Abstract
The aim of this review is to evaluate the studies available from reference systems and published congress contributions on the prophylactic treatment of idiopathic and cervicogenic headache with botulinum toxin A, and to classify these studies according to evidence-based medicine (EBM) criteria. The studies were analysed with respect to the study design, the number of patients enrolled, the efficacy parameters, and the significance of results. We used the following classification of EBM. I: randomized, controlled study with sufficient number of patients; II: well-designed, controlled study (or randomized, controlled study with insufficient number of patients, no exact diagnosis, missing data of botulinum toxin A dose); III: well-designed, descriptive study; IV: case reports, opinions of experts. For tension-type headache, two studies were found with negative evidence of I with respect to the primary endpoint. There are about as many positive as negative studies with evidence of II or III. For the therapy of migraine, one study with both negative and positive evidence of I, one in part positive study of II, and three positive studies classified as III are available. Two studies on cervicogenic headache with evidence of II and III are contradictory. In addition, we found several positive case reports. For patients with cluster headache, there are positive and negative case reports. We found one positive case report for the treatment of chronic paroxysmal hemicrania. As a result of this analysis, we consider no sufficient positive evidence for a general treatment of idiopathic and cervicogenic headaches with botulinum toxin A to date. Further studies are needed for a definite evaluation of subgroups with benefit from such treatment.
Introduction
For many years, botulinum toxin A has been used in the treatment of dystonic movement disorders and in diseases with pathologically increased muscle tonus. In particular, the focal dystonias and infantile cerebral palsy show a high benefit from treatment with botulinum toxin A. Therapy of idiopathic headaches or pain disorders (e.g. back pain) with botulinum toxin A is relatively new. Most experience exists for the treatment of tension-type headache, but the prophylactic effect on migraine has also been studied.
For a systematic evaluation of the efficacy of botulinum toxin A in headache disorders, well-defined treatment procedures and an exact diagnosis, for headache according to the criteria of the International Headache Society (IHS) (1), is mandatory. In this review, all available studies on the prophylactic treatment of idiopathic headache and cervicogenic headache with botulinum toxin A (published as full paper or abstracts) are analysed and ranked according to evidence-based medicine (EBM) criteria (2). The definitions for the different EBM classes are given in Table 1. In order to present the current overview, the studies themselves are ranked. In general, EBM criteria classify the study data of one disease or related groups of disorders rather than a single study. Because of this method, it is not possible to give definite treatment recommendations in this review. We consider evidence as sufficient if at least two positive level I studies without contradictory studies are available. A synopsis of all studies available is given in Table 2. This review has been published in part before (3).
Classification of published studies according to their scientific validity in evidence levels (according to the criteria of the Agency for Health Care Policy and Research; Perfetto and Morris 1996)

Studies on botulinum toxin A in the treatment of idiopathic or cervicogenic headache

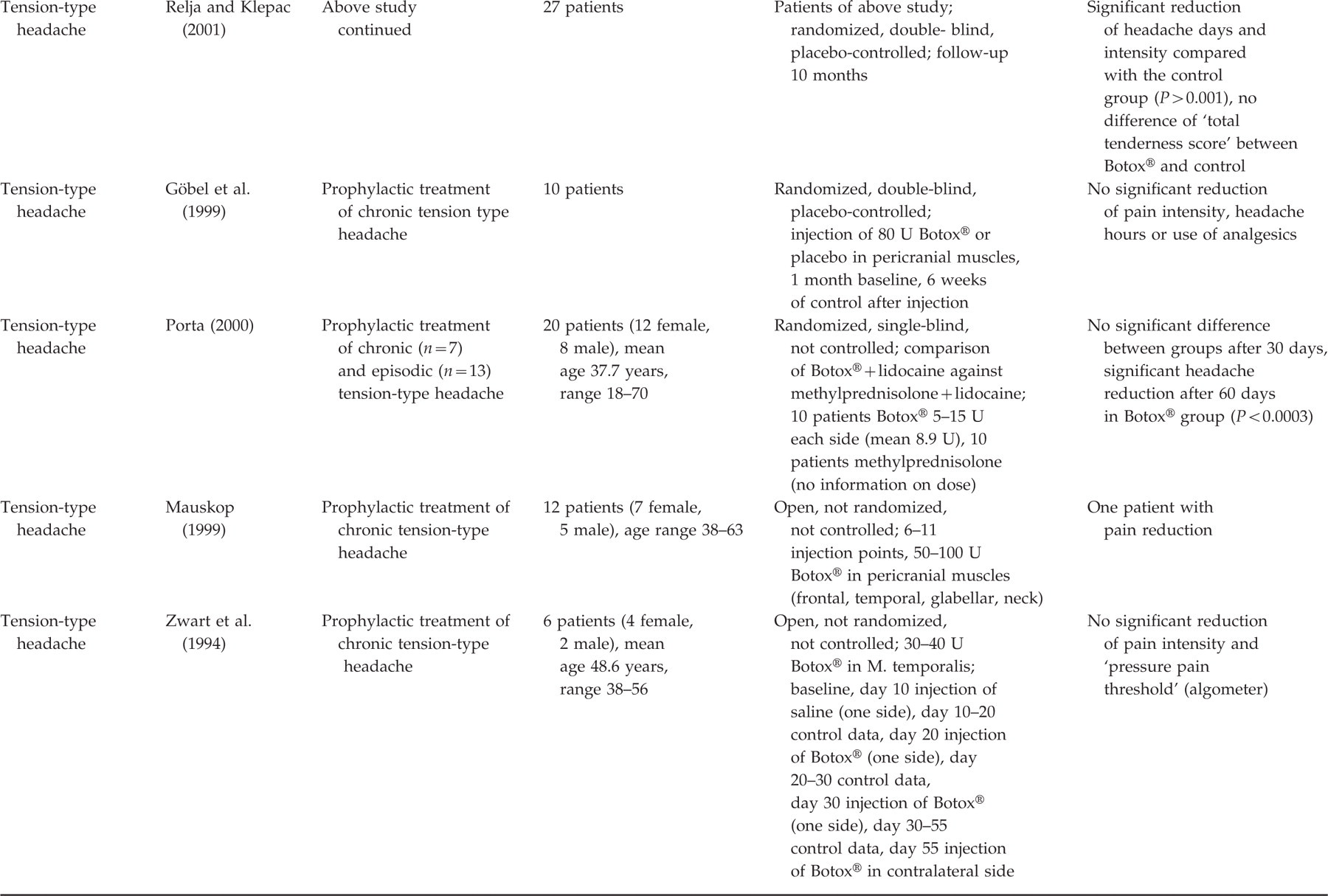




Tension-type headache
In five randomized, double-blind, and placebo-controlled studies on patients suffering from tension-type headache, contradictory results for the efficacy of botulinum toxin A were found. Schmitt et al. (4) enrolled 59 patients and could not find any significant difference between 20 U Botox® and placebo. Because of the high number of patients, an evidence level of IB is reached. The results of Burch et al. (5) could also not reveal a significant difference in headache frequency between treatment with Botox® (50 U) and placebo injected in pericranial muscles (41 patients). However, there was a significant decrease of pain intensity in the treatment group with Botox® compared with the control group. The study is ranked as IB. Rollnik et al. (6) studied the efficacy of 200 U botulinum toxin A (presumably Dysport®) against placebo. After 4, 8, and 12 weeks, the 21 patients showed no significant difference. Smuts et al. (7) showed a significant decrease of headache days in the 3 following months and, in month 3, a significant reduction of pain intensity in patients treated with 100 U Botox® compared with placebo (37 patients in follow-up period). In 27 patients randomized to placebo or the injection of 40–95 U Botox® every 3 months over a period of 10 months, the verum group experienced a significant reduction of headache frequency and intensity compared with the placebo group (8). These three studies reach an evidence level of II.
The design of the above studies mainly followed the recommendations of the IHS for studies on tension-type headache (9). In particular, the primary endpoint was the reduction of headache frequency. However, except for Schmitt et al. (4) and Burch et al. (5), the number of patients in the single subgroups is too small for evidence I. Furthermore, the dose of botulinum toxin A and the injection sites were not always standardized. The study design of Göbel et al. (10) was very similar to those above. No difference was found between botulinum toxin A and placebo in this study, but the number of patients was only 10. All additional studies of the prophylactic effect of botulinum toxin A on tension-type headache are either open, not controlled, have no exact diagnosis, have a too low number of patients, or do not define the reduction of headache frequency as the primary endpoint. The comparison between an injection of Botox® plus lidocaine and methylprednisone plus lidocaine (number of patients was 20) showed no significant difference after 30 days (11). After 60 days, the treatment group with Botox® suffered significantly less headache compared with the patients treated with methylprednisone plus lidocaine. Klapper et al. (12) enrolled 56 patients in their study. However, they separated into four subgroups, and no exact headache diagnosis (‘chronic headache’) was given. Treatment with botulinum toxin A in both frontal and suboccipital sites caused a significant reduction of headache compared with the other three subgroups. The open study of Robbins (13) with 79 patients treated with 24 U botulinum toxin A describes only chronic daily headache as diagnosis. After a control period of 3 months, 30% of the patients showed a good or moderate efficacy, 70% had no relief. Two studies of Relja (14, 15) either showed no significant results, the number of patients was under 16, or the design did not allow a statement on the evidence. Similar concerns are raised by the studies of Mauskop (16), Zwart et al. (17), Schulte-Mattler et al. (18), Smuts and Barnard (19), and Wheeler (20). Schulte-Mattler et al. (18) found a significant reduction of the ‘area under curve’. However, the defined primary endpoint, the reduction of headache days, was not influenced by botulinum toxin A. Rollnik et al. (21), in another study, examined the efficacy of 500 U botulinum toxin A (Dysport®) in chronic tension-type headache and also studied the EMG activity of different face and neck muscles. The study was placebo-controlled and parallel grouped (four patients in each group). Although EMG activity was significantly reduced by botulinum toxin A, there was no effect on the headache.
In summary, only two studies on the treatment of tension-type headache with an evidence of IB are found, the studies are negative. Further studies with similar design show too small a number of patients for an evidence level of I. As for evidence level of II, the number of positive and negative studies is the same. Only the studies ranked at level III show more positive than negative results.
Migraine
Silberstein et al. (22) enrolled 123 patients with migraine (diagnosis according to IHS criteria) with two to eight attacks per month. The study was randomized, double-blind, and placebo-controlled. The patients were treated with 25 U or 75 U Botox® against placebo. The follow-up control revealed a significant reduction of migraine frequency in month 3 and of pain intensity in months 2 and 3 in the 25 U group compared with placebo. Significantly more patients in the 25 U group had a reduction of >50% in month 3 compared with the control group. However, the patients treated with 75 U showed no significant results. According to the EBM criteria of the AHCPR (see Table 1), it is possible to separate this study into both a positive (for 25 U) and a negative (for 75 U) evidence. This evidence can be ranked as IB because of the high number of patients, the exact diagnosis, and a correct primary endpoint. Brin et al. (23) were able to show a significant reduction of pain intensity but not of migraine frequency. The study was randomized, double-blind, and placebo-controlled, but the differentiation of 56 patients into four subgroups, the unknown diagnosis criteria, and the unknown dose of botulinum toxin A do not support a classification better than evidence level II. The positive results of Binder et al. (24) can only be ranked as level III. Patients with migraine and with probable migraine were analysed together, the study was open and not controlled, and different doses of botulinum toxin A were used. Furthermore, some patients received several injections as acute and prophylactic treatment which resulted in a different frequency of the injections within the study. Mauskop and Basdeo (25) realized an open, not controlled study with 25 patients treated with botulinum toxin A in different doses and injection sites. The positive prophylactic effect of the patients can be classified as a positive study with an evidence level of III. Smuts and Barnard (19) treated different idiopathic headaches with botulinum toxin A including 19 patients with migraine. Positive results were found in 13 of 19 cases. There was no statement on significance, different doses were injected in different sites, and there was no randomization or control. This study can be ranked as only level III.
For the prophylactic treatment of migraine with botulinum toxin A, one study with both positive and negative evidence of level IB can be found. One study with an evidence of II presents significant positive results for pain intensity, but not for migraine frequency. Three positive studies can be ranked in evidence level III.
Cervicogenic headache
Freund and Schwartz (26) treated 24 patients with cervicogenic headache (criteria according to Sjaastad et al. (27)) and six patients with possible cervicogenic headache (a clear distinction between cervicogenic headache and tension-type headache was not possible) with 100 U Botox® or placebo. The control examination after 4 weeks showed a significant pain reduction in the Botox® group. The study can only be ranked as an evidence of III because of partially inexact diagnoses, small number of patients, and unknown injection sites. A further randomized, double-blind, placebo-controlled trial showed only a trend to a lower analgesic use and a trend to a reduction of pain duration in weeks 5–9 after the injection of botulinum toxin A (28). The results must be classified as a negative study of evidence level II. Beside these studies, only positive case reports are available (19, 29).
Cluster headache
The application of botulinum toxin A for the treatment of cluster headache was reported only as case reports. Robbins (30) treated four patients with chronic and two patients with episodic cluster headache with 24 U Botox®. Two patients experienced abrupt pain relief, two patients felt a moderate effect with a reduction of attack frequency, and two patients reported no effect. Smuts and Barnard (19) reported four patients, of whom two had a benefit after injection. In two cases of Freund and Schwartz (31), the patients were pain-free after injection, one patient after 6 days, the other after 9 days. The effect had a duration of several weeks, a second injection after recurrence was again successful. One successful case report on the treatment of chronic paroxysmal hemicrania was published by Göbel et al. (32)
Pathophysiological considerations
For the explanation of successful treatment of idiopathic headaches with botulinum toxin A, different hypotheses with different pathophysiological concepts can be found. Various authors discuss peripheral myogenic as well as central antinociceptive mechanisms (33–35).
A direct myotonolytic effect of botulinum toxin A as the underlying mechanism of a potential pain relief seems obvious, and is based on the experience that in dystonia the substance can offer a pain-decreasing effect independently from the anti-spastic effect in adults (36, 37) and in children (38). However, this hypothesis might not suffice as an explanation, because to date there is no evidence that a pathologically increased muscle tonus is the only reason for a primary headache. On the contrary, most experimental studies are not in concordance with this hypothesis (39). The concept of a peripheral mode of action is supported by the effect of botulinum toxin A on muscle spindles in experimental animal studies (40, 41). A direct affect of intra- and extrafusal γ-fibres was demonstrated, resulting in a reduced activity of α-motor neurons and a decreased muscle tonus.
Experimental studies on the central effect of botulinum toxin A were able to show that the substance is internalized in neurons and can be transported afferently, and that uptake in cultures of hippocampal neurons and astrocytes is possible (42, 43). This uptake in nociceptive neurons leads to a decreased release of neuropeptides (e.g. substance P) in an in vitro animal model (44). An increased release of neuropeptides as well as sensitization of trigeminal nociceptors of the first branch are well known mechanisms in idiopathic headaches (45).
Our current experimental knowledge about the central antinociceptive mechanisms of botulinum toxin A is based on animal models; human studies are missing. However, the studies also show that botulinum toxin A might have an impact on nociceptive (and antinociceptive) structures located in the brain-stem which exert influence on the pathophysiology of idiopathic headache.
Discussion
In this review, we analysed the current published data on botulinum toxin A in pain therapy. For classification, we used the EBM criteria as published by Perfetto and Morris (2), and modified them by including a limit of at least 20 subjects in a single subgroup. Although arbitrary, we think it is appropriate to demand this number of patients for evidence level I.
The analysis of the available studies and experience with botulinum toxin A in the treatment of idiopathic headache shows that a general efficacy of this therapy cannot be postulated to date. However, with respect to the often insufficient design of the trials, an inverse statement of a general inefficacy in idiopathic headache is not possible, either.
For tension-type headache, only two studies with an evidence level of I are available, with a negative result. Within the evidence levels of II and III, the number of positive and negative studies is about the same. It is necessary to take into consideration that the probability of publishing negative results or case reports is poorer.
Concerning the prophylactic treatment of migraine, one both positive and negative study (dependent on the dose) with an evidence level of I can be found. Beside this study, one level II and three level III studies found an efficacy of botulinum toxin A. This small number of studies does not allow a final evaluation.
For cervicogenic headache, no definite classification is possible to date, either. One negative study (level II) and one positive study (level III) show contradictory results. Several positive case reports can be added.
For patients with cluster headache, only case reports have been published to date. Approximately half of the patients experienced relief. Notably, the phenomenon that some patients were pain-free directly after the injection cannot easily be explained by the known pharmacological characteristics of botulinum toxin A. The treatment of cluster headache with botulinum toxin A must still be considered as purely experimental.
In conclusion, a study design according to the IHS recommendations would be helpful for a reliable classification of the studies. Botulinum toxin A might be a sufficient therapy for defined subgroups of patients with idiopathic headache, but well-designed and controlled studies with a high number of patients are needed for a valid evaluation. Furthermore, the known standard therapies in headache treatment need to be compared with botulinum toxin A. This can be performed in blinded double-dummy studies, which are, however, very difficult to design. It is nevertheless important for the development of treatment recommendations to discuss the advantages and disadvantages of this particular prophylactic procedure in comparison with oral medication (e.g. considering the therapeutic gain or the number needed to treat).
