Abstract
Cluster headache (CH) typically presents in clusters of attacks of intense (peri)orbital, unilateral pain. The distribution of the pain implies involvement of central and/or peripheral trigeminal pathways. These can be investigated by means of trigeminal somatosensory evoked potentials (TSEP) and blink reflexes (BR). We aimed to relate functional changes in trigeminal sensory pathways to the presence of cluster periods. TSEP and BR were performed in 28 episodic CH patients during a cluster period and repeated in 22 outside a cluster period. TSEP latencies (N1, P1 and N2) and amplitude (N1-P1 and P1-N2) and BR latencies (R1, R2 ipsilateral and R2 contralateral) were compared between sides, during and outside a cluster period and with healthy control data (n = 22). During a cluster period, N2 TSEP latencies were longer on the symptomatic side compared with the non-symptomatic side (27.2 ± 3.0 ms vs. 26.3 ± 3.4 ms, P = 0.02), and compared with the same side outside the cluster period (26.7 ± 3.1 ms vs. 25.1 ± 3.0 ms, P = 0.01). N1, P1 and N2 latencies on the symptomatic side in patients during the cluster period (14.8 ± 2.3 ms, 20.4 ± 2.5 ms and 27.2 ± 3.0 ms, respectively) were significantly longer than those of healthy controls (13.4 ± 1.9 ms, 18.8 ± 2.4 ms and 25.0 ± 2.6 ms, respectively, P < 0.03). Outside the cluster period, N1 latencies of both sides (15.3 ± 2.8 ms symptomatic side and 15.4 ± 2.6 ms asymptomatic side) were longer compared with controls (13.4 ± 1.9 ms, P < 0.04). TSEP amplitudes and BR latencies revealed no significant differences. We conclude that abnormalities of the afferent trigeminal pathway are present in patients with cluster headache, most prominent during the cluster period, and on the symptomatic side. This seems primarily due of changes within the higher cerebral regions of the system.
Keywords
Introduction
Cluster headache (CH) is characterized by frequent attacks of intense, strictly unilateral, periorbital pain behind or around one eye, accompanied by one or more ipsilateral symptoms of facial autonomic dysfunction. Attacks of CH usually last 15–180 min and typically are clustered in periods of weeks to months (‘clusters’) during which attacks may occur up to eight times per day. In between these cluster periods, patients are usually free of symptoms. In addition to the hypothalamus (1), other central and peripheral structures have been invoked to explain the pathophysiology of CH. The distribution of the pain suggests involvement of trigeminal somatosensory pathways, further supported by the activation of the trigeminovascular system during CH attacks (2).
Trigeminal somatosensory evoked potentials (TSEP) and blink reflex (BR) studies allow assessment of the central and peripheral parts of sensory trigeminal pathways, and we have shown them to be sufficiently reproducible and sensitive for use in repeated measurements (3). TSEP measurements assess the functional integrity of the trigeminal sensory traject, from peripheral afferents up to and including the cortex (4). The BR on the other hand is a brain-stem reflex, involving the first branch of the trigeminal nerve and motor efferents of the facial nerve (5).
In the present study, we used TSEP and BR to assess (i) changes during compared with outside a cluster period, (ii) differences between the symptomatic and non-symptomatic side, and (iii) differences between patients and healthy volunteers.
Methods
Study population
Patients were recruited from a nationwide scientific database, including CH and CH-like syndromes, established at the LUMC Department of Neurology since 1998. Patients with episodic CH according to the International Headache Society criteria, and with strictly one-sided CH, were included. Patients who were on prophylactic medication for CH, who had had surgical treatment of the trigeminal nerve or thermolesion of the sphenopalatine ganglion, or who had other neurological disease in addition to CH, were excluded from the study.
Design
Bilateral TSEP and BR were performed once during a cluster period, but not during the actual attack, and repeated several weeks to months later outside a cluster period. The time between the previous and subsequent attacks and the measurements was noted.
Twenty-eight patients were included and measured during the cluster period. Six of them could not be measured outside the cluster period, because of logistic reasons (n = 3), or because of an unusually long continuation of their cluster period (n = 3). Repeated TSEP and BR measurements were thus obtained in 22 patients. Data of healthy controls described in a previous paper were used for comparisons (3). The study was approved by the medical ethical committee and informed consent was given by all subjects.
Trigeminal somatosensory evoked potentials
Procedures have been described in detail elsewhere (3). In short, for TSEP measurements, subjects lay down with their eyes closed, keeping their facial and jaw muscles as relaxed as possible. The unaffected side was stimulated first. Electrical stimuli (400 at 3.7 Hz, 0.2 ms duration) were delivered to both sides on the corner of the mouth in all subjects at three times sensory threshold. EEG electrodes were placed at C5′ and C6′ of the 10–20 system (6), using Cz as the ground site and Fz as the reference site. Two individual trial blocks of 400 stimuli were averaged for contralateral and ipsilateral responses of both sides; cathode and anode were switched after 200 stimuli to combat stimulus artefact. Latencies of contralateral N1, P1 and N2 and amplitude (N1-P1 and P1-N2) were evaluated (Fig. 1).
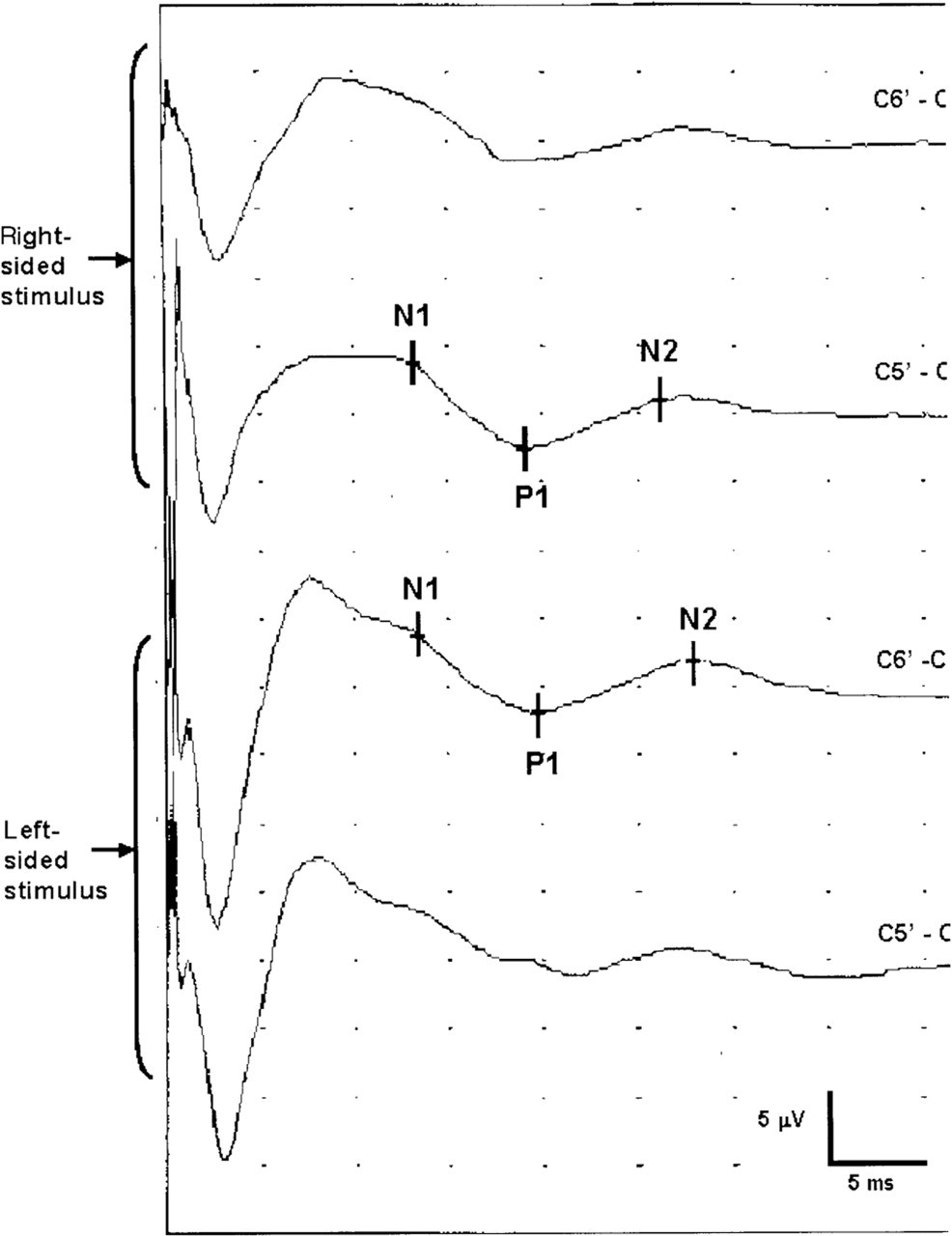
Representative example of a trigeminal somatosensory evoked potential measurement. N1, P1 and N2 latencies are indicated for contralateral responses after right-sided and left-sided stimulation.
Blink reflex
Subjects were seated on an examination chair with their eyes open. Electrodes were placed on the orbicularis oculi muscle, below the eye and lateral to the eye on both sides. The ground electrode was placed on the subjects’ forehead. The supraorbital nerve was stimulated directly over the supraorbital notch. The current intensity of the stimulus was set at 10 mA, but increased if necessary until stable, reproducible reflexes were obtained. Four blink reflexes were obtained on both sides, with unpredictable intervals of at least 10 s to minimize habituation. Signals were amplified and averaged. Responses were stored for off-line analysis. Latencies of R1 and R2 responses were evaluated with use of Matlab software. Amplitudes and area were not used, as we earlier found these impracticable for longitudinal studies (3).
The TSEP and BR responses were analysed in one run by one of the authors, who was not directly involved in the measurements and who was blinded for the time of the measurement, during or outside a bout, and for the affected side.
Statistical analysis
Side-to-side differences were evaluated in the 28 patients who were measured during the cluster period; the effect of the cluster period was evaluated in the 22 patients who were measured both during and outside the cluster period. Responses were also compared with healthy control data. We used the Wilcoxon signed ranks test for related samples statistics (during vs. outside cluster period and symptomatic vs. non-symptomatic side) and the Mann–Whitney test for independent sample statistics (patients vs. healthy controls). Spearman's correlation coefficient was used to assess the influence of the time between the previous and subsequent attacks on the measurements. P < 0.05 was considered statistically significant.
Results
Subject characteristics are listed in Table 1. The time between the two measurements ranged between 6 and 40 weeks. Mean age (SD) did not differ significantly between controls (40.1 (12.9) years) and CH patients (44.8 (8.6) years). The elapsed time from the beginning of a cluster period to the time of the first measurement was 4.1 ± 0.9 weeks. The second measurement was performed 10.0 (1.9) weeks after the last attack of the previous cluster period. TSEP and BR parameters and sensory threshold are shown in Table 2.
Subject characteristics

Data are expresses as mean (SD), or n (%).
Mean trigeminal somatosensory evoked potential (TSEP) and blink reflex (BR) latencies of patients and healthy controls
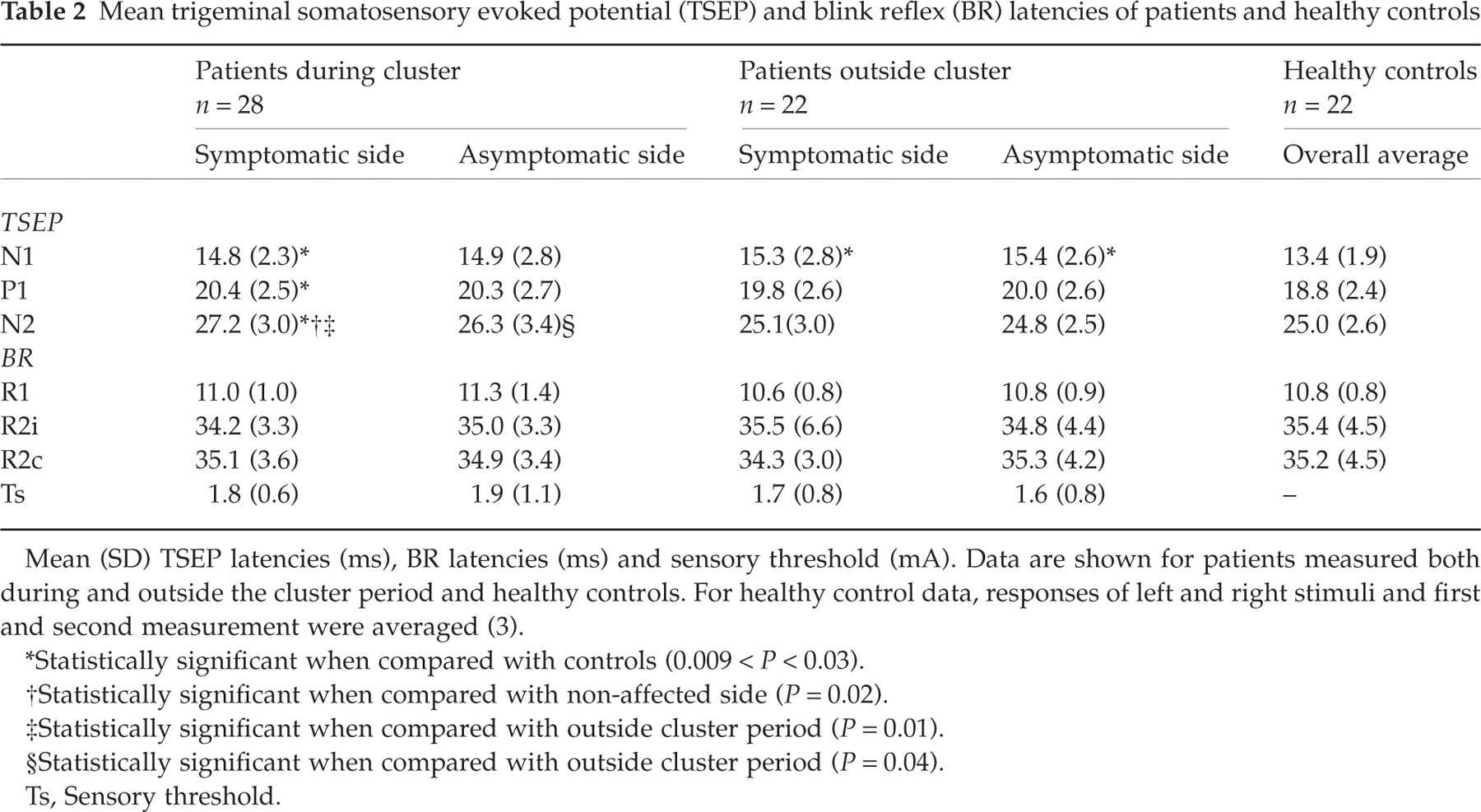
Mean (SD) TSEP latencies (ms), BR latencies (ms) and sensory threshold (mA). Data are shown for patients measured both during and outside the cluster period and healthy controls. For healthy control data, responses of left and right stimuli and first and second measurement were averaged (3).
∗Statistically significant when compared with controls (0.009 < P < 0.03).
†Statistically significant when compared with non-affected side (P = 0.02).
‡Statistically significant when compared with outside cluster period (P = 0.01).
§Statistically significant when compared with outside cluster period (P = 0.04).
Ts, Sensory threshold.
During the cluster period (n = 28), N2 latency was longer on the affected side than on the non-affected side (P = 0.02) and N1, P1 and N2 TSEP latencies were significantly increased on the affected side compared with controls (P < 0.03). When measurements during a cluster period were compared with those outside a cluster period (n = 22), N2 latencies of both sides were significantly longer during the cluster period than outside the cluster period (P = 0.01 and 0.04, respectively) (Fig. 2). Outside the cluster period, N1 latencies were significantly longer on both sides in patients compared with controls (P < 0.03), but no side-to-side differences were found. TSEP amplitudes revealed no significant differences.
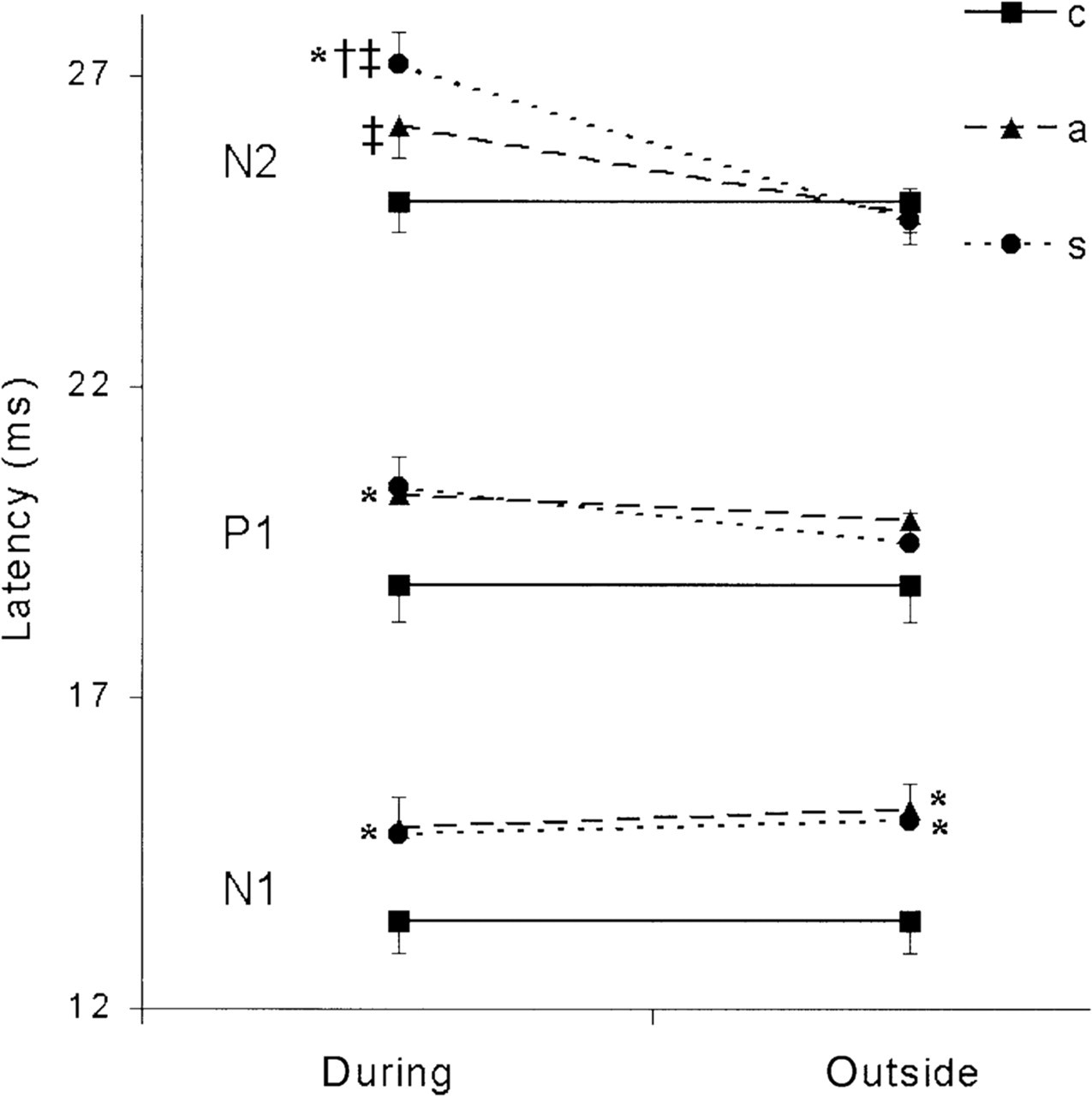
Mean trigeminal somatosensory evoked potential latencies. Mean latencies during and outside the cluster period of patients (symptomatic and asymptomatic side) and of healthy controls are shown (with SEM). For control data, latencies of both sides and both measurements were averaged. ∗Significant differences compared with controls. †Significant difference compared with the non-affected side. ‡Significant difference compared with the same side outside a cluster period.
No statistically significant difference was found between the sensory threshold of the affected vs. the non-affected side during or outside the cluster period. For BR data, no differences in R1 and R2 latencies were found.
There was no effect of the time that had passed since the last attack (mean 21 ± 4.3 h, P > 0.08), or the time until the next attack (mean 26 ± 5.4 h, P > 0.09) on any TSEP or BR parameters.
Discussion
In the present study we found increased TSEP latencies in CH patients compared with healthy controls, mostly during the cluster period. In addition, the increase was more pronounced on the symptomatic side during the cluster period. The partial normalization outside the cluster period implies a functional change of these pathways rather than a structural lesion. BR and TSEP share a peripheral afferent conduction pathway; as BR latencies were normal and TSEP latencies were not, a higher cerebral lesion in the trigeminal sensory system is more likely than merely peripheral or pontine changes. The fact that the N1 latencies further increased outside the bout, in contrast to N2 latencies, suggests that these two latencies may have altered independently.
Increased TSEP latencies have also been found in other studies in CH on the symptomatic side (7) and during attacks (8) and in trigeminal neuralgia patients (9–12). However, a temporal relationship with the cluster period was not assessed, as patients have not been measured repeatedly (7). Further, other methods and smaller patient groups were used (8), making comparisons between these studies and ours difficult. These differences notwithstanding, the studies share impaired stimulus processing of the trigeminal sensory system in patients with CH and trigeminal neuralgia, more pronounced on the affected side.
As many comparisons were made, correction of the P-values for multiple comparisons (e.g. Bonferroni's correction) could be considered appropriate. However, many of the comparisons are related. In such a case, the Bonferroni correction is too conservative. Due to the exploratory nature of the study, we decided not to correct for multiple comparisons, and therefore the reader should be aware of the increased type I error of the analysis (meaning that some of the significant findings would be considered less or not significant with a more conservative approach).
In accordance with some studies (7, 13), we found no differences in BR latencies comparing patients and controls, nor in comparisons within patients. In contrast, other studies did report asymmetrical blink reflex responses in CH patients. These studies did not evaluate differences within the same patient, or used other BR parameters, such as the R3 response, or BR threshold (14–16). However, these parameters have not been investigated in longitudinal studies. Increased recovery of R2 after paired supraorbital stimuli on the symptomatic side was found in one study, suggesting hyperexcitability of spinal trigeminal neurones (13).
TSEP studies evaluate the function and integrity of the sensory trigeminal system from the peripheral nerve up to and including the sensory cortex, while BR involves mainly peripheral nerves and brain-stem structures. Based on our results we suggest that peripheral trigeminal afferents or pontine structures are probably not involved in CH, as only TSEP responses showed changes. A central mechanism is in accordance with the recently demonstrated hypothalamic involvement in CH (1). However, we cannot conclude with certainty that the first division of trigeminal nerve is not affected in CH, since a discrepancy exists between the findings of BR measurements, which involve the first branch of the trigeminal nerve, and TSEP measurements involving the second and third branch.
Obviously, both BR and TSEP are generated by stimulation of sensory nerve fibres and are not specific for nociception (17, 18). Measuring pure nociception usually needs painful stimuli and may therefore not be preferable in patient studies. Recently, BR stimulation with a non-traumatic nociception-specific stimulator was performed in healthy volunteers (19), which probably allows evaluation of nociceptive transmission in a more comfortable way, and may be more sensitive for abnormalities in CH patients. On the other hand, our data may be used to hypothesize that sensory abnormalities in CH are not limited to nociception.
The trigeminal nerve and its central pathways have a pivotal role in the trigeminovascular system (TVS), known to be activated during CH attacks (20). The results of our study imply a permanent central dysregulation of sensory transmission in CH patients, more pronounced during the cluster period and on the attack side. Hypothetically, cortical sensory neurones may be more sensitive to external or internal stimuli during a cluster period, leading to a lower attack threshold. Central neuroplasticity, the capacity of neurones to change their function, structure or chemical profile (21, 22), may be responsible for this phenomenon.
As functional hypothalamic changes have been shown in CH patients during attacks (23), and functional abnormalities of the trigeminal sensory transmission system are found in this study, a pathophysiological model integrating these two systems could be of particular interest. Although it is unknown if the trigeminal sensory system is specifically controlled by the hypothalamus, a few studies suggest that the hypothalamus does affect general nociceptive control (24). The hypothalamus is known to receive nociceptive afferents from the brain-stem reticular formation. It also projects to the periacqueductal grey matter, which has inhibitory control on trigeminal and spinal nociceptive transmission (25).
We conclude that permanent abnormalities of the afferent trigeminal pathway are present in patients with CH, most prominent during the cluster period, and on the symptomatic side. As the responses of the BR showed no differences, a higher cerebral lesion in sensory transmission seems more likely than merely peripheral or pontine changes.
Footnotes
Acknowledgements
J.A.v.V. is supported by the Asclepiade foundation.
