Abstract
The objective was to assess the placebo response in randomized clinical trials of analgesics in the treatment of migraine attacks. We included placebo-controlled studies that used the criteria of the International Headache Society for the diagnosis of migraine and headache response as the primary efficacy parameter. In the 11 studies that qualified for inclusion, headache response occurred after placebo treatment in 7-50% of the migraineurs with an average placebo response rate of 30% (95% confidence interval (CI) 23-36). Two hours after treatment with placebo an average of 9% (95% CI 7-12, range 7-17%) of the patients were found to be pain free. In conclusion, the average headache response rate to placebo was 30% in randomized clinical trials of analgesics in migraine with a tremendous variation among studies. Placebo response rates vary with the choice of primary efficacy measure as well as patient characteristics and study design.
Introduction and methods
The aim of this review was to summarize the rates of placebo response in randomized clinical trials of analgesics in the acute treatment of migraine. The search for trials was done in Medline (1988–2001, restricted to the English language), using Keywords ‘analgesics’ and ‘migraine’. To be eligible for inclusion studies used the criteria of the International Headache Society for the diagnosis of migraine, were placebo controlled and used headache response as an outcome measure. We evaluated the two most widely used efficacy parameters: headache response, i.e. the proportion of attacks that go from moderate or severe pain to pain that is mild or absent within 2 h; and pain free, i.e. the proportion of patients free of pain at 2 h after treatment (1). The results are summarized in Table 1.
Headache response and pain-free rates following acute treatment of migraine with active drugs and placebo
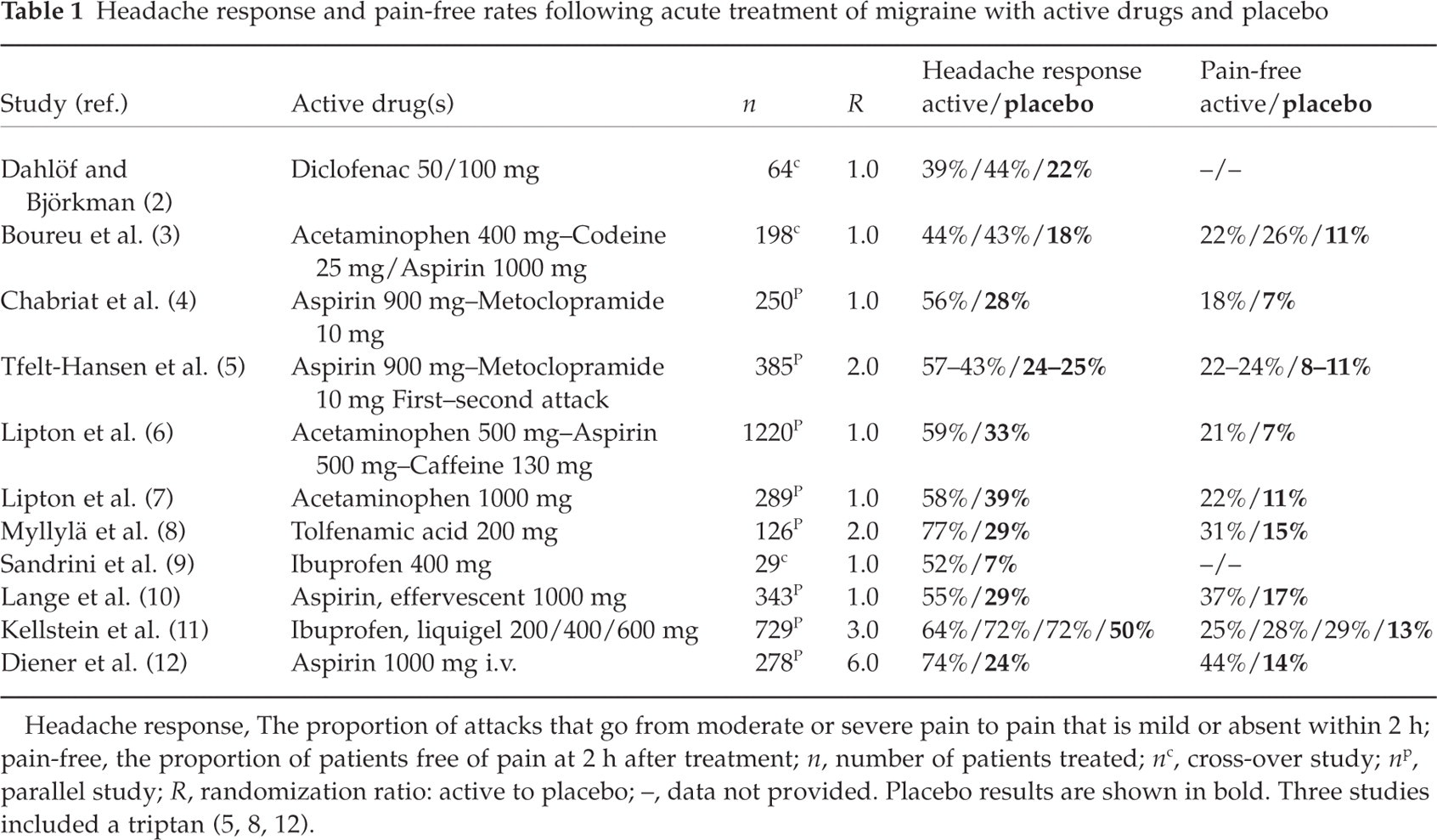
Headache response, The proportion of attacks that go from moderate or severe pain to pain that is mild or absent within 2 h; pain-free, the proportion of patients free of pain at 2 h after treatment; n, number of patients treated; n c, cross-over study; n p, parallel study; R, randomization ratio: active to placebo; –, data not provided. Placebo results are shown in bold. Three studies included a triptan (5, 8, 12).
We calculated the arithmetic means of responses of all the studies. The arithmetic means were weighted by the inverse of the variance of each study group (number of patients/(response)∗(1–response)). If there were data from more than one analgesic, different doses or more than one attack in a study, the arithmetic mean was calculated. Correlations were analysed by the test of Spearman. Differences in responses between groups were analysed by the unpaired t-test after weighting for differences in group size and by the test according to Mann–Whitney. Since the P-values derived from these tests differed marginally, only the P-values from the latter test are presented.
Results and discussion
Dahlöf and Björkman (2) found that treatment with diclofenac 50 mg and 100 mg resulted in headache response rates of 39% and 44% compared with 22% following treatment with placebo.
Boureu et al. (3) reported that acetaminophen 400 mg in combination with codeine 25 mg, aspirin 1000 mg and placebo resulted in positive headache responses in, respectively, 44%, 43% and 18% of the patients. The corresponding pain-free rates were 22%, 26% and 11%. Another French study evaluated the combination of lysine acetylsalicylate 1620 mg (equivalent to 900 mg of aspirin) and metoclopramide 10 mg (4). Positive headache response rates were 56% for active and 28% for placebo treatments. The corresponding pain-free rates were 18% and 7%.
In a European multicentre study, the combination of lysine acetylsalicylate 1620 mg (equivalent to 900 mg of aspirin) and metoclopramide 10 mg resulted in a positive headache response to the drug in 57% and 43% in the first and second attacks, respectively, compared with 22% and 24% to placebo in the first and second attacks, respectively (5). Pain-free rates for active drug were 22% and 24% in the first and second attacks, respectively, and for placebo 8% and 11% in the first and second attacks, respectively. This study included a comparison with triptan.
Lipton et al. (6) compared the combination of acetaminophen 500 mg, aspirin 500 mg and caffeine 130 mg with placebo, pooling the results of three separate clinical trials involving about 1200 patients in total. Patients with severe pain or vomiting were excluded. Fifty-nine percent of patients had a positive headache response following active treatment compared with 33% after placebo, while the pain-free rates were 21% and 7%. In a later study, Lipton et al. (7) compared oral acetaminophen 1000 mg with placebo. Patients with severe pain or vomiting were again excluded. Fifty-eight percent of patients had a positive headache response following active treatment compared with 39% after placebo, while the pain-free rates were 21% and 12%, respectively.
In a Finnish multicentre study, Myllylä and colleagues (8) found that 77% of migraineurs receiving tolfenamic acid rapid release 200 mg and 29% of patients receiving placebo had a positive headache response at 2 h. The pain-free rates were 31% and 15%. This study included a comparison with a triptan.
Sandrini et al. (9) compared a fast absorbed formulation of ibuprofen 400 mg with placebo. The headache response was 52% following treatment with ibuprofen and 7% after placebo.
Lange et al. (10) evaluated effervescent aspirin 1000 mg in 343 German migraineurs and found a headache response with aspirin and placebo of 55% and 29%, respectively, while pain-free rates were 37% and 17%.
An American multicentre study (11) evaluated a liquigel formulation of ibuprofen 200 mg, 400 mg and 600 mg and found headache response rates of 64%, 72% and 72% for active treatment and 50% for placebo. The pain-free rates were 25%, 28% and 29% for active treatment and 13% for placebo.
Diener and colleagues (12) compared lysine acetylsalicylate 1800 mg (equivalent to 1000 mg of aspirin) i.v. and placebo i.v. Treatment was given in out-patient clinics. Headache response rates were 74% and 24%, respectively, while pain-free rates were 44% and 14%. This study included a comparison with a triptan given subcutaneously.
Table 1 shows that a positive headache response within 2 h after placebo treatment was found in 7–50% of the patients, with an average response rate of 30% (95% confidence interval (CI) 23–36). A positive headache response to simple or combination analgesics was found in 39–77% of the patients, with an average response rate of 60% (95% CI 54–66). The mean proportion of pain-free patients 2 h after treatment with placebo and plain analgesics were 9% (95% CI 7–12) and 24% (95% CI 19–28), respectively. The placebo headache response and pain-free rates observed in these trials of analgesics are of the same magnitude as the placebo rates observed in triptan trials (13). The results show that the analgesic and placebo rates were lower for pain-free than headache response, which is expected since the outcome of pain free is hardest to attain. There was a marginally significant correlation between headache response to analgesics and placebo (Spearman, ρ= 0.43, P = 0.08) and a highly statistically significant and positive correlation between the proportion of patients pain free after 2 h following treatment with analgesics and placebo (Spearman, ρ= 0.91, P = 0.0006). These findings indicate that within individual studies the magnitude of the placebo response and the response to active drug are explained by shared factors, perhaps the characteristics of the study population or the study design. The findings should be interpreted cautiously, as three of the studies were cross-over studies and, hence, the response to drug and placebo were not collected in independent samples. It has to be emphasized that the above-mentioned placebo rates do not reflect only the placebo effect. The magnitude of the placebo effect can be evaluated only by including a natural history group because both active and placebo treatments may be followed by a spontaneous resolution of the attacks (14).
The study design considerably influenced the magnitude of the headache response rate. Thus, the placebo response was significantly lower for the three cross-over studies (mean response rate 16%, 95% CI 6–26) than for the eight studies that used the parallel group design (mean response rate 33%, 95% CI 28–39) (Mann–Whitney U-test, p = 0.02). The headache response rate to active treatment was also significantly lower for the three cross-over studies (mean response rate 44%, 95% CI 31–57) than for the eight studies that used the parallel group design (mean response rate 62%, 95% CI 57–67) (Mann–Whitney U-test, P = 0.02). The analgesic effectiveness is influenced by past experience with effective analgesic agents (e.g. conditioning) and expectations of the efficacy of the treatment (15). It is possible that when the patient knows that he or she will receive placebo at some time during the trial then expectations of the efficacy of the treatment will be lower.
The highest headache response rates to placebo were seen in the three American studies (6, 7, 11), which had a mean response rate of 36% (95% CI 28–45) compared with a mean response rate of 24% (95% CI 17–31) in the eight European studies (P = 0.03 in both t-test and Mann–Whitney U-test despite overlapping 95% CI). This may be explained in part by factors related to patient selection. All three American studies evaluated approved over-the-counter medications and not prescription drugs, a factor which may lead to enrolment of less severely affected patients. In addition, two of the American studies (6, 7) excluded the most disabled migraineurs, increasing the possibility that less severely affected more placebo-responsive patients were enrolled. Moreover, the American studies used population-based recruiting to a higher degree than the European studies. In addition, all the American studies used the parallel group design, which favours higher response to active drug and placebo. There was no significant correlation between the randomization ratio (ratio of the number of patients randomized to active or placebo treatment) and the headache response to placebo (Spearman, ρ= 0.19, P = 0.57). There was no statistically significant difference in the headache response between the three studies that included triptans (5, 8, 12) and the other studies (response rates of 25% (95% CI 5–47) vs. 30% (95% CI 23–38)). The only study in which placebo was given parenterally (12) had a placebo response rate similar to the other studies.
There was much less variation in magnitude of the placebo response between studies when pain free was used as the end point (mean 9%, range 7–17%) instead of headache response (mean 30%, range 7–50%). In contrast with the finding for headache response rates, there was no significant difference between the American and the European studies in the proportion of patients pain free 2 h after placebo (8%, 95% CI 5–12 vs. 10%, 95% CI 5–15) (Mann–Whitney U-test, P = 0.44). Two of the European studies did not measure the proportion of pain-free patients. These data indicate that pain free is a more robust outcome measure and should be preferred as primary end point in headache trials. In addition, the variability in analgesic effectiveness may also be due to differences in non-specific activation of endogenous opioid systems (16), and factors such as expectations of the outcome, suggestibility and conditioning may contribute to the magnitude of placebo analgesia (17).
In conclusion, a positive headache response was seen in between 7% and 50% of the migraineurs within 2 h after treatment with placebo, with an average headache response rate of 30%. Two hours after treatment with placebo an average of 9% (range 7–17%) of the patients were found to be pain free. The headache response rates were lower in studies that used the cross-over design than in parallel group studies and higher in studies that excluded patients difficult to treat. The variability between studies was lower when pain free rather than headache response was used as efficacy parameter. A high response to placebo was correlated to a high response to active drug.
