Abstract
Serotonin-1F receptor (5-HT1F) agonists may relieve acute migraine without vasoconstriction. We conducted a review of preclinical and clinical data that assessed the potential link between migraine and 5-HT1F activation. (i) A high correlation exists between the potency of various 5-HT1 receptor agonists in the guinea pig dural plasma protein extravasation assay and their 5-HT1F receptor binding affinity. (ii) 5-HT1F receptors are on the trigeminal system, and may participate in blocking migraine pain transmission through the trigeminal ganglion and nucleus caudalis. (iii) 5-HT1F receptors are located on glutamate-containing neurones and their activation might inhibit glutamate release; glutamate excess may play a role in migraine. (iv) Selective 5-HT1F receptor agonists (LY334370; LY344864) are effective in preclinical migraine models and are non-vasoconstrictive. (v) LY334370 is effective in acute migraine, and does not cause any symptoms/signs of coronary vasoconstriction. Preclinical experiments and clinical observations argue for a role of selective 5-HT1F agonists in migraine.
Introduction
The development of sumatriptan and the second-generation triptans over the last 10–15 years revolutionized the treatment of acute migraine. The discovery of sumatriptan was based on the belief that migraine pain is caused by cerebral vasodilatation, and that a vasoconstrictor would alleviate it (1). A more recent hypothesis of migraine posits that the attacks involve activation of dural afferents resulting in release of inflammatory neuropeptides such as calcitonin gene-related peptide (CGRP) and substance P (SP) (2, 3). The release of these substances induces an intense vasodilatation and neurogenic inflammation, leading to plasma protein extravasation (PPE) and mast cell degranulation (2). Sumatriptan and other class members, in addition to their vasoconstrictive properties, block neurogenic PPE (4–6).
Initial characterization of the triptans’ receptor binding profile demonstrated their high affinity for 5-hydroxytryptamine (5-HT)1B and 5-HT1D receptors (7–10). It was then believed that 5-HT1B receptor activation would mediate vasoconstriction, and 5-HT1D receptor activation would inhibit neuronal impulse transmission (11). Subsequent to cloning the 5-HT1F receptor (12), it was found that several triptans also display affinity for that receptor subtype (6, 13) (Table 1). These observations were particularly important because, unlike 5-HT1B, the 5-HT1F receptor does not mediate constriction of human vasculature (14–16). If 5-HT1F receptor agonism plays a role in migraine, selectively targeting this receptor could then provide an advantage over 5-HT1B receptor agonists by avoiding the liability of systemic and cerebral vasoconstriction. Several selective 5-HT1F receptor agonists, including LY334370 and LY344864 (Table 2), were developed to test that hypothesis.
Human 5-HT1 affinity of various triptans and related compounds that have been tested in the clinic for acute migraine

Data are compiled from different sources in the literature. Values in parentheses represent standard errors. NA, Not available.
Receptor profile of selective 5-HT1F agonists

GTPgS assay was used on cell lines that express human 5-HT1F receptors. EC50, Concentration at which 50% of the effect is observed; nM, nanomolar; ND, not done.
Properties of the 5-HT1F receptor and selective 5-HT1F receptor agonists
Receptor distribution
5-HT1F receptors have been identified on various relays in the trigeminovascular system, including the human trigeminal ganglion (TG) (15), the trigeminal nucleus caudalis (TNC) (17, 18), and cerebral vessels (15), and on non-trigeminovascular sites such as the neocortex and the hippocampus (19). In situ hybridization studies showed the presence of 5-HT1F receptor messenger ribonucleic acid (mRNA) in the guinea pig TG neurons (20). Low levels of mRNA for the 5-HT1F and 5-HT1D receptors have also been found on human cerebral and coronary vessels (15, 16).
Further characterization of the distribution of the 5-HT1 receptors in humans indicated the presence of 5-HT1B and 5-HT1D, but not 5-HT1F, receptors on cerebral microvascular smooth muscles, and expression of all three subtypes on astrocytes (21). These data, combined with pharmacological observations indicating lack of vasoconstrictive activity of selective 5-HT1F receptor agonists (see below), led Cohen et al. to suggest that this receptor subtype may be involved in blood–brain barrier physiology rather than vascular contractility (21).
Finally, a recent localization study using double immunohistochemical staining indicated that 5-HT1B/D/F receptor-positive neurons in TG are also glutamate-positive (22). This colocalization may be quite relevant to migraine if these receptors inhibit glutamate release, a potential culprit in migraine (23).
Biological activities
The biological activities of the 5-HT1F receptor have been investigated using the following migraine models: neurogenic dural PPE (6), inhibition of c-fos expression in the TNC (24, 25), and suppression of neuronal firing in TNC (26). Also, the effect of 5-HT1F receptor on contractility of systemic, dural and cerebral blood vessels has been investigated extensively (16, 26–29).
5-HT1F receptor and neurogenic inflammation
The trigeminal neurogenic inflammation model that quantifies dural PPE has been used as a surrogate of clinical efficacy in migraine (30, 31). Effective migraine therapies such as the triptans, aspirin, and dihydroergotamine block PPE, indicating that the model is sensitive to the action of pharmacologically distinct, anti-migraine compounds (32–36). Several experiments indicate that efficacy in the PPE model may have therapeutic relevance in migraine. First, extraparenchymal accumulation of radiolabelled albumin is observed during migraine attacks in humans (37). Second, stimulation of the trigeminal system causes release of CGRP and SP, which induce vasodilatation and PPE (31). Third, plasma levels of CGRP are elevated during migraine attacks (38–40) and normalize following the administration of sumatriptan (41). Finally, the administration of CGRP induces migraine in susceptible individuals (42). The above-described observations provide a good argument supporting neurogenic PPE as a valid preclinical migraine model, but it should be noted that (i) some investigational compounds that are effective in blocking neurogenic inflammation are ineffective in alleviating migraine pain (43–46), and (ii) although PPE is detected in the retina and dura of rats, it could not be demonstrated in the retina during migraine in humans (47). A sensitivity–specificity analysis of the value of PPE as a predictive model of efficacy in clinical migraine indicates high sensitivity and a 61% positive predictive value (Table 3).
Sensitivity of plasma protein extravasation model (PPE)
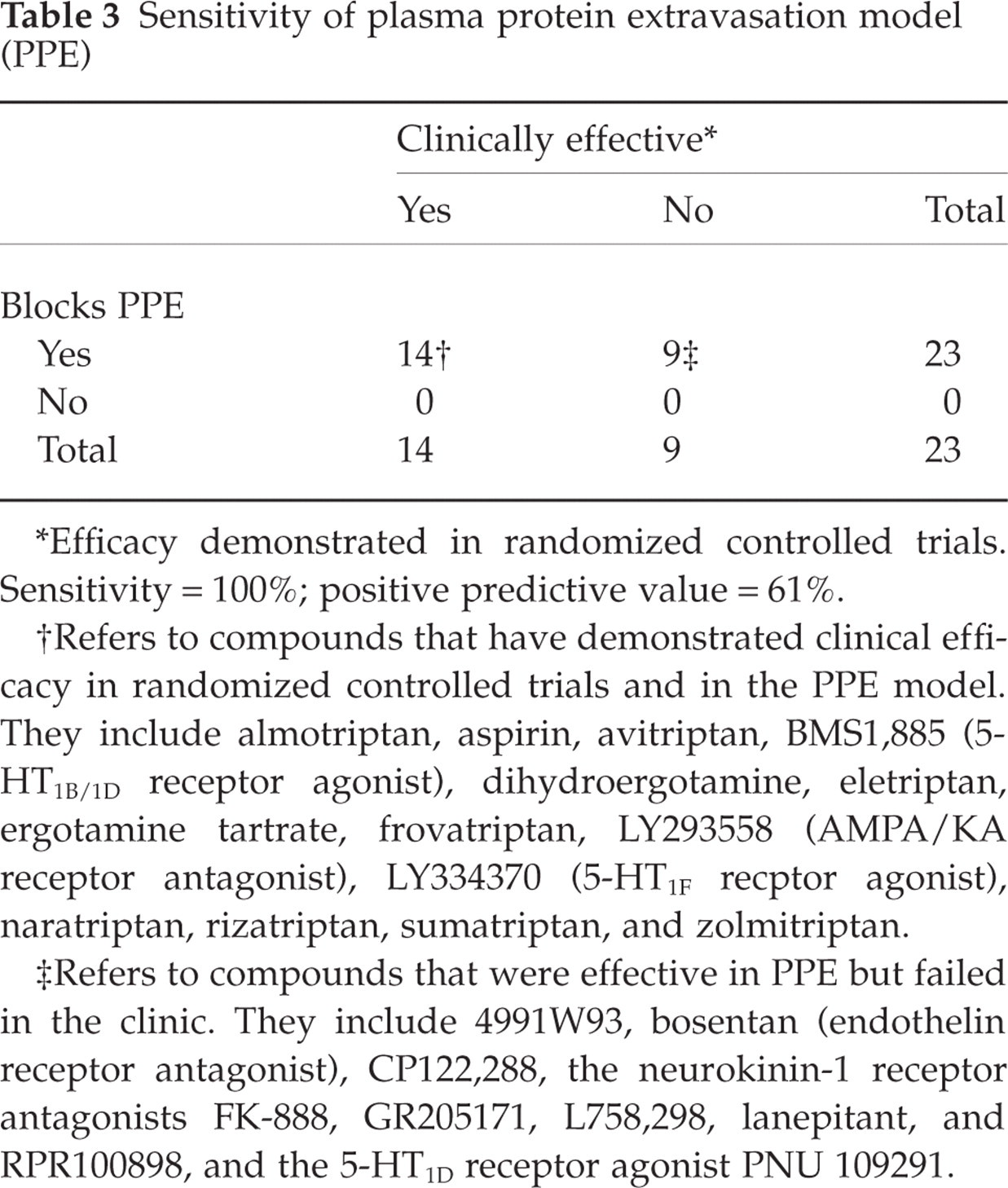
∗Efficacy demonstrated in randomized controlled trials. Sensitivity = 100%; positive predictive value = 61%.
†Refers to compounds that have demonstrated clinical efficacy in randomized controlled trials and in the PPE model. They include almotriptan, aspirin, avitriptan, BMS1,885 (5-HT1B/1D receptor agonist), dihydroergotamine, eletriptan, ergotamine tartrate, frovatriptan, LY293558 (AMPA/KA receptor antagonist), LY334370 (5-HT1F recptor agonist), naratriptan, rizatriptan, sumatriptan, and zolmitriptan.
‡Refers to compounds that were effective in PPE but failed in the clinic. They include 4991W93, bosentan (endothelin receptor antagonist), CP122,288, the neurokinin-1 receptor antagonists FK-888, GR205171, L758,298, lanepitant, and RPR100898, and the 5-HT1D receptor agonist PNU 109291.
The effects of several 5-HT1 receptor agonists on trigeminal stimulation-induced dural PPE were studied in guinea pigs (6). Each of the compounds studied inhibited PPE when administered intravenously, with the rank order of potency being LY334370, naratriptan, LY302148, LY306258 = zolmitriptan, dihydroergotamine, sumatriptan, rizatriptan, L-694,247. The correlation of potency (50% inhibitory dose (ID50)) and 5-HT1F receptor binding affinity was very high (r = 0.94), whereas no correlation with 5-HT1B or 5-HT1D affinities was observed (Fig. 1).

Correlation of receptor affinities with potency in inhibiting plasma protein extravasation (PPE) in guinea pigs for the following serotonergic agonists: LY334370, LY302148, naratriptan, LY306258, zolmitriptan, sumatriptan, DHE, rizatriptan, and L-694 247. Reproduced from Johnson et al. (6) with permission from Lippincott Williams & Wilkins. (a,b,c) Correlation plots of the compounds comparing their receptor affinity (on the abscissa) and i.v. potency in the PPE model (on the ordinate). (d) A similar analysis except that the abscissa represents the pEC50 values for the individual compounds in the whole cell cyclic AMP inhibition assay. The R-value and P-level are displayed for each correlation. The calculated line of best fit is displayed for statistically significant correlations. ∗Values plotted were the maximum concentrations tested. Without these points the r-values would be 0.88 and 0.84 and the P-levels would be 0.003 and 0.009 for (c) and (d), respectively.
The effects of LY344370 and sumatriptan on PPE in response to trigeminal stimulation were also studied following intravenous and oral administration in guinea pigs (LY334370 and sumatriptan) and rats (LY334370) (48). The results indicate that LY334370 is more potent in these experiments (Table 4). Also, sumatriptan po was fully effective in blocking PPE at 1 h, but not at 4 h, in guinea pigs, whereas LY334370 po was effective at 1, 4, 8, and 16 h but not at 24 h.
LY334370 vs. sumatriptan in plasma protein extravasation model

Plasma protein extravasation was induced following trigeminal stimulation. Test compound was administered 10 min (intravenous) or 60 min (oral) before stimulation. ID50, Dose that produces 50% inhibition of extravasation; ND, not done.
Finally, using a 35S-GTPγS (stable analogue of guanosine-5′-O-3-thiotriphosphate) functional assay developed for detection of the activated cloned human 5-HT1F receptor, it was determined that LY334370 had the greatest in vitro potency among the compounds tested, which included sumatriptan, zolmitriptan, naratriptan, rizatriptan, DHE, methysergide, and others (49). There was a significant correlation between 5-HT1F receptor agonist potency in this assay and the pID50 for inhibition of PPE in guinea pigs.
5-HT1F and inhibition of c-fos expression in the trigeminal nucleus caudalis
Activation of second-order neurons in the TNC by electrical stimulation of the TG is quantified by analysing the expression of the immediate early gene c-fos and/or its corresponding protein. In rats, oral administration of the selective 5-HT1F receptor agonists LY334370 and LY397584 significantly decreased the amount of Fos protein at 1 and 1000 µg/kg, respectively (24). A comparison of dose–response curves for LY334370 and selected triptans in the c-fos model is shown in Fig. 2. An additional selective and potent 5-HT1F agonist, LY344864, decreased c-fos expression induced by intracisternal injection of capsaicin in rats, another putative model of migraine (25).

Dose–response curves for LY334370 and selected triptans in the c-fos model. Assessment of fos protein immunoreactivity following electrical stimulation of the trigeminal ganglion in rats. Fasted Sprague-Dawley rats were pretreated orally with test compound or saline vehicle 1 h prior to trigeminal electrical stimulation. Animals were subsequently anaesthetized with sodium pentobarbital and placed in a stereotaxic frame where the trigeminal ganglion was stimulated unilaterally for 3 min. They were then removed from the stereotax and allowed to survive for 90 min before perfusion fixation with 4% paraformaldehyde and subsequent staining for Fos protein-like immunoreactivity. ∗P < 0.05 (vs. vehicle; one-way analysis of variance (ANOVA) test followed by Dunnett's comparison with control). ▪, LY334370; ▵, rizatriptan; •, naratriptan; ○, sumatriptan.
5-HT1F and inhibition of neuronal cell firing in the trigeminal nucleus caudalis
The effects of LY334370 on inhibition of nociceptive transmission, as measured by single-cell recording in the TNC, have also been investigated (25). LY334370 dose-dependently (0.3–3 mg/kg i.v.) inhibited neuronal firing in TNC following electrical stimulation of the dura mater without altering dural vessel diameter.
In summary, there is substantial evidence to support that 5-HT1F receptor agonists inhibit TNC neuronal activity, whether measured by c-fos immunohistochemistry or single-cell electrophysiological recording. The inhibitory effect of 5-HT1F agonists is a direct central phenomenon, in the absence of cerebral vasoconstriction. However, it should be noted that Dupuis et al. argued that LY334370 activates 5-HT1A receptors (50), which could imply that LY334370's central inhibition of TNC neurones may not be solely 5-HT1F receptor mediated since 5-HT1A agonism also inhibits TNC neurones (51). For reasons that are detailed below, we do not agree with the conclusions of Dupuis et al.
5-HT1F agonists and vasoconstriction
The contractile response of the dog saphenous vein was initially used as an experimental model to identify potential anti-migraine agents (1). This was based on the assumptions that dog saphenous and human cerebral arteries react in the same way to anti-migraine agents and that contractile responsiveness of human cerebral arteries is a surrogate for efficacy in migraine. However, more recent studies have indicated that the contractile potencies of a variety of serotonergic agonists in the saphenous vein model also correlate with their contractile potency in human coronary arteries (13). That discovery transformed the saphenous vein model into a tool for screening compounds with a potential cardiovascular liability.
The assessment of the potency of various 5-HT receptor agonists in contracting rabbit saphenous vein (RSV) has demonstrated a strong positive correlation between contractile potency and affinity at the 5-HT1B (r = 0.93, P < 0.003), but not at the 5-HT1F receptor (27). Similar conclusions were reached in RSV experiments using ergotamine, various triptans and two selective 5-HT1F agonists (27, 28). The 5-HT1F receptor agonists tested (LY334370 and LY344864) failed to induce vasoconstriction in concentrations up to 10−4 M (Fig. 3). Induction of modest baseline vascular tone by using PGF2α (potentiated RSV model) resulted in augmentation of the contractile responses, which paralleled the originally demonstrated contractile potency of the compounds tested. It is of note that, in the potentiated RSV model, some vasoconstriction was observed with LY344864, but only at concentrations above 10−5 M, which probably resulted in activation of 5-HT1B receptors as well.
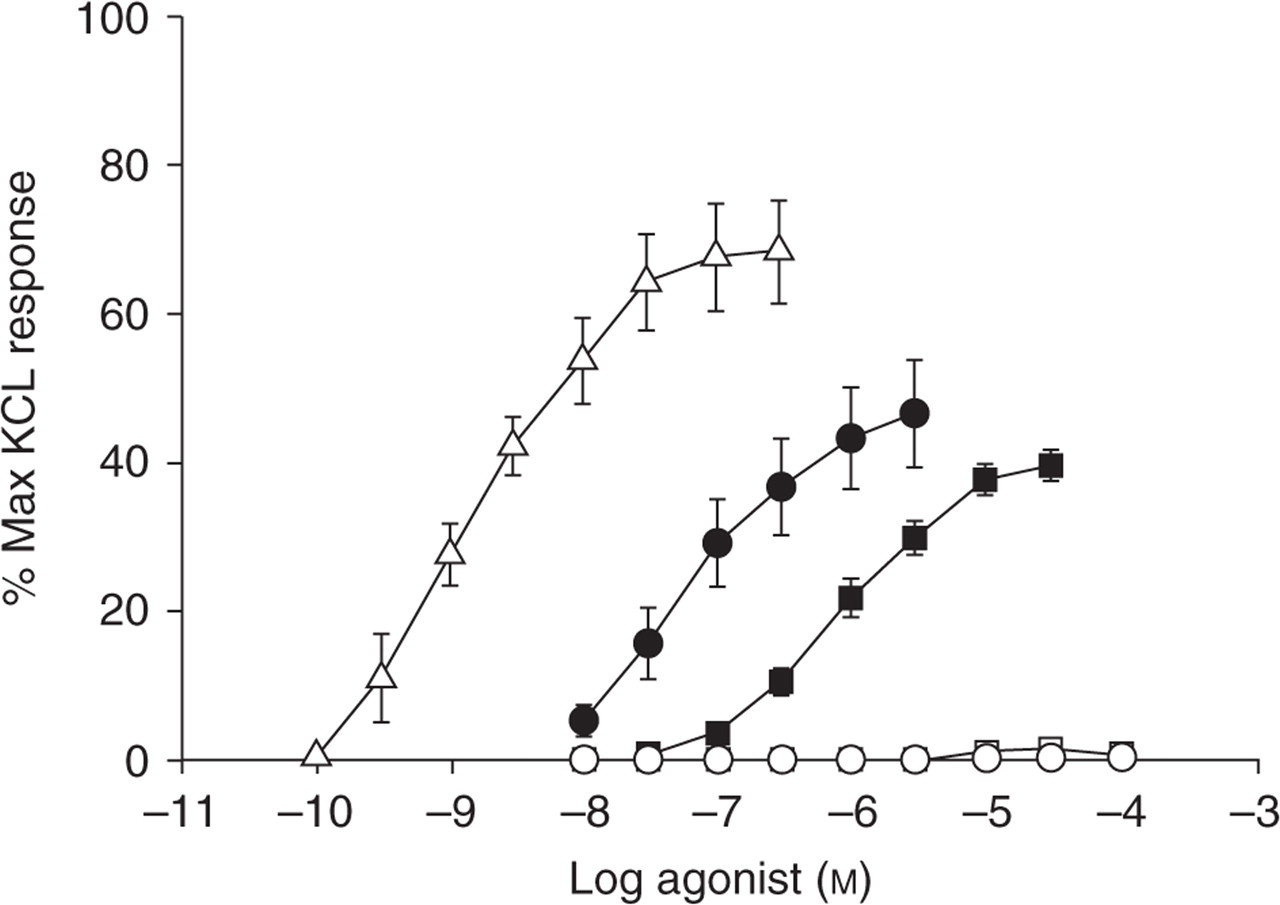
Rabbit saphenous vein contractile response to 5-HT, sumatriptan, ergotamine, LY334370 and LY344864. Reproduced from Cohen and Schenck (23) with permission from Nature Publishing. Points are mean values and vertical bars represent the standard error of mean for number of tissues (shown in parentheses). •, 5-HT (11); ▪, sumatriptan (13–15); ▵, ergotamine (3); □, LY334370 (4); ○, LY344864 (4).
In summary, preclinical vascular contractility experiments indicate that activation of the 5-HT1F receptor has no vasoactive effects.
LY334370, a selective 5-HT1F receptor agonist (Tables 1 and 2)
Further characterization of LY334370 indicated its high affinity for the 5-HT1F receptor (Ki = 1.6 nM) with more than 50-fold binding selectivity vs. all other 5-HT receptor subtypes tested except 5-HT1A (Ki = 11.9 nM). The Ki was greater than 1000 nM for a large number of other binding sites (receptors, ion channels, transporters) (52). Despite relatively high binding affinity to the 5-HT1A receptor, LY334370 exhibits functional selectivity for 5-HT1F over 5-HT1A, with agonist EC50 values for 35S-GTPγS binding of 1.08 nM and 1590 nM in cells expressing cloned human 5-HT1F and 5-HT1A receptors, respectively. Finally, LY334370 exhibits no 5-HT1A agonist activity in a number of in vivo tests, again illustrating functional selectivity for the 5-HT1F receptor (53). In summary, LY334370 is highly selective to the 5-HT1F receptor, a conclusion that contrasts with that reached by Dupuis et al. (50). Dupuis et al. used a rather high concentration (10 µM) of LY334370 to show 5-HT1A receptor-mediated stimulation of GTPγS binding in slices. At 10 µM LY334370, one would expect significant activation of the 5-HT1A receptor (Table 2: 50% activation at 1.6 µM). Also, Dupuis et al. did not find any 5-HT1F-mediated GTPγS binding in the brain slices, which is not surprising since the 5-HT1F receptor has low expression density in various brain regions. While we have been able to localize the receptor in brain using 3H-LY334370, the expression density is so low relative to the background binding of 35S-GTPγS that no 5-HT1F stimulation of 35S-GTPγS binding above background can be seen (unpublished data; manuscript in preparation) . In contrast, the 5-HT1A receptor is highly expressed in the brain. The difference in expression densities of these two receptors is illustrated historically by the fact that the 5-HT1A receptor expression density is such that this receptor was discovered and characterized in the brain before it was ever cloned, while the 5-HT1F receptor was unknown until it was cloned.
The preclinical pharmacological and toxicological profiles of LY334370 made it an attractive clinical candidate to test the hypothesis of 5-HT1F agonism in migraine. The compound was advanced in clinical development through phase 2 clinical trials. However, prior to initiation of phase 3 trials, preclinical toxicity studies indicated that the liver was a potential target for injury when LY334370 was administered to beagle dogs for longer than 1 month (data on file, Eli Lilly & Co.), and therefore clinical development was terminated. It is important to note that liver toxicity was not observed in rats who were exposed to LY334370, and liver enzyme elevation was not observed in humans who received LY334370. Also, animal liver toxicity was not observed with other selective 5-HT1F receptor agonists, and to our knowledge liver injury has not been reported with triptans that have high 5-HT1F receptor binding affinity such as naratriptan. Taken collectively, these observations suggested that the liver injury which was observed in dogs following LY334370 administration was likely a species-specific, drug-related, non-mechanism-related toxicity.
Clinical trials of the 5-HT1F receptor agonist LY334370
Early clinical trials of LY334370 provided solid evidence that the compound is safe and possesses no vasoconstrictive properties in humans (54, 55). Adverse events (AE) commonly reported in phase I oral and i.v. trials, at doses up to 200 and 20 mg, respectively, were transient asthenia, dizziness, somnolence and paresthesia. The AEs were typically of mild to moderate severity and lasting not more than 24 h. No changes in vital signs and no electrocardiographic (ECG) evidence of coronary ischaemia were observed.
In one study of 39 patients experiencing a moderate or severe migraine episode, LY334370 (20 mg, i.v.) was superior to placebo (P = 0.021) in the primary efficacy endpoint of headache response (i.e. the proportion of patients whose headache improved from a severity score of 3 or 2 to a score of 1 or 0 after 2 h) (56). The probability of pain response and pain-free (proportion of patients whose headache completely disappeared at 2 h) outcomes, as assessed by the random-effect proportional-odds model on (censored) score profiles for predicting time to response and time to pain free 0–24 h, were similar between the two doses (5 mg and 20 mg) of LY334370 and significantly better than placebo (odds ratio (OR) OR5mg-placebo = 19.3 (4.3, 87.4); P < 0.001; OR20mg-placebo = 32.5 (7.8, 135.7); P < 0.001; OR20mg-5mg = 1.7 (0.3, 9.5); P = 0.568). AEs in this pilot trial included somnolence/asthenia and numbness/paresthesia, which were more prevalent in the 20 mg LY334370-treated patients than in the placebo group. No other significant clinical, laboratory, or ECG abnormalities were observed. In particular, no chest symptoms or signs or symptoms of coronary ischaemia were observed.
A review of three phase I oral studies of LY334370, using doses ranging from 0.1 to 400 mg in 80 healthy volunteers and 61 migraineurs, indicated linear pharmacokinetics and efficacy at total plasma concentrations > 150 nmol/L (57). Of note is that LY334370 is highly bound to plasma protein (data on file at Eli Lilly and Co.). Somnolence and dizziness were observed at doses exceeding 80 mg.
The safety and efficacy of LY334370 were also assessed in three phase II studies using oral doses of 15–200 mg. In one of these studies, 20 mg (n = 22), 60 mg (n = 30), or 200 mg (n = 21) LY334370, or placebo (n = 26) were administered during a moderate or severe migraine (58). A positive result for the primary efficacy measure (sustained response, defined as patients who had improved from moderate or severe pain to mild or no pain after 2 h, with neither worsening from 2 to 24 h nor use of rescue medication) was seen in 8% of placebo patients, 14% of LY334370 20 mg patients (P = 0.392), 37% of LY334370 60 mg patients (P = 0.012), and 52% of LY334370 200 mg patients (P = 0.001) (Fig. 4). Also, the 60-mg and 200-mg doses were superior to placebo on secondary efficacy measures such as response rates at 2 h, pain-free rates at 2 h, and sustained pain-free rates. The AE profile in this phase II study was not different from that in the phase I programme (asthenia, dizziness, somnolence, paresthesia). Again, no ‘triptan-like’ symptoms (chest pressure, pain or tightness) were reported.

Efficacy results of a phase II clinical trial of oral LY334370 in acute migraine (51). Efficacy measures: 2-h response = response at 2 h; 2-h sustained response = sustained response 2–24 h; 2-h pain free = pain free at 2 h; 2-h sustained pain free = sustained pain free 2–24 h. Statistical analysis: treatment differences were analysed by Cochran–Mantel–Haenszel tests. The confidence intervals for binary outcomes were calculated with the normal approximation to the binomial distribution (2 SE). □, placebo; ▨, LY334370 20 mg;  , LY334370 60 mg; ▪, LY334370 200 mg. ∗P < 0.05 compared with placebo. Error bars represent 95% confidence intervals for the single treatment group.
, LY334370 60 mg; ▪, LY334370 200 mg. ∗P < 0.05 compared with placebo. Error bars represent 95% confidence intervals for the single treatment group.
Discussion
The underlying processes responsible for the initiation and propagation of migraine are not fully understood. Recent observations implicate the serotonergic system in migraine (59). To this end, a new class of neuronally selective drugs in development, namely the 5-HT1F receptor agonists, one of which (LY334370) was advanced into clinical development, have the potential to become novel anti-migraine therapies devoid of vasoconstrictive properties. As such, the 5-HT1F receptor agonists could provide a safety advantage over the efficacious 5-HT1B/1D receptor agonists (triptans) currently in clinical use, all of which are contraindicated in patients with, or at significant risk of, cardiovascular disease.
Cerebral vasodilation is probably one component of migraine pain, and it is likely that the triptans exert their anti-migraine efficacy at least partly through cerebral vasoconstrictor effects (5-HT1B receptor-mediated effect). However, triptans also inhibit neurotransmitter release from trigeminal nociceptive neurones (4, 5, 60, 61), and several triptans have modest to significant agonist activity at 5-HT1F receptors (62) (Table 1). The ability of serotonergic receptor agonists to inhibit PPE in guinea pigs is correlated with their affinity for 5-HT1F, but not 5-HT1B/1D receptors. Data demonstrating that selective 5-HT1F receptor agonists are potent inhibitors of neurogenic dural inflammation suggest that such compounds may thus inhibit the peripheral inflammatory processes believed by some to occur in migraine. Activation of 5-HT1F receptors in the afferent central limb of the trigeminal pathway may block transmission of nociceptive signals to the TNC, in which 5-HT1F receptor mRNA and 5-HT1F receptor binding sites have been identified (17, 18). 5-HT1F receptor agonists with low affinity for 5-HT1B/1D exhibit no vasoconstrictive effects at concentrations well in excess of those required to activate 5-HT1F receptors, suggesting that despite their presence in peripheral vessels and cerebral arteries, these receptors are not involved in contractile responses (21).
The positive results obtained in laboratory studies led to a number of clinical trials with a 5-HT1F receptor agonist. LY334370 was indeed effective in acute migraine without associated vasoconstrictor effects (54–58). Goldstein et al. postulated that LY334370 probably works by blocking 5-HT1F receptor-mediated nociceptive impulses centrally within the TNC in the absence of any appreciable cerebral vasoconstriction (58). This mechanism was suggested for several reasons. First, at efficacious doses, LY334370 causes CNS symptoms such as asthenia and somnolence (58), which argues for significant central penetration of the compound in humans. In addition, non-painful symptoms of migraine, including nausea, phonophobia, and photophobia, may originate centrally (63), and therapeutic doses of LY334370 effectively stopped all of these symptoms (58). Furthermore, 5-HT1B receptor-mediated cerebral vasoconstriction was unlikely to explain the efficacy of LY334370 since the highest efficacious free plasma concentration of LY334370 measured in clinical efficacy studies was substantially lower than concentrations that block neuronal activity in TNC without any effect on dural vessel diameter (26). Finally, free plasma concentrations at the efficacious 60-mg oral dose (32 nM) (58) and 5-mg i.v. dose (21 nM) of LY334370 (56) were significantly closer to the EC50 values for h-5-HT1F than h-5-HT1B receptor.
In conclusion, current laboratory and clinical data indicate that 5-HT1 receptor agonists may inhibit migraine processes through interaction with 5-HT1B, 5-HT1D, and/or 5-HT1F receptors present on trigeminal sensory nerves. Thus, selective 5-HT1F receptor agonists may provide an alternative to the 5-HT1B/1D receptor agonists or ‘triptans’ for the treatment of acute migraine pain without vascular liability or such untoward AEs as chest pain and tightness. In addition to directly testing the therapeutic role of 5-HT1F receptor selective agonists in migraine, it would be informative to conduct pharmacogenomic studies similar to that reported by Massen VandenBrink et al. (64), in order to better understand the link between genetic diversity on the 5-HT1F and other 5-HT1 receptor genes and the clinical response to potential anti-migraine therapies.
