Abstract
We describe the polysomnographic data of two patients with nocturnal headache attacks fulfilling the clinical criteria for hypnic headache syndrome. Two overnight polysomnographic studies were performed in each patient. Four nocturnal headache attacks were captured, all emerging from the REM phase of sleep. Our findings suggest a close relationship between the REM phase of sleep and the appearance of hypnic headache attacks.
Introduction
Hypnic headache syndrome (HHS) is a headache disorder, described in 1988 by Raskin (1), characterized by recurrent attacks occurring exclusively during sleep. HHS is considered a rare primary headache disorder and, at the time of this writing, approximately 50 patients with the disorder have been described in the literature. However, HHS is probably more common than previously thought (2).
Several authors have suggested that hypnic headache syndrome is a rapid eye movement (REM) sleep-phase phenomenon (1, 3). However, polysomnographic studies in patients with HHS provided conflicting results. Arjona et al. (4) reported the data concerning a single nocturnal headache attack associated with stage 3 slow wave sleep while Dodick (5) described a nocturnal attack during a REM period.
We report the clinical and polysomnographic data of two patients with HHS. Both the patients presented nocturnal headache attacks associated with the REM phases of the sleep cycle.
Case reports
Case 1
A 58-year-old-woman presented with a 6-year history of severe nocturnal headache. Headache attacks occurred only during nocturnal sleep and usually began between 2 and 4 h after she fell asleep. The pain awakened her from sleep once or twice each night, often lasted 30–40 min but has persisted for up to 2 h. The patient frequently reported being awoken with headache from a vivid dream. The pain was described as a throbbing headache of severe intensity, located in the left ocular and occipital regions. The headaches were seldom associated with mild nausea. No ptosis, lacrimation, rhinorrhea or other autonomic signs were reported.
During the attack, none of the following drugs were effective: paracetamol, sumatriptan, indomethacin and nimesulid. Previous therapeutic trials with propranolol, pizotifen, divalproex, carbamazepine, verapamil and amytriptiline were ineffective.
The medical history was unremarkable and her family history was negative for migraine. The results of the general physical and neurological examinations were normal. Standard blood chemistry, complete blood count and erythrocyte sedimentation rate were normal, as were EEG and CT of the head. Magnetic resonance imaging (MRI) of the brain showed signal changes in the white matter, suggesting small ischaemic lesions. The average blood pressure with ambulatory blood pressure measurement was 135/72 mmHg.
Overnight polysomnographies were recorded during two subsequent nights. The registrations were characterized by reduced sleep efficiency (67.0 and 60.8%). There was no evidence of significant sleep-disorder breathing (apnoea-hypopnea index (AHI) < 5 in both nights) or nocturnal oxygen desaturation. Only rare episodes of mild snoring were recorded. Total sleep time (TST) was 352.5 min in the first night and 275.5 min in the second one. Table 1 shows the sleep characteristics of both registrations. The patient reported two headache attacks during the first night. Both the attacks aroused the patient from a REM phase of the sleep (Fig. 1). The patient did not experience a headache during the second registration.
Polysomnographic data in patients with hypnic headache syndrome

SE, sleep efficiency (%); TST, total sleep time (min); NREM 1, % of non REM sleep stage 1; NREM 2, % of non REM sleep stage 2; NREM 3–4, % of non REM sleep stage 3–4; REM, % of REM sleep; SaO2, mean oxygen saturation (%); SaO2 REM, mean oxygen saturation during REM sleep (%); AHI, apnoea-hypopnea index; AHI, apnoea-hypopnea index during REM sleep.
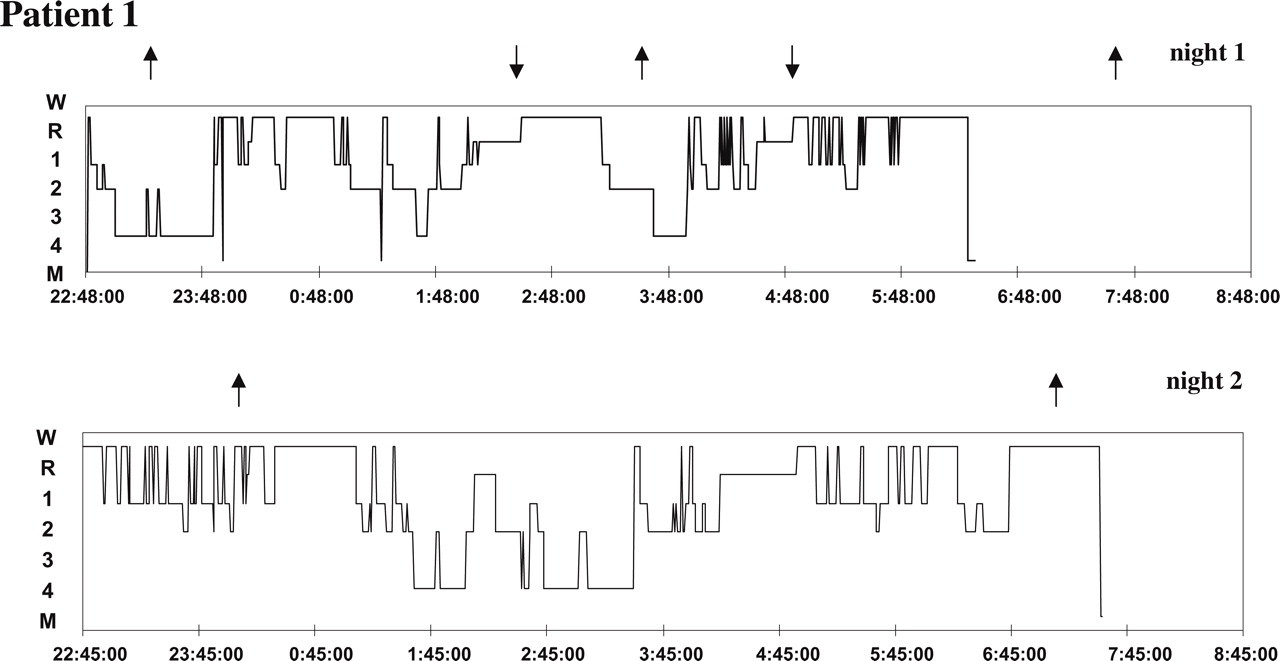
Hypnograms of the first patient with hypnic headache syndrome. W, wake; R, REM sleep, 1 2 3 4, stages of N-REM sleep, M, morning awakening. The arrowa indicate the onset of headache attacks.
Treatment was initiated with lorazepam, 2.5 mg, at bedtime, and during the next 2 weeks she experienced no nocturnal headache attacks. Afterwards, nocturnal headache attacks reappeared. Treatment with lithium, 300 mg at bedtime, was begun and headache severity and frequency decreased.
Case 2
A 49-year-old man presented with a 1-year history of headache attacks during sleep. They occurred both during nocturnal sleep and daytime naps. The pain was described as a headache of moderate to severe intensity, located bilaterally in the frontal and occipital regions. The headache attacks awakened the patient from sleep and lasted from 30 to 60 min The attacks recurred once to two times nigthly. The pain was not accompanied by autonomic signs (ptosis, nasal obstruction, lacrimation, rinhorrea, nasal or conjunctival redness, etc.).
During the attack, trials with multiple medications failed. Prophylactic therapies with propranolol, flunarizine and amytriptiline had been unsuccessful.
The patient reported a medical history fulfilling IHS criteria (6) for episodic tension-type headache. However, the tension-type headache attacks occurred infrequently and were less severe than the nocturnal headaches. The family history was negative for migraine or tension-type headache. Physical and neurological examinations were normal and the patient was in good general health. There was no pain over the posterior cervical region. Routine blood and urine analyses were normal. EEG and CT scan of the brain were normal. MRI of the brain showed no abnormality. The average blood pressure was 124/69 mmHg.
Overnight polysomnographies, recorded during two subsequent nights, were characterized by reduced sleep efficiency (56.2 and 45.9%). There was no evidence of significant sleep-disorder breathing or nocturnal oxygen desaturation. Total sleep time (TST) was 279.5 min in the first night and 286.0 min in the second one. Table 1 shows the sleep characteristics of both nocturnal registrations. The patient reported one headache attack each night. Both the headache attack began in a REM phase of the nocturnal sleep (see Fig. 2).

Hypnograms of the second patient with hypnic headache syndrome. W, wake; R, REM sleep, 1 2 3 4, stages of N-REM sleep, M, morning awakening. The arrows indicate the onset of headache attacks.
Therapy with lithium, 450 mg at bedtime, was started and headache attacks completely disappeared after 10 days of treatment.
Discussion
We reported the results of clinical and polysomnographic studies in two patients with headache attacks occurring during sleep. Both patients fulfilled proposed criteria for hypnic headache syndrome (7). We registered four nocturnal headache attacks and all the attacks were associated with a REM period.
The results of our study confirm the data of Dodick (5) who captured a hypnic headache attack in one patient arising out of REM sleep. In contrast, Arjona et al. (4), in a 79-year-old woman, registered a nocturnal headache episode during stage 3 slow wave sleep. However, the patient was being treated with venlafaxine when polysomnography was performed and is well known that antidepressant drugs alter sleep architecture (8). In the study of Dodick (5), the headache attack recorded during the REM sleep was associated with a severe oxygen desaturation due to obstructive sleep apnoea (OSA). Patients with OSA report frequently morning headaches and continuous positive airway pressure (CPAP) induces a marked improvement in headache (9, 10). In our patients no oxygen desaturation was observed, both in the REM and in the non-REM phases of the sleep, and the sleep respiratory patterns were normal in all the registrations. So our data rule out a role for oxygen desaturation in the genesis of hypnic headache.
The reasons for the association of hypnic headache with REM sleep are unknown. Several primary headache disorders, such as cluster headache, chronic paroxymal hemicrania, and migraine, occur often during the REM phase of sleep (11, 12).
Different areas in the brainstem and forebrain are important for the modulation and expression of sleep/wake cycle (13). Rapid eye movement sleep results from the activity of REM-sleep generating and REM-sleep inhibiting neurones. The serotoninergic neurones of dorsal raphe and locus coeruleus are a well known population of REM-sleep inhibiting neurones. Dorsal raphe neurones fire most rapidly during the waking state, decrease their firing rate during NREM sleep, and become silent during REM sleep. At the end of a REM sleep period, dorsal raphe neurones resume firing (14).
It is of interest to note that both dorsal raphe and locus coeruleus are deeply involved in the modulation of pain ascending pathways and in the initiation of migraine attacks (15, 16). Considering the role of serotonin in pain modulation, it could be speculated that the relative increase of 5-HT activity occurring at the end of REM sleep may precipitate the nocturnal headache attacks.
Several studies have shown that lithium is an effective drug for the treatment of patients with HHS (1, 3, 17). However, despite the wide clinical use of the drug in affective disorders and in cluster headache, the mechanism of lithium action is still unknown. Studies in experimental animals have shown that lithium exerts several effects on cerebral serotonin metabolism at the levels of precursor uptake, synthesis, storage, catabolism, release, and receptor–effector interactions (18, 19). Studies in humans generally suggest that lithium has a net enhancing effect on cerebral 5-HT functions (20). So, lithium may serve to correct an, as yet unknown, abnormality of cerebral 5-HT functions involved in the pathogenesis of HHS.
Polysomnographic studies both in healthy volunteers and in depressed patients showed that lithium administration significantly decrease REM sleep (21, 22). In addition, lithium therapy causes significant delays in the sleep-wake circadian rhythm. The relevance of these findings in understanding the pathogenesis of HHS deserves further studies.
In conclusion, we reported a close association between hypnic headache attacks and REM sleep. In addition, the results of our study suggests that routine use of polysomnography should be a useful tool to study patients with nocturnal headache attacks.
Footnotes
Acknowledgements
Supported by grants from the Ministry of University and Scientific and Technological Research (MURST) of Italy.
