Abstract
Stress is the most commonly reported trigger of an episode of chronic tension-type headache (CTTH); however, the causal significance has not been experimentally demonstrated to date. Stress may trigger CTTH through hyperalgesic effects on already sensitized pain pathways in CTTH sufferers. This hypothesis could be partially tested by examining pain sensitivity in an experimental model of stress-induced headache in CTTH sufferers. Such examinations have not been reported to date. We measured pericranial muscle tenderness and pain thresholds at the finger, head and shoulder in 23 CTTH sufferers (CTH-S) and 25 healthy control subjects (CNT) exposed to an hour-long stressful mental task, and in 23 CTTH sufferers exposed to an hour-long neutral condition (CTH-N). Headache developed in 91% of CTH-S, 4% of CNT, and 17% of CTH-N subjects. Headache sufferers had increased muscle tenderness and reduced pain thresholds compared with healthy controls. During the task, muscle tenderness increased and pain thresholds decreased in the CTH-S group compared with CTH-N and CNT groups. Pre-task muscle tenderness and reduction in pain threshold during task were predictive of the development and intensity of headache following task. The main findings are that stress induced a headache in CTTH sufferers, and this was associated with pre-task muscle tenderness and stress-induced reduction in pain thresholds. The results support the hypothesis that stress triggers CTTH through hyperalgesic effects on already increased pain sensitivity in CTTH sufferers, reducing the threshold to noxious input from pericranial structures.
Introduction
Psychological stress is the most frequently reported trigger of a chronic tension-type headache (CTTH) episode (1–3), and a number of studies have demonstrated increased headache activity following exposure to stressful mental tasks in headache sufferers (4–6). However, stress has not been experimentally shown to trigger headache in CTTH sufferers to date (i.e. using CTTH subjects in both stress and neutral conditions). Such an examination is required to establish the causal significance of stress in CTTH.
The mechanisms by which stress triggers headache in CTTH sufferers, if in fact it does, are not clearly understood. One suggestion is that stress may aggravate already increased pain sensitivity in CTTH sufferers, further reducing the threshold to noxious input from pericranial structures (7–9). Supporting this, we recently found a brief stress task reduced subsequent cephalic pain thresholds in CTTH sufferers compared with healthy controls (10). Leisted et al. (1) reported increased pain sensitivity following induced stress in a mixed sample of episodic (ETTH) and CTTH sufferers, and Janke et al. (4) found depressed ETTH subjects had increased pain sensitivity and were more susceptible to headache following induced stress than were euthymic ETTH subjects. No studies have examined pain sensitivity in an experimental model of stress-induced headache in CTTH sufferers. The present study therefore measured muscle tenderness, pain thresholds, and headache activity during prolonged mental stress in CTTH sufferers and healthy controls. The aim was to examine whether stress-induced headache is associated with hyperalgesic effects of stress on already increased pain sensitivity in CTTH sufferers.
Method
Subjects
Subjects were recruited via advertisements in local media and University of South Australia media requesting volunteers for a study on headaches. Written consent from each subject for study participation was obtained and the study was approved by the University's Human Research Ethics Committee. Potential volunteers underwent a diagnostic interview based on the International Classification of Headache Disorders, 2nd edn (ICHD-II) criteria (11) and a structured clinical interview to assess for current of past psychiatric or major medical conditions. Inclusion criteria for the CTTH group were: satisfying ICHD-II criteria for chronic tension-type headache, aged 18–65 years, not currently receiving (or having received in the last 12 months) intervention for headache, no psychiatric or major medical condition currently or in the last 12 months, no concurrent headache or pain symptoms or diagnoses (other than CTTH). Additionally, CTTH subjects were required not to be taking, or not have taken in the last 3 months, any analgesic medication other than ≤ 1000 mg daily of aspirin or paracetamol. No subjects reported taking prophylactic medication for headache. No subjects reported daily analgesic use. Eight subjects reported paracetamol use of ≤ 1000 mg up to 20 days per month. All subjects were required to have not taken any analgesic on the day of examination, and were required to be headache free at presentation for the experimental session. Six headache sufferers reported having a headache on the day of their scheduled session time and were subsequently rescheduled. Inclusion in the control group required additional criteria of no past or current chronic pain or headache diagnoses, fewer than five headaches in the last year, and none within the last 6 months. All recruited subjects completed the study procedures.
Procedures
The protocol involved exposing subjects to either an hour-long stressful mental task or an hour-long neutral condition, and measuring negative affect, headache, and pain sensitivity before, during and after task/neutral condition exposure. Headache subjects were randomly assigned to either the stress (CTH-S) or neutral (CTH-N) conditions, whereas all non-headache (CNT) subjects were exposed to the stress condition only, as we previously found no effect of the neutral condition on pain sensitivity in healthy subjects (S. Cathcart et al., unpublished data). To control for possible effects of the eight subjects who reported paracetamol use up to 20 days per month, these subjects were evenly distributed across CTH-S and CTH-N conditions. All sessions were conducted in an interview room at the School of Psychology, University of South Australia, between 09.00 and 17.00 h on Monday to Friday. The room was maintained at a constant 23°C. Subjects were given a debriefing interview at the conclusion of the laboratory session.
Stress task
The stress task was adapted from one previously demonstrated to induce stress and headache (5), and involved subjects solving anagrams and arithmetic problems, presented via computer monitor. Anagrams had three levels of difficulty: ‘easy’ anagrams were words of eight to 10 letters with two sets of adjacent letters presented in reverse order; ‘difficult’ anagrams were long words with letters presented in random order; and ‘insoluble’ anagrams were 9–11-letter words with letters presented in random order and one letter omitted so no solution existed. Previous research has shown subjects can solve most of the easy anagrams but few of the difficult anagrams (5). The arithmetic problems have been used in previous research by our group (10), and involve subtraction or addition of two-digit (e.g. 73–58), and three-digit (e.g. 576–283) problems. There was an equal amount of anagrams and arithmetic problems in each block, which were presented in random order.
The task involved presentation of each problem for 10 s on the monitor, during which subjects were required to solve the problem in their head (without the use of paper or pencil). Following the problem presentation screen, a screen with the words ‘intertrial interval’ was presented for 5 s, during which subjects verbalized their answer to the previous problem, or said ′pass′ if they did not know the answer. The experimenter remained in the room with the subject for the entire time and pretended to enter their responses into a computer.
The task was divided into 12 ‘blocks’ of 10 problems. Subjects were told they would receive performance feedback via the screen after each block, advising of their accuracy relative to previous participants, in the form of ‘below average’, ‘average’, or ‘above average’. In fact, feedback was not based on performance, with subjects receiving ‘below average’ for eight blocks, and ‘average’ for four blocks. Subjects were also advised that the duration of the task was based on their performance, with the shortest possible duration being 30 min (if they got all problems correct), and the longest duration 1.5 h (if they got all of the problems incorrect). In fact, the task lasted for 1 h, regardless of performance.
Neutral condition
The neutral condition involved subjects remaining seated in the interview room in an identical position to the ‘stress condition’ subjects (i.e. seated at a table in front of the monitor), and browsing supplied magazines and newspapers for 60 min. The experimenter remained in the room working on a computer, and did not engage with the subjects during the task. The monitor was blank for the duration of the neutral condition, except when subjects were requested to provide ratings of negative affect and headache, as below.
Measures
Self-report questionnaires
Prior to the task, subjects completed an in-house socio-demographic questionnaire detailed elsewhere (12). Subjects also completed the State-Trait Anxiety Inventory (13), and the Centre for Epidemiological Studies-Depression Scale (14).
Negative affect and headache (during stress task)
At the beginning of the task, and after every two blocks (10 min), subjects rated their overall negative affect according to a visual analogue scale (VAS) presented on the monitor. The screen read ‘Please rate your current overall level of Negative Affect (stress, anxiety, anger, or depression) according to the rating scale below’. The rating scale ranged from ‘0-Not at all’ (stressed, anxious, angry, or depressed) to ‘10-Extremely’ (stressed, anxious, angry, depressed). Between 0 and 10 were evenly spaced consecutive whole numbers from 1 to 9. Additionally, the descriptor ‘Somewhat’ was placed at approximately 2.5 on the VAS, ‘Moderately’ at approximately 5 on the VAS, and ‘Very’ at approximately 7.5. This composite ‘Negative Affect’ rating has been used in previous research and is based on correlation and factor analysis deriving common subjective headache triggers and responses to stress tasks (15). Immediately following the Negative Affect rating, subjects rated their headache intensity according to a six-point Likert scale previously used and detailed elsewhere (5), also presented on the monitor. The headache scale response categories are 0 = no headache; 1 = pain, but easily ignored; 2 = can be ignored at times; 3 = painful, but able to work; 4 = painful, work very difficult; and 5 = very painful, unable to work. Subjects in the neutral condition were presented the Negative Affect and Headache rating screens only, at corresponding times to those presented during the stress task conditions (i.e. every 10 min).
Muscle tenderness
Pericranial muscle tenderness (MT) was assessed by a qualified and experienced pain clinician (the senior author) using the standard method (16). Seven muscle and tendon insertions were palpated bilaterally: frontalis, temporalis medial belly, occipitalis belly, trapezius upper shoulder belly, trapezius superior insertion, sternocleidomastoid belly, sternocleidomastoid insertion. A total tenderness score was created for each subject by summing the seven bilateral scores. MT was recorded immediately before the task, after 30 min (six blocks), and immediately post task.
Pressure pain thresholds
Pain thresholds were assessed using an analogue pressure algometer (Wagner Instruments, FPK20; Wagner Instruments, Greenwich, CT, USA) with a circular silicon rubber tip measuring 0.79 cm2. Thresholds were recorded at the right-hand side of the body only, since we have previously demonstrated no laterality differences using this procedure and raters. Three locations were asessed: (i) the dorsal surface of the medial segment of the first phalange; (ii) a parietal location without overlying muscle; and (iii) the middle of the shoulder belly of the trapezius muscle. Pressure was increased at a rate of approximately 1 kg/s and subjects were asked to say ‘pain’ at the point the sensation first became painful, at which point pressure was released and the readout recorded. Thresholds were taken as the average of two measures taken 30 s apart. Pressure pain thresholds (PPTs) were recorded on four occasions: (i) pretask; (ii) after 20 min of task (four blocks); (iii) after 40 min of task (eight blocks); and (iv) immediately post task (12 blocks). Reliability of the PPT technique for repeat within-session assessment by the present rater has been confirmed elsewhere (17).
Statistical analyses
Statistical analyses were conducted using the Statistical Package for the Social Sciences (SPSS Version 15; SPSS Inc., Chicago, IL, USA) (18). Analyses of vbariance (
Random coefficient regression was used to examine effects of the stress and neutral tasks on pain sensitivity, negative affect, and headache activity. Random coefficient regression is a mixed model analysis and the analysis is subject specific rather than population averaged. More variance is therefore explained than using conventional repeated-measures
Finally, ordinary least squares (OLS) regression analyses were used to identify: (i) measures predictive of stress inducing a headache episode (assessed with logistic regression); and (ii) measures predictive of stress-induced headache intensity (assessed with OLS linear regression).
Results
Socio-demographic and clinical measures
Socio-demographic and clinical data for chronic tension-type headache subjects and healthy controls

Speilberger State-Trait Anxiety Inventory.
Centre for Epidemiological Studies—Depression scale.
Structured diagnostic interview of headache characteristics.
CNT, healthy control group; CTH-S, chronic tension-type headache subjects exposed to stress task; CTH-N, chronic tension-type headache subjects exposed to neutral task.
Negative affect during task
Figure 1 shows the mean overall negative affect rating for the three groups during the stress and neutral conditions. There was a significant group effect, F
2,68 = 4.10, P < 0.05, a significant time effect, F
1,68 = 109.66, P < 0.01, and a significant group–time interaction, F
2,68 = 30.94, P < 0.01. A direct comparison of parameter estimates (19) indicated negative affect ratings were lower for the CTH-N group compared with both CNT (t = 2.40, P < 0.05) and CTH-S groups (t = 2.57, P < 0.05). Comparison of parameter estimates for group × time affects indicated a greater increase in negative affect during the task in the CTH-S (t = 7.17, P < 0.01, d = 1.90) and CNT (t = 6.45, P < 0.01, d = 1.64) groups compared with the CTH-N group. Bonferroni adjusted post hoc tests indicated negative affect ratings were significantly higher in the CTH-S and CNT groups compared with the CTH-N group from the 10-min time point onwards (all P < 0.01), whereas there was no difference between CTH-S and CNT groups (all P > 0.10).
Negative affect in headache and healthy control subjects during stress task and neutral conditions. CTH-S, chronic tension-type headache subjects exposed to stress task; CTH-N, chronic tension-type headache subjects exposed to neutral condition; CNT, healthy control subjects exposed to stress task. *CTH-N < CTH-S and CNT, P < 0.05.
Headache during task
Twenty-one subjects in the CTH-S group (91%) developed a headache during the task, compared with one CNT subject (4%) and four CTH-N subjects (17%). χ2 analyses indicated significantly more CTH-S subjects developed a headache compared with both the CTH-N and CNT groups (both P < 0.01), whereas the difference between CNT and CTH-N groups was not significant (P > 0.05). Figure 2 shows the mean headache ratings for the three groups during the stress and neutral conditions. There was a significant group effect, F
2191 = 3.08, P < 0.05, a significant time effect, F
1423 = 239.62, P < 0.01, and a significant group–time interaction, F
2423 = 96.69, P < 0.01. Comparison of parameter estimates indicated headache ratings increased significantly faster in the CTH-S group compared with the CTH-N group (t = 10.84, P < 0.01, d = 2.64), whereas group × time effects between CNT and CTH-N were not significant (t = −1.96, P = 0.06). Bonferroni adjusted post hoc tests indicated headache intensity ratings were significantly higher in the CTH-S group than in the CNT and CTH-N groups at all time points from 20 min to post-task (all P < 0.01), whereas there were no differences between CTH-N and CNTgroups on headache intensity measures at any time point (all P > 0.10).
Headache intensity in headache and healthy control subjects during stress task and neutral conditions. CTH-S, chronic tension-type headache subjects exposed to stress task; CTH-N, chronic tension-type headache subjects exposed to neutral condition; CNT, healthy control subjects exposed to stress task. *CTH-S > CTH-N and CNT, P < 0.05.
Pressure pain thresholds
Figure 3 shows the mean PPTs at the finger (PPTF) for the three groups during the stress and neutral conditions. There was a significant group effect, F
2,68 = 6.71, P < 0.01, a significant time effect, F
1,68 = 14.15, P < 0.01, and a significant group–time interaction, F
2,68 = 5.63, P < 0.01. Comparison of parameter estimates indicated that the CNT group had higher finger pain thresholds compared with the CTH-N group (t = 3.49, P < 0.01), whereas there was no significant difference between the CTH-S and CTH-N groups (t = 0.83, P > 0.10). Comparison of parameter estimates for group × time effects indicated that finger pain thresholds for the CTH-S group decreased significantly more during the task than thresholds in the CTH-N group (t = −3.07, P < 0.01, d = 0.73), whereas group × time effects between the CNT and CTH-N groups were not significant (t = −0.41, P > 0.10). Bonferroni adjusted post hoc tests indicated finger pain thresholds were significantly higher in the CNT group than in the CTH-S and CTH-N groups at all time points (all P < 0.02), whereas there were no differences between the CTH-N and CTH-S groups on finger pain thresholds at any time point (all P > 0.10).
Pressure pain threshold at finger in headache and healthy control subjects during stress task and neutral conditions. CTH-S, chronic tension-type headache subjects exposed to stress task; CTH-N, chronic tension-type headache subjects exposed to neutral condition; CNT, healthy control subjects exposed to stress task. *CNT > CTH-N and CTH-S, P < 0.05.
Figure 4 shows the mean PPTs at the head (PPTH) for the three groups during the stress and neutral conditions. There was a significant group effect, F
2,68 = 6.41, P < 0.01, a significant time effect, F
1,68 = 50.45, P < 0.01, and a significant group–time interaction, F
2,68 = 12.83, P < 0.01. Comparison of parameter estimates indicated that the CNT group had higher head pain thresholds compared with the CTH-N group (t = 3.34, P < 0.01), whereas there was no significant difference between the CTH-S and CTH-N groups (t = 0.59, P > 0.10). Comparison of parameter estimates for group × time effects indicated that head pain thresholds for the CTH-S group decreased significantly more during the task than thresholds in the CTH-N group (t = −5.01, P < 0.01, d = 0.83), whereas group × time effects between the CNT and CTH-N groups approached significance (t = −1.89, P = 0.06). Bonferroni adjusted post hoc tests indicated head pain thresholds in the CNT group were higher than in the CTH-S group at all time points (all P < 0.02), and higher than the CTH-N group at pre-task and 20-min recordings (all P < 0.05). There were no differences between the CTH-N and CTH-S groups on head pain thresholds at any time point (all P > 0.10).
Pressure pain threshold at head in headache and healthy control subjects during stress task and neutral conditions. CTH-S, chronic tension-type headache subjects exposed to stress task; CTH-N, chronic tension-type headache subjects exposed to neutral condition; CNT, healthy control subjects exposed to stress task. *CNT > CTH-S at all time points, P < 0.05. #CNT > CTH-N at pre-task and 20-min time points, P < 0.05.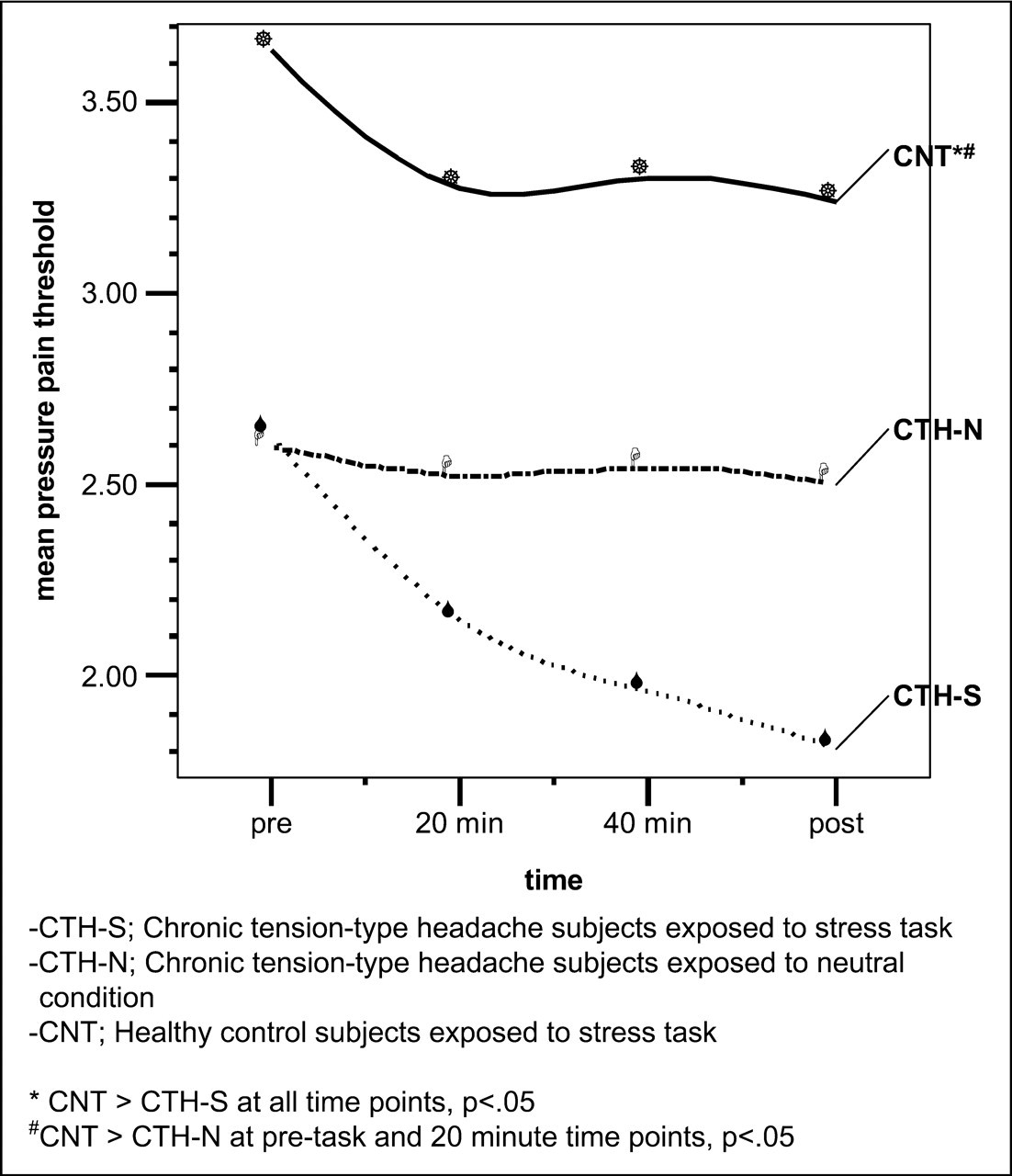
Figure 5 shows the mean PPTs at the shoulder (PPTS) for the three groups during the stress and neutral conditions. There was a significant group effect, F
2,68 = 3.73, P < 0.03, a significant time effect, F
1,68 = 36.93, P < 0.01, and a significant group–time interaction, F
2,68 = 20.44, P < 0.01. Comparison of parameter estimates indicated that the CNT group had higher shoulder pain thresholds compared with the CTH-N group (t = 2.31, P < 0.03), whereas there was no significant difference between the CTH-S and CTH-N groups (t = −0.07, P > 0.10). Comparison of parameter estimates for group × time effects indicated that shoulder pain thresholds for the CTH-S group decreased significantly more during the task than thresholds in the CTH-N group (t = −6.09, P < 0.01, d = 0.80), whereas group × time effects between the CNT and CTH-N groups were not significant (t = −1.42, P > 0.10). Bonferroni adjusted post hoc tests indicated shoulder pain thresholds in the CNT group were higher than in the CTH-S group at all time points (all P < 0.03), but not higher than the CTH-N group at any time point (all P > 0.10). The CTH-S group had higher shoulder pain thresholds than the CTH-N group at post-task measurement (P < 0.01).
Pressure pain threshold at shoulder in headache and healthy control subjects during stress task and neutral conditions. CTH-S, chronic tension-type headache subjects exposed to stress task; CTH-N, chronic tension-type headache subjects exposed to neutral condition; CNT, healthy control subjects exposed to stress task. *CNT > CTH-S, P < 0.05. #CTH-N > CTH-S at post-task time point, P < 0.05.
Muscle tenderness
Figure 6 shows the mean pericranial MT ratings for the three groups during the stress and neutral conditions. There was a significant group effect, F
2,90 = 12.20, P < 0.01, a significant time effect, F
1139 = 23.81, P < 0.01, and a significant group–time interaction, F
2139 = 12.39, P < 0.01. Comparison of parameter estimates indicated MT was higher in the CTH-N group than in the CNT group (t = −4.38, P < 0.01), whereas there was no significant difference between the CTH-N and CTH-S groups (t = −0.26, P > 0.10). Comparison of parameter estimates for group × time effects indicated a greater increase in MT during the task in the CTH-S group than in the CTH-N group (t = 4.52, P < 0.01, d = 0.44), whereas there was no significant difference between the CNT and CTH-N groups (t = 0.52, P > 0.10). Bonferroni adjusted post hoc tests indicated MT ratings were significantly higher in the CTH-S and CTH-N groups than in the CNT group at each time point (all P < 0.01), whereas there were no differences between the CTH-S and CTH-N groups (all P > 0.10).
Pericranial muscle tenderness in headache and healthy control subjects during stress task and neutral conditions. CTH-S, chronic tension-type headache subjects exposed to stress task; CTH-N, chronic tension-type headache subjects exposed to neutral condition; CNT, healthy control subjects exposed to stress task. *CNT < CTH-N and CTH-S, P < 0.05.
Relationships between stress, pain and headache
Logistic regression was used to predict development of headache during the task from negative affect and pain sensitivity, while linear regression was used to examine if negative affect and pain sensitivity could predict the post-task intensity of induced headache. Stepwise entry procedures were used to find a model containing significant predictor variables. As well as including absolute levels of pain sensitivity in the regression analyses, we also calculated change scores (difference between pre- and post-task measurements) in MT and pain thresholds, to examine if magnitude of stress-induced change in pain sensitivity affected development of headache.
Logistic regression predicting stress-induced headache from affect and pain sensitivity

Pre-task pericranial muscle tenderness.
Pressure pain threshold at the shoulder at 40 min into stress task.
Change in pressure pain threshold at shoulder from pre to post task.
Linear regression predicting stress-induced headache intensity from affect and pain sensitivity

All P < 0.01.
Change in pressure pain threshold at shoulder from pre to post task.
Pre-task pericranial muscle tenderness.
Negative affect rating at 30 min into task.
Pressure pain threshold at the head at 40 min into task.
Discussion
Task effects on negative affect and headache
The present results indicate that the stress task successfully increased negative affect in the headache and control groups. The lack of change in the CTH-N group indicates that this increase can be attributed to the task. We found no baseline or task reactive differences in negative affect between the CTH and control subjects exposed to the stress task. These results are consistent with and extend previous findings in other headache diagnostic groups (21) to suggest CTH sufferers are not more emotionally reactive to prolonged stress than are healthy subjects.
Of the CTH sufferers exposed to the stress task, 91% developed a headache, compared with only 17% of headache sufferers in the neutral condition, and 4% of the control subjects. This is consistent with previous studies demonstrating increased headache activity in ETTH sufferers (4) and mixed headache samples during induced stress (5). However, only one previous study (6) was an experimental design incorporating a headache group not exposed to the stress task. The present study is the first experimental demonstration of stress-induced headache in CTTH sufferers, thereby confirming stress as a trigger in CTTH. We cannot conclude the induced headache in our study represents a true episode of CTTH; however, the debriefing interview confirmed that the induced headache was similar to headache subject's usual headache, in terms of intensity, pain quality, location, and differential diagnostic features (e.g. no features of migraine or other headache diagnosis symptoms).
Additional to the stress task inducing a headache in most of the CTH-S sufferers, we also found headache intensity to increase throughout the course of the task, in conjunction with increasing negative affect. This result supports previous findings that mental stress not only triggers a headache, but also aggravates existing headache (22), although conclusions are tentative since we did not include a CTTH group who discontinued task exposure after headache onset. Previous studies, however, show headache intensity declines following cessation of a stress task.
Task effects on muscle tenderness
Consistent with previous research, the headache groups in the present study reported higher pericranial MT compared with the healthy controls (7,9,16). We also found a within-subject increase in MT following task in the CTH-S group. The lack of change in the CTH-N group indicates that this increase can be attributed to the task. To our knowledge, this is the first experimental demonstration that stress increases MT in CTTH sufferers. Janke et al. (4) measured MT before and after induced stress in depressed ETTH sufferers; however, although visual inspection of their data indicates post-task increase in MT, within-subject analyses were not reported.
Although increased MT is a common finding in CTTH sufferers, it is unclear if this is a cause, concomitant or consequence of headache. MT has been found to increase during the headache episode in some studies (23,24), but not others (25,26). In our study, headache was induced in the majority of CTH-S subjects within 30 min, whereas MT was unchanged at 30 min but increased at 60 min. Hence, although our results indicate increased MT during headache, the development of headache was not secondary to immediate increase in MT. Rather, our results suggest the MT increased secondary to some other factor, such as stress response to the task, headache, or increased pain sensitivity during the task.
Task effects on pressure pain thresholds
Consistent with previous findings, the present study found cephalic and extracephalic PPTs were lower in the CTTH compared with the healthy control subjects (27–30). This difference was observed both before and after exposure to the stress task, indicating an already existing increase in general pain sensitivity in the CTTH sufferers.
In a recent study, we found a brief stress task reduced cephalic PPTs in CTTH sufferers to a greater extent than in healthy controls (10). Extending those findings, the present study found pain thresholds reduced during the hour-long stress task in both CTTH and control groups, with a greater decrease in the CTH-S compared with the CTH-N and CNT groups. The lack of change in the CTH-N group suggests the decrease in the CTH-S group can be attributed to the task. Pain thresholds decreased within 20 min of task exposure, whereas headache developed from 20 min onwards in most CTTH sufferers. It is therefore unlikely that the reduction in pain thresholds during the task was secondary to headache. Rather, the results support the suggestion that stress aggravates already increased general pain sensitivity in CTTH sufferers.
Mechanisms underlying effects of stress on pain sensitivity
Pain thresholds decreased during the mental task in both the healthy controls and CTTH sufferers, with a greater reduction in the CTTH group. The finding of a reduction in healthy subjects is consistent with previous findings (31–33), and suggests a functional mechanism for stress-induced hyperalgesia. Logan et al. (31) and others (34) suggest such effects may represent a warning mechanism for increasing challenge to the organism, facilitating elaboration of coping behaviours.
How then can the presently observed group differences in effects of stress on pain sensitivity be explained? Since we found no group differences between CTTH and control subjects on measures of anxiety, depression, or affective reactivity during the task, the result cannot be explained by these factors. Rather, our results indicate an enhanced hyperalgesic response to ‘normal’ stress reactivity in the CTTH subjects.
Olesen (8) proposed sensitization in the trigeminal nucleus caudalis as a model for integrating myofascial and supraspinal contributions to TTH. This nucleus receives both facilitatory and inhibitory descending input from supraspinal structures, as well as convergent input from myofascial structures of the pericranial region. In the present study, stress from the task could have aggravated already existing peripheral and/or trigeminal sensitization in the CTTH sufferers through descending facilitation or reduction of inhibition, resulting in an enhanced hyperalgesic effect of the task on MT and/or PPT in the CTH-S group. Additionally, the stress task could have aggravated already sensitized supraspinal/cortical pathways in the CTTH group (35). It is also possible that the stress task aggravated an already existing cognitive bias to pain in the CTTH sufferers (36). A recent study in rat found stress enhanced pronociceptive effects of immune mediators in peripheral tissue (37). It is possible therefore that the present stress task increased pain sensitivity at the periphery, instead of, or in addition to, affecting central pain processing. Further research is needed to examine these possibilities.
Relationships between negative affect, pain sensitivity, and headache
We used logistic regression to predict development of a headache during the task from measures of negative affect and pain sensitivity. The results indicated that 80% of cases could be correctly classified from measures of pre-task MT, PPTS at 40 min, and magnitude of PPTS change from pre to post task. Further, increasing MT and decreasing PPT significantly increased the odds of developing a headache during stress task exposure. It is notable that a combination of pre-task muscle tenderness and subsequent reduction in pain thresholds during the task were predictive of headache development. This provides strong support for the hypothesis that stress triggers headache in CTTH sufferers through aggravating already increased pain sensitivity, reducing threshold to nociceptive input from pericranial structures. It is also consistent with and extends a previous study in which daily stress was associated with increased MT and reduced pain thresholds in CTTH sufferers (38).
Additionally, we used linear regression to examine if affect and pain sensitivity were related to the intensity of induced headache following the task. Pre-task MT, increased negative affect at 30 min, decreased PPTH at 40 min, and magnitude of PPTS change from pre to post task, were all significant predictors of headache intensity. This suggests that pre-task MT and a decrease in pain threshold during stress are related not only to the development of stress-induced headache, but also to the intensity of the developed headache. As with the logistic model, it is notable that pre-task MT and subsequent reduction in pain thresholds during the task were both predictive of headache intensity. Stress may aggravate headache intensity through reducing threshold to nociceptive input and enhancing the pain from already tender pericranial structures.
Limitations to the present study
A number of limitations in this study warrant mention. We only used a subjective measure of stress in the form of a compound measure of negative affect. However, this measure has been empirically derived in previous research to reflect affective factors important in the relationship between stress and headache (15). Further, differences between headache and control subjects have generally been found on subjective but not physiological indices of stress (21). Similarly, we used self-report of pain sensitivity, which presents limited information on the pain system. However, self-report represents maximal integration of the stress and pain systems, and most previous research has used self-report to demonstrate increased pain sensitivity in CTTH sufferers [e.g. (1,2,23,24)], and hyperalgesic effects of stress on pain sensitivity in healthy humans [e.g. (31–33)]. Indeed, previous research has found effects of stress on pain report but not nociceptive reflex (39,40). Finally, as reported, eight of the headache subjects in the present study reported paracetamol use up to 20 days per month. Hence, our sample may have included a subset of subjects with possible medication overuse headache. Although these subjects were evenly distributed across CTH-S and CTH-N groups, it remains a possibility that this subset of subjects influenced the headache vs. control group comparisons. We were unable to examine this possibility, however, due to the subset sample size being too small for reliable analysis. Future research specifically examining medication overuse headache would be valuable.
Conclusions
The present study is the first experimental demonstration of stress-induced headache in CTTH sufferers. We also found prolonged stress increased pain sensitivity in healthy subjects and CTTH sufferers, with this effect being greater in CTTH sufferers. The main finding is that baseline levels of MT, and stress-induced decrease in pain threshold, were both predictive of the development and intensity of stress-induced headache. Taken together, the results support the hypothesis that stress triggers headache in CTTH sufferers through aggravating already increased pain sensitivity, reducing threshold to nociceptive input from pericranial structures.
