Abstract
The objective of this study was to evaluate and compare the efficacy and safety of single doses of acetaminophen (paracetamol) 1000 mg and naproxen 375 mg vs. placebo over a six-hour period in the treatment of tension-type headache. The treatments were compared in a randomized, double-blind, multicentre, placebo-controlled study. Efficacy was evaluated using four standard analgesic summary endpoints (the sum of pain intensity differences from baseline, the maximum pain intensity from baseline, the sum of the pain relief scores, and the maximum pain relief score). Both acetaminophen 1000 mg and naproxen 375 mg were significantly superior to placebo (P ≤ 0.009 and P ≤ 0.021, respectively) but not significantly different from each other (P ≥ 0.498) for these four endpoints. For example, the mean sum of pain intensity differences from baseline was 9.14 ± 0.34 for acetaminophen 1000 mg and 8.81 ± 0.35 for naproxen 375 mg compared with 7.42 ± 0.34 for placebo. Other efficacy endpoints (percentage of responders (pain reduced to none) at two hours, onset of meaningful relief, time to use of rescue medication and subject's overall impression of study medication) showed similar trends. A significantly larger mean pain intensity difference from baseline was observed for acetaminophen 1000 mg (1.13) than for naproxen 375 mg (0.95) (P = 0.036) at one hour after treatment. There was no significant difference among the treatment groups in the incidence of adverse events (P = 0.730). In summary, the results of this well-controlled, double-blind study demonstrate that over-the-counter acetaminophen 1000 mg and prescription naproxen 375 mg are effective and well tolerated in the treatment of tough (moderate-to-severe) tension-type headache.
Introduction
Tension-type headache, also known as muscle contraction, stress, essential or psychogenic headache, is the most commonly experienced type of headache. Overall, 78% of the general population reports having experienced a tension-type headache during their lifetime (1). Tension-type headaches are reported more frequently by women (88%) than by men (69%) (1). The Headache Classification Committee of the International Headache Society (IHS) introduced the term ‘tension-type headache’ in 1988 and defined the diagnostic criteria for classification (2). Tension-type headaches have been classified in two clinical forms: episodic, for which the pain is present for less than 15 days per month or chronic, for which pain is present for at least 15 days per month (for at least six months). Tension-type headaches are typically characterized by a pressing/tightening quality of mild or moderate intensity, are bilateral in location, and do not worsen with routine physical activity.
These headaches are largely self-treated and many consumers do not consult a physician. Results of a survey of 1573 adults 15 years of age or older indicated that 90% of 783 tension-type headache sufferers use over-the-counter analgesics and that 55% do not consult a physician (3). In another study that surveyed 274 adults 18–65 years of age with frequent headaches (≥12 per year), 56% of 108 tension headache sufferers medicated with acetaminophen (paracetamol), 15% with aspirin, and 9% with ibuprofen (4). The remaining respondents used other single-ingredient or combination products and only 1% used prescription medication.
Acetaminophen is a widely used and clinically proven nonsalicylate para-aminophenol derivative with analgesic and antipyretic properties that has been available over-the-counter in the United States since 1959. Acetaminophen, at a dose of 650 mg up to six times a day or 1000 mg up to four times a day (daily maximum of four grams), is indicated for the temporary relief of minor aches and pains associated with headache, muscular aches, backache, minor arthritis pain, common cold, toothache, menstrual cramps and for the reduction of fever. Acetaminophen has recently been shown to be effective in the treatment of migraine (5). Numerous studies have demonstrated the efficacy and safety of acetaminophen 1000 mg vs. placebo for the treatment of tension-type headaches (6–13).
Naproxen is a prescription nonsteroidal anti-inflammatory drug (NSAID) with analgesic and antipyretic properties. Naproxen sodium, the sodium salt of naproxen, is absorbed more rapidly than prescription naproxen and was approved for over-the-counter use in the United States in 1994. Naproxen sodium, at a dose of 220 mg every eight to 12 h (daily maximum of 660 mg), is indicated for the same over-the-counter conditions as acetaminophen. Three studies (14–16) have demonstrated the efficacy of naproxen sodium at doses ranging from 275 mg to 550 mg in the treatment of tension-type headaches.
The present study was designed to evaluate and compare the efficacy and safety of single doses of acetaminophen 1000 mg and naproxen 375 mg vs. placebo over a six-hour period in the treatment of tension-type headache.
Methods
Study procedures and design
This randomized, double-blind, single-dose, placebo-controlled study was conducted in 1993 by 19 investigators throughout the United States. Study participants included both current patients of the investigators as well as new recruits. This study was approved by institutional review boards appropriate for each investigator and all study participants gave written informed consent to participate.
Outpatient volunteers with a clinical history of tension-type headaches, which had been generally relieved in the past with over-the-counter analgesics were recruited. The inclusion criteria for participation in the study were:
men or women 18 years of age or older;
history of acute tension-type headaches of at least moderate intensity on a scale of none, mild, moderate, moderately severe, or severe that met at least two of the following characteristics (a pressing, tightening, nonpulsating quality; possible inhibition but not prohibition of activity; bilateral or variable location; not aggravated by physical activity) derived from the diagnostic criteria established by the International Headache Society, required treatment with over-the-counter analgesics and occurrence between four and 10 times per month, and were not associated with nausea, vomiting, photophobia, phonophobia, or auras (to exclude subjects with migraine headaches);
history of response to treatment of acute tension-type headaches with over-the-counter analgesics;
medical history, physical and neurologic examinations inconsistent with organic disorders associated with headaches.
Table 1 provides a comparison of the IHS criteria for diagnosis of episodic tension-type headache and the study headache inclusion criteria.
Comparison of the IHS criteria for diagnosis of episodic tension-type headache and the inclusion criteria for this study

Subjects were excluded if they had a history of any of the following:
vascular (migraine or cluster) headaches;
recurrent sinus headaches;
headaches related to food or excess alcohol;
withdrawal headaches from substances such as caffeine or nicotine;
headaches due to other underlying pathology or related to head or neck trauma;
alcohol abuse, drug dependency, or psychiatric disease;
use of daily nonsteroidal anti-inflammatory drugs, other analgesics, low dose aspirin prophylaxis, anticoagulants, or psychotropics;
continuous daily headaches;
headaches unresponsive to treatment with over-the-counter analgesics;
headaches related to menses;
a sensitivity or allergy to acetaminophen, aspirin, or nonsteroidal anti-inflammatory drugs;
peptic ulcer disease, inflammatory bowel disease, gastrointestinal bleed, unstable clinically significant cardiovascular disease, clinically significant renal or hepatic disease, coagulation disorders, unstable diabetes, pancreatic disease, uncontrolled hypertension, seizures, cerebral vascular ischaemia, infarct, haemorrhage or other central nervous system disease, unstable metabolic disease, current malignancy, or active tuberculosis; and prior gastrointestinal surgery which could influence absorption, metabolism or excretion of study medication.
Also excluded were those with an active viral or flu-like illness or currently receiving antibiotics and females that were pregnant or lactating.
Each subject was randomly assigned to one of the following three treatment groups according to a computer-generated randomization schedule allocated in blocks of nine: acetaminophen 1000 mg, naproxen 375 mg or placebo. The assigned study medication was dispensed at the investigative site after eligibility was confirmed and written informed consent was obtained. Subjects in all three treatment groups received a blister card containing two capsules that were identical in colour, size and shape. During the study, neither the subjects, the investigators, nor personnel directly involved in monitoring the study or reviewing the data knew the treatment assignments.
Subjects were required to be experiencing an acute tension-type headache of at least moderate severity before ingesting the study medication. Subjects were instructed to record in a diary the date and time of ingestion, pain intensity before treatment and pain intensity and pain relief after treatment recorded at 0.25, 0.5, 0.75, 1, 2, 3, 4, 5 and 6 h. Pain intensity was recorded on a scale of 0–4 as no pain, mild, moderate, moderately severe, or severe. In addition, subjects recorded pain relief (compared with the pain level before treatment) on a scale of 0–4 as no relief, a little, some, a lot, or complete relief. Stopwatches were used by the subjects to record the time to onset of meaningful pain relief. At the end of the six-hour measurement interval or at the time of use of rescue analgesia, subjects evaluated and recorded their overall impression of the study medication on a scale of 0–4 as poor, fair, good, very good or excellent.
Subjects were allowed to use rescue medication after one hour if their pain remained at or returned to the level before treatment; time of use of rescue medication was recorded. For subjects with conditions that required drug therapy, those conditions and the medications used were recorded on the case report form. Subjects returned to the study site for a follow-up visit within 48 h after use of the study medication. Study personnel reviewed the completed diary with the subject to ensure that all required information had been recorded and to clarify any reported adverse events.
Outcome measures
Treatments were compared using the following four standard analgesic summary measures to assess efficacy: the time-interval weighted sum of pain intensity differences from baseline (SPID), the maximum pain intensity difference from baseline occurring over the observation period (MAXPID), the time-interval weighted sum of the pain relief scores (TOTPAR), and the maximum pain relief that occurred during the observation period (MAXPAR). Efficacy was also assessed by comparing treatments at each measurement interval from 0.25 to six hours for pain intensity differences from baseline and pain relief scores, time to onset of meaningful relief, time to use of rescue medication, subject's overall impression of study medication, and the percentage of subjects that responded (had a reduction in pain intensity to mild or none) by two hours. Subjects used a stopwatch to measure the time to meaningful pain relief; subjects that did not experience meaningful relief were assigned a time of six hours.
Statistical considerations
Demographic and baseline characteristics were compared among treatment groups using a one-way analysis of variance for continuous variables and a χ2 test for categorical variables. A chi-square test was also used to compare adverse event rates.
The effect of baseline pain intensity level on measures of efficacy was accounted for through the use of stratification in the analyses of variance. There were three pairwise comparisons of interest for the efficacy analysis: acetaminophen 1000 mg vs. placebo, naproxen 375 mg vs. placebo, and acetaminophen 1000 mg vs. naproxen 375 mg. Pairwise comparisons between each treatment pair were made at the 0.05, two-tailed alpha level. The baseline pain intensity levels of moderately severe and severe were combined to form one level; moderate baseline pain formed a second level.
A two-way analysis of variance was used to evaluate the four analgesic summary measures. Pairwise comparisons between treatments for pain intensity difference from baseline and pain relief over time were analysed using repeated measures analyses of variance. The Peto-Peto-Wilcoxon test was used to test for differences between treatments for the time to onset of meaningful relief and the time to use of rescue medication. Analysis of variance was also used to evaluate subjects' overall impression of the study medication. The IHS guidelines (17) recommend use of the percentage of subjects with headaches resolved at two hours as an efficacy endpoint; this endpoint was not defined a priori. The Cochran-Mantel-Haenszel test was used for the analysis of percentage of responders at two hours.
All study pain scores subsequent to the use of rescue medication were set to the pain intensity ratings given by the subject at the time rescue medication was used. Pain relief scores were set to zero subsequent to the use of rescue medication.
Linear interpolation was used to estimate a score when a subject failed to make a pain intensity or pain relief evaluation within five minutes of the 0.25, 0.5 or 0.75 h assessments, or within 15 min of the one through six-hour assessments. Missing data (≤ two consecutive time points) were also estimated by linear interpolation if bounded by two pain intensity or pain relief ratings or if the missing data were at the end of the study period.
Results
Participant flow and follow-up
A total of 963 subjects were enrolled in the study. Overall, 900 of the 915 subjects who took study medication completed the study. Figure 1 summarizes the randomization profile and the status of subjects in the study; the number of subjects who completed the study, who withdrew and their primary reason for withdrawal are shown. Table 2 shows the demographic characteristics, use of concomitant medication and baseline pain intensity levels for all subjects included in the intent-to-treat analysis. Race and age were comparable for the three treatment groups. There was a significant difference in gender among the treatment groups, with the placebo group having a lower percentage of women. Overall, 646 (71.8%) subjects treated in the study were women, which is consistent with the female preponderance in the tension-type headache population. The average age of the subjects was 33.9 years and 815 (90.6%) subjects classified themselves as Caucasian. There were no significant differences among treatment groups in the percentage of subjects that used concomitant medications or in baseline pain intensity levels.
Demographic characteristics, use of concomitant medication and baseline pain intensity level by treatment group

∗Statistical comparisons were performed using a one-way analysis of variance for continuous variables and a chi-square test for categorical variables.
†Significantly different among treatment groups (P=0.004).

Profile and flow diagram for subjects enrolled in the randomized control trial.
Efficacy endpoints
Table 3 provides the least squares means and standard errors for the analgesic summary endpoints (SPID, MAXPID, TOTPAR, and MAXPAR) and shows the P-values of the pairwise treatment comparisons. Both acetaminophen 1000 mg and naproxen 375 mg provided significantly more relief than placebo (P≤0.009 and P≤0.021, respectively) but were not significantly different from each other (P≥0.498) for these four endpoints.
Summary efficacy pain relief endpoints by treatment group expressed as least squares means±standard errors

APAP, Acetaminophen; SPID, Time-interval weighted sum of the pain intensity difference from baseline; MAXPID, Maximum pain intensity differences from baseline occurring during the observation period; TOTPAR, Time-interval weighted sum of the pain relief scores; MAXPAR, Maximum pain relief score occurring during the observation period.
†Significantly different vs. placebo, P<0.021.
Figure 2 shows the least squares means of pain intensity differences from baseline by treatment group over the six-hour study period. Comparisons at each measurement interval demonstrated that the acetaminophen 1000 mg group had a significantly larger mean pain intensity difference from baseline than the placebo group from hours one through six (P≤0.024) while the naproxen 375 mg group had a significantly larger mean pain intensity difference than the placebo group from hours two through six (P≤0.023). In addition, the acetaminophen 1000 mg group had a significantly larger mean pain intensity difference from baseline than the naproxen 375 mg group at hour one (P=0.036). Figure 3 shows the least squares means of pain relief over time by treatment group. Comparisons at each measurement interval revealed a similar pattern to that observed for pain intensity differences from baseline; the acetaminophen 1000 mg group had a significantly larger mean pain relief score than the placebo group from hours one through six (P≤0.015) while the naproxen 375 mg group had a significantly larger mean pain relief score than the placebo group from hours two through six (P≤0.001). However, no significant differences between the acetaminophen 1000 mg group and the naproxen 375 mg group were observed for mean pain relief scores.

Least squares means of pain intensity differences from baseline by treatment group, ▪ acetaminophen 1000 mg; ▴ naproxen 375 mg; • placebo. ∗P≤ 0.024 acetaminophen 1000 mg or naproxen 375 mg vs. placebo, †P=0.036 acetaminophen 1000 mg vs. naproxen 375 mg based on repeated measures analysis of variance.
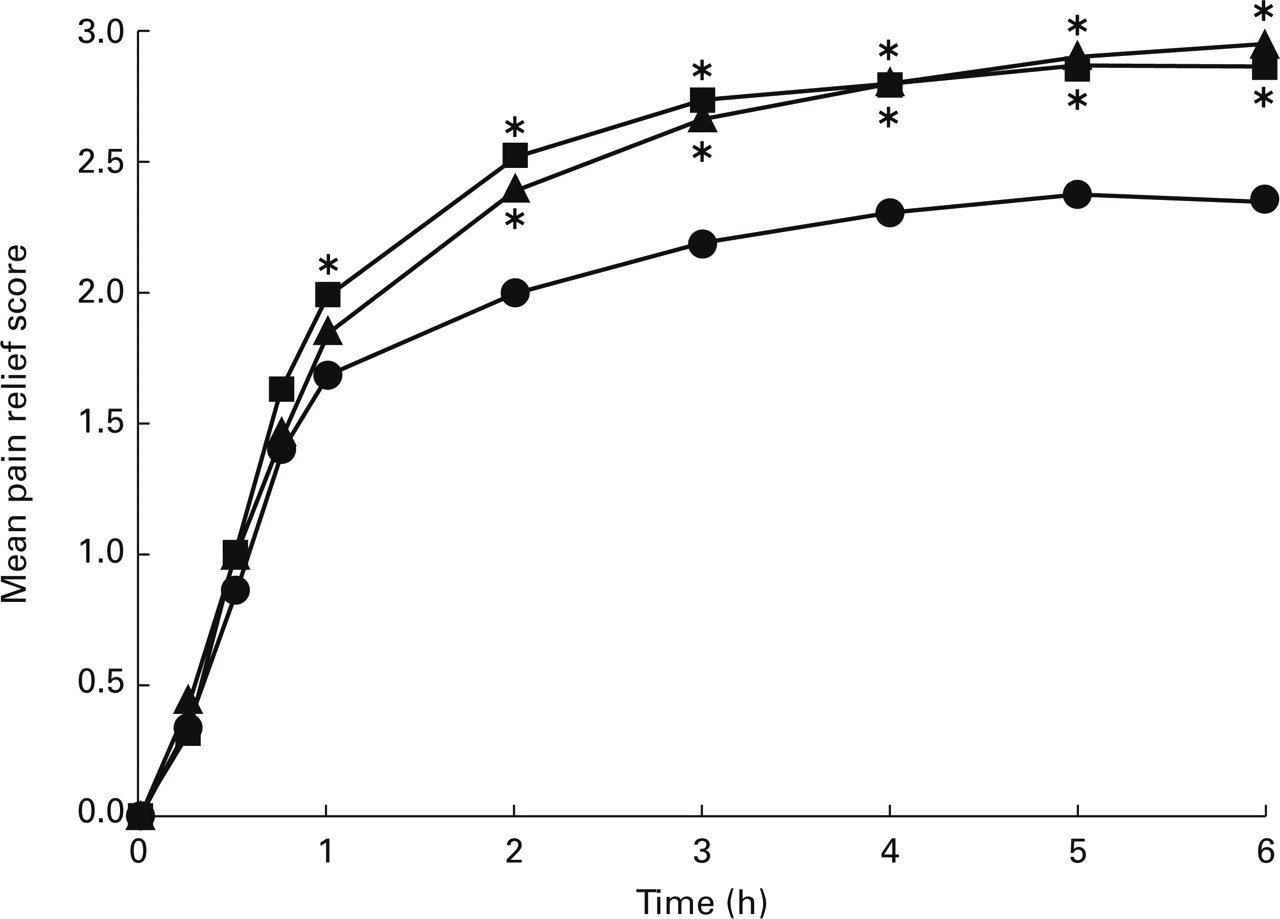
Least squares means of pain relief over time by treatment group, ▪ acetaminophen 1000 mg; ▴ naproxen 375 mg; • placebo. ∗P≤0.015 acetaminophen 1000 mg or naproxen 375 mg vs. placebo based on repeated measures analysis of variance.
Table 4 provides data for the secondary efficacy endpoints. The percentage of subjects with headaches completely resolved (reduced to none) at two hours was 36.8% for the acetaminophen 1000 mg group, 31.5% for the naproxen 375 mg group and 25.9% for the placebo group. Although the percentages were larger, a similar pattern was observed for the percentage of subjects with headache pain reduced to mild or none at two hours. Pairwise comparisons between treatments for both pain reduced to mild or none and for pain reduced to none demonstrated that acetaminophen 1000 mg was significantly superior to placebo (P≤0.008) while naproxen 375 mg was not (P≥ 0.097); however, there was no significant difference between acetaminophen 1000 mg and naproxen 375 mg (P≥0.164).
Secondary efficacy endpoints by treatment group

APAP, acetaminophen; LSM, least squares means.
†Significantly different vs. placebo, P<0.032.
‡Pain severity score <1.5.
The mean time to use of rescue medication is also presented in Table 4. Subjects in both the acetaminophen 1000 mg and naproxen 375 mg groups had significantly longer mean times to rescue (324.0 min and 326.4 min, respectively) than subjects in the placebo group (308.4 min) (P=0.020 and P=0.009, respectively) while there was no significant difference in mean time to use of rescue medication between the two active treatments (P=0.751). In addition, the percentage of subjects using rescue medication was similar for acetaminophen 1000 mg and naproxen 375 mg (17.4% and 16.6%, respectively) and both were less than the 25.6% of subjects who used rescue medication in the placebo group (data not shown).
As shown in Table 4, subjects in both the acetaminophen 1000 mg and naproxen 375 mg groups had significantly shorter median times to onset of meaningful pain relief than subjects in the placebo group (P=0.015 and P=0.032, respectively), while there was no significant difference between the two active treatments (P=0.686). The least squares means for the subject's overall impression of the study medication at the end of the six-hour study period are also presented in Table 4. Pairwise comparisons showed that subject's mean overall impression of acetaminophen 1000 mg and naproxen 375 mg were significantly higher than for placebo subjects (P=0.024 and P=0.015, respectively) but were not significantly different from each other (P=0.845).
Safety
All subjects who took study medication (n=915) were included in the analysis of adverse events. No serious adverse events were reported in this study. Adverse events reported by 1% or more of the subjects in any treatment group are summarized in Table 5. Overall, 10.5% of subjects reported adverse events, with no significant differences among treatments (P=0.730). The most common adverse events were in the digestive system (mainly dyspepsia and nausea). Dyspepsia was reported by 3.7% of subjects in the naproxen 375 mg group compared with 1.9% of subjects in the acetaminophen 1000 mg group and 1.3% of subjects in the placebo group; this difference was not statistically significant (P=0.163). Nausea was reported by 3.0% of subjects in the naproxen 375 mg group compared with 1.6% of subjects in the acetaminophen 1000 mg group and 2.6% of subjects in the placebo group; this difference was also not statistically significant (P=0.490).
Incidence of adverse events by body system reported in 1% or more of subjects in any treatment group

APAP, acetaminophen.
Discussion
This was a large (n=963) randomized, double-blind, single-dose, placebo-controlled study of subjects with tension-type headache. The trial was designed to evaluate the efficacy and safety of a single dose of over-the-counter acetaminophen 1000 mg (vs. placebo) in the treatment of tension-type headache. Prescription naproxen 375 mg was chosen as an active comparator treatment in this tension headache model because these subjects experienced tough (moderate-to-severe pain intensity) headaches.
Our analysis of baseline characteristics revealed a significant difference in gender among groups (P=0.004), with the placebo group having a lower percentage of women. A recent dental pain study failed to demonstrate a gender effect on analgesic response (18). In our study, randomization was stratified by pain intensity, which would mitigate any potential confounding effect of gender or other factors that may have an effect on pain intensity.
The age of subjects in this study ranged from 18 to 87 years, with the mean age being similar for the acetaminophen, naproxen, and placebo groups (33.2, 34.6 and 33.8 years, respectively). Overall, only 8.6% (n=77) of the 900 patients that completed the trial were >50 years and 2.1% (n=19) were >60 years. Three subjects were 70 years of age or older (70, 79, and 87 years). IHS guidelines suggest that tension-type headache in subjects >50 years may be due to an underlying organic disease that mimics tension-type headache (17). Although this could be a concern, our physician investigators were required to screen all participants with thorough medical histories, exams and neurologic testing to rule out headaches caused by organic disorders prior to enrolment and randomization.
In this study, acetaminophen 1000 mg and naproxen 375 mg were shown to be statistically superior to placebo for all predefined analgesic efficacy endpoints (SPID, MAXPID, TOTPAR, and MAXPAR). The percentage of subjects who used rescue medication as well as the mean time to use of rescue medication in the placebo group compared to those in both active treatments also support the efficacy of over-the-counter acetaminophen 1000 mg and prescription naproxen 375 mg in the treatment of tension-type headaches.
Acetaminophen 1000 mg was effective for the treatment of tension-type headache in this study; results were consistent with several previous double-blind trials (6–13). Acetaminophen 1000 mg was superior to placebo from hours one through six for pain intensity differences (PID) from baseline and for pain relief over time. In addition, acetaminophen 1000 mg was shown to be superior to naproxen 375 mg at one hour after treatment for PID from baseline (P=0.036). Further, while there was no statistically significant difference between acetaminophen 1000 mg and naproxen 375 mg, only acetaminophen 1000 mg was statistically superior to placebo (P=0.003) for the percentage of subjects with headache pain reduced to none at two hours.
Naproxen 375 mg was superior to placebo from hours two through six for PID from baseline and for pain relief over time; results were consistent with several previous double-blind trials that used naproxen sodium (14–16). These study findings differ from the expected onset of relief statement provided in the product label, which states that acute pain relief can begin within one hour for naproxen.
One published study (14) compared a lower dose of acetaminophen (650 mg) to a higher prescription dose of naproxen sodium 550 mg (500 mg of naproxen) in the treatment of moderate-to-severe tension headache. Miller and colleagues evaluated 124 subjects and reported that naproxen sodium 550 mg provided more analgesia than acetaminophen 650 mg or placebo.
It has been suggested that the results obtained from the use of a five point categorical pain scale (none, mild, moderate, moderately severe, and severe) might have been different if the study had utilized a four point categorical pain scale (none, mild, moderate, severe) suggested by IHS Guidelines (17). The data were reanalysed by combining the categories of moderately severe and severe and recalculating the affected endpoints (SPID, MAXPID, and the mean pain intensity differences). The results of the reanalysis were essentially the same with both acetaminophen and naproxen remaining significantly different from placebo (SPID P<0.004; MAXPID P<0.016). While this reanalysis cannot precisely predict how patients would have categorized their pain intensity on a four point categorical scale, it does suggest that the results would have been similar if a four point scale had been used and supports our conclusion that both acetaminophen 1000 mg and naproxen 375 mg are effective in the treatment of tension-type headache.
Acetaminophen 1000 mg and naproxen 375 mg were both well tolerated in this single-dose six-hour study. No serious adverse events were reported. There was no statistically significant difference in the incidence of reported adverse events among the three treatment groups; 10.5% of treated subjects reported an event. The most commonly reported events were gastrointestinal (mainly dyspepsia and nausea).
In summary, the results of this well-controlled, double-blind study demonstrate that over-the-counter acetaminophen 1000 mg and prescription naproxen 375 mg are effective and well tolerated in the treatment of tough (moderate-to-severe pain intensity) tension-type headache. Acetaminophen 1000 mg was shown to be significantly superior to naproxen 375 mg at one hour after treatment for pain intensity difference from baseline.
Footnotes
Acknowledgements
This study was conducted, analysed and supported by McNeil Consumer & Specialty Pharmaceuticals, Fort Washington, PA. We are grateful for and acknowledge the team of investigators who conducted the clinical research at their individual sites: Dan Bressler, MD, San Diego, California; Larry Gilderman, DO, Pembroke Pines, Florida; David Ginsberg, DO, Harleysville, Pennsylvania; Claude Goswick, MD, Bryan, Texas; Mark Greenbaum, MD, Sacramento, California; Dan Henry, MD, Salt Lake City, Utah; Lawrence Jenkyn, MD, Lebanon, New Hampshire; Barry Marmorstein, MD, Bellevue, Washington; Frank Nieto, MD, Everett, Washington; Nona Niland, MD, Austin, Texas; Douglas Owens, MD, Greer, South Carolina; J. Joseph Prendergast, MD, Redwood City, California; Lance Rudolph, MD, Albuquerque, New Mexico; Gary Ruoff, MD, Kalamazoo, Michigan; Harry Serfer, DO, Hollywood, Florida; Mark Sontag, MD, Redwood City, California; David Stryker, MD, Albuquerque, New Mexico; Brian Trafficante, MD, Portland, Oregon; David Williams, MD, Daytona Beach, Florida.
