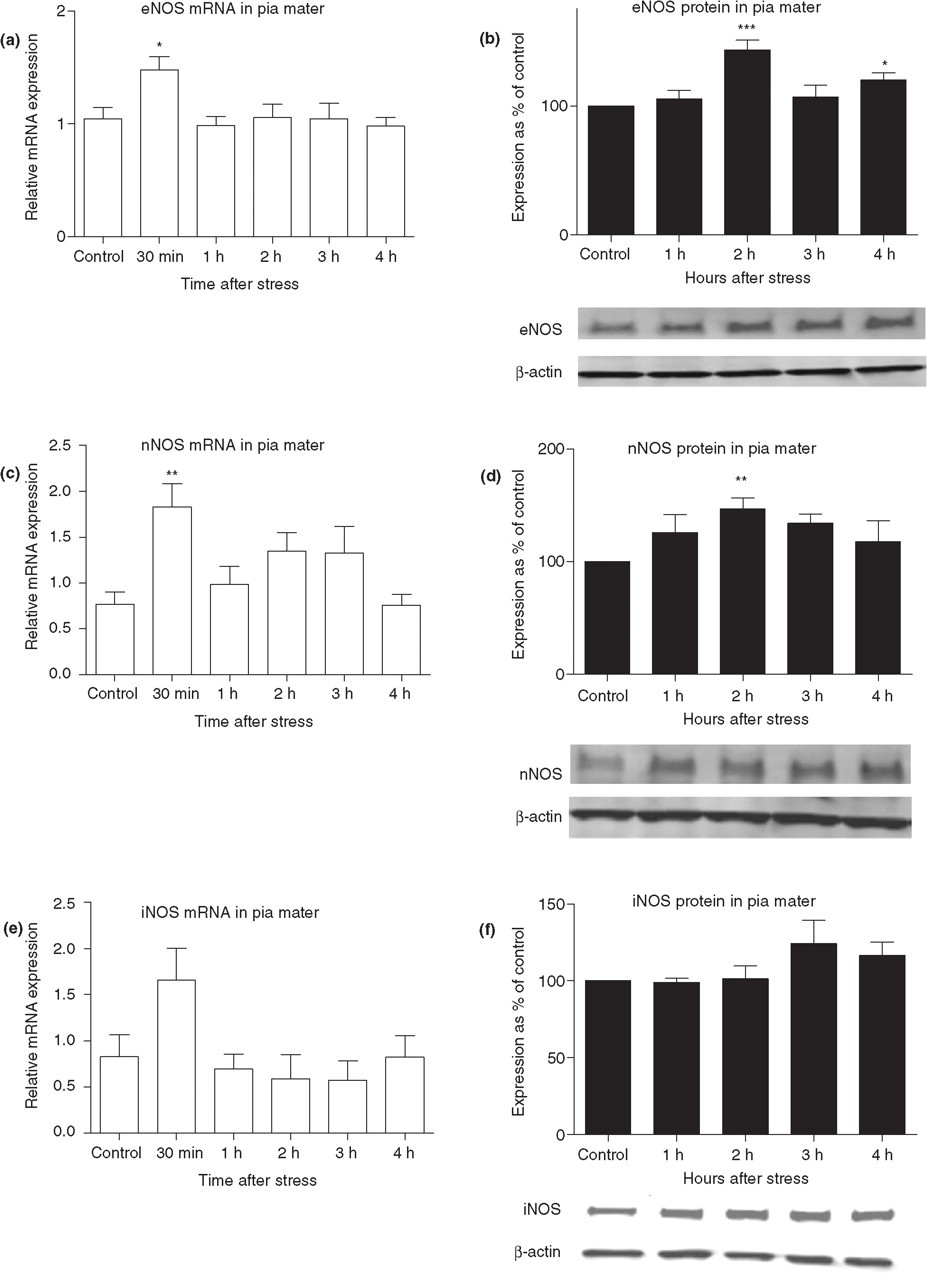
eNOS mRNA and protein expression in rat dura mater was investigated at different time points after air stress exposure. The expression of eNOS mRNA was significantly up-regulated by 1.9-fold 3 h after air stress (P < 0.05, n = 5) when compared with the control group (Fig. 4a). However, eNOS protein expression was significantly increased when compared with controls, 1 h after stress by 43 ± 20% (P < 0.05, n = 6), 2 h after stress by 58 ± 10% (P < 0.001, n = 12) and 3 h after stress by 33 ± 8% (P < 0.05, n = 6) (Fig. 4b). Neither dural nNOS mRNA nor protein was significantly altered after air stress (Fig. 4c,d). A 2.8 times increase in iNOS mRNA level was detected 30 min after the end of air stress, but this increase was found to be statistically non-significant (P = 0.07) (Fig. 4e). No iNOS protein expression could be detected using ordinary Western blot analysis, so an avidin–biotin amplification step was employed. Dural iNOS protein expression after stress did not differ significantly from the expression in control animals (Fig. 4f).

Nitric oxide synthase (NOS) mRNA and protein expression in rat dura mater at different time points after air stress. Endothelial (e)NOS (a), neuronal (n)NOS (c) and inducible (i)NOS (e) mRNA expression is presented as relative to a calibrator sample (mean ± SEM, n = 5–7). eNOS protein expression (b) (n = 6–12), nNOS protein expression (d) (n = 5–12) and iNOS protein expression (f) (n = 9 in the 2-h group and otherwise n = 3) data are presented as percentage of control animal NOS expression after being corrected for the density of the β-actin bands. This was performed for each experimental round. The NOS and corresponding β-actin bands from representative Western blots are shown. Note that an amplification step was applied in order to detect the iNOS bands. Significance of the differences at specific time points: ∗P < 0.05; ∗∗∗P < 0.001.
NOS expression in pia mater after air stress
The eNOS mRNA level in pia mater was significantly increased by 1.4-fold 30 min after air stress (P < 0.05, n = 6) (Fig. 5a). eNOS protein expression was increased by 43 ± 7% 2 h after stress when compared with controls (P < 0.001, n = 12) and by 20 ± 6% 4 h after stress (P < 0.05, n = 7) (Fig. 5b). nNOS mRNA expression was significantly increased by 2.4-fold (P < 0.01, n = 6) 30 min after air stress (Fig. 5c), while protein expression was increased by 34 ± 8% 2 h after stress (P < 0.01, n = 15) (Fig. 5d). iNOS mRNA expression 30 min after the end of air stress was twice the average level detected in the control animals, but the increase was found to be statistically non-significant (P = 0.08, n = 7) (Fig. 5e). The iNOS proteins were not detectable using ordinary Western blotting, so an avidin–biotin amplification step was employed. Pial iNOS protein expression did not increase after stress (Fig. 5f).

Nitric oxide synthase (NOS) mRNA and protein expression in pia mater at different time points after air stress. Endothelial (e)NOS (a), neuronal (n)NOS (c) and inducible (i)NOS (e) mRNA expression is presented as relative to a calibrator sample (n = 5–7). eNOS protein expression (b) [n = 3 (3 h) to 12 (2 h)], nNOS protein expression (d) (n = 7–15) and iNOS protein expression (f) (n = 3–7) data are presented as percentage of control animal NOS expression after being corrected for the density of the β-actin bands. This was performed for each experimental round. The NOS and corresponding β-actin bands from representative Western blots are shown. Note that an amplification step was applied in order to detect the iNOS bands. Data are presented as mean ± SEM. Significance of the differences at specific time points: ∗P < 0.05; ∗∗P < 0.01; ∗∗∗P < 0.001.
Effects of air stress on plasma corticosterone concentrations
In order to verify that the rats were stressed from the air stress treatment, the plasma corticosterone levels elicited in control rats and rats exposed to 1 h/day of air stress for either 1, 2 or 3 days were determined (Fig. 6). The basal median corticosterone level measured in six control rats was 35 ng per ml plasma. A significant difference was revealed between the corticosterone level in control animals and animals exposed to 1 day of air stress, a median value of 506 ng per ml plasma (P < 0.01, n = 6). After 2 and 3 days of air stress median corticosterone levels were increased to 296 (P < 0.05, n = 6) and 390 ng per ml plasma (P < 0.05, n = 6), respectively. The results indicate that a gradual adaptation to the stress exposure had occurred. Interestingly, there may be low and high responders to stress exposure as the corticosterone measurements seemed to be divided into two groups.
Plasma corticosterone levels in plasma of control rats and rats exposed to 1 h/day of air stress for one, two or three consecutive days, n = 6. A line is placed at the median (∗∗∗P < 0.001; ∗∗P < 0.01; ∗P < 0.05).
NOS protein expression in dura and pia mater after air stress and sumatriptan treatment
It was investigated whether treatment with the migraine drug sumatriptan was able to antagonize the increased protein expression of eNOS and nNOS, seen 2 h after air stress exposure. In rat dura mater, no significant difference was detected between eNOS expression in stressed rats subjected to either vehicle (saline) or sumatriptan treatment. When compared with the controls, dural eNOS expression was increased by 64 ± 14% after air stress and vehicle (P < 0.01, n = 8) and 52 ± 14% after air stress and sumatriptan treatment (P < 0.05, n = 8). No significant difference in dural nNOS expression could be detected between the control group and the different treatments. Moreover, air stress had no significant impact on dural iNOS expression and treatment with sumatriptan did not cause any change in basal iNOS expression. No significant difference was found between pial eNOS expression in stressed rats receiving vehicle and stressed rats administered sumatriptan. The detected eNOS expression in pia mater was significantly increased, in rats receiving both vehicle (53 ± 12%, P < 0.01, n = 6) and sumatriptan treatment (42 ± 12%, P < 0.05, n = 6), when compared with expression in control animals. Similarly, no significant difference was found between pial nNOS expression in stressed rats administered vehicle and sumatriptan, respectively. When compared with the control group, expression of nNOS in pia mater was significantly increased 2 h after air stress in rats receiving vehicle (51 ± 14%, P < 0.05, n = 8) but not in rats receiving sumatriptan treatment (44 ± 14%, P > 0.05, n = 8). Air stress had no significant effect on iNOS protein expression in pia mater and treatment with sumatriptan did not alter iNOS expression.
Discussion
A wide range of naturally occurring and experimental migraine-triggering factors are known, but the underlying mechanisms for the initiation of a migraine attack remain unclear. That NO may be involved in the initiation of migraine attacks is emphasized by the ability of NO donors to induce a migraine attack in migraineurs occurring on average 5.5 h after the infusion (12). Applying this migraine model to the rat, a dural neurogenic inflammation including macrophagal iNOS protein expression 4, 6 and 10 h after the infusion was detected (24). Although stress is the most common trigger of migraine attacks, only limited insight into the link between stress and migraine has been acheived (1, 2). Consequently, the aim of the present study was to examine the relation between a mild non-traumatic stress and NOS expression in the migraine-relevant tissues dura mater and pia mater.
Contrary to our expectations, rat dural iNOS expression was not significantly altered by stress. Increased iNOS expression has been reported in rat cortex after 6 h of immobilization stress (20). Furthermore, during acute immobilization stress (30 min) dural mast cells are activated and release proinflammatory mediators (25). Equally, pial iNOS expression was not affected by air stress, implying that iNOS is not involved in the stress response in the investigated tissues.
Still, eNOS protein expression was significantly increased in rat dura mater (including the middle meningeal artery and its branches) 1, 2 and 3 h after air stress exposure, while the corresponding mRNA expression was not significantly increased until 3 h after air stress. It is possible that the transiently increased eNOS protein expression could be due to a translational regulation mechanism or a decrease in eNOS protein breakdown. A translational regulation of eNOS has been described previously, where a 70% increase in protein expression was found in endothelial cells 2 h after hypoxia, although the detected eNOS mRNA level remained unaltered (26). A decrease in eNOS protein degradation has been demonstrated in endothelial cells, where high-density lipoprotein increased the amount of eNOS protein by 250% solely by increasing the protein half-life from approximately 81 to 270 min (27). In rat pia mater, eNOS mRNA expression was significantly increased 30 min after air stress, followed by a significantly increased protein expression 2 and 4 h after stress. That the human organism's response to stress involves eNOS is demonstrated in a human study, where release of NO was found to be involved in the forearm vasodilator response to mental stress (28).
Air stress treatment had no significant effect on dural nNOS mRNA and protein expression. In rat pia mater, however, nNOS mRNA was significantly increased 30 min after air stress which preceded a significant increase in nNOS protein detected 2 h after stress. It has previously been demonstrated that nNOS expression can be affected by stress as an increased nNOS protein expression was detected in the rat pituitary and hypothalamus after 6 h of immobilization stress (21).
Measurements of plasma corticosterone levels showed a significant increase in air-stressed rats, indicating that the animals reacted to the treatment with an adequate stress response. A slight habituation to air stress occurred as the corticosterone response declined after the first day. Rats are known to adapt to repeated stress exposure, resulting in a decrease in measured plasma corticosterone levels after the first day (29). Since the corticosterone level was highest after 1 day of stress, it may seem to be the best day for examining NOS expression. This possibility was not tested in the present study. We tried to model the human situation, where stress has been reported to occur after one to four repeated days with increased incidence of daily hassles and, based on this information, 3 days of repeated mild stress seemed an appropriate animal stress model (3–6).
Interestingly, there seemed to be low responders and high responders to air stress. An individual difference between rats in the response to stress has been described previously (29). Male Sprague-Dawley rats (an outbred strain like the Wistar rats used in the present study) displayed individual differences towards exposure to immobilization stress (1 h). The increase in plasma corticotropin (ACTH) and corticosterone was significantly higher in the high-response group immediately after and also post stress when compared with the low-response group (29). It may be speculated whether the occurrence of low and high responders explains the somewhat high variation in NOS expression found after stress in the present study.
In our hands, treatment with the 5-HT1B/D agonist sumatriptan did not attenuate the increased eNOS and nNOS protein expression in stressed rats. Triptans have been demonstrated to act at several migraine-relevant sites, but which of the several mechanisms is responsible for aborting migraine pain remains uncertain. Experiments have demonstrated that triptans act to constrict meningeal blood vessels via activation of 5-HT1B receptors located on smooth muscle cells (30), inhibit the release of calcitonin gene-related peptide (CGRP) from trigeminovascular nerves (31), and in case of blood–brain barrier disruption, triptans could inhibit nociceptive transmission within the brain stem trigeminal nucleus caudalis (TNC) by preventing sensory neurotransmitter release (32, 33).
The sumatriptan dose used in the current study was 0.3 mg/kg i.p., a dose able completely to block CGRP release as well as dural neurogenic inflammation in rats (34). In a recent study, pretreatment with 0.6 mg/kg s.c. sumatriptan did not attenuate the increased nNOS expression seen in TNC 4 h after administration of high-dose GTN (10 mg/kg s.c.) (35). In a similar study, however, sumatriptan pretreatment (0.4 mg/kg i.v.) succeeded in attenuating the increased nNOS expression seen in the perivascular nerve fibres, trigeminal ganglia and TNC 1 h after infusion of high-dose GTN (10 mg/kg i.v.) to rats (36). These contradictory results indicate that timing and the manner of drug administration may influence the findings. In the present study, sumatriptan was administered after the stress exposure, which was based on acute migraine treatment, where sumatriptan has little efficacy if it is taken at the time of the aura, i.e. before the headache phase (37). Furthermore, in man, s.c. sumatriptan is effective within 1 h after administration (38, 39). Therefore, the 2 h between sumatriptan administration and sacrifice of the rats should be adequate for the drug to affect the increase in NOS expression. The lack of effect on the stress-induced increase in eNOS and nNOS expression indicates that the increase in expression of eNOS and nNOS is located upstream to the site of action of sumatriptan. Another possibility is that the increased expression is not directly linked to the pathway leading to migraine pain.
It has been reported that migraineurs have an increased basal NO production, as evidenced by the significantly increased nitrite and nitrate levels in the plasma of migraine patients outside of attacks (40, 41). It can be speculated whether the NO-cGMP hyperactivity predisposes migraineurs to attacks and thus the increased NOS expression seen as a response to stress may act to aggravate the abnormality. Migraineurs are known to be genetically predisposed towards migraine, meaning that their condition unfortunately cannot be mimicked in either rats or healthy volunteers. It is to be hoped that in future, the nature of these predisposing factors will be revealed and thus the link between these factors and migraine-triggering stimuli such as stress will be clarified.
In conclusion, we have for the first time shown that air stress causes an increase in eNOS expression in rat dura and pia mater. Furthermore, an increased level of nNOS was found in rat pia mater after air stress. In contrast, no change in iNOS expression could be observed. Treatment with the migraine drug sumatriptan had no influence on the stress-induced increase in NOS expression. Thus, the role of the present findings in relation to migraine pathophysiology remains unclear.
Footnotes
Acknowledgements
The authors are deeply indebted to Kenneth Beri Plough MSc for excellent technical assistance and to statistician Lene Theil Skovgaard for her assistance with the statistical analysis. This work is supported by the Danish Medical Research Council (grant no. 9902883), the Lundbeck Foundation and the Hede Nielsen Foundation.
