Abstract
Electrophysiological methods may help to unravel some of the pathophysiological mechanisms of migraine. Lack of habituation is the principal and most reproducible interictal abnormality in sensory processing in migraineurs. It is found in evoked potential (EP) studies for every stimulation modality including nociceptive stimuli, and it is likely to be responsible for the increased intensity dependence of EP. We have hypothesized that deficient EP habituation in migraine could be due to a reduced preactivation level of sensory cortices because of hypofunctioning subcortico-cortical aminergic pathways. This is not in keeping with simple hyperexcitability of the cortex, which has been suggested by some, but not all, studies of transcranial magnetic stimulation (TMS). A recent study of the effects of repetitive TMS on visual EP strongly supports the hypothesis that migraine is characterized by interictal cortical hypoexcitability.
With regard to pain mechanisms in migraine, electrophysiological studies of trigeminal pathways using nociceptive blink and corneal reflexes have confirmed that sensitization of central trigeminal nociceptors occurs during migraine attacks.
Keywords
Introduction
The common migraine phenotypes are heterogeneous and appear to be complex genetic disorders in which susceptibility genes and environmental factors interact (1–5). Both neuronal and vascular components are relevant in migraine pathophysiology and probably inter-related (6–9). The neuronal structures involved are the cerebral cortex, the brainstem (periaqueductal grey matter, aminergic nuclei), and both peripheral and central components of the trigeminovascular system. The sequence of activation and the relative role of these structures are still controversial.
Several psycho-physical studies of the visual system tend to support this hypothesis: more intense illusions to grating patterns (10), faster low-level performance on psychophysical visual tests (11) and the known clinical hypersensitivity to environmental light stimuli (12). From these findings, it was proposed that there could be a generalized interictal hyperexcitability of the cerebral cortex in migraine, more pronounced in visual areas because of its neuronal density (13), and possibly favouring the occurrence of spreading depression that is thought to be responsible for the migrainous aura (14). By contrast, other psycho-physical tests of the visual system were thought to reflect hypoexcitability of the visual cortex (15).
The methods of electroneurophysiology are particularly appropriate for the study of migraine pathophysiology because they are atraumatic and able to detect functional abnormalities. During the past decade, numerous studies have yielded interesting, but partly controversial results. It seems timely to review those that may disclose dysfunctions of the trigeminal system and/or phenotypic markers of the interictal vulnerability to migraine: evoked potentials, electromyography and transcranial magnetic stimulation.
Evoked potentials
Evoked and event-related potentials have been extensively studied in migraine during the past 30 years. Almost every modality of stimulation has been used, but particularly visual and auditory stimuli. Both modality-specific evoked responses and ‘cognitive’ potentials such as CNV and P300 have been examined. Various interictal and ictal abnormalities have been reported, but some results appear contradictory. Besides patient- and disease-related variables, methodological differences may in part account for these contradictions.
Visual evoked potentials
In all early studies of flash-evoked visual potentials (16–18), except one (19), higher amplitudes have been found in migraineurs than in controls. Early visual evoked potential (VEP) components have been described as reduced on the side opposite to the aura (20).
Pattern-reversed-visual evoked potential (PR-VEP) studies in migraine show more heterogeneous results (Table 1). On the one hand, most studies have (n = 12) reported normal amplitudes (3,5,7,18,21–27). On the other hand, increased amplitudes have been found by several investigators between attacks (8,21,28–33) or in temporal proximity to an attack (34), and decreased amplitudes by some (4, 35). Similarly, PR-VEP latencies were increased in some studies (28,34,36,37), but decreased in others (2, 29).
Pattern reversal VEP studies

MO, migraine withou aura; MA, migraine with aura; ME, migraine aura without headache; ETTH, episodic; CTTH, chronic tension-type headache.
A certain degree of asymmetry in VEP amplitudes (4,32,38,39) and latencies (2, 38) has also been described, often on the symptomatic side. PR-VEP amplitude may decrease with duration of the disease (8), but this was not confirmed in another study (9).
Although patients affected by migraine with aura had globally similar results as migraineurs without aura (5,8,18,22,24,27,29,34,36), both reduction (4) and increase (32) of the P100 amplitude have been described on the side of the visual aura.
The difference in results can be explained in part by methodological differences; spatial frequency (40), temporal frequency or number of averages varied between studies (see Table 1). Some discrepancies may be patient-related. Diagnostic groups were less well-defined in studies performed before the Headache Classification of the International Headache Society (41) became available in 1988. More importantly, evoked cortical responses undergo profound modifications up to 24 h before and during migraine attacks (see below). While in most studies there was a sufficient interval between the recordings and the last attack, controlling for the occurrence of an attack within 24 h after the recordings was rare (7, 27). Taken together, classical studies of averaged PR-VEP did not show any consistent abnormality for the CNS pathophysiology of migraine.
Spectral fast Fourier analyses of the electroencephalograph (EEG) during steady-state flash stimulation (SS-VER) have shown concordantly that the fundamental F1 component (42–45) and the alpha band (46) are more powerful in migraineurs than in healthy volunteers. As with simple flash VEP, interhemispheric asymmetries have also been found (47). During the attack, using steady-state VEP, a reduced amplitude of the F1 component has been found (48).
These results are in line with the pioneering finding of increased photic driving of the EEG (49), and suggest that sustained visual stimulations induces more synchronous net activity in the visual cortex in migraineurs between attacks.
Compared with all other electrophysiological data obtained in migraine, those published on habituation of evoked potentials are more concordant. The first detailed studies of habituation were performed using VEP and showed that amplitudes of the N1–P1 and P1–N2 components (3,25,26) (Table 2) remained unchanged, or increased during repetitive stimulation in migraineurs between attacks, while they decreased in healthy volunteers. Interictal lack of habituation in migraine was not confirmed as such in two studies. In the first one (40), only a trend for a N1–P1 habituation deficit was found in migraine with aura for the lower spatial frequency; technical differences (e.g. a higher pattern contrast) may explain the difference. In the second study (27), the majority of the patients had an attack within 24 h following the recording, and while habituation was clearly present in healthy volunteers, this was not the case in patients between attacks. Red light, supposed to represent the most effective stimulus for the visual cortex, induced VEP potentiation in healthy subjects, but not in migraineurs (6), indicating that increased excitation is not responsible for the reversal of the normal habituation pattern. The degree of VEP habituation was negatively correlated with amplitude in the first block of averaged responses (7), which suggests that the lack of habituation in migraine may be due rather to a decreased cortical preactivation level. The habituation deficit of VEP was very similar in related parent–child pairs of migraineurs, but not in unrelated pairs (50), which demonstrated its familial, most probably genetic, character. The interictal lack of habituation of PR-VEP normalizes during the attack (51).
Habituation of visual evoked cortical responses

MO, migraine withou aura; MA, migraine with aura; ME, migraine aura without headache; ETTH, episodic; CTTH, chronic tension-type headache.
Taken together, these results indicate that lack of habituation of VEP is an interictal endophenotypic marker in many migraineurs and that it characterizes the information processing all along the visual pathways from the early to the later stages.
Auditory evoked potentials
The studies of short latency, i.e. brainstem auditory evoked responses (BAER) provide varying results in migraine. Many of them are normal interictally (27,38,52–54), but in some studies, increased latencies (especially for wave V) (55, 56) have been found, particularly during the attack (52, 53). Interaural asymmetries of cortical responses or BAER have been mentioned by some (55), especially in migraine with aura (57), as well as a negative correlation between low sound (55dB) discomfort threshold and wave IV–V amplitude (27).
Conversely, the few studies of cortical long-latency auditory evoked potentials are concordant in showing no significant difference between migraineurs and controls with regard to N1, P2 and N2 component latency or amplitude (27, 56).
Only two studies have explored habituation of cortical auditory evoked potentials. The first reported a potentiation of N1–P2 amplitude at high, but not at low stimulus intensities, contrasting with habituation in healthy volunteers (58). This was not confirmed in another report (27), possibly because of methodological differences. In a recent study presented in abstract form (59), we measured simultaneously the intensity dependence of auditory N1–P2 and habituation for each stimulation intensity. We replicated our finding of a greater potentiation for high- compared with low-intensity stimulations in migraineurs, as opposed to habituation or absence of amplitude change for all stimulation intensities in controls.
The only available study of gating of the auditory P50 response (60) showed a marked reduction of gating in patients compared with healthy volunteers, which suggests that the lack of habituation in migraine might result in part from a precortical dysfunction. Contrary to schizophrenic patients in whom deficient P50 gating is well-documented and associated with a low amplitude of the first response, the control P50 response was of normal amplitude in migraineurs.
The finding of a pronounced intensity dependence of auditory evoked potentials (IDAP) in migraine (58) raised interest, because intensity dependence is thought to be inversely related to central serotonin neurotransmission in humans (61–63) and in animal models (64, 65). Increased IDAP is thus another example of abnormal information processing in interictal migraine, with a well-investigated biological background. Further evidence for IDAP as a surrogate marker for central serotonergic neurotransmission has come from a study (66) showing that dexfenfluramine, a drug that increases serotonergic activity, decreases IDAP, while zolmitriptan, a 5-HT-1B/D receptor agonist that is able to decrease brain serotonin via presynaptic inhibition of its release, increases IDAP. Interestingly, increased IDAP found interictally normalizes during the attack (51). IDAP abnormalities that were correlated with personality profiles were found to be associated with lower serotonergic transmission in migraine, but not in post-traumatic headache (26).
Two independent studies (50, 67) found evidence for a familial influence on IDAP in migraineurs, pointing towards a genetic background. However, a direct genetic link is still to be proven and further studies on the extent of familial influences are missing.
Interestingly, when investigated in the same migraine patients, degrees of amplitude–stimulus function slopes reflecting IDAP and PR-VEPs habituation deficits are not significantly correlated (7), although predictions of a neuronal network model would suggest a parallel modification by modulating the efficacy of excitatory or inhibitory synaptic transmission (68).
To summarize, increased IDAP in migraine may chiefly be another consequence of lack of habituation of cortical responses at high-intensity stimulations. In spite of its well-established link to serotonergic transmission, IDAP cannot be used for diagnostic purposes because of its limited repeatability in both pathophysiological (69) and in pharmacological studies (70).
CNV and P300
Contingent negative variation (CNV), a slow negative cortical potential appearing during a reaction time task with a warning and an imperative stimulus, is thought to reflect certain aspects of cortical information processing (71) and to characterize the cortical activation level (72). Its early component is considered to indicate the level of expectation and to be modulated by noradrenergic systems. Its late component is thought to be related to motor readiness and to be under dopaminergic control (73).
Consistent studies from different laboratories have shown that CNV amplitude is increased in migraineurs between attacks, mainly in those suffering from migraine without aura (74–78) (Table 3). This increase is more pronounced for the early component in migraineurs compared with healthy volunteers or tension-type headache patients (78), which can be interpreted as disturbed attention and possibly reflects an overweight of excitatory vs. inhibitory processes. Taking age as a variable into account, Kropp et al. (79) showed a decrease in early CNV amplitude with ageing in healthy volunteers but not in migraineurs, which they interpreted as a disturbance of cerebral maturation. Disease duration also had an effect on CNV abnormalities in migraineurs (80). A strong familial influence on CNV parameters was reported by Siniatchkin et al., who found abnormalities not only in migraineurs but also in healthy subjects with a positive family history for migraine (81).
Contingent Negative Variation (CNV)
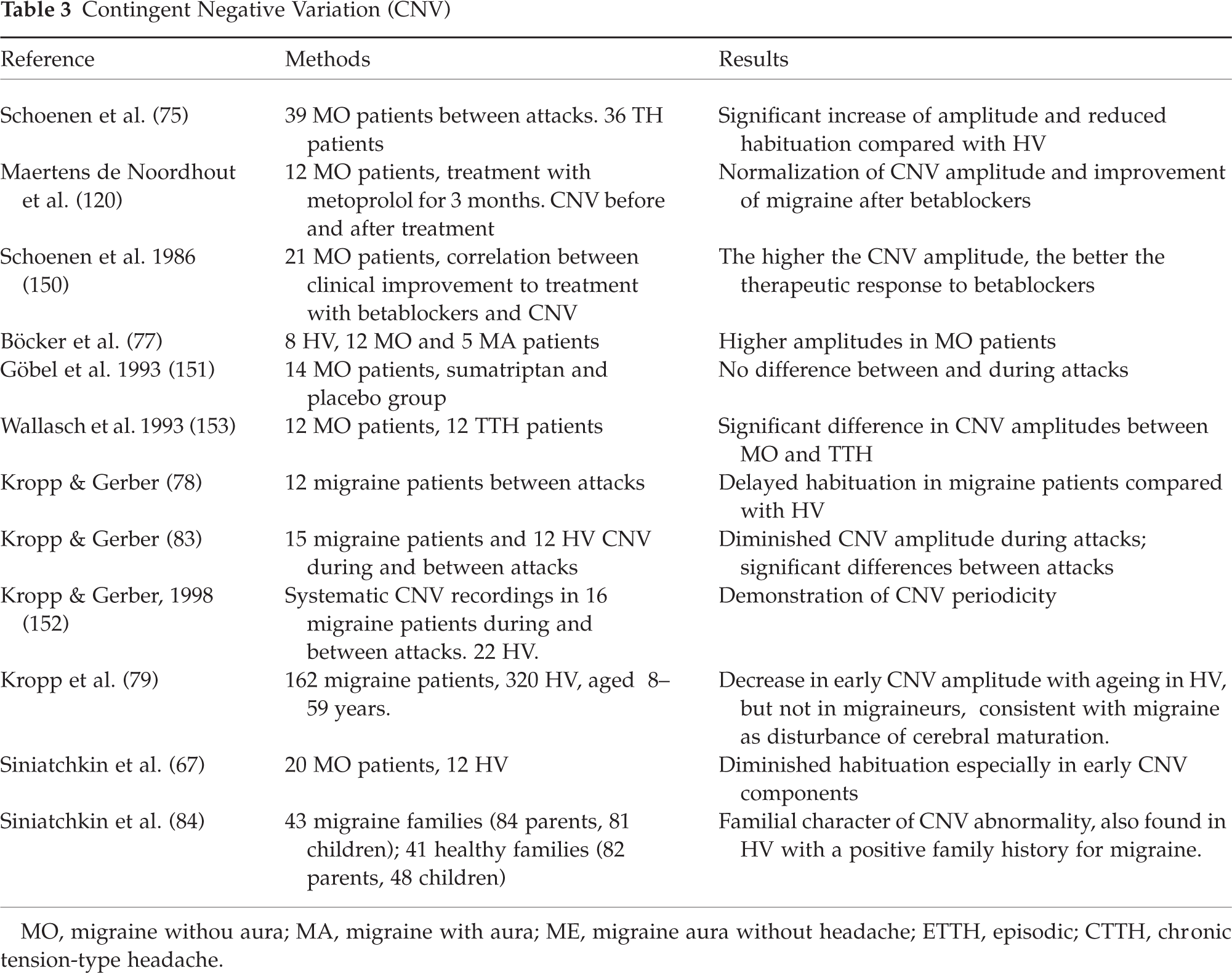
MO, migraine withou aura; MA, migraine with aura; ME, migraine aura without headache; ETTH, episodic; CTTH, chronic tension-type headache.
Like visual and auditory cortical evoked potentials (vide supra), CNV habituation is reduced between attacks (78,82,83), especially for the early component (78, 84). The lack of CNV habituation in migraineurs does not depend on age, whereas the ‘normal habituating behaviour’ of CNV in healthy volunteers is reached only in adulthood, suggesting that the electrophysiological abnormality in migraine could be the functional counterpart of a disturbance of cerebral maturation (79).
The P300 component of the classical ‘oddball paradigm’ gives conflicting results. Apart from one study (85), reduced P300 amplitude and a prolonged latency have been found when an auditory stimulus is used (56, 86). In a paradigm using visual stimuli, however, there were no differences in P300 amplitude between groups, but habituation, as assessed by the increase of latency during trial repetition, was significantly reduced in migraineurs (87–89).
The P3a component in the passive ‘oddball’ paradigm reflects automatic processing of a ‘novel’ stimulus. In one study, its normal habituation pattern was replaced by potentiation in migraine (90).
Discussion
The majority of evoked and event-related potentials studies in migraine have shown two abnormalities: increased amplitudes of averages of large numbers of trials and lack of habituation in successive trial blocks. At first sight, increased amplitudes of cortical evoked responses would favour the hypothesis of cortical hyperexcitability in migraine between attacks (13). One must keep in mind, however, that evoked responses are averaged over a large number of stimulations and that for low numbers of trials amplitudes were lower, not higher, than in healthy volunteers. The suggestion has been made therefore that the increase of EP amplitude found in some studies was not due to cortical hyperexcitability, but merely to the lack of habituation of the responses during sustained stimulation (3). In the strict physiological sense, hyperexcitability indeed indicates that the threshold to obtain a response is decreased and/or that a greater response is induced by a given suprathreshold stimulus.
Subsequently, lack of habituation is the most consistent abnormality found in migraineurs. Although habituation is a complex neurobiological phenomenon, it might crucially depend, for cortical evoked activation, on the preactivation excitability level. According to the ‘ceiling theory’ (91), most often applied to explain the occurrence of an ‘augmenting’ or ‘reducing’ response to increasing stimulation intensities, a low preactivation level of sensory cortices allows a wider range of suprathreshold activation before reaching the ‘ceiling’ and initiating a ‘reducing’ response, i.e. habituation. This theory, applied to EP findings in migraineurs, would explain both the low first-block amplitude for most EP and the lack of habituation on trial repetition. There is evidence that the preactivation level of cortical excitability depends on the so-called ‘state-setting, chemically addressed connections’ that originate in the brainstem and involve serotonin and noradrenaline as transmitters (61, 92). Low interictal activity of these systems, expecially of the raphe–cortical serotoninergic pathway, could indeed be responsible for the observed electrophysiological abnormalities in migraineurs (93). If this hypothesis is correct, one would expect that decreasing cortical activation would produce lack of habituation in healthy volunteers, and vice versa, that increasing activation in migraineurs would normalize their habituation pattern. This was indeed recently demonstrated in our laboratory by using repetitive transcranial magnetic stimulation (rTMS); high-frequency rTMS of the occipital cortex, known to activate the underlying cortex, was followed by a normalization of VEP habituation in migraineurs (Fig. 1) while low-frequency rTMS, which has an inhibitory effect, induced a VEP habituation deficit in normal controls (Fig. 2; 94).

Healthy volunteers (n = 24): mean PR-VEP N1–P1 amplitudes (symbols) in six successive blocks of 100 averaged responses and corresponding linear regression slopes (lines) before (dashed lines) and after (continuous lines). 1 Hz or 10 Hz rTMS. Note that 1 Hz rTMS, which inhibits the underlying cortex, induces dishabituation, while 10 Hz rTMS, thought to activate the cortex, has little effect (with permission from Bohotin et al. Brain 2002, 125:1–11.)
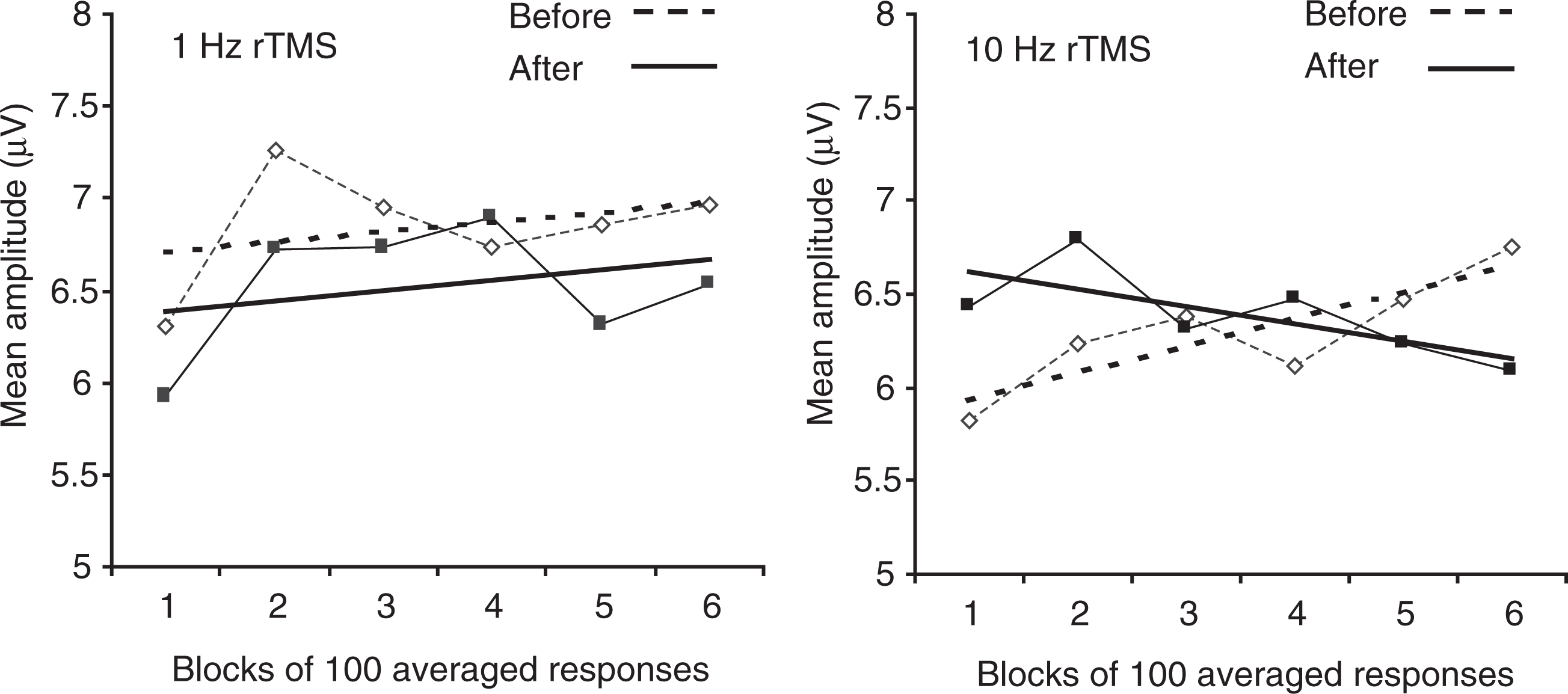
Migraine without aura patients (n = 16): mean PR-VEP N1–P1 amplitudes (symbols) in six successive blocks of 100 averaged responses and corresponding linear regression slopes (lines) before (dashed lines) and after (continuous lines). 1 Hz or 10 Hz rTMS. Note that 1Hz rTMS has little effect while 10 Hz rTMS induces normal habituation (with permission from Bohotin et al. Brain 2002, 125:1–11.)
The precise relationship between interictal abnormal cortical information processing and migraine pathogenesis remains to be determined. The possibility that dysfunctional monoaminergic nuclei in the brain stem may play a causative role in migraine attacks (95), and may be responsible independently for interictal lack of habituation of evoked potentials as an epiphenomenon, cannot be excluded. If one keeps in mind, however, that cortical habituation is also a protective mechanism against overstimulation (96) and that the mitochondrial phosphorylation potential as studied by MR spectroscopy is reduced interictally in the brain of migraineurs (97–99), lack of habituation may favour, under certain circumstances, a metabolic dysequilibrium, which would lead to activation of the major pain-signalling system of the brain, the trigeminovascular system (100). Preliminary results using functional MRS during prolonged visual stimulation suggest that there are at least two clinically and metabolically different subgroups of migraine with aura patients: those in whom the baseline lactate level in the occipital cortex is normal, but increases excessively during stimulation and those with increased lactate levels at baseline that, as in healthy subjects, are not modified by the visual stimulation (101). The relationship between function and metabolism is thus likely to be complex in migraine.
In the headache-free interval, migraineurs present subtle cognitive dysfunctions that may contribute to the burden of the disorder. For instance, memory defects have been found, and are thought to be related to strategically and organizationally defective aspects of learning (102). Whether these learning deficits are related to the habituation deficit in cortical information processing shown with electrophysiological methods remains to be demonstrated. Such a relationship would not be surprising, as the phenomenon of habituation is considered to play a pivotal role in learning processes (103).
The ictal normalization of evoked potential amplitudes and habituation suggests that there is, in close temporal proximity to the migraine attack, an increase in cortical preactivation levels, probably due to enhanced activity in raphe–cortical serotonergic pathways. Interestingly, the ictal normalization of visual-evoked P3 habituation is accompanied by a decrease in platelet serotonin content (89) and two PET studies have shown brainstem activation during the migraine attack (95–104).
According to the biobehavioural theory of migraine (105), this ictal normalization of electrophysiological findings could be part of a homeostatic process.
Electromyographic techniques
At first sight, electromyography is not supposed to be of help in investigations on the pathophysiology of migraine; nevertheless, some interesting results have arisen from the application of specific EMG techniques.
Trigeminal networks
Among the first EMG methods used in migraine is the blink reflex, which is a useful tool to investigate the trigeminal system by stimulation of the ophthalmic division of the trigeminal nerve. It provides contrasting results. Some authors found a prolonged latency of the R2 component (106), which was interpreted as a marker of brainstem dysfunction; other studies did not confirm this finding (107, 108). The R3 component shows a reduced latency interictally (109) and an increased area on the pain side during migraine attack, which is reversed by zolmitriptan (110). Unfortunately, because of the uncertainty about the anatomical and functional organization of this component, these findings cannot be easily interpreted. Recently, an increased R2 area was found in migraineurs on the side of the headache and especially during the attack, with a new device allowing a more specific stimulation of Aδ and C fibres (111).
Another method to study the blink reflex is electrical stimulation of the cornea. A lower threshold for the corneal reflex is found interictally in migraineurs compared with controls. This reduction is bilateral even in migraineurs presenting strictly unilateral headache (112).
Exteroceptive suppression of the voluntary temporal muscle activity has been studied independently by two groups. After stimulation of the labial commissure (108, 113), its late component ES2 was not different between migraineurs and controls. Its occurrence after index finger stimulation (113) was more prevalent in migraineurs, suggesting hyperexcitability of spino-bulbar pathways.
Taken together, electrophysiological investigations of trigeminal networks in migraine are in favour of segmental hypersensitivity and/or decreased control of descending inhibitory pathways.
Ischaemia test
Electromyograph (EMG) recordings during ischaemia of the upper limb induce abnormal spontaneous motor unit discharges in migraineurs, which is negatively correlated with red blood cell magnesium levels (114). This is thought to reflect neuromuscular hyperexcitability.
Single- fibre electromyography
Based on the fact that the P/Q Ca2+ channels that are dysfunctional in familial hemiplegic migraine (FHM) because of mutations in the CACNA1A gene (1) play a crucial role in acetylcholine release at the neuromuscular junction, we have studied single-fibre electromyography (SFEMG) in migraine and found interesting abnormalities in subgroups of patients. When compared with a group of healthy subjects without personal or familial history of headache, some migraineurs with aura showed subclinical impairment of neuromuscular transmission (115–117). This is especially true for patients suffering from auras characterized by sensorimotor symptoms, language disturbances and/or disturbed equilibrium (115) or from prolonged auras (116). An extensive study on subgroups of migraineurs affected by migraine without aura, migraine both with and without aura, migraine with typical aura and migraine with prolonged aura showed that neuromuscular abnormalities increased among migraine subgroups proportionally to the presence and duration of the aura (117). These mild abnormalities of neuromuscular transmission found in subgroups of migraineurs disappear during treatment with acetazolamide (118).
We postulate that the SFEMG abnormalities might be a consequence of dysfunctional motor nerve P/Q Ca2+ channels in subgroups of migraineurs whose clinical presentation is, in the migraine spectrum, close to that of familial hemiplegic migraine.
Transcranial magnetic stimulation
TMS has in theory the advantage of assessing directly excitability of the underlying cortex. It may thus be a tool of choice to search for cortical dysfunctions in migraine. TMS studies of both the motor and the visual cortex have been performed. The former has the advantage of relying on an objective measure, the motor-evoked potential in peripheral muscle, the latter using until recently the subjective perception of phosphenes. We will review and discuss the results of the various studies performed in migraineurs with TMS of motor or occipital cortices.
Motor cortex studies
Data of published studies are summarized in Table 4. The first study of motor cortex TMS in migraine was published in 1992 (119). To overcome the problem of large interindividual motor threshold (MT) variability, we investigated migraine patients with symptoms always located on the same side, which allowed use of the other side as an additional control. In that pilot study conducted between attacks, we observed that MT was significantly increased on the affected cortical side of patients with migraine with auras (MA) compared with normal subjects or to the unaffected side. No MT differences were observed between normal subjects and patients with unilateral migraine without aura (MO) or between the normal and affected side of MO patients. Moreover, in that study, the maximal amplitude of motor-evoked potentials (MEP) expressed as a ratio to maximal response to peripheral nerve stimulation (MEPmax/Mmax) was significantly reduced on the aura side of the body in MA patients. Abnormally high MTs were reported later in menstrual migraine without aura (120), not only in the interictal phase but also during attacks.
Transcranial magnetic stimulation (TMS) of motor cortex

MO, migraine without aura; MA, migraine with aura; HV, healthy volunteers.
∗: significant difference.
These results were not confirmed in a study by van der Kamp et al. (121), who in contrast found increased MEP amplitudes and reduced MT between attacks of patients with MA or MO. They also reported a positive correlation between MEP amplitudes and attack frequency but they did not control for the occurrence of an attack in the days following the recordings. In a subsequent paper (122), the same authors reported increased interictal MT and reduced MEP amplitudes on the side of motor deficits in patients with FHM. These results were very similar to those obtained in our first study of patients with unilateral MA.
In a subsequent paper (123), we studied a larger group of MA (n = 12) and MO patients (n = 19) with attacks occurring on either side, ensuring that TMS was performed at least 3 days after the previous or before the next attack. Here again, significantly higher mean MTs were observed during contraction in MA patients than in controls. Maximal MEP/MMax values were normal in MA as well as in MO patients, whose attacks were not always located on the same side. Other parameters such as the EMG silent period (SP) elicited by motor cortex stimulation and intracortical inhibition tested with paired TMS (124, 125), were also considered. Both were normal in MA or MO patients. We replicated our results in a recent study using a more focal stimulation with a figure-of-eight coil (126), but this time the trend for an MT increase in migraineurs did not reach the level of statistical significance.
By contrast, Aurora et al. (127) found that the cortical silent period was significantly shorter in MA patients than in controls. There was however, no control in this study for the possible occurrence of a migraine attack within 24 h after the recordings.
Finally, others have found no significant changes of MT to paired stimulations (128) or silent periods (129) in patients with MA or FHM.
Visual cortex studies
The conflicting results obtained in studies of phosphenes induced by occipital TMS are summarized in Table 5. Aurora et al. (130), using TMS over the occipital lobe, reported an abnormally high prevalence of magnetophosphenes in migraine with aura patients between attacks (11/11 MA compared with 3/11 normal subjects). Similar prevalence differences (100% in MA, 47% in controls) were reported by Brighina et al. (128). Moreover, the threshold at which phosphenes appeared (PT), was much lower in MA patients than in controls (130). The authors confirmed these results in a subsequent study of 15 migraineurs (14 MA) and 8 controls (131). Aguggia et al. (132) also found a significant decrease of PT in MA (58.3%) compared with controls (83.7%) or tension-type headache (81.9%). In another study (133), a reduced phosphene threshold was reported in MA and in MO, but the proportion of patients who experienced TMS-induced phosphenes, though lower in migraineurs, was not significantly different from controls. The same group found that prophylactic treatment with valproate was able to increase PT in MA, but not in MO (134), and that the ability of a TMS pulse over the occipital cortex to suppress visual perception was reduced in MA patients (135), which was interpreted as reflecting reduced activity of inhibitory circuits in the occipital cortex of migraineurs.
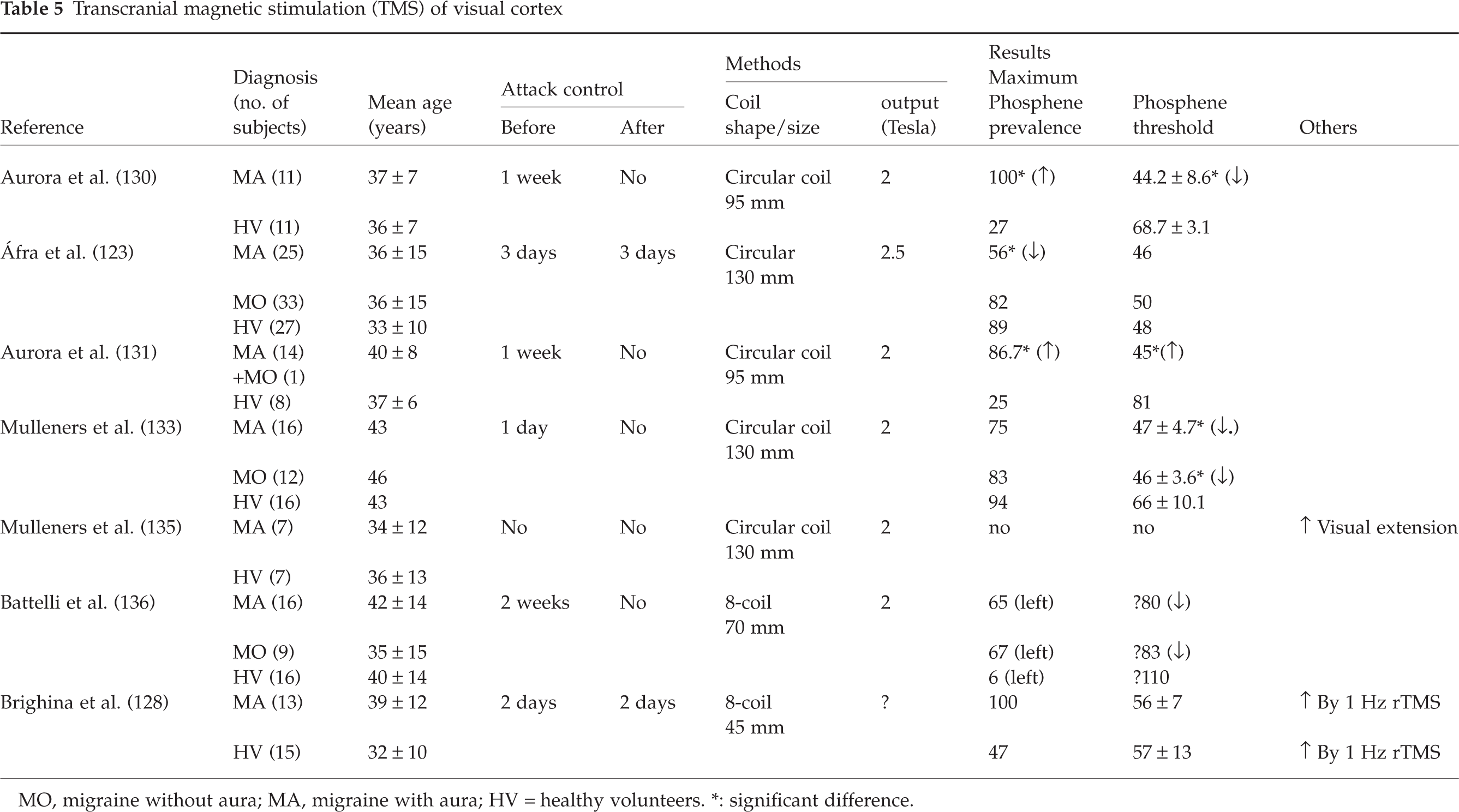
MO, migraine without aura; MA, migraine with aura; HV = healthy volunteers.
∗: significant difference.
Significantly lower phosphene thresholds for TMS over visual area V5 have been found in migraine both with or without aura compared with controls (136). A study published in abstract form by Young et al. (137) also concluded that phosphene thresholds for occipital TMS were lower in migraine with aura (36 ± 3%) or without aura (40 ± 6%) than in healthy subjects (55 ± 9%). Taken together, these studies were thought to favour the hypothesis of visual cortex hyperexcitability.
By contrast, we (123) have obtained rather opposing results using a similar methodology and a circular coil; the prevalence of phosphenes was significantly lower in MA patients than in controls (10/18 vs. 17/19), while no differences were found between controls and MO patients (18/22). Among subjects reporting phosphenes, mean thresholds for magnetophosphenes were similar in all groups. We replicated these findings using more focal visual cortex stimulation with a figure-of-eight coil (126). Others have failed to find significant differences between migraineurs and healthy subjects in phosphene thresholds for TMS over the primary visual cortex (128, 138). Interestingly, in the latter study, phosphene thresholds tended also to be higher in MA (71.04%) and MO (74.21%) than in controls (62.51%).
In a recent study (94), we have used VEP amplitude and habituation as a more objective measure than phosphenes to assess excitability changes induced in the visual cortex by rTMS at two different frequencies. In normal subjects, 1 Hz rTMS, which is known to inhibit the underlying cortex, reduced amplitude in the first block of 100 averaged responses and induced lack of habituation over successive blocks (Fig. 1). By contrast, in MA and MO patients between attacks, 10Hz rTMS, which most often activates the underlying cortex, increased first-block VEP amplitude in migraineurs and transformed their lack of habituation into a normal habituation pattern (Fig. 2). This study therefore strongly supports the concept that the visual cortex in migraineurs is hypo- and not hyper-excitable between attacks.
Discussion
On the basis of the studies described above, the use of TMS to assess motor and visual cortex excitability seems to have yielded conflicting results. Some discrepancies could be due to methodological differences, which may be device- and patient-dependent. With two exceptions (129, 136) where a figure-of-eight coil was used, all published studies were performed with a circular coil. The size of the coil, however, differs, for instance being much smaller in the studies of Aurora et al. The two types of coils differ substantially; a figure-of-eight coil produces a focal stimulation under the centre of the coil while a circular coil causes diffuse stimulation of the underlying cortical area (139, 140). It is thus likely that a larger cortical area is stimulated with the circular coils. In addition, the human cortex is sensitive to the direction of current flow in the coil, and with the circular coil this effect is more pronounced. Other technical differences between the methods used for TMS may contribute to the variance of results, but are sometimes difficult to evaluate based on available information: biphasic or monophasic magnetic pulses, maximum stimulator output, etc.
With regard to patient selection, it must be kept in mind that dramatic changes of evoked cortical responses, and thus of cortical excitability, occur 24 h before and during the attack (51, 83). While occurrence of the last attack before the recording can be checked by history, occurrence of an attack within 24 h after the recording has to be controlled by other means such as telephone calls to the patient. This was done in a restricted number of studies. In addition, cortical excitability fluctuates with hormonal variations during the menstrual cycle (141). In our studies we avoided such hormonal influences by recording all females at mid-cycle.
Nevertheless, some general lines can be drawn, particularly for motor cortex excitability. With the notable exception of one study (121) the general findings were reduced interictal motor cortex excitability in several forms of migraine: (i) unilateral or bilateral migraine with aura (119 123); (ii) menstrual migraine without aura (120); and (iii) familial hemiplegic migraine (122). These findings do not favour the hypothesis (13) of a permanent cortical hyperexcitability in migraine. In addition, excitability changes do not seem to result from dysfunction of cortical inhibitory interneurones, which were found to be normal in MA and MO patients (123, 129). Although such studies cannot be transposed to visual areas, these results do not favour the hypothesis of loss of GABAergic interneurones secondary to repetitive ischaemic insults (142). Moreover, in all studies except one, EMG silent periods to motor cortex stimulation were also found to be normal. Although the origin of this silent period remains debated (143), this point also argues against abnormalities of inhibitory output pathways of the motor cortex. Thus, it seems that the increase of MT in several subtypes of migraine corresponds to decreased excitability of large pyramidal neurones, as changes in spinal motoneurone excitability are unlikely in migraine.
Published studies on visual cortex excitability seem to give contradictory results. Although their exact generator remains a matter of debate, phosphenes elicited by brief, intense magnetic pulses directed to the occipital area of the brain are probably due to activation of the primary visual cortex and/or of subcortical sites such as the optic radiations adjacent to the posterior tip of the lateral ventricles (133). One puzzling result in several studies (128,130,131,136) is the very low prevalence of phosphenes elicited in the control group (3/11, 2/8, 1/16, 7/15), while all previous studies conducted in normal subjects report a much higher prevalence, usually between 60 and 80%(144–147). Although one cannot exclude that migraineurs were part of ‘healthy volunteers’ in the latter studies, the normal prevalence rates found in our studies (123, 126), where subjects with a personal or family history of migraine were excluded, are similar; 89 and 64%.
Contrasting with our finding (123, 126) of a significantly higher PT in both groups of migraineurs, Aurora et al. (130, 131) and Mulleners et al. (133) reported a significantly lower PT. In a recent study where TMS was applied laterally over visual area V5 (136), thresholds for magnetophosphenes were also lower in migraine patients. Results of the latter study are difficult to compare with all previous studies in which midline occipital TMS was used. It must be pointed out that in studies (130,131,136) reporting reduced magnetophosphene thresholds, subjects who had no phosphenes were not taken into account for the threshold calculations. In fact, if one assumes that subjects without phosphenes have at least a 100% threshold, a recalculation of the figures in the paper of Mulleners et al. (136) will, for instance, increase the mean phosphene threshold in the migraine without aura group from 46 to 55%, and in the migraine with aura group from 47 to 60.2%, while in the healthy volunteer group it would increase only from 66 to 68%. Needless to say, the differences between groups would lose statistical significance after such recalculations. On the other hand, some subjects may be resistant to induction of phosphenes by TMS and arbitrarily assigning them a PT of 100% may increase the risk for a type 2 error. This was done, nevertheless, in the study of Battelli et al. (136), in which a threshold value of 110% was assigned to 94% of control subjects who reported no phosphenes. Considering that only one-third of migraineurs had no phosphenes in that study, there is little doubt that the results of PT were biased towards higher values in controls. Moreover, interpretation of the results of this study (136) may be confounded by the modulatory effect of attention and expectation on extrastriate visual areas (148).
It may be premature to consider decreased thresholds for magnetophosphenes in migraine as evidence for increased excitability of the visual cortex. The fact that opposing results were obtained in different laboratories with similar methods suggests that phosphenes are too subjective and variable to be used to measure excitability of the visual cortex. Alternative, more objective and reliable methods should be developed. We have shown, for instance, that PR-VEP can be used to assess excitability changes of the visual cortex induced by rTMS (94). Using this method and analysing PR-VEP habituation, we have obtained evidence in favour of hypoexcitabiliy, and not hyperexcitabilty.
Conclusions
To summarize, studies of evoked potentials and transcranial magnetic stimulation disclose abnormalities of cortical information processing and excitability in both types of migraine between attacks. There seems to be a clear familial and predisposing character to some of these electrophysiological characteristics. Although their exact significance remains a matter of discussion, they underline at least that migraine cannot be reduced to a ‘trigeminovascular disorder’. Besides the studies of magnetophosphenes that have yielded contrasting results, chiefly because of lack of reliability of the method, most recent electrophysiological investigations of cortical activities in migraine favour lack of habituation and decreased cortical excitability as the predominant dysfunctions. Both abnormalities may be linked to each other, if one applies the ‘ceiling theory’, postulating that a reduced preactivation excitability level of sensory cortices will be responsible for a lack of habituation of cortical evoked responses during repeated stimulations. Whether the latter plays a role in migraine pathogenesis by increasing metabolic demands in the migrainous brain, which has a reduced mitochondrial energy reserve, remains to be proven. Further studies are necessary to better define the precise nature of CNS dysfunction in migraine and to determine whether the cortical electrophysiological patterns allow, like other neurophysiological tests (117, 149), the identification of subgroups of migraineurs in whom correlations can be established with specific genotypes, responses to prophylactic agents or interictal cognitive disturbances.
