Abstract
Lanepitant, a potent non-peptide neurokinin-1 receptor antagonist, inhibits neurogenic dural inflammation, and may have a role in migraine therapy. This study evaluated the effect of lanepitant taken daily for migraine prevention. Patients with migraine headaches with and without aura by International Headache Society classification criteria were enrolled in a 12-week double-blind, parallel design study comparing the effect of 200 mg qd lanepitant (n = 42) and placebo (n = 42) on reduction of migraine frequency. The primary outcome measure was response rate, i.e. the proportion of patients with a 50% reduction in days of headache. Of the 84 patients enrolled, 90.5% were female. The endpoint response rate for lanepitant-treated patients (41.0%) was not statistically significantly (P = 0.065) greater than that for placebo-treated patients (22.0%). No efficacy variables differed significantly between treatments, except for response rates at month 3 (P = 0.045). Higher plasma concentrations were no more effective than lower concentrations. In this study lanepitant was not effective in preventing migraine, but was well tolerated. These results do not support a role for NK-1 antagonism in migraine prevention.
Introduction
The exact pathogenesis of migraine is unclear. One hypothesis is that migraine pain is caused by the release of inflammatory peptides, primarily substance P and calcitonin gene-related peptide (CGRP), onto the dural vasculature peripherally resulting in nociceptor sensitization and central pain transmission (1–4). According to this hypothesis, substance P induces dural inflammation and increases sensitization to migraine headache pain by stimulating NK-1 receptors (5). Thus, NK-1 receptor antagonists may have a role in migraine therapy.
Lanepitant is a high-affinity, non-peptide, competitive NK-1 receptor antagonist that acts both peripherally and centrally. It is effective in the guinea pig model of dural inflammation (5, 6) at a dose of 1 mg/kg, lasting 8 h and longer at higher doses; but it proved ineffective in treating acute migraine (7). It is also effective in blocking licking behaviour in the late phase of the formalin test for at least 24 h after dosing, indicative of central penetration (8).
In man (unpublished data), lanepitant has a large variability in plasma concentrations. It undergoes saturable first pass metabolism, such that lanepitant attains disproportionately higher concentrations with increasing dose. The absorption after a fatty meal is 40% of the absorption when fasted by the area under the curve for plasma concentration. The half life is 12–20 h. Steady state levels are attained within a week.
We hypothesized that lanepitant failed as acute migraine therapy because: (i) dural inflammation may be more resistant to treatment once underway; (ii) oral absorption might be compromised by gastric stasis during the migraine attack so that plasma concentrations were insufficient; and/or (iii) inhibition by NK-1 antagonists alone might be insufficient to interrupt a migraine headache. These theories led us to propose that NK-1 antagonists might be more appropriate in migraine prevention. Hence we studied daily doses of 200 mg of lanepitant and placebo in the prevention of migraine headaches over a 3-month period.
Methods
Design
The design of the study is shown in Fig. 1. This was a 3-month, randomized double-blind, parallel-design, placebo-controlled study. Patients who met study inclusion and exclusion criteria and who signed an Institutional review board-approved informed consent were entered into the trial. Patients were at least 18 years old with at least a 1-year history of migraine (9). For at least 3 months prior to entry, patients had three to eight migraine headaches per month, but no more than 15 days of headache per month. Women could not be pregnant or lactating. Patients received 4 weeks of single-blind placebo treatment prior to randomization, then received oral doses of lanepitant or placebo for up to 3 months. Patients recorded migraine headaches, headache severity, duration, associated symptoms and severity, and treatment in a migraine diary. In addition, Patient Global Evaluation (PGE), Clinical Global Impression (CGI), and Acceptability were assessed at visit 5 or at the discontinuation visit.

Study design.
Both spontaneous report and the Symptom Assessment for Analgesia (SAA) questionnaire (10) were collected at every visit to assess adverse events. The SAA solicits adverse events including sleep, appetite, gastrointestinal, cardiovascular, neurologic, and others associated with the use of opiates, non-steroidal anti-inflammatory drugs, and central nervous system penetrant agents. Vital signs were recorded at all visits. Laboratory tests (including haematology, clinical chemistry and urinalysis) were done at visits 1, 2, 3, 4, and 5 or at the time of discontinuation. Plasma lanepitant concentration was assessed at visits 3, 4, and 5, or at time of discontinuation. A 12-lead ECG was obtained at visits 1 and 5 or at discontinuation.
Statistical analysis
Sample size was estimated to be 38 patients in each treatment group completing at least 1 month of study drug therapy, providing 80% power at the 0.05 level one-sided. This was based upon a meta-analysis of propranolol vs. placebo indicating a reduction in migraine frequency of 43% and 14%, respectively (11).
Response is defined as a decrease in migraine frequency (corrected for a 30-day month) of at least 50% from baseline. Response rates were assessed using the Cochran–Mantel–Haenszel statistic, stratified by investigator. Tests of treatment effects for efficacy variables were two-sided (Fig. 2).

(a) Mean number of migraine attacks (standardized to 30-day periods) by month. (b) Mean headache response rate (50% reduction in migraine frequency from baseline) by month. □, Lanepitant 200 mg; ▪, placebo.
The actual time interval was corrected to a 30-day month. For example, if a patient's interval from visit 4 to visit 5 was 22 days, then frequency of migraines during the 22 days was multiplied by 30/22 to correct or standardize the data to a frequency of migraines per 30-day month.
Treatment comparison P-values for percent reduction variables, PGE, and transformed CGI are from a two-way analysis of variance model (
Treatment-emergent signs and symptoms (TESS) were defined as any event reported during the double-blind portion of the study (study period II) that occurred for the first time after baseline or worsened in severity relative to baseline. The frequency and percent of TESS, reasons for study discontinuation, and the SAA were presented for each treatment group and compared using Pearson's χ2 test.
Results
Patient characteristics
Table 1 summarizes baseline demographic characteristics for all 84 randomized patients. There were no significant group differences in any of the assessed baseline variables.
Summary of patient characteristics
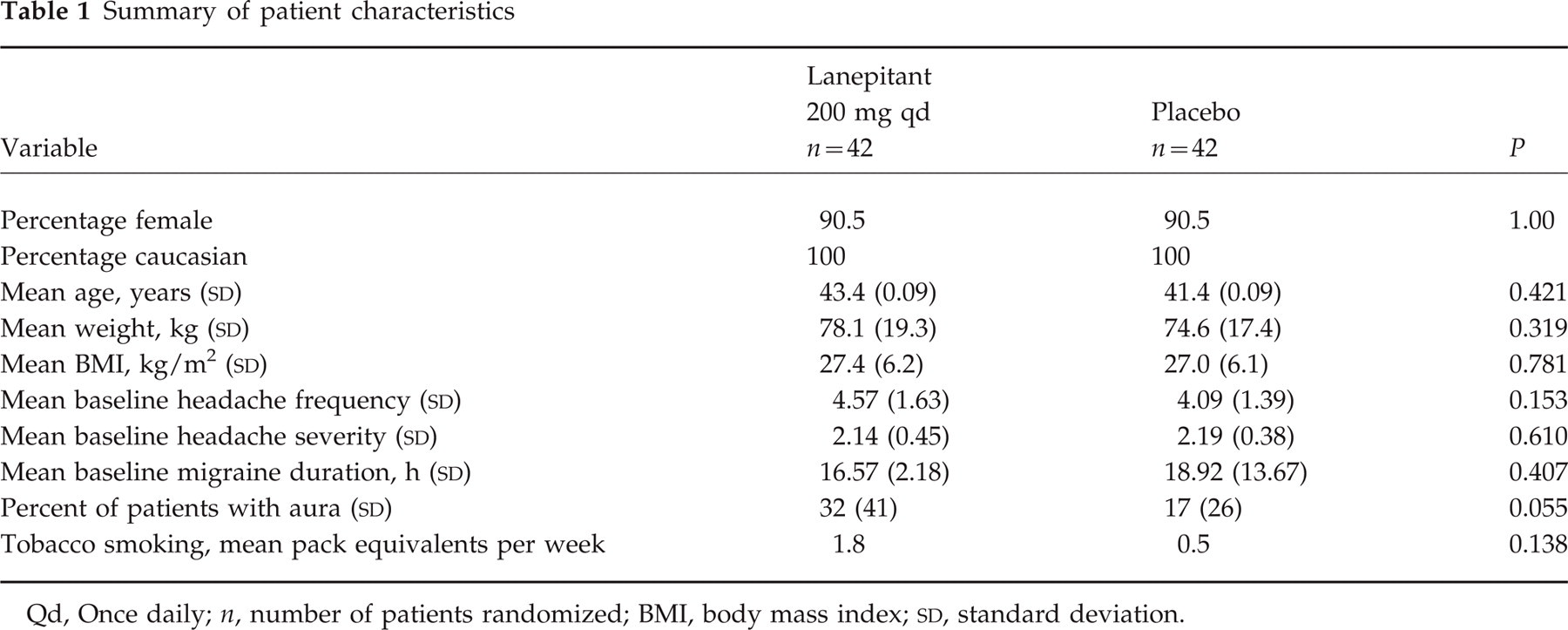
Qd, Once daily; n, number of patients randomized; BMI, body mass index;
Efficacy results
The endpoint response rate tended (P = 0.065) to be greater for lanepitant (41.0%) than for placebo (22.0%). Although not significant for the first or second months, the response rate at 3 months was statistically significantly (P = 0.045) greater for lanepitant (40.6%) than for placebo (17.4%) (Fig. 2). The corresponding reduction in mean migraine frequency were 0.45 for lanepitant and 0.40 for placebo. The average response over all 3 months was also numerically greater (P = 0.333) for lanepitant (20.0%) than for placebo (11.4%). Neither of these differences was statistically significant.
However, there was no statistically significant effect of lanepitant over placebo (Table 2) in percent reduction of migraine frequency, either for endpoint frequency analysis (P > 0.570) or for the average frequency for months 1 through 3 (P = 0.191). There was no statistically significant treatment difference in migraine frequency at month 3 (P = 0.977). At month 3 there was a statistically significant decrease in the mean percent of migraines with aura for lanepitant-treated patients (LS means 11% vs. 23%, respectively; P = 0.040) and a numerical decrease in the mean percent of throbbing migraine headaches for the lanepitant-treated patients relative to placebo-treated patients (LS means 71% vs. 82%, respectively; P = 0.092).
Effect of lanepitant and placebo on endpoint and average migraine headache frequency normalized to a 30-day interval

n, Number of patients randomized; LS, least squares; CI, confidence interval.
There was no statistically significant treatment difference observed for CGI Severity, CGI Efficacy Index or PGE.
Furthermore, of the migraines that occurred there was no statistically significant difference in migraine-associated conditions (disability, duration, photophobia, nausea, or migraine severity). However, duration of migraines that occurred were an average of 3.18 h (based on least squares means) shorter in the lanepitant-treated group than in the placebo-treated group (P = 0.096).
Steady-state plasma concentrations were determined from samples collected at visits 3, 4, and 5. There was a very large mean inter- and intrapatient (median 19.9 and range from < 0.1 to 285.0 ng/ml) variability. There was no relationship between effect and plasma concentration.
Safety
Four patients treated with placebo, who had confusion, insomnia, and lab abnormality, and two patients treated with lanepitant, who had nausea and heart palpitations, discontinued from the study early due to an adverse event.
Treatment emergent adverse events were reported by 22 (52.4%) of the patients treated with lanepitant and by 17 (40.5%) of patients treated with placebo (P = 0.274). The treatment emergent adverse events that were more frequently reported by lanepitant-treated patients were headache (11.9% for lanepitant vs. 0% for placebo; P = 0.021), back pain (11.9% vs. 2.4%; P = 0.080), diarrhoea (11.9% vs. 2.4%; P = 0.090), nausea (7.1% vs. 0%; P = 0.078), and pain (7.1% vs. 0%; P = 0.078).
Three events by SAA were more frequent for lanepitant-treated patients than for placebo-treated patients: interrupted sleep (21.4% vs. 7.1%; P = 0.061), nausea (23.8% vs. 9.5%; P = 0.079), and hot flushes (16.7% vs. 4.8%; P = 0.078). One event was less frequent for lanepitant-treated patients: difficulty falling asleep (14.3% vs. 31.0%; P = 0.068).
Vital sign and laboratory evaluations
There were no statistically significant treatment effects for changes in vital signs from baseline to endpoint. Baseline to endpoint changes in leucocytes (− 0.00 billion/l for lanepitant and −0.31 billion/l for placebo; P = 0.020), segmented neutrophils (0.082 billion/l vs. −0.209 billion/l; P = 0.024), and calcium (− 0.014 mmol/l vs. −0.061 mmol/l; P = 0.024) were not deemed clinically relevant. Although at endpoint more patients treated with lanepitant compared with those treated with placebo had treatment-emergent abnormal urinary microscopic exam for erythrocytes (P = 0.048) and leucocytes (P = 0.004), this result was interpreted as likely to be spurious since similar effects were not seen in prior studies.
Discussion
The study results suggest that lanepitant is ineffective in preventing attacks of migraine with or without aura. Nevertheless, the proportion of patients with at least a 50% reduction in headache frequency during their final month of therapy tended to be greater for those treated with lanepitant than for those treated with placebo. This difference in response rates was statistically significant for the patients who were treated for 3 months, although the difference in mean reduction in migraine frequency between treatments was clinically irrelevant. These observations suggest that use of response rate, although a standard measurement of efficacy in most migraine prevention trials, can be too liberal since it can show a treatment effect when mean differences are marginal. This corroborates the recommendation of the International Headache Society that mean changes in frequency be used (9).
Although the observed data may represent a false-negative result due to a potentially smaller true effect of lanepitant than expected, the 3-month response was not correlated with lanepitant plasma concentrations. The lack of a concentration response relationship suggests that lanepitant is ineffective in preventing migraine headaches and not due to insufficient plasma concentrations that may have been due to the variability in absorption and metabolism or due to non-compliance.
Lanepitant penetrates the central nervous system as indicated by the inhibition of licking behaviour in the formalin model. Although the extent of brain penetration is low (12, 13), based upon rat plasma concentrations at doses effective in the formalin model, human plasma concentrations taken at random during steady-state dosing in this trial, and the 50-fold greater affinity of lanepitant for human than for rat NK-1 receptors, the amount present could have been adequate to prevent migraines if the isolated mechanism of NK-1 antagonism were sufficient to prevent migraines. However, it is not known whether the central transport of lanepitant in humans is similar to that of rats.
Lanepitant was well tolerated, as evidenced by the relative lack of more discontinuations for adverse events, treatment-emergent adverse events, SAA events, vital signs, and laboratory results by lanepitant-treated patients than placebo-treated patients. In evaluating adverse effects of treatment, the use of the SAA appears to be complementary to the use of spontaneous adverse events, especially for small trials that have limited power for detection of adverse effects.
Failure of dapitant and lanepitant to treat acute migraine (7, 13) or lanepitant to prevent attacks (current study) suggests that NK-1 antagonists are insufficient as sole therapy for migraine. Whether an NK-1 antagonist in combination with other mechanisms might provide a synergistic effect is as yet untested.
Footnotes
Acknowledgements
The authors thank Drs Benjamin Frishberg of Rockville, MD and Randall R. Stolz of Evansville, IN for their work in this trial, Dr Ouhang Wang for his statistical assistance, and Dr Nabih Ramadan for his discussions and suggestions. This work was sponsored by Eli Lilly and Company.
