Abstract
In order to investigate the c-fos response within the trigeminal nucleus caudalis (Sp5C) after noxious meningeal stimulation, capsaicin (0.25, 0.5, 1 and 5 nmol) was administered intracisternally in urethane (1 g/kg) and α-chloralose (20 mg/kg) anaesthetized male mice. Capsaicin induced a robust and dose-dependent c-fos-like immunoreactivity (c-fos LI) within Sp5C. C-fos LI was observed within laminae I and II of the entire brain stem from the area postrema to C2 level, being maximum at the decussatio pyramidum level. The area postrema, solitary tract, medullary and lateral reticular nuclei were also labelled. The 5-hydroxytryptamine1B/1D/1F receptor agonist sumatriptan (0.01, 0.1, 1 and 10 mg/kg), administered intraperitoneally 15 min before capsaicin stimulation (1 nmol), decreased the c-fos response within Sp5C, but not within solitary tract. The novel specific 5-hydroxytryptamine1F agonist LY 344864 (0.1 and 1 mg/kg, i.p.) significantly decreased the c-fos LI within the Sp5C as well. These findings suggest that intracisternally administered capsaicin activates the trigeminovascular system and that the pain neuro-transmission can be modulated by 5-hydroxytryptamine1B/1D/1F receptors in mice. Thus, the availability of this model in mice, taken together with the possibility of altering the expression of specific genes in this species, may help to investigate further the importance of distinct proteins in the neurotransmission of cephalic pain.
Introduction
The unmyelinated A-δ and C-fibres of the trigeminal nerve transmit painful stimuli from the meninges to the trigeminal nucleus caudalis (Sp5C) within the brain stem, via the trigeminal ganglion (TG) cells. Their peripheral endings surround as a plexus the meningeal vessels (1) and contain vasoactive peptides, mainly substance P (SP) and calcitonin gene-related peptide (CGRP). This neurovascular network is defined as the trigeminovascular system (2). In laboratory animals, activation of this system induces a sterile inflammatory response within the meninges associated with the release of the neuropeptides SP and CGRP (the so-called neurogenic inflammation) (3). Centrally, within the Sp5C cells, nuclear changes are seen as well, such as expression of c-fos protein (4).
C-fos protein is a transcription factor expressed by the immediate-early response c-fos gene within neurones and glial cells. Early studies showed that c-fos-like immunoreactivity (c-fos LI) within the Sp5C can be used as a marker of cephalic sensory activity (5). After delivery of noxious stimuli to the periphery, c-fos is expressed by cells in lamina I and II of the Sp5C, which contain the pain-receiving and transmitting zones of the brain stem (6). Experimentally, c-fos within the Sp5C is induced after stimulation at various sites of the trigeminovascular system, such as the meninges, the TG and the superior sagittal sinus (SSS) (4).
Meningeal stimulation was first achieved by injecting autologous blood into the subarachnoid space of rats. This technique, relevant to the headache seen after subarachnoid haemorrhage in humans, induced c-fos LI within Sp5C by activating the peripheral endings of C-fibres (7). Intracisternal (i.c.) capsaicin was used in another paradigm for induction of c-fos LI in Sp5C in guinea pigs (8) and rats (9). Electrical stimulation of TG (10) or SSS (11) also induces c-fos LI in Sp5C of rats and cats, respectively. Using these paradigms, it has been shown that c-fos expression is modulated via 5-HT1B/1D (12, 13), NMDA (9), non-NMDA (14) and GABAA (15) receptors. Recently, using the technique of i.c. capsaicin injection in rats, we have shown that c-fos expression in Sp5C cells is also regulated by the 5-HT1F receptors (16).
The aim of this study was to establish a model of trigeminovascular activation in mice and to show that c-fos LI is also modulated by the 5-HT1B/1D/1F receptors in this species. The availability of transgenic and knockout technology in mice will help to elucidate the role of specific proteins (e.g. receptor subtypes) in pain neurotransmission within the trigeminovascular system.
Materials and methods
Animal preparation
Male mice (129/SVEvTacBR; 25–30 g; Taconic Farms, MA, USA) were housed under diurnal lighting conditions and allowed food and water ad libitum. The animals were anaesthetized with intraperitoneal (i.p.) urethane (1 g/kg) and α-chloralose (20 mg/kg) and were injected intraperitoneally 15 min later with either drug or drug-vehicle. After 15 min, the capsaicin solution (see Drugs) was injected into their cisterna magna (over 5 min) via a Hamilton syringe with the aid of a stereotaxic frame, after a surgical opening was made in the region between the scalp and C1. To avoid capsaicin leakage, the needle was removed 10 min after the injection was over. The animals were placed into a homeothermic blanket and remained anaesthetized for 2 h until euthanasia was performed via a pentobarbital overdose (120 mg/kg, i.p.), followed by perfusion via the ascending aorta with 0.9% saline (10 ml), and 4% formaldehyde (20 ml) in 0.1
C-fos immunohistochemistry
Tissue sections were processed as free floating sections with the avidin–biotin procedure using commercially available kits. Sections were incubated in 0.3% hydrogen peroxide (Sigma Chemical Co., St Louis, MO, USA) in 0.1
Cell counting
C-fos-positive nuclei were counted by an observer naive to the treatment group (D.D.M.) and confirmed (in randomly selected sections) by another investigator (M.S.R.) blind to the treatment groups. C-fos LI cells induced by increasing doses of capsaicin (see Capsaicin treatment) were counted in laminae I, II of Sp5C within 34 serial sections from AP caudal to C2 level and then the average number of c-fos LI cells per 25-µm section was calculated. After drug treatment, c-fos LI cells within Sp5C were counted using the weighted-average method within 12 selected sections (see below). An assessment of the extent of c-fos LI in area postrema (AP), solitary tract (Sol), lateral (LRt) and medullary reticular nuclei (Md; visible in three serial sections) was performed as well. In these nuclei the average number of labelled cells per section was calculated. To identify AP, Sp5C, LRt, Md, and Sol we compared the c-fos LI staining with the histology shown in the Sidman, Angevine and Pierce Atlas (17).
Weighted average method
The number of c-fos LI-labelled cells in the entire Sp5C was estimated using the weighted average method described previously in guinea pigs and rats. Based on the observation that c-fos LI was maximal at the levels 0.4–1.2 mm caudal to the lowest level of AP and then decreased linearly on either side of this level (Fig. 1), four 25-µm sections were counted at each of three levels from AP (the most caudal level) to 0.15 (caudal to AP), 0.65–0.8 and 1.95–2.1 mm. The mean values from each of the three levels were designated X1, X2 and X3, respectively (Fig. 2). The values X1, X2 and X3 correspond to the heights AB, DC and EF, respectively (Fig. 2). The total number of labelled cells within the two trapezoids, ABCD and CDEF, can be estimated by the formula 12(× 1 + × 2)/2 for ABCD and 22(× 2 + × 3)/2 for CDEF. The sum of the two values is then divided by the total number of sections (34) to obtain the weighted average. To validate the weighted average method in mice, brain stem sections from 15 animals receiving capsaicin i.c. (1 nmol) plus drug-vehicle (normal saline, n=7), or sumatriptan (1 mg/kg, n=4), or LY 344864 (1 mg/kg, n=4) were analysed by both the weighted average sampling method and the counting of positively stained cells in Sp5C in every third section (34 sections).

C-fos response within the entire trigeminal nucleus caudalis (Sp5C), from the area postrema (AP) to C2 level, after intracisternal administration of increasing doses of capsaicin (three animals per dose). Capsaicin caused a robust induction of c-fos immunoreactivity (c-fos LI) within laminae I and II, as detected 2 h after capsaicin injection. Each dot represents the mean number of c-fos LI cells per 25-µm section at each level of Sp5C. Error bars represent the standard error. Numbers at the abscissa represent the distance from the most caudal aspect of AP (zero point) in mm.

The distribution of c-fos immunoreactivity (c-fos LI) within laminae I and II in the entire trigeminal nucleus caudalis (Sp5C), from the caudal parts of area postrema (AP) to C2 level, after intracisternal injection of 1 nmol capsaicin. The maximum of c-fos LI was observed at −0.75 mm level, caudal to AP, close to the decussatio pyramidum level. Each dot represents the mean number of c-fos LI cells per 25-µm section at each level of Sp5C obtained from four representative mice. Error bars represent standard error. Numbers at the abscissa represent the distance (mm) from the most caudal aspect of AP (zero point). For analysis, a weighted average method was used to estimate c-fos LI cell number based on mean values obtained at co-ordinates AB (x1), CD (x2) and EF (x3; four sections at each of three levels). The weighted average per mouse was estimated by the total area of the two trapezoids (ABCD and DCFE) divided by the number of sections (34).
Capsaicin treatment
The dose–effect relationship of capsaicin on c-fos LI within mouse brain stem nuclei was studied in preliminary experiments. Capsaicin diluted in artificial CSF (see Drugs) was administered intracisternally to 12 drug-vehicle-treated mice (normal saline) at four different concentrations (10 µl of 25, 50, 100 and 500 µ
In preliminary experiments we investigated the time course of c-fos LI induction within Sp5C and found that 2 h post-surgery c-fos LI reached the highest level.
Drug treatment
Drug vehicle (normal saline, i.p., n=9), or sumatriptan, 0.001 mg/kg (n=6), 0.01 mg/kg (n=4), 0.1 mg/kg (n=5), 1 mg/kg (n=6) and 10 mg/kg (n=4), or LY 344864 0.1 mg/kg (n=5) and 1 mg/kg (n=4) were injected intraperitoneally in anaesthetized mice.
Drugs
The capsaicin solution (8-methyl-N-vanillyl-6-nonenamide; Sigma) was made fresh 2 h before use. Capsaicin (1.55 mg) was diluted in 1 ml of saline : ethanol : Tween-80 (8 : 1 : 1). The solution was further diluted into artificial CSF (132 m
Systemic physiology
Physiological monitoring was carried out in three randomly selected animals from each group (drug-treated, sumatriptan 1 mg/kg and LY 344864 1 mg/kg). After anaesthesia with i.p. urethane and α-chloralose, a catheter (PE-10, internal diameter 0.28 mm; Becton Dickinson, Sparks, MD, USA) was advanced into the left femoral artery. The animals were placed into the stereotaxic frame followed by the usual procedure as described above. The effect of i.c. capsaicin injection (1 nmol) on mean arterial blood pressure (MABP) and heart rate (HR) was measured for 60 min after treatment. MABP and HR were monitored using Mac/Lab8 data acquisition system (AD Instruments, Medford, MA, USA) equipped with an ETH 400 transducer amplifier. The effect of capsaicin (1 nmol) on arterial pH, PaCO2 and PaO2 was also studied by measuring blood samples (10 min after i.p. drug treatment, 10 and 30 min after capsaicin i.c. injection) using a Corning 178 from Ciba-Corning Diagnostics apparatus (Medford, MA, USA). Core temperature was maintained at 36–37°C by a homeothermic blanket (Harvard Apparatus no. 551, South Natick, MA, USA).
Statistical analysis
Data are expressed as a weighted average±standard error of cells per 25-µm section. The weighted averages were compared by one-way
Results
Physiological parameters after capsaicin treatment
Baseline values are shown in Table 1. (i) Vehicle: intracisternal injection of saline did not significantly change HR, PaO2 or PaCO2, whereas 10 min after i.c. capsaicin injection HR decreased (8%) and PaO2 increased (up to about 30%), slowly returning to baseline within 30 min. (ii) Sumatriptan-treated animals: 10 min after i.p. sumatriptan treatment, PaCO2 increased (40%, P<0.05), PaO2 decreased (26%), and pH decreased from 7.36 to 7.27; HR also increased; 30 min after capsaicin treatment, PaO2 returned to baseline levels and PaCO2 remained higher (35% approximately); HR decreased after capsaicin treatment and then increased again (P<0.05); pH was stable at 7.27. (iii) LY 344864-treated animals: HR decreased by 11% after capsaicin treatment (P<0.05), returning to baseline levels 40 min later; no other significant changes were observed.
Heart rate (HR), mean arterial blood pressure (MABP), PaCO2, PaO2, HCO3 and pH in animals treated with drug-vehicle (normal saline, i.p.), sumatriptan (i.p. 1 mg/kg) and LY 344864 (1 mg/kg)

Values were measured in three randomly selected animals from each group, at four different time-points: (i) baseline, before any treatment (animals with intracisternal catheters only); (ii) 10 min after i.p. drug treatment (before capsaicin); (iii) 10 min after intracisternal capsaicin administration (1 nmol); and (iv) 40 min after capsaicin treatment.
∗Significant difference compared with baseline.
Capsaicin-induced c-fos expression within mice brain stem nuclei
In capsaicin vehicle-treated animals only very few c-fos LI-labelled cells were found at each level of the brain stem (up to five cells, without a specific pattern of distribution). Sp5C was not labelled.
Capsaicin dose-dependently induced c-fos LI within laminae I and II of Sp5C (Figs 1 and 3). The AP, Sol, Md and LRt were also labelled, as well as the leptomeninges (Fig. 3). No positive correlation between the dose of capsaicin and the number of c-fos LI per section was found in these nuclei. After capsaicin treatment (1 nmol) the mean number of c-fos LI was 150 ± 10, 95 ± 18, 55 ± 8, 16 ± 3 and 29 ± 7 cells per section in Sp5C (the entire nucleus, from AP to C2), AP (the most caudal parts), Sol, Md and LRt, respectively. C-fos LI was present bilaterally and most intensely within dorsal than ventral aspects of laminae I, II at each sampled level of Sp5C. The magnitude of c-fos LI expression was greatest at −0.75 mm (caudal to AP, very close to the decussatio pyramidum level) with a mean of 355 ± 30 cells per section (Fig. 2). At the level of AP the mean number of c-fos LI-labelled cells in Sp5C was 118 ± 47 and at C2 level was 62 ± 13 cells per section.
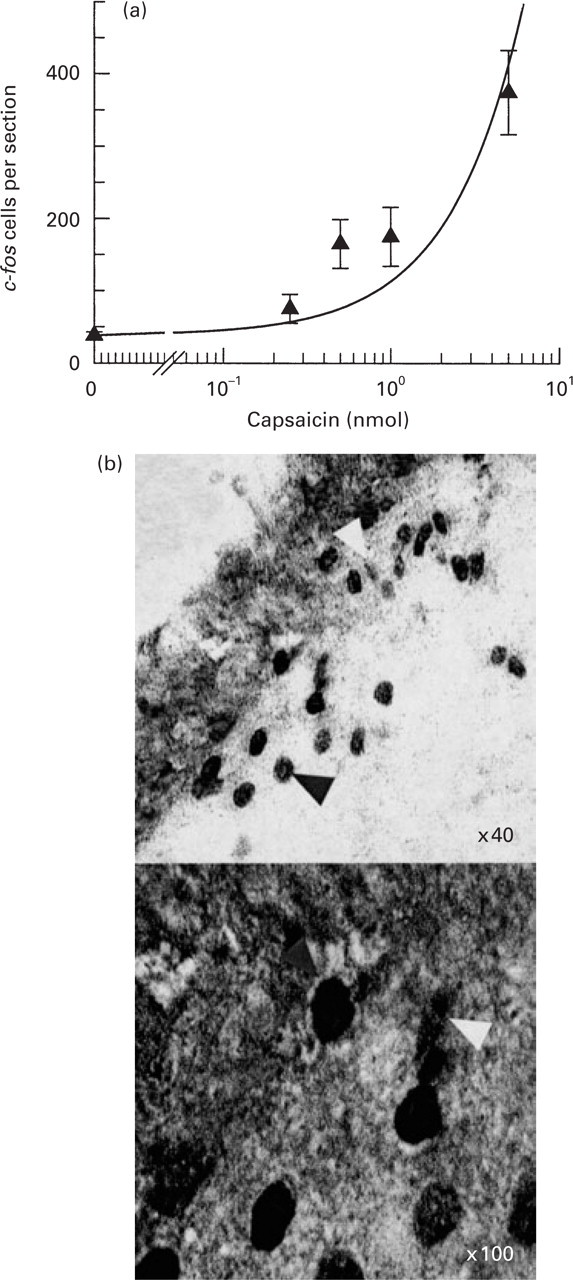
(a) C-fos immunoreactivity (c-fos LI) within the trigeminal nucleus caudalis (Sp5C) after intracisternal administration of several doses of capsaicin. Solid triangles represent the mean of c-fos LI-expressing cells within the entire Sp5C, as estimated by the weighted average method (four animals at each dose of capsaicin). Capsaicin caused a dose-dependent c-fos LI response within the Sp5C of mice. (b) Two photomicrographs showing c-fos LI cells (dark arrows) within Sp5C after intracisternal capsaicin (1 nmol), at the decussatio pyramidum level of a vehicle-treated representative animal. For counting × 40 magnification was used (for example, in the upper picture, mag. × 40, 18 c-fos LI cells were counted). The white arrow shows the type of staining that was not counted as c-fos LI cell.
Validation of the weighted average method
The numbers obtained counting every third section (34 sections for each animal) were similar to those obtained using the weighted average method of counting four sections at each of three representative levels of brain stem, in seven drug-vehicle-, four sumatriptan-, or four LY 344864-treated animals. Average numbers of cells per section were similar for the two methods and the differences between each were ≤10%. In addition, the decrease caused by drug treatments was also very similar using both cell counting methods, the differences being ≤3% (Table 2).
Validation of the weighted average method
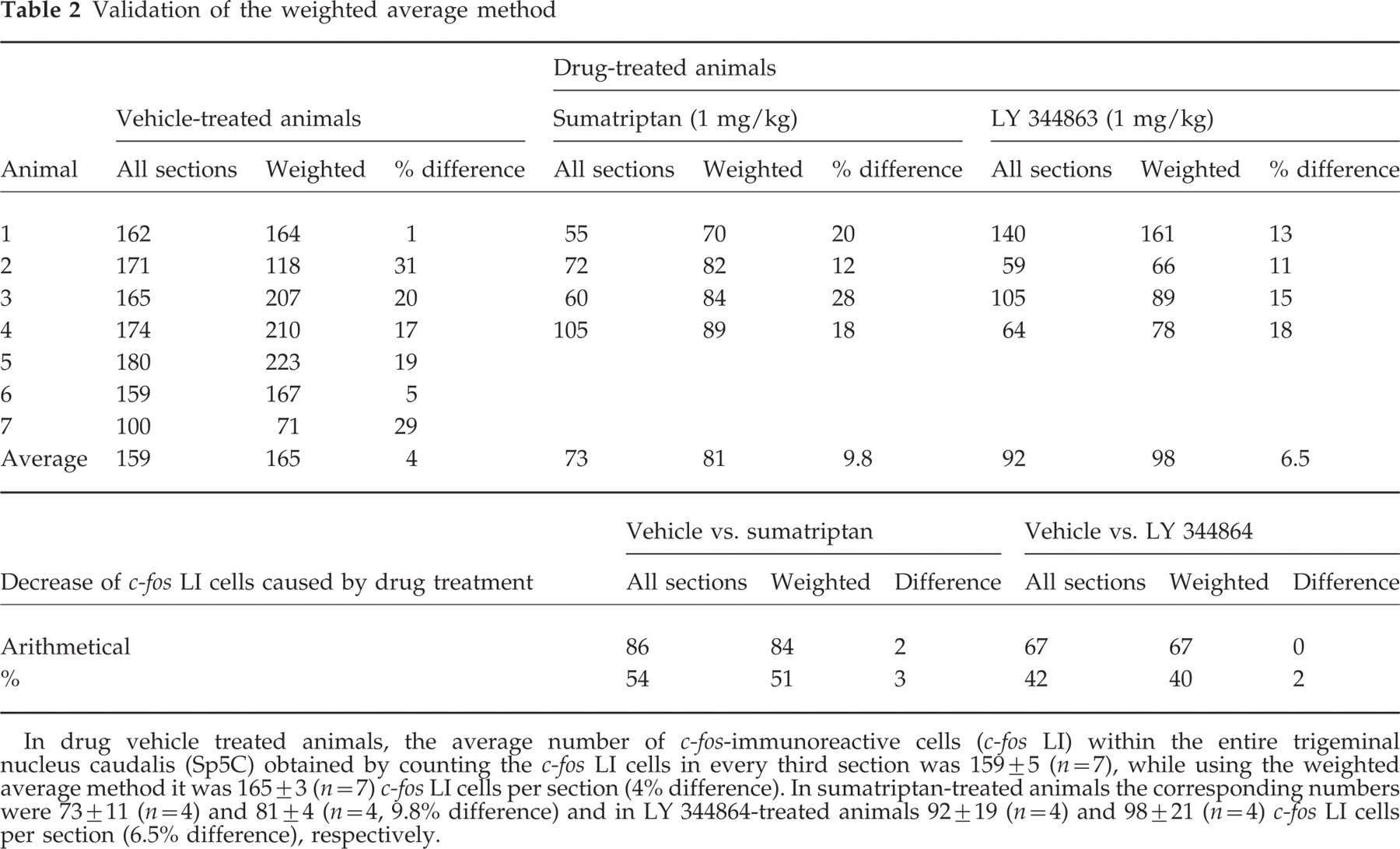

In drug vehicle treated animals, the average number of c-fos-immunoreactive cells (c-fos LI) within the entire trigeminal nucleus caudalis (Sp5C) obtained by counting the c-fos LI cells in every third section was 159 ± 5 (n=7), while using the weighted average method it was 165 ± 3 (n=7) c-fos LI cells per section (4% difference). In sumatriptan-treated animals the corresponding numbers were 73 ± 11 (n=4) and 81 ± 4 (n=4, 9.8% difference) and in LY 344864-treated animals 92 ± 19 (n=4) and 98 ± 21 (n=4) c-fos LI cells per section (6.5% difference), respectively.
Drug treatment
Sumatriptan and LY 344864 dose-dependently reduced capsaicin-induced c-fos LI expression within Sp5C (Figs 4 and 5). Treatment with sumatriptan (1 mg/kg) and LY 344864 (1 mg/kg) did not alter the number of c-fos LI cells within AP, Sol, Md, and LRt (data not shown).

Photomicrographs of three sections (25 µm) taken from the brain stem of two representative mice. In the left panel the animal was treated with drug-vehicle (normal saline, i.p.) plus intracisternal capsaicin (1 nmol), and in the right panel the animal was co-treated with sumatriptan (1 mg/kg, i.p.) and intracisternal capsaicin (1 nmol). The upper sections were taken from the most caudal level of area postrema (AP) (level 0), the mid sections from the decussatio pyramidum level (0.65 mm caudal to AP), and the lower sections from the C2 level (2.0 mm caudal to AP). Capsaicin induced c-fos LI bilaterally within the laminae I and II of the trigeminal nucleus caudalis (Sp5C), AP, solitary tract (Sol), medullary reticular (Md) and lateral reticular (LRt) nuclei (left panel). The maximum of c-fos LI within Sp5C was observed at the decussatio pyramidum level. Pretreatment with sumatriptan significantly reduced the number of c-fos LI within laminae I, II at each level of brain stem, but not within the deeper laminae (Sol, Md; right panel).

Histograms showing the mean of capsaicin-induced c-fos immunoreactive cells within the entire trigeminal nucleus caudalis (Sp5C) of mice after treatment with drug vehicle (□), or several doses of either sumatriptan (▪) or LY 344864 (hatched). Error bars represent the standard error. ∗Significant difference (P<0.05) compared with drug vehicle. Both sumatriptan and LY 344864 significantly decreased the c-fos LI response within the Sp5C.
Discussion
We first showed that i.c. capsaicin induces c-fos LI expression within the Sp5C of mice in a dose-dependent manner. The pattern of the c-fos LI-expressing cells in all the brain stem nuclei is similar to that previously observed in rats. We have also validated the weighted-average technique of counting c-fos LI cells within the entire Sp5C. Then we have shown that the anti-migraine drug sumatriptan that displays high affinity to 5-HT1B/1D/1F receptors, and LY 344863, a selective 5-HT1F receptor agonist, inhibit the c-fos LI response within Sp5C, but not in other brain stem nuclei, after noxious meningeal irritation with i.c. capsaicin.
The distribution of c-fos LI within Sp5C and other brain stem nuclei
The pattern of c-fos LI within brain stem nuclei in mice induced by i.c. capsaicin was similar to that previously reported in rats after either capsaicin or autologous blood i.c. injection (7, 9), or in guinea pigs after i.c. capsaicin (8). The rostro-caudal distribution was similar to that reported in other species. The magnitude of expression was greatest at the level of decussatio pyramidum in mice, a level similar to the region with maximal expression in guinea pigs and rats (−2.47 and −2.05 mm caudal to obex, respectively) (8, 9). At more caudal levels, the number of c-fos LI cells gradually decreased. As in rats (9), capsaicin induced c-fos LI within Sp5C in a dose-dependent manner. As in rats and guinea pigs, other brain stem nuclei were labelled in addition to Sp5C, including AP, Md, Sol and LRt (7–9). The somatotopic and laminar distribution of either facial or dural sites has been extensively investigated in reports by Strassman and colleagues (18–21). However, this is the first report of the pattern of c-fos LI expression in the mouse brain stem after noxious meningeal stimulation. Furthermore, the physiology data recorded showed that neither the surgical procedure nor the drug administration significantly altered the cardiovascular and respiratory function of the animals, parameters that induce c-fos LI expression. Therefore, inhibition of c-fos LI within Sp5C is likely to be attributable to direct drug effect, making this model reliable for future pharmacological studies.
Most anaesthetics induce c-fos LI expression within brain stem nuclei (e.g. barbiturates in the Sp5C (22), urethane in Sol and the intermediate reticular formation (19)). Because our aim was primarily to investigate c-fos LI expression within Sp5C, we chose as anaesthetic a mixture of urethane and α-chloralose. Urethane was used in previous experiments done in rats (9, 14, 16), whereas α-chloralose is used for c-fos LI studies within cat Sp5C (11, 23). We found in preliminary studies that, while large doses of urethane alone (up to 5 g/kg) were needed to induce anaesthesia in mice, addition of 20 mg/kg α-chloralose allowed significant reduction of the amount of urethane required to maintain areflexia. This anaesthetic mixture was well tolerated by the animals.
C-fos expression within mouse Sp5C is modulated by 5-HT1B/1D/1F receptor subtypes
Serotonin exerts its effects by binding to G protein-coupled receptors classified into six major classes (5-HT1,2,4,5,6,7) according to their pharmacological properties, primary structures and transduction mechanisms (24, 25). Among these, only a few have been shown in functional studies to be involved in pain neurotransmission within the trigeminovascular system: the 5-HT1B, 5-HT1D, 5-HT1F and 5-HT2B subtypes (4, 26–28). The distribution of 5-HT1 subtypes within the human trigeminovascular system is selective. 5-HT1B mRNA and receptor immunoreactivity, but not 5-HT1D mRNA, nor receptor immunoreactivity, is densely localized within smooth muscle and less in the endothelium of both the cerebral and coronary blood vessels (29–32). This vascular distribution has been shown to mediate the vasoconstrictive properties of the triptans (33–35). In contrast, the 5-HT1D receptor is localized exclusively within the neuronal tissue and lacks vasoconstrictive properties (32, 35). Although 5-HT1F receptor is localized within both the neuronal tissue (trigeminal ganglion and Sp5C), and vascular tissues (31, 36), it is devoid of vasoactive properties (37, 38). Based on these characteristics, and considering the significant cardiovascular side-effects of triptans (39), 5-HT1D and 5-HT1F receptors have become attractive targets for the development of new anti-migraine drugs with improved safety profile.
LY 344864 is a novel 5-HT1 receptor agonist that displays considerable selectivity for 5-HT1F (pKi=8.1) vs. 5-HT1B (pKi=6.3) and 5-HT1D (pKi=6.2) binding sites (40). The drug is not vasoactive and inhibits neurogenic dural inflammation in rats with an ID50 value of 0.6 ng/kg (2 pmol/kg, i.v.) (40). LY 344864 and other selective 5-HT1F receptor agonists also reduce plasma protein extravasation in guinea pigs (41, 42). Sumatriptan inhibits neurogenic inflammation with an ID50 of 20 ng/kg (5 nmol/kg, i.v.) and 27 ng/kg (6.8 nmol/kg, i.v.) in guinea pigs and rats, respectively (43, 44). We have previously reported that LY 344864 and sumatriptan inhibit capsaicin-induced c-fos LI within Sp5C in rats at doses higher than 0.3 and 0.01 mg/kg, s.c., respectively (or with an ID50 of 0.6 mg/kg (1.5 µmol/kg, s.c.), and 0.04 mg/kg (0.1 µmol/kg, s.c.)) (16). Thus, both drugs are more potent in inhibiting the neurogenic inflammation than c-fos LI within Sp5C, in rats. The 5-HT1A/5-HT1B receptor antagonist SDZ 21-009 (45) prevented the effects of sumatriptan, but not of LY 344864 on the c-fos LI response after i.c. capsaicin, indicating that stimulation of the 5-HT1F subtype is sufficient to inhibit the trigeminovascular pathway in rats (16). In the present study, we investigated the role of 5-HT1B/1D/1F receptor subtypes in the same paradigm of cephalic pain, but in a different rodent species, and we found that the same receptor populations modulate trigeminal nociception in mice. As in rats, LY 344864 decreased c-fos LI at doses ≥0.1 mg/kg, while sumatriptan attenuated c-fos LI expression within Sp5C at doses >0.01 mg/kg, s.c. 5-HT1F subtype receptors have been detected in the trigeminal ganglion and trigeminal nucleus. Activation of these receptors in the ganglion would lead to presynaptic inhibition and in the trigeminal nucleus to direct inhibition of second order neurones.
Like LY 344864, LY 334370 is a 5-HT1F receptor agonist with approximately 100-fold selectivity over the 5-HT1B and 5-HT1D subtypes (42). Shepheard and colleagues found that LY 334370 inhibited evoked neuronal responses within rat Sp5C induced by electrical stimulation of the dura matter at doses >1 mg/kg, i.v. (46). In a preliminary report, LY 334370 also showed efficacy in inhibiting c-fos LI within Sp5C in rats, induced by electrical stimulation of the trigeminal ganglion (47). Interestingly, LY 334370 showed significant efficacy in aborting migraine attacks in a recent preliminary report from a phase II, double-blind, placebo-controlled clinical trial (48). These data, together with preclinical data from our laboratory on the LY 344864 compound, indicate that the 5-HT1F receptor subtype plays an important role in modulating transmission in the trigeminal system. Future drugs targeting this subtype may have anti-migraine efficacy without the vascular effects that currently restrict the use of triptans.
In conclusion, we have developed a novel rodent model using c-fos LI as a marker for noxious trigeminovascular activation. This paradigm has previously been used in rats as well as guinea pigs, and was successfully applied to show the involvement of various receptors in the modulation of pain neurotransmission within the trigeminovascular system (4). Considering the possibility of selectively altering the expression of specific genes in mice species, this model could be useful in investigating the role of various proteins in trigeminal pain.
Footnotes
Acknowledgements
This study was supported by the Hellenic Navy, General Staff (D.D.M.), by the American Association for the Study of Headache/Glaxo Wellcome Clinical Research Award 1998–99 (M.S.R.) and by the NIH (NS35611) (C.W.).
