Abstract
A central sensitization has been advocated to explain chronic daily headache (CDH) due to sustained peripheral sensitization of algogenic structures responsible for sustained trigeminovascular system activation. Several mechanisms have been suggested to underlie central sensitization, but have been poorly investigated in CDH. They involve N-methyl-D-aspartate (NMDA) receptor activation and nitric oxide (NO) production and supersensitivity and increased and maintained production of sensory neuropeptides. The present study supports the above pathogenic mechanisms demonstrating a significant increase in glutamate and nitrite levels in the CSF of CDH patients, without a significant difference between patients without and those with analgesic overuse headache (P < 0.0001 and P < 0.002). The increase in CSF nitrites was accompanied by a significant rise in the CSF values of cyclic guanosine monophosphate (cGMP) in patients in comparison with controls (P < 0.0001). A statistically significant correlation emerged between visual analogic scale (VAS) values and glutamate, nitrites and cGMP. Although substance P (SP) and calcitonin gene-related peptide (CGRP), and to a lesser extent neurokinin A, were significantly increased in CSF compared with control subjects, their values did not correlate with glutamate, nitrites and cGMP levels in CSF in the patient group. The present study confirms the involvement of glutamate-NO-cGMP-mediated events underlying chronic head pain that could be the target of a new therapeutic approach which should be investigated.
Keywords
Introduction
Few studies have attempted to clarify the biochemical basis of chronic daily headache (CDH) (1–3). Neurotransmitter and intracellular mediators have also been scarcely investigated in medication overuse headache on a daily or almost daily basis (4–6).
One of the putative candidates in the development and maintenance of head pain is glutamate, through the interaction with its own receptors which include inotropic, cation-selective, ligand-gated receptors and metabotropic G-protein-coupled receptors (7–9).
It is well known that N-methyl-
Calcium entry into sensory neurones in the dorsal horn and the caudal part of the trigeminal nucleus induces activation of nitric oxide (NO) synthase, leading to the synthesis of NO and the activation of soluble guanylate cyclase which causes the increase in the soluble intracellular messenger cyclic guanosine monophosphate (cGMP) (15).
Both glutamate and diffusible NO can also affect the nociceptor terminals and lead to a further release of glutamate and enhance the release of sensory neuropeptides (in particular substance P, but also calcitonin gene-related peptide (CGRP)) from presynaptic neurones, therefore contributing to the development of hyperalgesia and maintaining central sensitization (7, 16).
It has been suggested that the above mechanisms underlie CDH evolving from a previous history of migraine or tension-type headache, but they have not until now been investigated. Moreover, no studies have been carried out to assess the involvement of the excitatory amino acid glutamate, NO pathway activation and the intracellular messenger cGMP in medication overuse headache.
The hypothesis of the study was that the glutamate–NO–cGMP cascade underlies the pain state in CDH, and that repeated activation of this system could be the basis for the headache becoming chronic.
The present study therefore aimed to investigate variations in the levels of glutamate and those of stable NO end-products, which are nitrites, in the CSF of patients affected by CDH without analgesic overuse and patients affected by analgesic overuse daily headache compared with control subjects. The intracellular messenger of NO, cGMP, was also determined in the CSF of the same patients. The CSF levels of the above parameters have been correlated with the sensory neuropeptides substance P (SP), CGRP and neurokinin A (NKA).
Patients and methods
Patients
Inclusion criteria were: a previous history of migraine without aura and the presence of headache for at least 15 days per month for at least 6 months (14). On the basis of the above assumption, 25 consecutive patients attending the Headache Centre of the Neuroscience Department of the University of Perugia were admitted to the study.
All patients could be diagnosed as suffering from transformed migraine according to Silberstein et al. criteria (17, 18). When applying IHS criteria for migraine without aura, only eight patients could be classified. The remaining 17 patients could not be IHS classified as migraine without aura, in eight cases because the headache was essentially continuous, and in nine because they did not have sufficiently associated features or because of a missing pain factor. None of these patients assessed fulfilled the criteria for chronic tension-type headache. All patients were asked about symptomatic drug intake. To define analgesic overuse the criteria suggested by Silberstein et al. (17) were used: simple analgesic use> 1000 mg/ASA/acetaminophen > 5 days/week; combination analgesics > 3 tablets/day > 3 days/week.
On the basis of the above definition, patients could be subdivided into two groups: patients with CDH without analgesic overuse (n = 10) and medication overuse headache patients (n = 15). Among the latter, 10 patients used more than one simple analgesic, and five used both simple and combination analgesics.
The monthly drug intake averaged 69.3 ± 14.2 (mean ± SD) tablets or suppositories. None of the patients used drugs containing codeine and/or took preventive medication. They did not take analgesics for other chronic painful conditions, with the exception of chronic headache.
Details of patient groups are given in Table 1.
Characteristics of patients and control subjects

∗The chronic daily headache (CDH) patients were given a diary to record, on a daily basis, the occurrence, severity and duration of the headache episodes. They were asked to make one assessment of their headache episodes by means of a visual analogic scale (0–100 score) during the 3 months of diary recording.
The study protocol was approved by the Ethics Committee of the Municipality of Perugia, and all patients gave written consent to the study. They were admitted to the Neurological Clinic of the Neuroscience Department of the University of Perugia to undergo lumbar puncture. All patients were in pain at the time of lumbar puncture.
Control CSF specimens were also obtained from 20 age- and sex
Methods
Sample collection
CSF (10 ml) was collected in polypropylene tubes containing EDTA (1 mg/ml) and kallikrein (500 IU/ml). For nitrite, cGMP and sensory neuropeptide determination, CSF samples were centrifuged at 4°C and CSF aliquots immediately frozen at −80°C. For glutamate determination, CSF samples were deproteinized with ice-cold 100 g/l trichloroacetic acid (TCA; 1 : 20 by volume); after centrifugation (3000
Determination of NO2 – CSF levels
Oxidation products of NO, nitrite and nitrate, into which this radical is rapidly converted in solution, were determined as total NO2 –, after enzymatic reduction of nitrate to nitrite, by fluorimetric high-performance liquid chromatography (HPLC) analysis with precolumn hydralazine derivatization, according to the method of Noda (19), with minor modification.
Briefly, CSF samples were thawed and an aliquot (0.1 ml) of the sample was incubated with 20 µl of buffer containing 50 m
Determination of glutamate CSF levels
Glutamate CSF levels were measured by HPLC, using o-phthalaldehyde precolumn derivatization and electrochemical plus fluorometric detection. A BioRad pump apparatus coupled to a Coulochem 5100A electrochemical detector (ESA Inc., Chelmsford, MA, USA) with a coulometric analytic cell (Model 5011) and a Jasco FP-920 fluorescence detector (Jasco Int.) was used with a C18 Rosil HL (150 × 4.6 mm i.d.; BioRad Laboratories Ltd, Hemel Hempstead, UK) reversed-phase column. The phosphate buffer, prepared from 25 mmol/l potassium phosphate and 150 ml/l acetonitrile, was applied as the mobile phase. The flow rate was 1.0 ml/min. Glutamate content was evaluated after comparison with an external glutamate calibration solution. Data in CSF specimens were expressed as µmol/l.
Sensory neuropeptides and cGMP CSF levels
SP, CGRP, and cGMP were eluted with 60% acetonitrile in 0.1% trifluoroacetic acid in SEP-C18 columns activated with 0.1% trifluoroacetic acid and 60% acetonitrile in 0.1% trifluoroacetic acid. Eluates were dried with a centrifugal concentrator (Supervap PL-CC-180). Residues were dissolved in radioimmunoassay (RIA) buffer and determined with RIA kits (Peninsula Laboratories GmbH, Belmont, CA, USA).
Standards for the above substances were dissolved in 0.1 mol/l phosphate buffer pH 7.5, containing 0.1% bovine albumin, 0.01% sodium azide and 500 kIU/ml kallikrein.
SP, NKA and CGRP human antisera were obtained from rabbit and were specific for the C-terminal end for all three neuropeptides.
SP antiserum cross-reacts with SP 2-11, SP 3-11, SP 4-11 and SP 5-11 (100%), and also SP 6-11 and SP 7-11, though only minimally. Cross-reactivities with endothelin 1, NKA and neuropeptide K are 0.5%, < 0.01% and 0.01%. The detection limit for SP is < 1 pmol/l.
NKA antiserum cross-reacts with kassidin (100%), porcine neuropeptide K (100%) and neurokinin B (80%), but shows a cross-reactivity of < 0.05% and 0.02% with SP and physalahemin, respectively. No cross-reactivity for the same antiserum is present with neuropeptide Y (porcine), neuromedin B (porcine), ACTH (human) and Arg8 Vasopressin. The minimum amount of immunoreactive NKA detectable by the assay is < 1 pmol/l. The intra- and interassay variabilities for the NKA radioimmunoassay are 5% and 8%, respectively.
CGRP antiserum showed a cross-reactivity with rat and chicken CGRP (100%), but not with other neuropeptides. The intra- and interassay variabilities were 3% and 6%, respectively. The detection limit of the assay is < 1 pmol/l.
Human cGMP antisera were obtained from rabbit. The intra- and interassay variation coefficients were 5% and 7%, and 4% and 6%, respectively, for the two cyclic nucleotides. The anti-cGMP does not show cross-reactivity with 5′-AMP, 5′-ADP, 5′-ATP, 3′,5′-cAMP, 5′-GMP, 5′-GDP, or 5′-GTP. The detection limit for cGMP is < 1 pmol/l.
Specificity data on SP, CGRP, NKA and cGMP are provided by Peninsula Laboratories.
Data were expressed as fmol/ml for SP, CGRP, NKA, and as nmol/ml for cGMP.
Statistical analysis
All values were expressed as mean ± 2 SEM. One-way within-subjects
Pearson correlation coefficients among CSF parameters and between these parameters and VAS values, duration of chronic headache and number of days with headache/month were also calculated.
Five percent for two-sided tests was chosen as a minimum level of statistical significance.
Results
Chemical analysis of the CSF revealed values of proteins, glucose, and lactate within the normal range both in patient and control groups, and the absence of cellular elements
Glutamate levels in the CSF of CDH patients were significantly higher than those measured in CSF of the control group (Fig. 1) (P < 0.0001). No statistically significant differences emerged between mean glutamate levels in the CSF of CDH patients without analgesic overuse and patients with analgesic overuse headache (Table 2).
Mean ± SEM of CSF values of NO2 –, cyclic guanosine monophosphate (cGMP) and glutamate in all patients, in chronic daily headache (CDH) patients without analgesic overuse and patients with analgesic overuse headache

Statistical significance vs. controls:
∗ P < 0.0001;
∗∗ P < 0.002;
∗∗∗ P < 0.0001;
† P < 0.0001;
†† P < 0.001;
††† P < 0.0002;
‡ P < 0.002;
‡‡ P < 0.003;
‡‡‡ P < 0.0001.
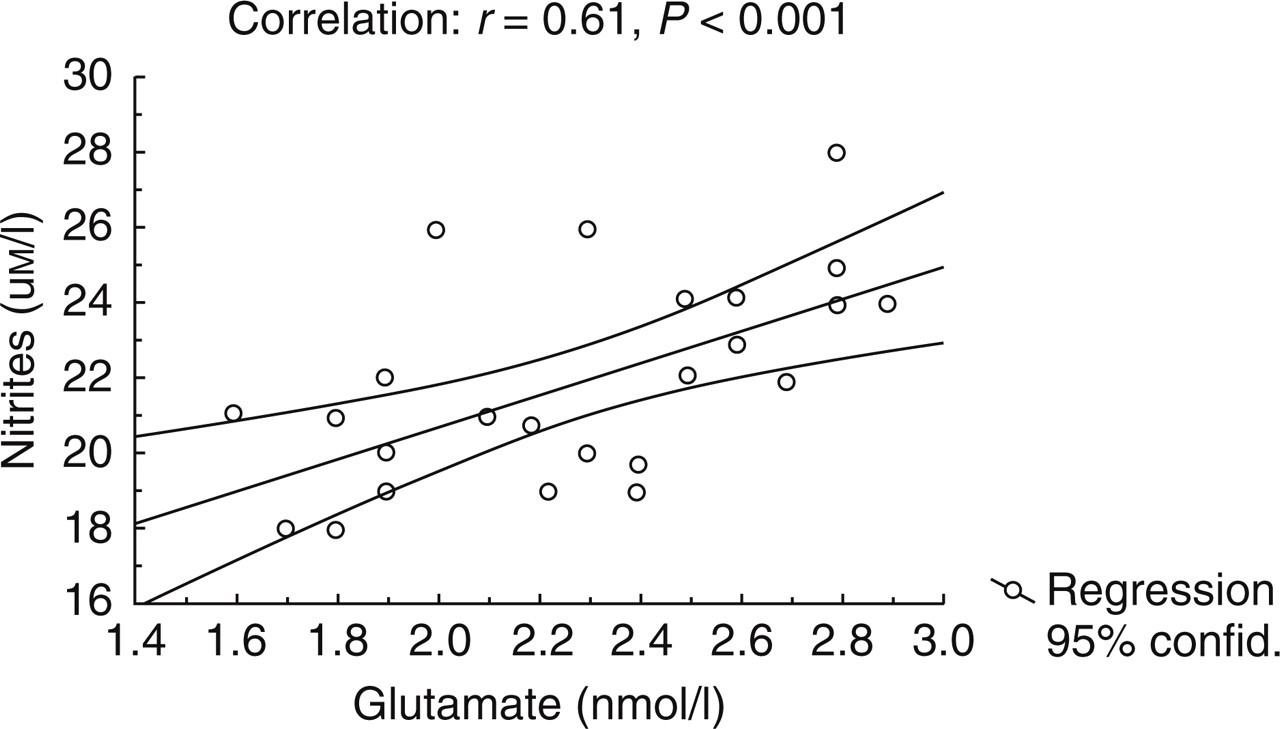
Correlation between glutamate and nitrite levels (both expressed in µmol/l) in CSF of patients with chronic daily headache.
A parallel increase in NO end-products in the CSF of CDH patients was found compared with the control group (P < 0.002), without a significant difference between the two patient groups (Table 2).
The increase in CSF nitrites was accompanied by a significant rise in CSF values of cGMP in patients in comparison with controls (P < 0.0001). The mean cGMP values of CDH patients without analgesic overuse were similar to those of patients with analgesic overuse headache (Table 3).
Mean ± SEM of CSF values of substance P (SP) and calcitonin gene-related peptide (CGRP) in all patients, in chronic daily headache (CDH) patients without analgesic overuse and patients with analgesic overuse headache

Statistical significance vs. controls:
∗ P < 0.001;
∗∗ P < 0.002;
∗∗∗ P < 0.003;
† P < 0.0005;
†† P < 0.001;
††† P < 0.002;
‡ P < 0.003;
‡‡ P < 0.004;
‡‡‡ P < 0.005.
In the CSF of the entire patient group glutamate and nitrite levels (Fig. 1), as well as nitrite and cGMP levels (Fig. 2), were significantly correlated. Consequently cGMP values showed a significant correlation with glutamate values (R = 0.70, P < 0.001).
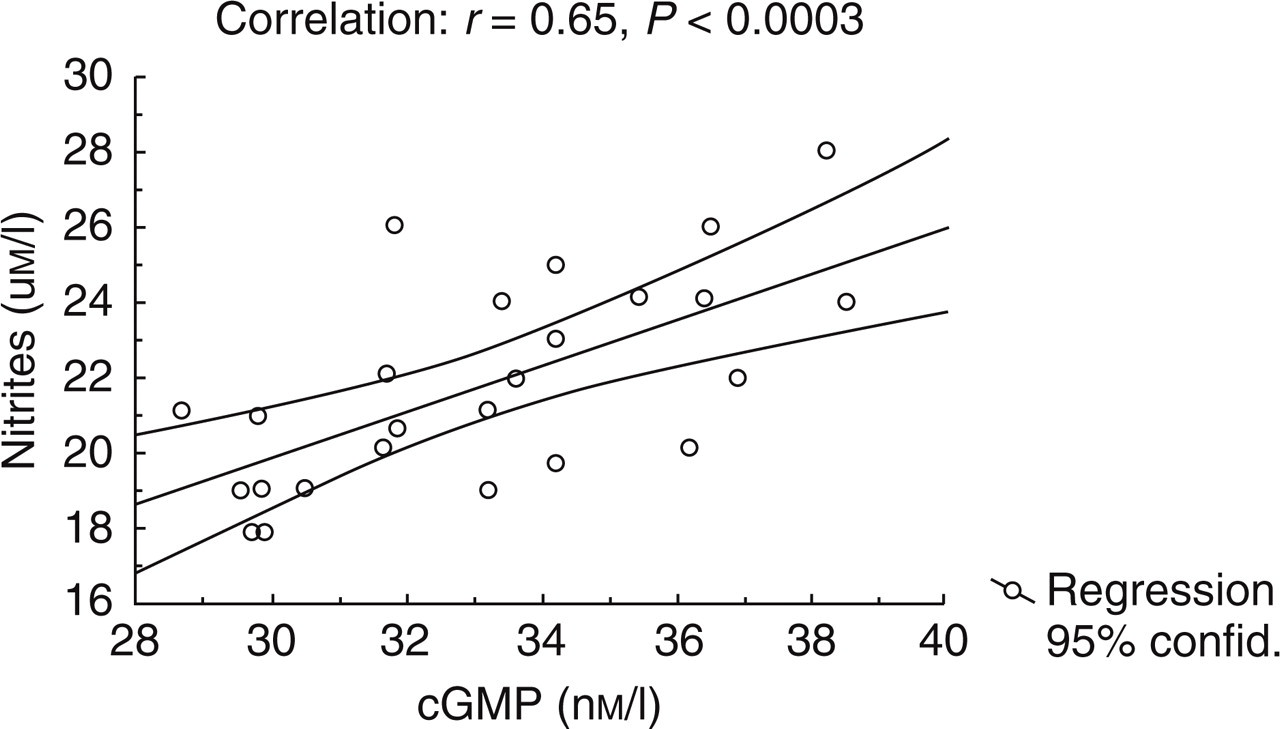
Correlation between nitrite (µmol/l) and cyclic guanosine monophosphate (cGMP) (nmol/l) levels in CSF of chronic daily headache patients.
Moreover, a statistically significant correlation emerged between VAS values and glutamate (R = 0.62, P < 0.008), nitrites (R = 0.54, P < 0.02) and cGMP (R = 0.75, P < 0.0001).
No significant correlation was found between the above CSF parameters and duration of CDH.
Although SP, CGRP and NKA were significantly increased in CSF compared with control subjects (P < 0.001, P < 0.0002 and P < 0.003, respectively) (Table 3), their values did not correlate with glutamate, nitrites and cGMP levels in CSF in the patient group. A slight but significant correlation was found between their values and patient VAS scores (SP: R = 0.44, P < 0.04 and CGRP: R = 0.49, P < 0.05), but not with chronic headache duration.
Discussion
Patients with CDH can often be encountered in headache specialty centres. This term indicates a heterogeneous group of headaches which includes different forms that occur daily, or almost daily over a prolonged period of time (17, 18, 20). The nosography of these forms is still a matter of debate.
Several epidemiological studies were carried out with the aim of identifying the clinical features of CDH (21–24). Some authors proposed a revision of the current criteria underscoring the need to categorize the most frequent subgroup of disorders, i.e. chronic tension-type headache and the so-called transformed migraine, not only on the basis of their prevailing clinical characteristics but also by taking into account their evolution (17, 18).
Most CDH patients overuse symptomatic medication, particularly analgesics, ergotamine, and more recently triptans, leading to a medication overuse headache, alternatively named drug-induced headache (25–28). These patients often respond to drug withdrawal with a significant reduction of head pain.
Apart from the difficult nosography and variability in the clinical features of CDH, one of the most important concerns is the pathophysiological substrate underlying the process by which head pain becomes chronic. Few studies of this have been carried out. Moreover, it has been hypothesized that patients with medication overuse headache could have different mechanisms underlying chronic head pain.
Central sensitization is believed to be one of the most relevant putative mechanisms underlying chronic painful conditions, including chronic head pain.
In conditions of chronic pain, nociceptors become sensitized, discharge spontaneously, and produce an ongoing and sustained painful condition. The resulting hyperalgesia and prolonged spontaneous pain are due both to increased sensitivity of peripheral nociceptors (primary hyperalgesia) and to facilitated spinal cord transmission (secondary hyperalgesia, receptive field expansion and allodynia) (29). Hyperexcitability of dorsal horn neurones and neurones of the trigeminal nucleus caudalis is first triggered by an increased neuronal barrage into the central nervous system (‘wind-up’) (30).
Prolonged firing of C-fibre nociceptors causes the release of glutamate, which acts on NMDA and non-NMDA receptors in spinal and supraspinal sites (31). Activation of NMDA receptors makes the spinal cord neurones more responsive to all nociceptive and also non-nociceptive inputs, resulting in central sensitization. Central transmission and hyperexcitability are mediated by excitatory amino acids (aspartate and glutamate), and by neurokinins (in particular SP) and other sensory neuropeptides (CGRP) acting via NMDA and non-NMDA receptors and neurokinin receptors, respectively (32).
NO has been demonstrated to play a crucial role in central sensitization in animal models of pain. NO formation within dorsal horns and the trigeminal nucleus caudalis by nociceptive afferent fibres is triggered by the activation of excitatory amino acid receptors, which results in an increased activity of nitric oxide synthase (NOS), mainly through Ca2+-dependent mechanisms (16).
It is well known that NO diffusing from the post-synaptic production site to the presynaptic glutamatergic fibres increases glutamate release which, on the other hand, acting on NMDA and non-NMDA receptors, further activates the NOS enzyme via Ca2+ increase, with a mechanism which can be defined as ‘long-term potentiation’.
These concepts taken from studies in experimental animal models of pain can be theoretically transferred to chronic head pain, but evidence of the above mechanisms needs to be clearly obtained in the patients affected (33).
In particular, a central sensitization, consisting of an increased neural excitability at different levels of the CNS, due to a prolonged nociceptive input from painful head structures (mainly vascular), is believed to play a role in CDH evolving from an episodic migraine, but the biological basis of chronic head pain has been poorly identified. No hypotheses have been formulated in this regard for medication overuse headache.
Based on the above findings from animal models of pain, the present study aimed at investigating the levels of the excitatory amino acid glutamate and end-products of NO metabolism, nitrites, and the NO messenger cGMP, in the CSF of patients with CDH without analgesic overuse and patients with analgesic overuse daily headache. CSF levels could better reflect changes in central structures involved in head pain processing in these patients compared with those in peripheral blood.
Some discussion of methodological considerations is necessary when CSF glutamate, nitrite and cGMP levels are chosen as biochemical markers of excitatory function and NO pathway in the CNS. Because the levels of free excitatory amino acids have been demonstrated to correlate with their levels in the brain, a reliable measure of their concentration in CSF is a potential useful tool for investigating synaptic excitatory transmission (34).
Nitrite levels in biological fluids have been demonstrated to be useful indicators of the activity of the L-arginine/nitric oxide pathway in humans, and have been suggested as a useful clinical marker for monitoring variations in NO production in many pathological conditions as well as in the effects of treatment on this production (35).
Due to its instability, NO produced as a consequence of NOS activation is, in fact, rapidly oxidized to nitrites, which are stable for several hours in plasma and CSF. The precise amount of nitrites which are converted to nitrates in CSF has not been established, whereas it has been clearly demonstrated that their basal concentration in whole blood is low, and that of nitrates is approximately 100 times higher due to the oxidation of nitrites by haemoglobin contained in erythrocytes. We measured NOx activity in CSF using HPLC, after the conversion of nitrites to nitrates. Nitrate levels in the diet have been demonstrated to affect the levels of NOx, particularly in blood samples, but the exact influence of nitrates ingested with the diet on NOx levels in CSF has not been clearly demonstrated. Although a better way to minimize this effect is to keep subjects on a nitrate-restricted diet for 5 days, we did not apply this restriction to patients because of the difficulty of applying the same restriction in control subjects, therefore avoiding the introduction of another variable which could influence the difference among groups.
Potential sources in the brain which could be advocated as the origin of NO, and consequently nitrites as its stable metabolites, are neural cells and glial or inflammatory cells, due to the activation of a constitutive and inducible NOS, respectively (31).
The intracellular messenger of NO is cGMP and its measurement has also been suggested as a marker of NO metabolism in biological fluids, including CSF. Although this messenger is mostly intracellular, it is possible to detect small amounts of this cyclic nucleotide also in CSF, not only in the case of increased production, but also in control individuals using sensitive RIA or enzymatic immunoassays with detection limits of a few fmoles.
In CSF the potential source of cGMP can be both neural and glial/inflammatory cells, as for NO. In our study, no leucocytes were found in CSF samples from either patients or controls. Their potential intervention in cGMP production can therefore be excluded and a prevailing neural origin hypothesized.
Glutamate levels have been poorly investigated in CDH (36) and have never been assessed in medication overuse headache. NO involvement was suggested in migraine by the results of the experimental model of nitroglycerin (NTG)-induced headache and the use of the NO inhibitor L-NG methylmonoarginine hydrochloride (L-NMMA) which appeared to relieve migraine attacks significantly (37–40). Data on NO involvement in chronic daily headache come from the NTG-induced headache and L-NMMA administration model, whereas increased NO endogenous production has been clearly shown in a platelet model in CDH patients (2, 41–43).
The present study supports the increase in glutamate release in the CSF of patients affected by CDH without medication overuse and medication overuse headache patients compared with controls, without a significant difference between the two patient groups. This release is accompanied by a parallel increased production of NO, as shown by increased levels of its end-products.
Both glutamate and NO levels appear to be significantly correlated, indicating a close relationship between activation of NMDA and non-NMDA receptors and intracellular production of NO in spinal and supraspinal sites implicated in chronic sensitization in the above patients.
Among the structures involved in central sensitization, the trigeminal nucleus caudalis and also the upper spinal cord may be hypothesized to be involved in increased glutamatergic transmission and NO production. Some sites of the brain stem producing both facilitatory and inhibitory influences on spinal dorsal horns and the trigeminal nucleus caudalis could also be implicated. Among them the rostral ventromedial medulla, other brain stem areas and periaqueductal grey matter play a central role in nociception (44, 45). At supraspinal levels, however, the role of NO is debatable, with contrasting findings regarding the nociceptive and anti-nociceptive effect of NO on pain sensation (46, 47). This aspect should be further clarified in future research.
In this study, the intracellular messenger cGMP appears to contribute to sustained nociception, as suggested by its increase, parallel to that of glutamate and nitrites, in the CSF of our patients.
Several findings in experimental models of pain suggest its production in the case of sustained glutamate receptor activation followed by persistent production of NO in nociceptive processing in both spinal and supraspinal pain pathways (48).
In our research we found a significant increase in the CSF levels of sensory neuropeptides SP, CGRP and NKA. This increase could express their release from the small-diameter C nociceptors in the spinal cord and the trigeminal nucleus caudalis as a consequence of their continuous activation related to the chronic pain state. They can contribute to evoking excitation in second-order neurones acting on their specific receptors.
In our research the increased levels of glutamate and nitrites, as well as those of cGMP, did not parallel the increase in sensory neuropeptides.
These negative findings do not exclude an interaction between sensory neurones producing and releasing SP, CGRP, NKA and glutamate receptors located in the presynaptic terminals of small-diameter nociceptors in the spinal cord and the trigeminal nucleus caudalis. In these sites they are believed to facilitate and prolong nociceptive input by increasing the release of sensory neuropeptides. In addition, the potentiating effect of sensory neuropeptides on glutamate-evoked responses in central sensitization can also not be excluded.
It can be hypothesized that persistent nociception in CDH and medication overuse headache can result from the recruitment of ionotropic glutamate receptors and perhaps the facilitation of slow calcium channels by metabotropic receptors, and, at the same time, by the summation of slow excitatory potentials mediated by SP and other sensory neuropeptides. The latter represent additional mechanisms underlying sensitization of head pain pathways which, although influencing NMDA activation, play a separate, independent role in synaptic plastic changes accompanying sensitization of pain pathways in the upper spinal cord and trigeminal nucleus caudalis.
The above mechanisms could be related to pain per se, and may not be specific for chronic daily headache or medication overuse daily headache, as suggested by recent findings in another painful condition, i.e. fibromyalgia (49).
Our data concur with previous findings of alteration in central nociceptive circuits in patients with CDH, consisting of an amplification and temporal summation of ‘second pain’ (50).
In the same patients oral administration of a weak antagonist of NMDA receptors, dextromethorphan, has been shown to reduce the increased temporal summation of second pain. This effect was also augmented by the contemporaneous administration of magnesium (51).
These data therefore suggest, although indirectly, the involvement of excitatory amino acids in central nociception in CDH patients, which was clearly demonstrated by our results, and the possibility of influencing head pain by inhibiting excitatory amino acid transmission in central neuronal circuits involved in the processing of nociceptive information.
On the basis of the assumption that CDH is characterized by an increased activity of excitatory amino acid pathways, the use of antagonists of N-methyl-
Finally, based on the demonstration that NOS inhibition reduces central sensitization in models of chronic pain and efficacy in chronic tension-type headache, the analgesic effect of L-NMMA, a non-selective NOS inhibitor, and its usefulness in clinical practice need to be established in CDH. The use of selective inhibitors for neural and endothelial forms of NOS can also be proposed for clarifying the peripheral and central components involved in chronic head pain evolving from a previous history of migraine (54).
