Abstract
5-HT1D (but not 5-HT1B)-receptor immunoreactivity (i.r.) can be detected on trigeminal fibres within the spinal trigeminal tract of the human brainstem. The present study used immunohistochemical and morphometric techniques to determine the proportions of trigeminal fibres expressing substance P, CGRP or 5-HT1D-receptor immunoreactivities. Co-localization studies between 5-HT1D-receptor and substance P- or CGRP-i.r. were also performed. Brainstem material was obtained with consent (four donors) and the total number of immunoreactive fibres within the trigeminal tract was estimated using random field sampling. A greater proportion of fibres (>1 μm diameter) expressed CGRP-i.r. (80 ± 6%) compared with substance P-i.r. (46 ± 7%) or 5-HT1D-receptor-i.r. (25 ± 1%). 5-HT1D-receptor-i.r. was co-localized on some CGRP- or substance P-i.r. fibres. This suggests that 5-HT1D-receptors can regulate the release of CGRP and substance P and may be relevant to the clinical effectiveness of 5-HT1B/1D-receptor agonists in the treatment of migraine and other cranial pain syndromes.
Introduction
Triptans are 5-HT1B/1D-receptor agonists and at least part of their therapeutic anti-migraine activity has been attributed to their ability to inhibit trigeminal activation, often considered as one of the hallmarks of migraine headache pain (1–4). This action of triptans is well established in experimental preclinical assays. In anaesthetized animals, trigeminal activation evoked by electrical stimulation of the perivascular fibres causes neurogenic vasodilation of meningeal arteries and also increased firing of neurones within the trigeminal nucleus caudalis (5–7). In addition, high-intensity stimulation of the trigeminal ganglion results in plasma protein extravasation within the dura mater (8) and increased c-fos expression (a marker for neuronal activation) in the caudal nucleus (9). The neurogenic vasodilation response is mediated via the release of endogenous CGRP (5, 6) and extravasation and increased c-fos expression by substance P (9). The ability of triptans to block both CGRP and substance P-mediated events (10, for review 11) suggests that these 5-HT1B/1D-receptors are located prejunctionally on substance P and/or CGRP-containing sensory fibres.
Previous immunohistochemical studies have shown that in rat trigeminal ganglia there is co-localization of CGRP and 5-HT1B-receptor immunoreactivities (12). However, there are species differences (13). Previous studies in our laboratory have shown that in man, whilst there is expression of both 5-HT1B/1D-receptor immunoreactivities in trigeminal cell bodies, there is selective expression of 5-HT1D-receptor immunoreactivity on meningeal perivascular fibres and on the central projections which terminate in the trigeminal nucleus caudalis (14).
Therefore, the primary aim of the present study was to determine the neuropeptide content of these 5-HT1D-immunoreactive trigeminal fibres by co-localization of the 5-HT1D-immunoreactivity (i.r.) with either CGRP or substance P immunoreactivity. In addition, we performed quantitative studies within the spinal trigeminal tract to examine the proportions of trigeminal fibres which express either 5-HT1D-, CGRP- or substance P-i.r.
Methods
Blocks of medulla oblongata were obtained post mortem (with consent) as routine histopathological samples from Addenbrooke's Hospital, Cambridge, UK. The collection of human material was approved by the local Ethics Committee. Quantification of the proportion of fibres expressing either 5-HT1D-, CGRP- or substance P-i.r. was performed in transverse sections of the spinal trigeminal tract and the co-localization studies were performed using sections obtained from higher in the brainstem at approximately the level at which the descending fibres enter the tract to allow maximum visualization of fibres in the longitudinal orientation.
Preparation of sections for immunohistochemistry
Following fixation (10% formal saline) blocks of medulla were embedded in paraffin wax, sectioned (7 µm), collected on to Fisher Superfrost/plus slides (Merck, Poole, UK) and air-dried. All sections (including those for single immunostaining and those for co-localization studies) were incubated (37°C, overnight), de-waxed (Histoclear), rehydrated in graded alcohols and endogenous peroxidases inhibited by immersion in 0.3% H2O2 in physiological buffered saline (PBS, Sigma, Dorset, UK; 30 min, room temperature). Following washing (3 × 5 min per wash) in PBS, sections were subjected to antigen-retrieval techniques (15) by immersion in Citra solution (pH 6.0; Biomen, Wokingham, UK) and microwaving (full power, 5–20 min, Panasonic 800 W), and then cooled under running water.
Estimation of number of 5-HT1D-, CGRP- and substance P-i.r. fibres within the spinal trigeminal tract: immunostaining procedure
Adjacent sections were used for immunostaining with 5-HT1D, CGRP, substance P antibodies and PGP9.5 as a neuronal marker. The source of the antibodies and working dilutions were: 5-HT1D-receptor, rabbit polyclonal MSD/Affiniti, 1 : 300; CGRP, rabbit polyclonal, Amersham, 1 : 1000; substance P, rat monoclonal Chemicon 1 : 3000; PGP9.5, rabbit polyclonal, Biogenesis, 1 : 1000. The sections were exposed to the primary antibody (overnight, 4°C, saturated humidity), washed (3 × 5 min, PBS) and then exposed to the secondary antibody (biotinylated goat anti-rabbit (for CGRP) or anti-mouse (for substance P) IgG, Vectastain Elite Kit, 1 h, room temperature). Following washing (3 × 5 min, PBS), sections were incubated in Avidin–Biotin Complex (Vectastain Elite Kit, 1 h, room temperature), washed (3 × 5 min, PBS) and immersed in di-amino benzidine solution (Menarini, Wokingham, UK). The reaction was terminated by immersion in distilled water. Light microscopy was performed on a Leica (DMRB) microscope using normal bright field illumination.
Estimation of the number of 5-HT1D-, CGRP- and substance P-i.r. fibres within the spinal trigeminal tract: morphometric procedure
Morphometric techniques were used to estimate the proportion of fibres in the spinal trigeminal tract expressing either 5-HT1D-, CGRP- or substance P-i.r. The area of the spinal trigeminal tract was defined under low magnification (× 100) and then immunoreactive profiles were estimated by applying uniform random sampling. An unbiased counting frame (900 µm2) was used and 10 uniform random fields within the area of the spinal trigeminal tract were counted. Individual fibres were visualized under high magnification (× 1000) and a single ‘dot’ of immunoreactivity was assumed to represent a single fibre. This procedure was followed for spinal trigeminal tract obtained from four individual donors. The mean area of the trigeminal nucleus caudalis (at the same level within the brainstem in the four donors) was calculated as 32 mm2 and this value used to estimate the total numbers of immunoreactive fibres. Note: fibres with diameter <1 µm were beyond the limits of accurate resolution using the current microscopy methodology.
Co-localization of 5-HT1D-i.r. and neuropeptides
Sections were first prepared for immunocytochemistry as described above. Then for the first layer 5-HT1D-receptor antibody, sections were incubated in 5% normal goat serum (5% NGS, in PBS, >30 min, saturated humidity) and then transferred to 5% NGS containing the primary 5-HT1D-receptor antibody (overnight, 4°C, saturated humidity). Following washing (PBS, 3 × 5 min), sections were incubated (1 h, room temperature) with the secondary antibody biotinylated goat anti-rabbit serum (1 : 200 Vectastain Elite Kit), washed (5 × 30 min, PBS/Triton buffer) and incubated (1 h, room temperature) in a modified 0.1
Then for the second layer neuropeptide antibodies, the same sections were again incubated in 0.3% H2O2/PBS (60 min) to inhibit any residual unreacted peroxidases, and endogenous proteins were blocked by incubation in either 5% normal goat or rabbit serum (1 h, room temperature) for experiments with the CGRP or substance P antibodies, respectively. The sections were transferred to either 5% normal goat or rabbit serum (as appropriate) containing either anti-CGRP or anti-substance P antibodies (overnight, 4°C, saturated humidity chamber). The sections were washed (3 × 5 min, PBS), incubated (1 h, room temperature) with the biotinylated secondary antibody (goat anti-rabbit 1 : 200 or rabbit anti-rat 1 : 200 for the CGRP or substance P studies, respectively; Vectastain Elite Kit) and washed (3 × 5 min, PBS). The immunoreactive signal to second antibody was visualized using the Tyramide Signal Amplification protocol of the Renaisance Kit (DuPont/NEN, described above), using the rhodamine tyramide label (red fluorescence).
Immunofluorescence was visualized on the Leica DMRB using epifluorescence with appropriate filters. Photographs were taken (1600asa film) with no winding on between single colour fluorescence exposures. Only the two colours (green and red) corresponding to 5-HT1D and substance P or CGRP were recorded.
Results
Co-localization of 5-HT1D-i.r. and neuropeptides in human spinal trigeminal tract
Fibres within the human trigeminal tract were shown to express substance P- or CGRP-i.r. No attempts were made to co-localize substance P and CGRP, although the total numbers of the substance P- plus CGRP-containing fibres was greater than the total number of PGP9.5-immunoreactive fibres suggesting that some fibres contain both neuropeptides. Some of the substance P and some CGRP-i.r. fibres were shown to co-localize with 5-HT1D-immunoreactivity (see Fig. 1 for representative findings).
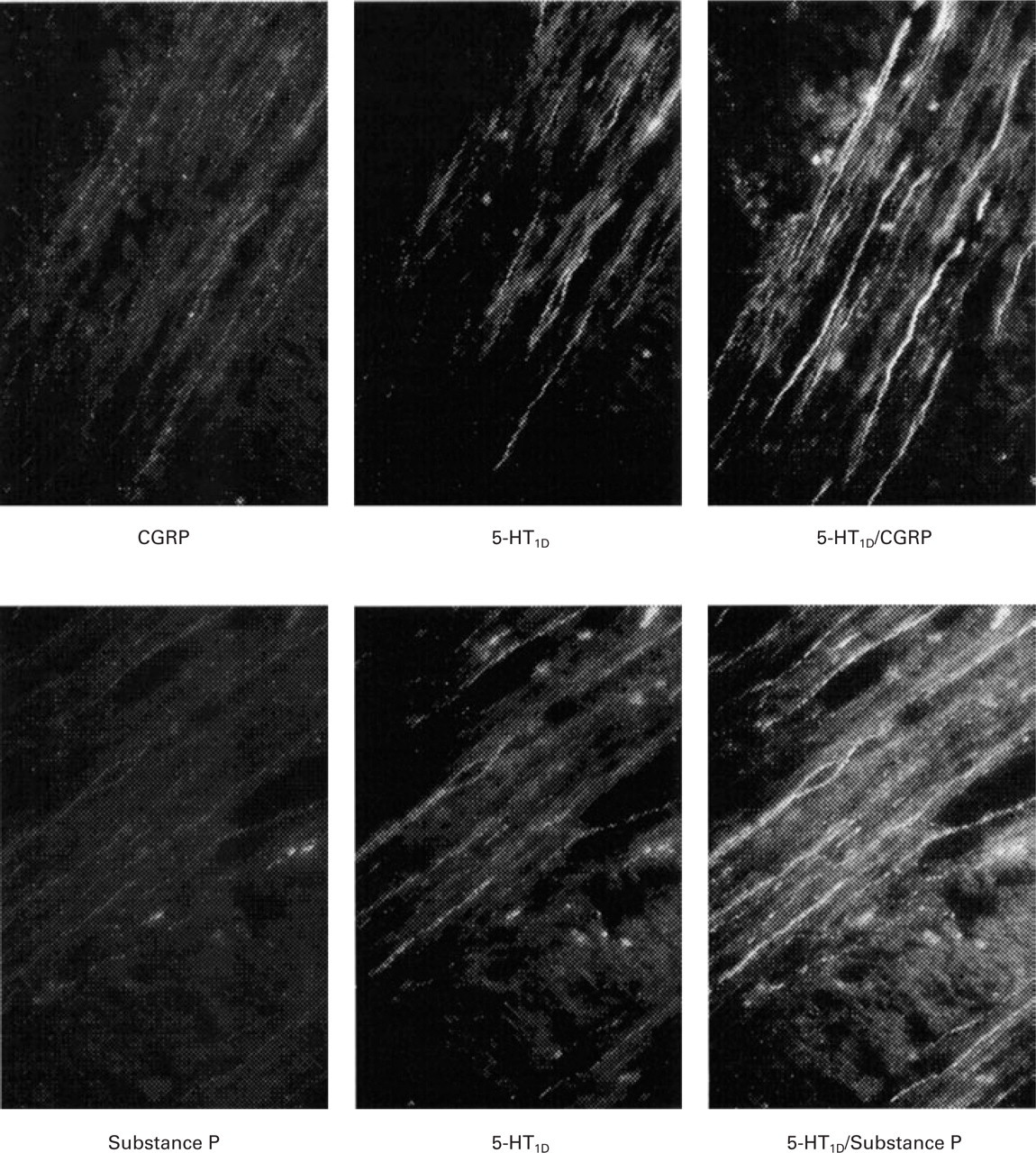
Typical photographic examples of the co-localization of 5-HT1D-receptor and neuropeptide immunoreactivity in human spinal trigeminal tract. Brainstem sections (7 µm) were stained with either CGRP (upper row, left panel) or substance P primary antibodies (lower row, left panel) and immunoreactivity was visualized using rhodamine tyramide (red fluorescence). Sections were also stained with the 5-HT1D-receptor primary antibody and immunoreactivity was visualized using fluorescein tyramide (green fluorescence, middle panels). Co-expression of 5-HT1D-receptor immunoreactivity with either CGRP or substance P is given by yellow/orange fluorescence (right panels). Immunofluorescence was visualized using a Leica DMRB microscope using epifluorescence with appropriate filters.
Estimation of the numbers of 5-HT1D-, CGRP- or substance P-containing fibres within the spinal trigeminal tract
Figure 2 shows a representative section to indicate the brainstem level at which the quantitative studies were performed and shows typical fibre immunostaining. The mean results determined using morphometric techniques are shown in Fig. 3 and Table 1. PGP9.5 was used as a marker to estimate the total number of fibres. Most of the PGP9.5-immunoreactive fibres were of ‘small’ diameter (approximately 2 µm); however, some ‘larger’ diameter fibres (approximately 5 µm) were also observed. (Substance P-containing, unmyelinated c-fibres are of 0.2–1.5 µm diameter (16) and therefore were largely excluded from the present analysis.) The density of PGP9.5-immunoreactive fibres was high, and it is possible that some of the ‘larger’ 5-µm fibres may have masked the presence of smaller fibres in the vicinity. Therefore the absolute numbers of nerve fibres may be an underestimate.
Numbers of trigeminal fibres (with diameters >1 µm) within the human spinal trigeminal tract expressing substance P, CGRP, 5-HT1D-receptor or PGP9.5-immunoreactivity
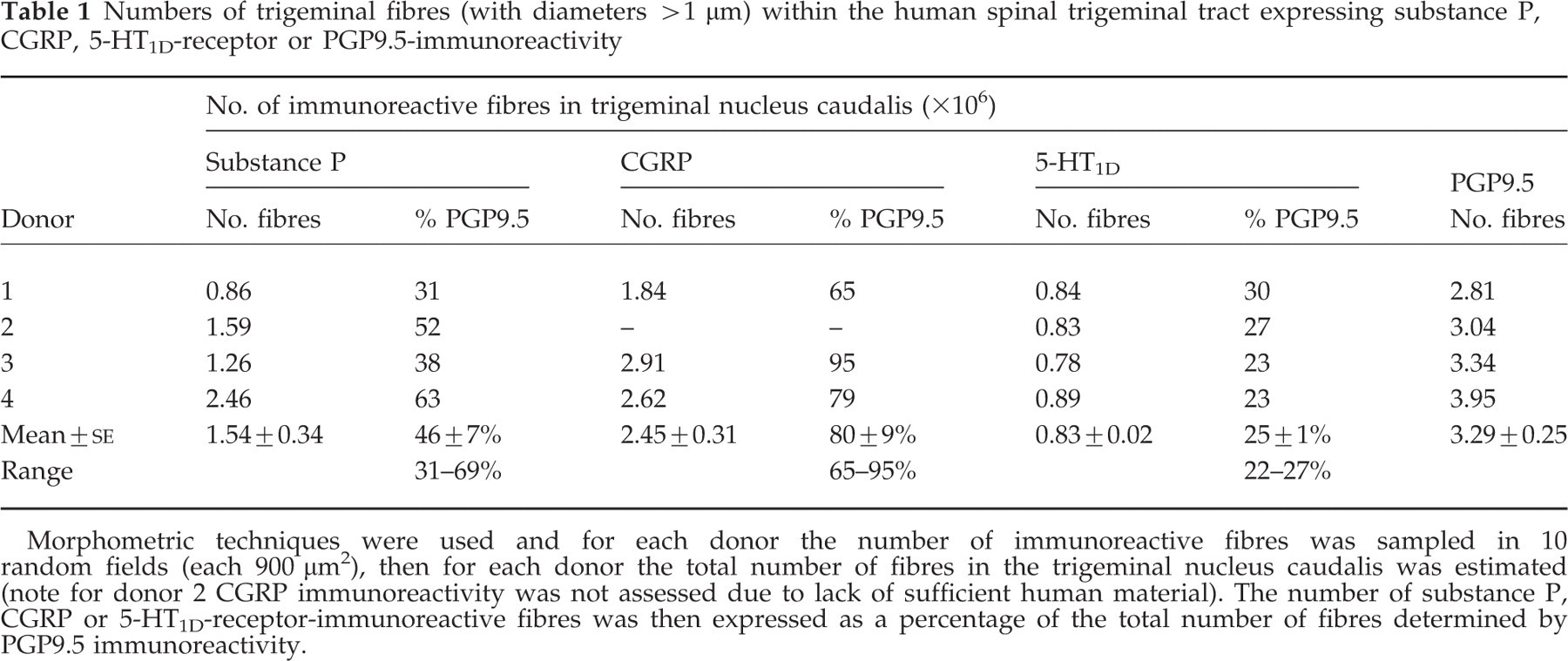
Morphometric techniques were used and for each donor the number of immunoreactive fibres was sampled in 10 random fields (each 900 µm2), then for each donor the total number of fibres in the trigeminal nucleus caudalis was estimated (note for donor 2 CGRP immunoreactivity was not assessed due to lack of sufficient human material). The number of substance P, CGRP or 5-HT1D-receptor-immunoreactive fibres was then expressed as a percentage of the total number of fibres determined by PGP9.5 immunoreactivity.

The left-hand panel shows a low power photograph of the DAB immunostaining in the spinal trigeminal tract which is representative of the sections used for the morphological studies. The right-hand panel shows a typical high-power photomicrograph (original mag. × 1000) of a section used for counting ‘dots’ of immunoreactivity.
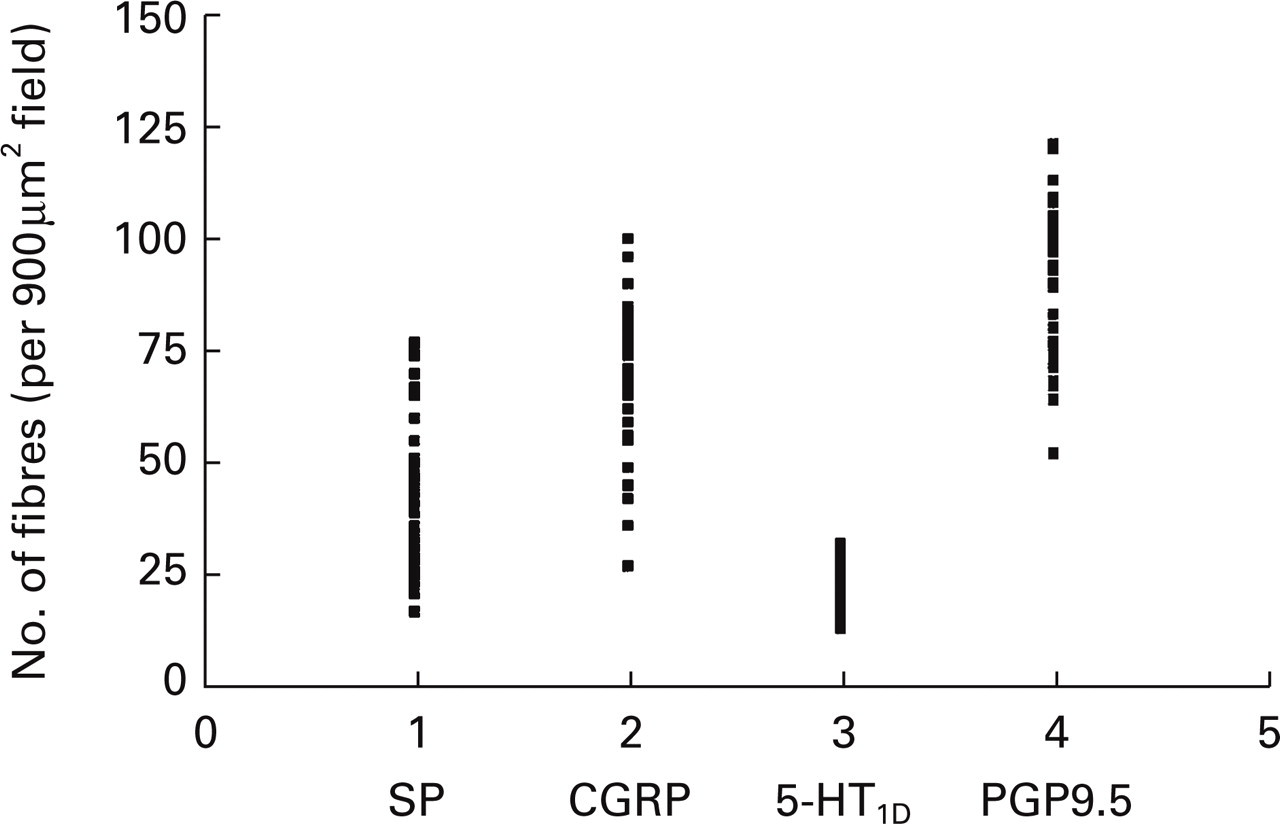
A scattergram indicating the numbers of substance P, CGRP, 5-HT1D-receptor and PGP9.5-immunoreactive fibres (>1 µm diameter) detected in the trigeminal nucleus caudalis. The number of immunoreactive fibres per uniform random field (900 µm2) was counted. Each point shows a fibre density estimate in brainstem sections (10 per antibody) obtained from four donors (except for donor 2, where CGRP immunoreactivity was not assessed due to lack of sufficient human material).
Substance P-i.r. was detected on some of the ‘large’ (5 µm) diameter fibres and also on some of the ‘small’ (approximately 2 µm) diameter fibres. The proportion of PGP9.5-immunoreactive fibres (>1 µm) containing substance P was 46 ± 7% (mean±
CGRP-i.r. was detected on some ‘small’ (approximately 2 µm) diameter fibres but was not detected on any of the ‘large’ (5 µm) fibres. The proportion of fibres of PGP9.5-i.r. fibres (>1 µm) containing CGRP was 80 ± 9% (mean±
5-HT1D-i.r. was detected on some of the ‘large’ (5 µm) diameter fibres and also on some of the ‘small’ (approximately 2 µm) fibres. The proportion of PGP9.5-i.r. fibres (>1 µm) expressing 5-HT1D-receptor immunoreactivity was 25 ± 1% (mean±
Discussion
Co-localization studies
This study suggests the co-localization of 5-HT1D-receptor immunoreactivity on CGRP-i.r. and on substance P-i.r. fibres within the human spinal trigeminal tract. The findings are in agreement with those by Hou et al. (17) in the human trigeminal ganglia. Not all substance P-containing fibres co-expressed 5-HT1D-i.r. and not all CGRP-containing fibres co-expressed 5-HT1D-i.r. The CGRP antibody used (anti-rabbit CGRP) was selected since it gave good-quality staining for the quantification studies. However, for the co-localization experiments the 5-HT1D-receptor and CGRP antibodies were raised in the same species, giving rise to the possibility that the apparent co-localization was due to antibody cross-reactivity, although precautions were taken to minimize this. However, whilst this is possible, it is likely that a substantial part of the co-localization was real, since the immunosignals did not always overlap and some CGRP-i.r. fibres did not co-express 5-HT1D-i.r. This is in general agreement with Hou et al. (17), who showed that in human trigeminal ganglia (using antibodies raised in different species) there was co-localization of CGRP and 5-HT1D-i.r. in medium sized neurones with the majority (96%) of 5-HT1D-i.r. neurones co-expressing CGRP-i.r.
CGRP is a potent, long-lasting vasodilator, and neurogenic vasodilation may be an important factor in migraine pathogenesis. The present study suggests that within the spinal trigeminal tract CGRP-positive trigeminal fibres co-express 5-HT1D-i.r., i.e. 5-HT1D-receptors are positioned to fulfil their role as an inhibitory receptor in regulating the CGRP release from sensory fibres. Although it has been previously shown that 5-HT1D-i.r. is present on perivascular fibres within the dura mater (14), it was not possible to conduct similar co-localization studies in dura mater, due to low fibre density which did not allow satisfactory double immunostaining, and it can only be assumed the co-localization seen on central fibres also applies to peripheral trigeminal fibres. The release of CGRP centrally may have important consequences in nociceptive processing, but it is difficult to assess the relative contributions of 5-HT1D-receptor-mediated inhibition of CGRP release from central terminals vs. inhibition of peptide release from peripheral perivascular terminals. The role of CGRP in regulating brainstem excitability is not well defined and is hampered by the lack of suitable, non-peptide CGRP-receptor antagonists. Recently PNU-142633, a selective, peripherally acting 5-HT1D-receptor ligand (that would rely on inhibition of neurogenic vasodilation or extravasation for its anti-migraine activity), was reported to be ineffective in the clinic (18). However, this ligand had weak efficacy in activating 5-HT1D-receptors (19) and a fully efficacious agonist may be a more successful strategy that requires testing clinically.
The present study demonstrated the expression of 5-HT1D-i.r. on substance P-containing fibres within the spinal trigeminal tract, suggesting that 5-HT1D-receptors can regulate substance P release. Inhibition of substance P release may not be important for the relief of migraine headache pain. It seems unlikely that activation of nociceptive, substance P-containing fibres is the primary driver of migraine headache pain, since there is no evidence for elevated substance P levels in migraineurs (20) and NK1-receptor antagonists have failed as acute or prophylactic migraine therapies (21–23). In experimental animals NK1-receptor antagonists failed to inhibit neurogenic vasodilation within the meninges or increases in facial blood flow evoked by stimulation of trigeminal pathways, yet these responses were inhibited by CGRP-receptor blockade (5, 24). Interestingly, increased substance P levels are found in saliva from patients with tension-type headache (25) and in cerebrospinal fluid from patients with trigeminal neuralgia (26). Thus, the presence of 5-HT1D-receptors on substance P-containing trigeminal fibres may suggest the use of 5-HT1B/1D-receptor agonists in the treatment of other cranial pain syndromes. Indeed, patients with tension-type headache responded to sumatriptan (27).
Quantification studies
In animals, afferent fibres innervating the dura mater have been characterized electrophysiologically as Aδ or C-fibres (7, 28) with diameters in the expected range of 1–5 µm and 0.2–1.5 µm, respectively (16). It has been reported that in man more than 90% of fibres within the trigeminal nucleus are small diameter fibres, i.e.<4 µm (29). However, Usunoff et al. (30) showed that some thicker fibres >6 µm can be detected. In the present study, quantification of the proportion of fibres (>1 µm, probably myelinated Aδ fibres) expressing substance P, CGRP or 5-HT1D-immunoreactivity was performed in the trigeminal tract.
Myelinated Aδ fibres contain both CGRP and substance P (31–33). In the present study in human spinal trigeminal tract, CGRP-i.r. was detected only in ‘smaller’ diameter fibres (approximately 2 µm) and the majority of these fibres expressed CGRP. The proportion of CGRP-i.r. fibres was higher (approximately two-fold) than the proportion of substance P-containing fibres, suggesting that substance P does not always co-localize with CGRP in fibres of this size. This is consistent with the greater numbers of CGRP-containing cell bodies in the trigeminal ganglia (34).
Substance P-i.r. was found on both ‘larger’ diameter fibres (i.e. 5 µm) and ‘smaller’ (2 µm) fibres. The possibility that the larger fibres represent bundles of smaller fibres can be discounted since the majority of smaller fibres (2 µm) were CGRP-positive and none of the larger ‘5-µm’ fibres was CGRP-positive, therefore it seems likely that the 5-µm fibres represent true fibres. The identity of the larger substance P-positive fibres is not known. They may be ascending second-order trigeminal neurones projecting to the hypothalamus (35). They are unlikely to be primary sensory afferents, although in guinea pig it has been shown that a small proportion of cutaneous Aα/β (6–12 µm diameter) fibres express substance P-i.r. (33). Further studies are required to clarify this issue.
5-HT1D-i.r. was detected on approximately 25% of PGP9.5-i.r. fibres within the spinal trigeminal tract, and this was relatively consistent (range 22–27%) across donors. It is tempting to speculate that the relatively low proportion of 5-HT1D-i.r. fibres and the interindividual variation of the numbers of CGRP fibres (65–95% across donors) may impact on the ability of 5-HT1D-receptors effectively to regulate CGRP release. This in turn may underlie the variability associated with therapeutic response to the triptans in acute migraine.
The study has a number of caveats, nevertheless the experiments are important since they were carried out in human brainstem and are important for understanding migraine pathophysiology, the therapeutic activity of 5-HT1B/1D-receptor agonists and the development of novel anti-migraine strategies. First, the co-localization experiments were performed in the spinal trigeminal tract, where it was possible to view fibres in longitudinal sections and enhance the visualization of the co-localized immunoreactivities. The area of the brainstem studied was likely to contain small diameter myelinated and unmyelinated fibres, and it was not possible to specifically examine meningeal afferent fibres. With respect to the treatment of migraine, inhibition of neuropeptide release from meningeal afferents within the perivascular region is often considered important for inhibition of neurogenic vasodilation or extravasation. Other experimental studies show that following trigeminal stimulation, triptans inhibit facial flushing (a CGRP-mediated response) and prevent extravasation in facial tissues (11), suggesting that 5-HT1B/1D-receptors are located on divisions of the trigeminal nerve which innervate these cranial structures. Inhibition of neuropeptide release from non-meningeal afferents with the trigeminal nucleus caudalis may also be important for anti-migraine activity. Cumberbatch et al. (36) showed with the caudal nucleus that there was facilitation or sensitization of neurones, receiving convergent inputs from both the meninges and face. Therefore, the inhibition of the central release of CGRP (including meningeal and non-meningeal afferents) may be important for anti-migraine activity.
Second, it should be remembered that the absence of neuropeptide or receptors in a particular fibre may reflect that the level of peptide or receptor expression is too low to be detected by the antibody used. In addition, due to the technical limitations of light microscopy the fibres studied are >1 µm diameter and probably represent small myelinated Aδ-sensory fibres, and consequently a large proportion of unmyelinated, mainly substance P-containing fibres are excluded from the quantitative analysis, and therefore the role of substance P may be underestimated.
Conclusion
In the human spinal trigeminal tract, immunocytochemical studies showed that 5-HT1D-immunoreactivity was expressed on some substance P- or CGRP-containing fibres (with diameters in the 1–5 µm range). The presence of 5-HT1D-receptors on CGRP-containing fibres is relevant to the anti-migraine activity of brain penetrant 5-HT1B/1D-receptor agonists, whereas the expression of 5-HT1D-receptors on substance P-containing trigeminal fibres may be more relevant to other disorders, e.g. tension-type headache.
Footnotes
Acknowledgements
Thanks to David Shaw and Reyhana Kajee for carrying out most of this experimental work.
