Abstract
Primary headaches are closely related to sleep. Modifications in the patterns of arousal during sleep have been reported in migraine, especially in the nights preceding a headache attack. We aimed at evaluating the pattern of arousal from sleep in a group of patients affected by sleep-related migraine. We enrolled 10 consecutive patients, three males and seven females, aged between 20 and 62 years, who presented frequent attacks of migraine without aura (more than five per month), closely related to sleep (more than one-half of the attacks occurred during sleep, causing an awakening). A control group was studied, matched for age and sex. Patients and controls underwent a full-night polysomnographic study, following adaptation; arousal pattern was studied by the scoring of the high-frequency EEG arousal and by the cyclic alternating pattern (CAP). Migraineurs showed a lower CAP rate in non-rapid eye movement (NREM) sleep and, in particular, a lower number of A1 phases (low-frequency, high-amplitude EEG bursts) compared with the controls. Migraineurs also showed a lower index of high-frequency EEG arousals during rapid eye movement (REM) sleep. The reduction in the CAP rate indicates a lower level of arousal fluctuation in NREM sleep. The reduced arousal index in REM suggests a dysfunction in neural structures involved in both the control of REM sleep and the pathophysiology of migraine, such as the hypothalamus and the brainstem.
Introduction
Headache has an intimate relationship with sleep (1–3). Nocturnal headaches can be the consequence of disrupted sleep (4); conversely, some primary headaches may appear selectively during night sleep and may cause sleep disruption (3). Biochemical and functional imaging studies have reported potential headache generators that may colocalize with areas of sleep regulation (3, 5, 6). Cluster headache and hypnic headache are closely related to rapid eye movement (REM) sleep (2, 3), whereas nocturnal migraine attacks occur in REM and during slow wave sleep (7–9). Within the physiological sleep processes, a close relationship has been described between primary headaches and the mechanisms of arousal. Various anatomical and neurochemical systems are involved in arousal during sleep (10) and several arousal-related structures can be impaired in primary headaches: brainstem (6, 11), hypothalamus (12) and thalamo-cortical excitatory cholinergic afferents (13). In addition, a reduction of arousal in the night preceding attacks has been observed in migraine (14–16), hypnic headache (17) and cluster headache (18).
EEG arousals during sleep can be detected and quantified in accordance with the definition of the American Sleep Disorders Association (ASDA) (19) and also in accordance with the definition of microarousal proposed by the cyclic alternating pattern (CAP) model (20). The ASDA (19) includes in the definition of ‘EEG arousal’ only low-voltage fast rhythms (Fig. 1); within this conceptual framework, EEG arousals represent a harmful feature for sleep and are considered markers of sleep disruption (21). The CAP model broadens the concept of microarousal to include high-amplitude, low-frequency EEG bursts. Therefore, the CAP is a periodic EEG activity of non-rapid eye movement (NREM) sleep, consisting of sequences of transient electrocortical events (‘phases A’) separated by intervals of ≤1 min (‘phases B’) (Fig. 2). Three subtypes of A phases can be distinguished: A1 (high-amplitude, low-frequency bursts); A2 (mixed frequencies); and A3 (high-frequency arousals). CAP phases A1 are the EEG expression of a special kind of anti-arousal process, which is activated to counterbalance the effect of arousal; in this view A1 subtypes express the effort of the brain to preserve sleep: if this effort fails, then an overt, high-frequency EEG arousal occurs (21). Cyclic, spontaneous arousal fluctuation during NREM sleep (22) occurs during the processes of synchronization and desynchronization of the brain cortex and is necessary for the ‘building’ of a normal sleep structure; for this reason, an amount of CAP is ‘structural’ during normal NREM sleep. Arousal fluctuations also occur in response to endogenous or external perturbations (22, 23). For a more detailed description of the CAP model and its implications in the sleep dynamics, see Terzano and Parrino (22) and Halasz et al. (21).

High-frequency EEG arousal in non-rapid eye movement sleep. Polysomnographic tracing from patient 1. F4-A1, C4-A1, P4-A1, O2-A1, C3-A2: EEG traces; Roc and Loc: right and left electrooculograms; EMG: submental and intercostal muscles surface electromyography; Airflow, Abd., Thor.: airflow, thoracic and abdominal effort; ECG: electrocardiogram. Time calibration for is in the bottom right-hand corner.

Cyclic alternating pattern scoring in 3 min of non-rapid eye movement sleep. Polysomnographic tracing from patient 1. F4-A1, C4-A1, P4-A1, O2-A1, C3-A2: EEG traces; Roc and Loc: right and left electrooculograms; EMG: submental and intercostal muscles surface EMG; Airflow, Abd., Thor.: airflow, thoracic and abdominal effort; ECG: electrocardiogram. Time calibration is in the bottom right-hand corner.
The CAP model, since it refers exclusively to NREM sleep, gives no information on the occurrence of arousals in REM, when migraine attacks are most likely to occur (3).
The aim of the present study was to investigate the activity of the arousal systems during NREM and REM sleep in migraine patients who presented headache attacks occurring exclusively or prevalently during sleep.
Methods
Patients
We enrolled in the study 10 consecutive patients of both sexes (seven women and three men), aged between 20 and 62 years (mean 41.9 ± 13.9), fulfilling the criteria of the International Classification of Headache disorders 2nd edition (ICHD-II, (24)) for migraine without aura (MoA). We selected patients with a high frequency of attacks (five or more per month) and who presented more than 50% of their attacks during sleep, causing an awakening. Other inclusion criteria were the absence of prophylactic treatment during the study or in the previous 3 months, absence of pharmacological treatment of any kind in the month prior to the sleep study [with the exception of triptans or non-steroidal anti-inflammatory drugs (NSAIDs) administered for the acute treatment of attacks], absence of other medical or psychiatric disorders, absence of history of severe head trauma and absence of previous history of sleep disorders of any type. No patient fulfilled the ICHD-II (24) criteria for hypnic headache. All subjects were asked to fill in a migraine diary for 2 weeks before and 2 weeks after the polysomnographic (PSG) recording. Hamilton scales for anxiety and depression were ≤5 in all patients. The main clinical data concerning the patients’ group are summarized in Table 1.
Main clinical features of the migraine patients

NSAIDs, Non-steroidal anti-inflamatory drugs. Frequency of migraine is expressed as the number of attacks per month.
Ten healthy volunteers were also enrolled in the study in order to constitute a control group matched for age and sex with the patients (seven women and three men, aged between 18 and 61 years, mean age 43.2 ± 16.9, Student's t-test for age differences P = 0.828). All patients and controls gave written informed consent to participate. The study was performed in agreement with the Declaration of Helsinki and was approved by the local Ethics Committee.
Polysomnography
Patients and controls underwent a full-night laboratory nocturnal video-PSG following adaptation. In order to avoid any influence of acoustic stimuli on the sleep microstructure (23), patients and controls slept in a sound-proof room. PSGs were recorded by a Micromed System (Micromed s.r.l., Mogliano Veneto (TV), Italy) ‘98 digital polygraph. Montages included eight EEG leads applied to the following locations: Fp1, Fp2, C3, C4, T3, T4, O1, O2; reference electrodes applied to the left (A1) and right (A2) mastoids; two electrooculographic (EOG) electrodes applied to the cantus of each eye, surface electromyography (EMG) of submental and intercostal muscles, airflow measured by oronasal thermocouple, thoracic and abdominal effort, EKG (V2 modified derivation) and peripheral haemoglobin saturation. Sleep monitoring lasted from 23.00 h to 07.00 h on the next morning. A technician was present for data acquisition and video monitoring was performed throughout the registration.
Sleep analysis
Sleep recordings were analysed by a computer and sleep stages were visually classified by an expert physician according to the criteria of Rechtschaffen and Kales (25). The analysis of sleep-related respiratory events was made visually by an expert scorer.
Sleep microstructure
Sleep microstructure was evaluated by means of the detection of the arousals and the analysis of CAP. Arousal were visually detected and quantified in accordance with the ASDA rules (19); separate arousal indexes (number of arousals/time) were calculated for the sleep period time, NREM sleep and REM. In order to evaluate the dynamics of arousal, arousal fluctuations during sleep were quantified by means of CAP. CAP scoring was performed visually, according to the criteria of Terzano et al. (20). We quantified, within NREM sleep stages, the number of CAP sequences, of phases A (divided into the three subtypes A1, A2 and A3), the mean duration of phases A and B and the percentage of NREM sleep occupied by CAP. This ratio (CAP duration/NREM sleep), referred to as CAP rate, is the expression of the percentage of NREM sleep spent in a state of arousal instability.
Data analysis
Data concerning the macro- and microstructural sleep parameters in the two groups (patients and controls) were compared by means of the Mann–Whitney U-test, followed by Bonferroni correction for multiple comparisons, with an adjusted significance level of ≤0.005.
Results
Headache diaries
The diaries of migraine attacks completed by the patients in the month of the sleep study (2 weeks before and 2 weeks after) showed that all patients had at least five migraine attacks during this interval. No patient presented migraine attacks in the 48 h before or after the sleep study.
Sleep macrostructure
All patients had a normal night's sleep; no patient had a migraine attack in the night of the sleep study. Patients and controls did not show snoring nor other sleep-related breathing abnormalities. One possible cause of arousal during sleep is upper airway resistance syndrome (UARS). In our sample, no patient or control subject showed flow limitation at airflow curve with flow decrease <30% of normal breath, which Guilleminault et al. considered a criterion for UARS (26). The use of an oesophageal balloon to monitor inspiratory effort is currently the gold standard for the diagnosis of UARS (27), but this technique is able potentially to induce some discomfort to the patient, which could potentially bias our results.
Table 2 lists the mean values and standard deviations of the main macrostructural sleep parameters evaluated in two groups, and the P-values resulting from Mann–Whitney's U-test. No significant differences were found between the migraine and control groups in any of the parameters considered.
Mean values and standard deviations (SD) of polysomnographic (PSG) parameters in migraineurs and controls

SPT, Sleep period time.
P-values express the results of the comparison between the two groups performed by means of Mann–Whitney U-test.
Sleep microstructure
Table 3 lists the mean values and standard deviations of the main microstructural sleep parameters evaluated in two groups, and the P-values resulting from the Mann–Whitney U-test.
Mean values and standard deviations (SD) of arousal analysis
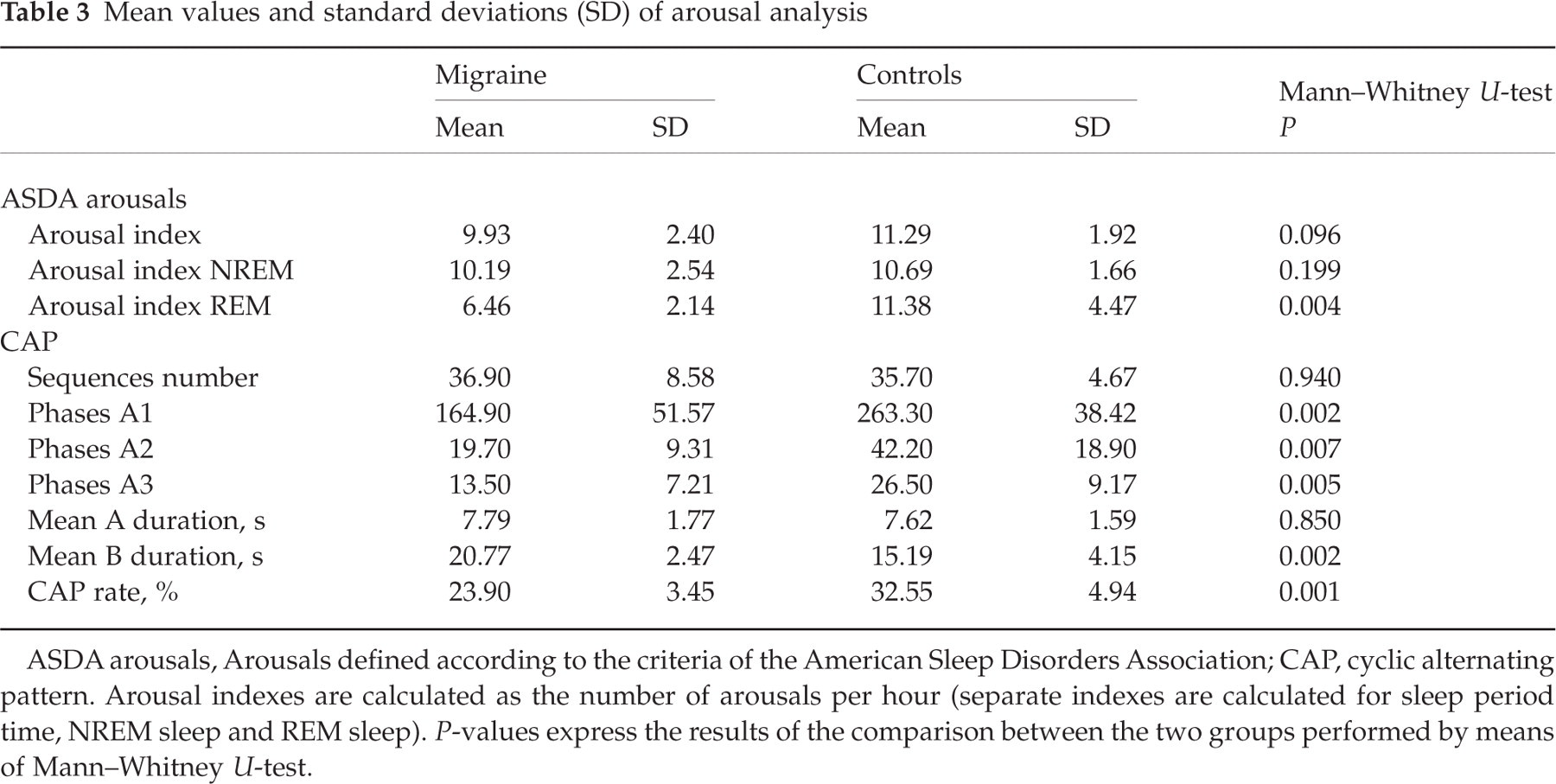
ASDA arousals, Arousals defined according to the criteria of the American Sleep Disorders Association; CAP, cyclic alternating pattern. Arousal indexes are calculated as the number of arousals per hour (separate indexes are calculated for sleep period time, NREM sleep and REM sleep). P-values express the results of the comparison between the two groups performed by means of Mann–Whitney U-test.
ASDA arousal indexes and CAP scores in the control group showed similar values to those reported in the literature for normal subjects of the same age and sex (28, 29).
As concerns the high-frequency EEG arousals (19), the arousal index, calculated over the entire night's sleep, was slightly, but not significantly, lower in the migraine (M) group compared with the controls (C) (mean arousal index M = 9.9 ± 2.4 events/h; C = 11.3 ± 1.9 events/h; Mann–Whitney U-test P = 0.096). A significant reduction in the arousal index in REM was found in the M group (mean arousal index M = 6.4 ± 2.1 events/h; C = 11.4 ± 4.5 events/h; Mann–Whitney U-test P = 0.004).
The most relevant CAP index, CAP rate, was significantly lower in the migraineur patients than in controls (CAP rate M = 23.9 ± 3.4%; C = 32.5 ± 4.9%; Mann–Whitney U-test P = 0.001). The decreased CAP rate in migraineurs was mainly due to the decreased number of low-frequency EEG bursts (A1, P = 0.002); a decrease, not reaching significance levels, between the two groups was observed in the number of high-frequency (A3, P = 0.005) and mixed-frequency (A2, P = 0.007) bursts. The decrease in A2 and A3 subtypes did not reach statistical significance, probably because of the small number of subjects in the sample. A2 and A3 subtypes largely correspond to high-frequency EEG arousal, but some of them are not included in the CAP analysis because of the scoring criteria, which exclude events occurring at the end of each sequence (20).
Discussion
The first finding of the present study is the reduction of CAP in patients with prevalently nocturnal MoA, in presence of a normal sleep macrostructure. This CAP reduction involved prevalently the A1 phases subtypes, less so the high-frequency EEG arousals. One main role of CAP A1 fluctuations is to buffer the effect of perturbations occurring during NREM sleep (20, 21). It can therefore be speculated that the reduction of CAP expresses a reduced efficacy of such mechanisms of processing of incoming inputs during sleep in migraine. Disinhibition of the somatosensory system has been observed in children with migraine (30), and the facilitation, or lack of inhibition, of afferent signals is considered one of the pathogenic mechanisms of migraine (31). Sensory dysmodulation in migraine is due to a dysfunction in subcortical structures that are involved in the control and gate of sensory neural traffic, which results in an excess throughput in sensory thalamus (6, 31, 32).
The second finding is the reduction of occurrence of high-frequency cortical arousals, as defined by the ASDA (increase of fast EEG activities and increase in muscle tone), during REM.
Nocturnal migraine attacks are more frequently associated with REM sleep and impaired arousal in REM has been reported in other primary headaches, such as cluster headache (18). Migraine attacks have also been reported in NREM. In particular, migraine has been reported in association with prolonged sleep duration and with excessive amounts of deep slow-wave sleep (stages III and IV NREM) (7, 8), all conditions indirectly associated with reduced activity of the arousal systems.
As concerns arousal, arousal modification in migraine patients has been described, especially in nights preceding an attack. In particular, Goder et al. (14) reported that the number of arousals, REM density, α power in REM sleep and β power in SWS (Slow Wave Sleep) are reduced in the nights preceding a migraine attack. Strenge et al. (15) performed a non-linear analysis of sleep EEG and observed loss of dimensional complexity in the second sleep cycle of a ‘migraine night’. Bruni et al. (16), using wrist actigraphy, showed a reduction of nocturnal motor activity of children during the night preceding the migraine attack. They interpreted their results as due to reduced cortical activation in migraineurs during the nights before the attack.
Our study substantially confirms these findings, despite several methodological differences: essentially, we selected adult patients with a high frequency of migraine attacks, occurring prevalently during sleep; moreover, we applied a peculiar model of arousal evaluation, the CAP model, which reflects a dynamic, oscillatory process, rather than an overall amount of specific frequencies.
In the interpretation of our findings, they must be considered to result from the observation of a selected population of patients, limited in number, with frequent (>5/month) MoA studied during pain-free nights, and whose migraine logs did not report attacks in the 48 h preceding or following the PSG night. Our study characterized the PSG changes of decreased arousal using indexes of decreased arousal rates and decreased CAP rate on migraine-free nights in these patients with frequent MoA. Another possible limitation is the wide age range of the patients and controls; it is well known that age-related variations of CAP indexes can occur (29).
The brain actively generates arousals by means of several arousal systems. Each of these systems has ascending projections to the cortex (which stimulate cortical activation and induce fast EEG activity) and descending projections to the spinal cord (which stimulate motor activation and induce high EMG activity) (10). Arousal systems are located within the brainstem, the thalamus, the hypothalamus and the basal forebrain; they utilize diverse neurotransmitters or modulators and they are redundant, so that any single neural system is not necessary and may be sufficient for arousal (10). A reduction of the cortical arousals reflected by the EEG might therefore reflect a general hypoactivity of the arousal systems. Since it is well known that nocturnal migraine attacks occur prevalently in REM (2, 3), we could hypothesize a functional impairment of some neural structures responsible for the control of REM sleep, such as the hypothalamus (33) and the brainstem, which are also involved in the pathophysiology of migraine (6, 12).
The reduction of spontaneous arousal during sleep may at first suggest a condition of cortical hypoexcitability. It is widely recognized that migraine events are triggered by brain cortical mechanisms, and that they are due, in particular, to a reduced habituation of cortical neurons to incoming sensory stimuli (34). On the other hand, it is still debated whether the brain cortex in migraine is hypoexcitable (13) or hyperexcitable (35). The hypoarousability observed in our migraineurs may express a condition of cortical hypoexcitability, and in particular a hypofunction of thalamocortical circuitries, which are involved in the genesis of CAP (22). In any case, it cannot be excluded that hypoarousal is due to hypofunction of subcortical arousal systems, located in the hypothalamus or brainstem, since the EEG modification in sleep is the final result of distinct pathways all producing modification of brain cortical activity.
In conclusion, we believe that the reduced reactivity to incoming stimuli, of internal or external origin, expressed by the low CAP rate during sleep, and the hypoactivity of arousal systems during REM, may be pathophysiological mechanisms, or at least predisposing factors, for the occurrence of migraine attacks during sleep.
