Abstract
The aim of the study was to evaluate the laser-induced suppression periods of the temporalis muscle in patients with tension-type headache, compared with the pattern of temporalis activity suppression induced by electrical stimulation.
Fifteen patients with chronic and 10 with episodic tension-type headaches were selected. Suppression periods were recorded simultaneously from both temporalis muscles using both electrical stimuli and CO2-laser stimuli.
A significant reduction in the later electrically induced suppression period was found in both tension-type headache groups. Laser stimulation induced a first suppression period (LSP1) with a latency of about 50 ms in all patients. The features of LSP1 were similar across groups.
The LSP1 should correspond to the first suppression period induced by electrical stimulus, which is partly a nociceptive response, whereas the second period seemed negligibly linked with the activation of pain-related afferents, though probably their activation may contribute to increase the reflex duration and to emphasize abnormalities in tension-type headache.
Introduction
Electrical or mechanical stimulation in the oral region causes reflex inhibition of the jaw-closing muscles, the temporalis (or masseter) inhibition reflex (exteroceptive suppression period, ESP). This reflex inhibition appears as an early and a late phase of suppression (1) (silent periods, referred to as ESP1 and ESP2, respectively). The late temporalis ESP2 is reduced, on average, in patients with chronic tension-type headache (2), though this finding was not confirmed by more recent studies (3). ESP2 is produced by the activation of brain-stem interneurones, which inhibit the motoneurones of the jaw-closing muscles via a polysynaptic neural net (4). The location of the interneurones responsible for ESP2 is uncertain, but they probably belong to the bulbar reticular, which receives afferents from limbic structures, the orbito-frontal cortex, the nucleus raphe magnus, and the periaqueductal grey matter. All these structures are involved in the pain control system, and the reduced duration of ESP2 in chronic tension-type headache has been attributed to inadequate control of the inhibitory interneurones exerted by the serotoninergic raphe magnus nucleus and the periaqueductal grey matter (2). Although analysis of the ESP is considered to be an interesting method for objective and standardized analysis of the pain mechanisms involved in headache (5, 6), the nociceptive nature of ESP2 is contentious. Recently, a study performed blindly with a large sample of carefully diagnosed patients with chronic tension-type headache (CTTH) failed to reveal any abnormalities of ESP2 (3), so the biological significance of ESP2 requires elucidation. Probably because electrical stimuli may produce both tactile and nociceptive inputs, it remains controversial whether the first, the second, or both components are nociceptive. Recently, purely nociceptive reflexes to radiant heat stimulation of the facial skin were described: the laser blink in the orbicularis oculi and the laser suppression period (LSP) in the masseter (7–9). The threshold and onset latency of the LSP consisted of an electromyographic (EMG) inhibition period: the latency was assessed at almost 50 ms (8) or 70 ms (7) and its significance was controversial, resembling the electrically induced ESP1 (8) or ESP2 (7). The aim of the study was to re-evaluate the LSP in the temporalis muscle in normal controls and in a selected group of patients with tension-type headache, compared with the temporalis activity suppression pattern induced by electrical stimulation (ESP).
Methods
Subjects
Twenty-five patients with tension-type headache were selected: 15 were affected by chronic tension-type headache and the remainder by the episodic form (ETTH), according to the International Headache Society (IHS) criteria (10). Ten healthy controls were also included. All subjects gave their informed consent before their inclusion in the study, which was approved by our department. They included four men and 21 women, aged 25–53 years: the duration of illness varied from 4 to 17 years in CTTH group (mean 7.5 ± 5.1 years) and from 3 to 18 years in ETTH series (mean 8.2 ± 6.5 years) and the frequency of headache was 16–20 days/month (mean 18.3 ± 2.9 days) in CTTH and 2–9 days/month in ETTH group (mean 6.2 ± 2.2), over the last six months. All patients had been attending the practice for at least 12 months. Ten sex- and age-matched controls (7 women and three men, aged 22–46 years), with no history of headache or other craniofacial pain according to IHS criteria (10), were selected. All subjects underwent a standardized interview and clinical, neurological, and psychiatric examinations. Patients with general medical, neurological or psychiatric diseases, according to DSM-IV (11), and patients who were taking psycho-active drugs or prophylactic treatment for headache, or who were assessed as having overused analgesic drugs in the previous two months, were excluded from the study.
All selected headache sufferers were asked to attend the recording session in a pain-free state and to avoid taking any medication for at least the preceding 48 h.
Procedure
EMG signals were recorded simultaneously from both temporalis muscles by surface electrodes, with the active electrode over the anterior belly of the muscle and the reference electrode in front of the tragus. A grounded electrode was fixed in the middle of the forehead. All values for interelectrode impedances were less then 5 kΩ. Before applying a stimulus, subjects were requested to maximally contract both temporalis muscles, checking the effect of contraction by visual feedback. A pretrigger interval of 100 ms was used. The electrical stimulus (single pulses of 200 µs duration) was applied percutaneously by surface electrodes at the mental foramen. Before the assessment, electrical stimulation was administered at 2 mA and increased in 2-mA steps. A series of 20 stimuli for each intensity level were delivered, to define the individual detection threshold (DT) and the pain threshold (PT). Both the subject and the experimenters wore protective goggles or glasses during data acquisition with laser stimulation. The laser stimulus was a single pulse generated by a CO2 laser (wavelength 10.6 µm; Neurolas, Electronic Engineering, Florence, Italy). The beam diameter was 2.5 mm and the duration of the stimulus pulse was 45 ms. Signals were amplified, filtered (bandpass 5–2000 Hz), and stored using a biopotential analyser (Micromed System 98, Mogliano Veneto, Italy). Analysis time was 300 ms and the sampling rate was 4096 Hz. The sites irradiated were shifted slightly for each stimulus, to minimize the effects of skin burns, nociceptor fatigue, and central habituation.
To define a threshold for detection (LDT) and a threshold for pain (LPT) for laser stimulation, single stimulus-pulses were presented at six to eight different intensities, starting at an intensity of 1.5 W. A series of 20 stimuli at each intensity level were presented at intervals of 20–40 s. The intensity was increased in increments of 1.5 W. After the presentation of each stimulus, the subject was requested to report the quality of sensation, using a visual analogue scale (VAS) in which 0 represented no pain, and 10 represented the most severe pain imaginable. The subjective LDT was defined arbitrarily as the intensity at which the subject rated the perceived sensation at level 2 (corresponding to a warm or hot sensation) for at least 50% of the total number of stimulus presentations. Similarly, the LPT was defined as level 4 (pinprick sensation followed by a burning sensation) for at least 50% of stimulus presentations. We stopped increasing the stimulus intensity when level 6 was reached, corresponding to moderate pain.
We chose to consider the electrical ESP elicited at the intensities corresponding to the individual pain sensation and the LSP elicited at an intensity available to obtain in all subjects a pain sensation over the threshold, corresponding to the level 6 of the VAS, according to Ellrich et al. (8). After threshold determination, the temporalis inhibitory reflex was evoked on both sides by 10 electrical and laser pulses using interstimulus intervals of 30 s. The laser and electrical induced SP were recorded in a random order, and a time of at least 30 m was interposed between the recording sessions. Both electric and laser stimuli were delivered on both sites in a random order, and the ipsilateral and controlateral muscular responses were recorded. The 10 trials for each stimulus series were rectified and averaged off-line. The mean amplitude of the 100-ms pretrigger EMG activity was taken to be 100%. An ESP was defined, according to the criteria of Cruccu et al., (12) as more than 50% depression of EMG activity. The latency and duration of ESP1, ESP2, LSP1, and LSP2 were measured blindly with regard to patients, mode of stimulus, side of recording at the points at which rectified EMG activity exceeded 80% of the pretrigger EMG, according to Ellrich et al. (8). One-way
Results
The mean intensity of the electrical stimulus corresponding to the individual DT was 4.5 ± 0.9 mA in CTTH patients (n = 15), 3.8 ± 0.6 mA in ETTH patients (n = 10), 4.2 ± 0.6 in control subjects (n = 10) on the right side, and 4.4 ± 0.8 mA in CTTH, 3.8 ± 0.6 mA in ETTH, and 3.6 ± 0.8 mA in control subjects for stimulation of the left side. The mean electrical PT was 4.6 ± 1 × DT in CTTH patients, 4.7 ± 1.9 × DT in ETTH patients, 5.2 ± 1.6 in controls on the right side, and 4.7 ± 0.9 × DT in CTTH, 5.5 ± 1.9 in ETTH, 6 ± 1.6 × DT in the control group when the left side was stimulated. One-way
Onset latencies and durations of suppression periods by ipsilateral and contralateral laser and electrical stimuli in chronic tension type patients, episodic tension type patients and controls.
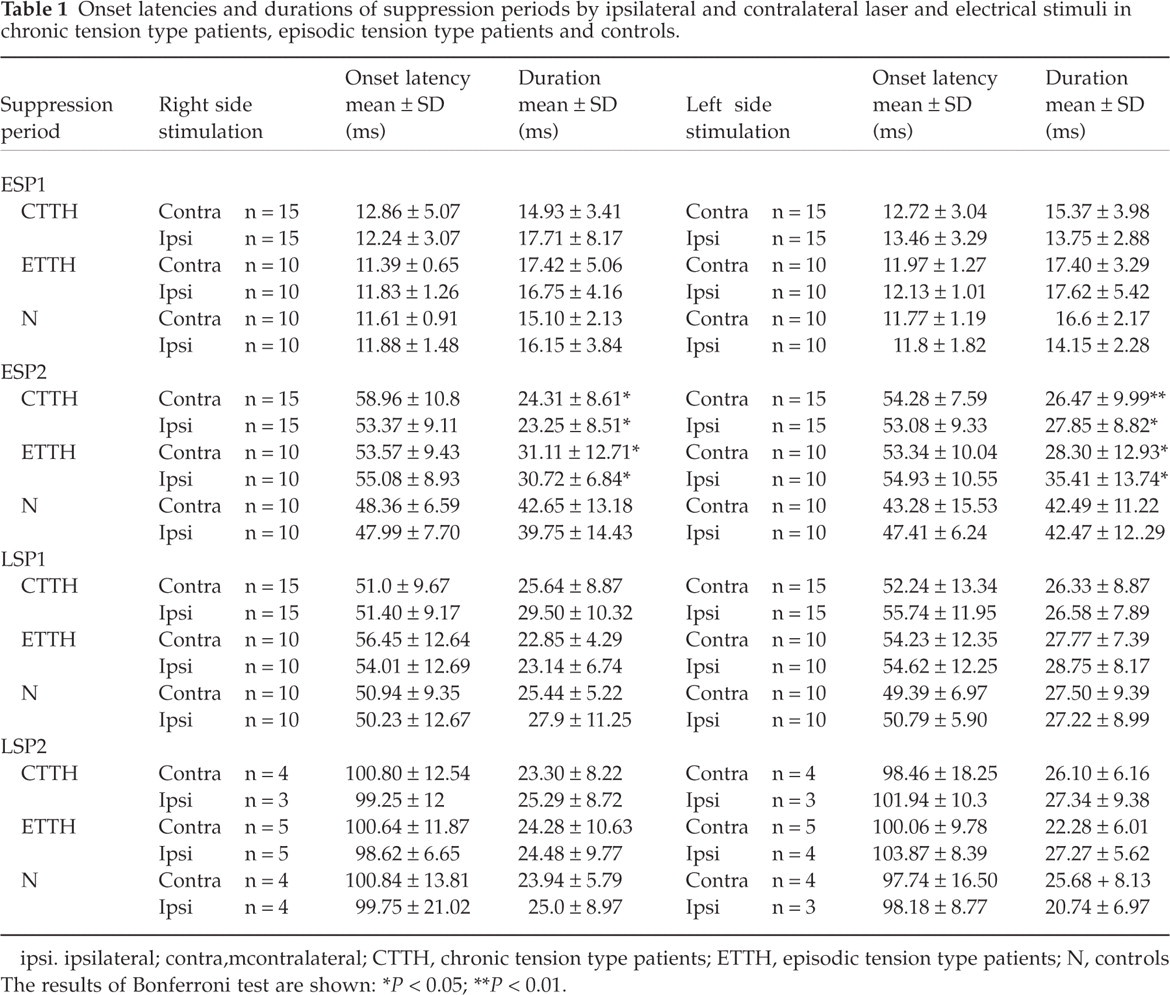
ipsi. ipsilateral; contra, mcontralateral; CTTH, chronic tension type patients; ETTH, episodic tension type patients; N, controls The results of Bonferroni test are shown:
∗ P < 0.05;
∗∗ P < 0.01.

Temporalis inhibitory reflex induced by electrical and laser stimuli with right-side stimulation. The average for 15 patients with chronic tension type headache and 10 normal controls. One rectified and averaged sweep was considered for each case. a, b and A, B are the ipsilateral and contralateral responses to electrical stimulation, respectively; c, d and C, D are the ipsilateral and contralateral responses to laser stimulation, respectively.
In all cases, a first suppression period with an onset latency of almost 50 ms was induced by ipsilateral and contralateral laser stimulation, and its features were similar across groups (Table 1). The differences in onset latency of ipsilateral LSP1 and ESP1 were 37.37 ± 9.75 ms in the CTTH group, 41.91 ± 12.9 in the ETTH group, and 38.35 ± 8.99 in controls for right-side stimulation, and 40.34 ± 6.4 ms in the CTTH group, 38.02 ± 8.8 ms in the ETTH group, and 39.31 ± 6.69 ms in controls for left-side stimulation. A later suppression period, at almost 100 ms, was observed in a few patients and controls: among tension type headache groups, it was detectable only in eight patients for the right side and in seven for the left side stimulation, moreover four and three control subjects showed this later response when the right and left sides were, respectively, stimulated (Table 1).
Discussion
According to previous findings by Ellrich et al. (8), a suppression period with a latency of around 50 ms was induced in all patients by laser stimulation, and a second period with latency values of about 100 ms was detected only in some patients and controls. These suppression periods were undoubtedly induced by Aδ fibre stimulation. We are not in keeping with results by Cruccu et al. (7) about LSP1 latency. The scoring of the suppression period is always problematic because they do not begin and end abruptly. Thus, differences in manual scoring procedures are probably also a source of variability in results across studies. The different reflex features may also be subtended by the variability of the intensity and duration of the stimulus, though we adjusted the stimulus features one step over the subjective pain perception, according to Ellrich (8) and also Cruccu & Romaniello (7) found the individual LSP threshold in a range of 4–10 times over the perceptive threshold; moreover the duration of the laser stimulus employed in our study was longer in respect with that reported by Cruccu et al. (7). Comparing the laser-induced responses with those elicited by electrical stimuli, the differences in onset latency were consistent with the range of latency of the nociceptor response (13). Therefore, according to Ellrich et al. (8), the first laser-induced period should correspond to the electrically induced ESP1. The second laser-induced suppression period was evident in a few patients and controls, and may correspond to the electrically induced ESP2 (8). Differently, Cruccu & Romaniello (7) claimed that the LSP1 may correspond to the electrically induced ESP2 on the basis of its recovery after conditioning stimulus. In our study the features of the LSP1 were similar across groups, and the small sample numbers did not permit statistical evaluation about the LSP2. The normal duration of LSP1 in our series should disclaim its affinity with ESP2, which appeared reduced in both ETTH and CTTH.
The results of this study confirm previous findings of a reduced ESP2 duration in patients with tension-type headache (2), although some researchers failed to detect this pattern in selected groups. In a recent study performed with a large series of patients with tension-type headache using a blind design and methods to standardize the elicitation and scoring of the reflex response, no ESP2 variable differed significantly between tension-type headache sufferers and controls (6). An abnormal ESP2 duration was frequently reported in tension-type headache, probably because it is an unsteady pattern, depending upon the variability of the scoring procedures and intensity of stimulation. In this study, we chose not to follow the European Headache Federation criteria that stipulate a stimulation of 20-mA intensity in all cases. According to Ellrich et al. (8), the reflex should be elicited by intensity levels intermediate between the subjective perception of the stimulus and the pain sensation. Therefore, we estimated the intensity level corresponding to the individual PT required to elicit the reflex: the activation of pain-related afferents may contribute to increase the reflex duration (14) and to emphasize the duration reduction in tension-type headache: in our control subjects, the ESP2 was longer-lasting than the values reported by Lipchik et al. (3).
The ESP2 is based upon a polysynaptic circuit and it is induced predominantly by the activation of Aβ fibres as shown in previous studies about the stimulus intensity to evoke the reflex (8); a modulation by limbic areas at the cortical level and by the periaqueductal grey matter, raphe magnum nucleus, and lateral reticular nucleus in the brainstem was supposed on the basis of animal studies (15, 16). These structures are involved in the endogenous control of pain, which dysfunction may emerge with strong stimulus intensities, causing subjective discomfort. According to Bendtsen (17), the pathophysiological mechanism of tension-type headache is based on central sensitization phenomena, which may reflect a failure of the antinociceptive system causing the reduced duration of ESP2.
All considering, the use of facial reflexes in the study of pain-related disorders may improve the understanding of the pathophysiologic mechanisms and surely the ESP abnormalities contributed to suggest the central origin of pain in tension-type headache (6). The employment of stimulation methods specific for nociceptive afferents may enhance the soundness of the procedures, but a clear statement of the causes of the variability of the responses is a precondition for further studies.
Footnotes
Acknowledgements
We wish to thank Online English for the technical assistance in English style. The study was supported by Italian Minister of University and Research, Biomedicine Project, cluster C04.
