Abstract
Migraine is characterized by an elevated contingent negative variation (CNV) in adults and children. In the present study the movement-related potential preceding self-initiated movements, the Bereitschaftspotential, was investigated in 30 children (mean age 12 years) who were suffering from migraine and tension-type headache and in 16 healthy age-matched controls. Children pressed a button 80 times with the right index finger while movement-related potentials were recorded from frontal and central electrodes. Whereas healthy children evidenced positive movement-related potentials at left and midline positions, children with migraine and tension-type headache showed negative movement-related potentials at midline leads without lateralization. Negativity was even more pronounced in cases of migraine with than without aura symptoms.
Introduction
Slow cortical potentials have been assessed repeatedly in migraine by means of the contingent negative variation (CNV), which is recorded during a forewarned reaction time task (1). Studies have found indication of an elevated negativity between the warning stimulus and the imperative stimulus in adult migraine patients (2–5), although there have also been failures to replicate this result (6). Reports of increased amplitudes concerned mainly the early CNV component in migraine patients (5, 7, 8). The elevated CNV is evident during the intervals between migraine attacks and appears to be attenuated during attacks (5, 9, 10). There is disagreement as to the effect of aura symptoms on the CNV, with Böcker et al. (4) finding an increased CNV in migraine patients without aura and Maertens de Noorhout et al. finding none (2).
In migraine children, Besken et al. (11) reported a similarly increased CNV. Comparing children and adults, Kropp et al. (12) reported an increased early CNV in both migraine groups as compared with two groups of healthy children and adults. There are also attack-related changes of early CNV amplitudes in children (10). Additionally, these authors found evidence of retarded habituation of the CNV. This has previously been suggested to be a contributing factor to the elevated CNV amplitudes (7). In a recent report on CNV habituation in a large sample of patients with migraine without aura ranging from 8 to 59 years, Kropp et al. (12) found that indicators of reduced habituation were most prominent in adult migraineurs.
Slow potentials related to self-initiated movement have so far not been investigated in migraine. Prior to self-initiated movement, a negative potential shift, the Bereitschaftspotential (BP), has been shown to occur at C3 (13). Two components have been distinguished (14); the first one, which starts about 1 s before the execution of the movement, shows a bilateral symmetric distribution. The magneto-encephalographic (MEG) equivalent of this BP component showed a bilateral shift over frontal and central regions, likely to be generated by the supplementary motor area (SMA) (15). Functionally, this first BP component has been associated with unspecific motor preparation. The second BP component starts at about 500 ms before movement onset and has been attributed to the preparation of the specific movement. In simple finger movements, the second component has a contralateral predominance (16). Localization of the MEG motor field of different movements confirmed the somatotopic organization of the primary motor cortex (17, 18). There is thus agreement that the late BP component is primarily generated by the contralateral primary motor cortex (19).
Unlike the smooth negative shift usually observed in adults, children's BP has often been reported to exhibit a positive deflection (20–22) that occurs between 600 and 150 ms before movement onset. A failure to find positivity has also been reported (23). The functional significance of the positivity is as yet unclear.
The aim of the present study was to investigate the influence of migraine on the BP in children with and without aura symptoms.
Methods
Participants
Thirty children suffering from migraine (17 boys, 13 girls) and 16 healthy children (7 boys, 9 girls) took part in the study. Their mean ages were 11.6 (SD=2.1) and 12.1 (SD=1.7) years, respectively. All were right-handed apart from four children in the migraine and two in the control group, as assessed by the handedness subscale of the Luria-Christensen Neuropsychological Test Battery (24).
Migraine diagnosis was established with a semi-structured interview with the children and one or both of their parents. All children of the migraine group fulfilled the diagnostic criteria of migraine according to the International Headache Society (25) and had suffered two migraine attacks within the last 4 weeks. Twenty-four children had additional tension-type headache and 12 suffered from migraine with aura. The mean duration of the migraine disorder was 4.6 years (SD=2.8). Children completed a daily headache diary for a period of 4 weeks. The mean weekly headache frequency was 2.0 (SD=1.5), with a duration median of 8.2 h (range: 1–97 h) and a mean pain intensity score of 4.7 (SD=1.8) on a visual analogue scale of 1–10.
All subjects and one of their parents gave their informed consent to participate in the study.
Apparatus
EEG signals were recorded with a Nihon Khoden Neurofax 4418G with a time constant of 10 s and a low pass filter of 30 Hz from six electrode sites, F3, Fz, F4, C3, Cz and C4, referenced to linked ears. The skin was prepared with a gel (Omniprep, Weaver Inc, USA) and Ag-AgCl electrodes were attached with Grass EC-2 EEG-paste. Vertical EOG was recorded from above and below the right eye for the correction of blink artifacts. Additionally, EMG was recorded from the musculus flexor digitorum superficialis of the right arm. The EMG signal was amplified and integrated by a Grass Model 7 Polygraph (Model-7 P3 AC), rectified and integrated with a time constant of 0.05 s. Electrode impedance was kept below 5 KΩ. Data were continuously digitized with 200 Hz by a Keithley AT2914 AD-card and stored on a computer for off-line analysis.
Procedure
Subjects were seated 1.8 m in front of a panel with a red LED. After attachment of the electrodes, the light of the room was dimmed and subjects were instructed to fixate the red light. They were given some practice trials in which they were trained to press the button infrequently, i.e. with an interval exceeding 4 s. Trials with intervals of less than 2.5 s were discarded. Eighty trials were recorded in one single session.
Data reduction and analysis
EOG correction was performed for blink artifacts (26). Trials with signal drifts in excess of 50 µV were rejected and EEG data were visually screened for artifacts caused by muscle activity or electrode drifts. The EEG was digitally filtered off-line with a low pass filter with −3 dB, at 9 Hz (27).
Trials were averaged over a time window of 1700 ms before to 200 ms after the button press. After artefact correction, there was a mean of 48.6 (SD=18.0) trials in healthy children and 46.8 (SD=18.2) in migraine children left for inclusion in the averaged BP. EOG and integrated EMG trials were also averaged. BP averages were referenced to the first mean 200 ms and the following mean BP epochs were submitted to further analysis: 1400–1200, 1000–800, 600–400 and 200–0 ms before the button press.
Statistical analysis
Two movement-related potential epochs (600–400 and 200–0 msec) were compared between groups and electrodes by means of a
Results
There were no significant group differences with regard to age or sex composition. Mean EOG and EMG activity during the last 200 ms before the button press were entered into analysis of variance comparing groups. There were no significant group differences with regard to mean EOG (F (1,44)=0.03; healthy children: 9.9 µV (SD=8.1); migraine: 10.5 µV (SD=11.8)) nor to EMG activity (F (1,44)=2.49; healthy children: 12.6 µV (SD=7.0); migraine: 17.9 µV (SD=12.3)). EMG and EOG did not correlate with any of the motor-related potential amplitudes.
Group comparison of migraine and healthy children (Fig. 1)
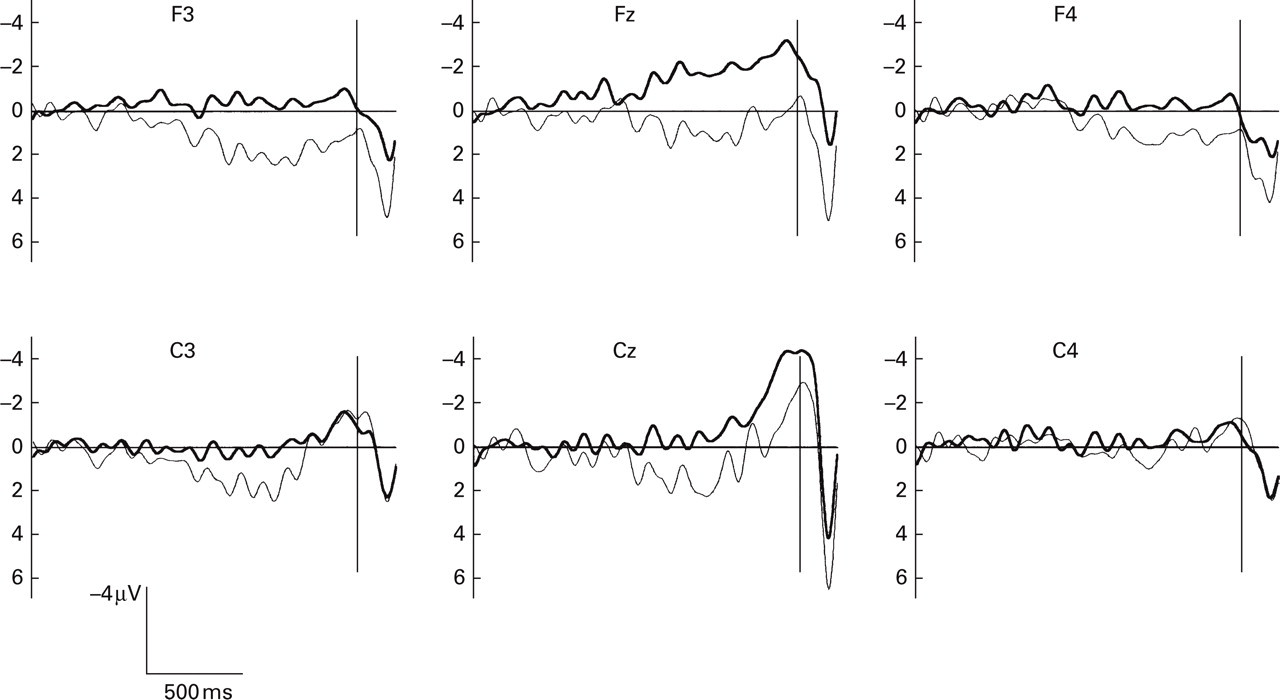
Grand averages of the movement-related potentials of healthy (—) and migraine children (—). The vertical line at the right indicates the time of the electrical contact of the button. Migraine children show the most pronounced negativity at the midline leads, whereas healthy children exhibit left-lateralized positivity half a second before the button press and positive shifts at frontal leads.
Healthy children showed an overall positive BP that was significantly different from the negative BP of migraine children (F (1,44)=4.65, P < 0.04). The group × epoch–electrode interaction was significant (healthy/migraine children: F (7,308)=2.54, P < 0.02).
Furthermore, analyses of variance were carried out comparing group (2) × epoch (2) at individual electrode sites (Fig. 2). Migraine children showed a negative BP that differed significantly from the healthy children's BP at F3 (F (1,44)=5.96, P=0.019), Fz (F (1,44)=6.77, P=0.013) and Cz (F (1,44)=4.29, P=0.044). The increase in negativity over epochs was again significant at all central positions and none of the group × epoch effects were significant.
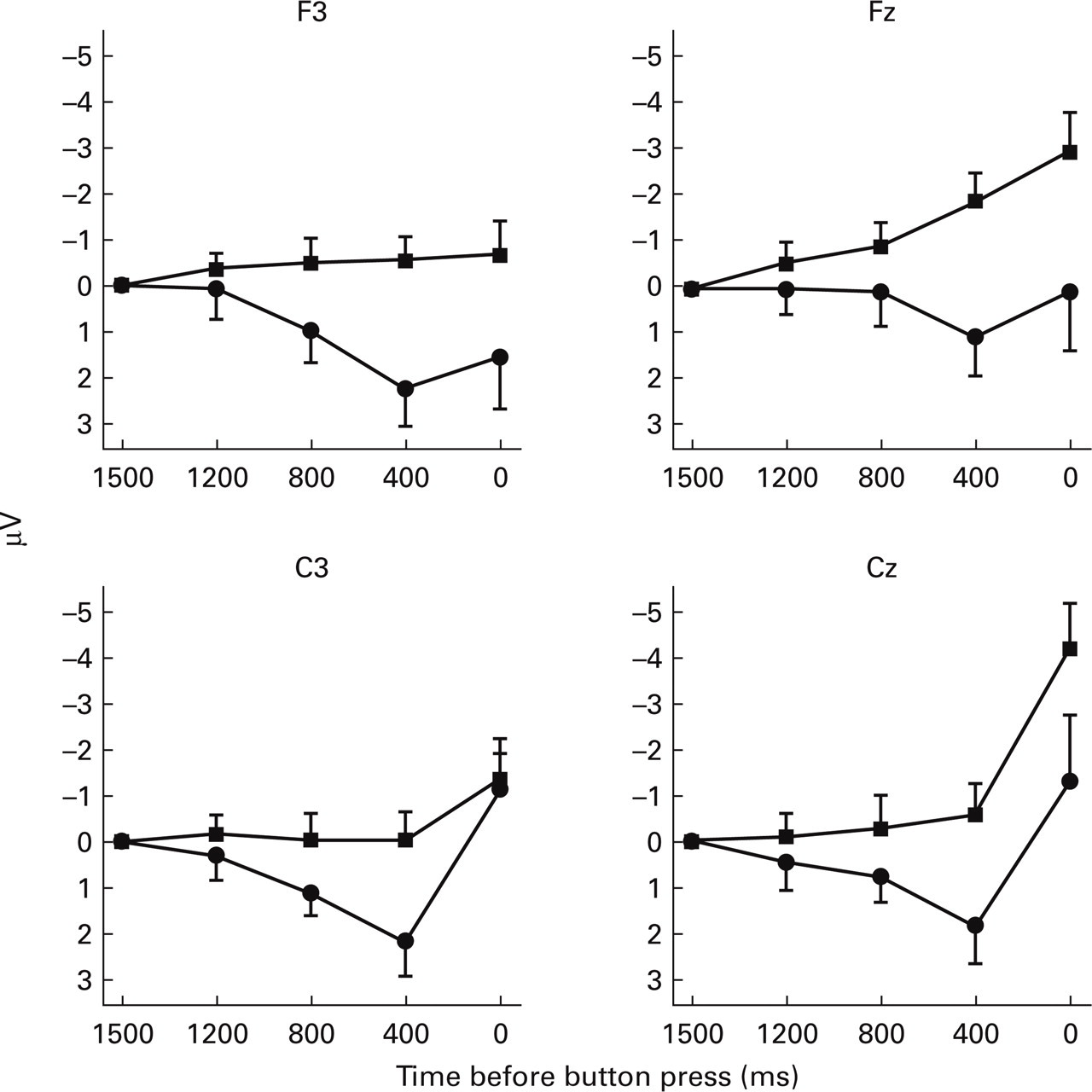
Mean amplitudes of the motor-related potentials of epochs: 1700–1500 ms (baseline), 1400–1200 ms, 1000–800 ms, 600–400 ms and 200–0 ms, before the button press. ▪ migraine group; • control group.
Migraine with and without aura symptoms (Fig. 3)
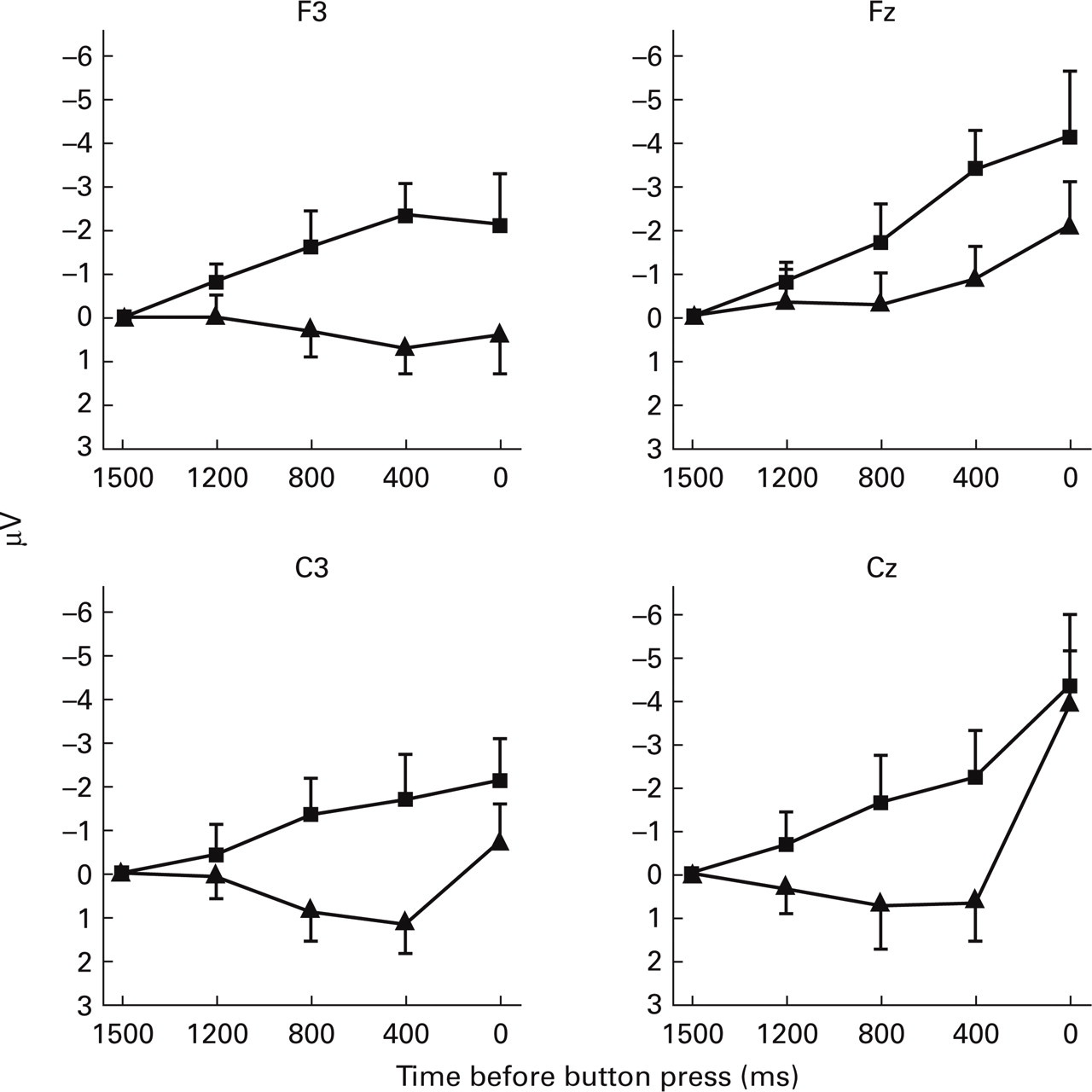
Mean amplitudes of motor-related potentials (epochs see Fig. 2) in migraine children with (▪) and without (▴) aura.
Migraine children were divided into groups with (n=12) and without (n=18) aura symptoms (Fig. 3). Groups were compared with regard to BP epochs (2: 600–400 and 200–0 ms) by electrodes (6: F3, Fz, F4, C3, Cz and C4) as within-subject factors. Migraine children with aura showed more negative motor-related potentials than those without (group: F (1,28)=6.68, P=0.02). Subsequent group (2) × epoch (2) comparisons of separate electrode positions revealed greater negativity at F3 (F (1,28)=7.36, P=0.02) and C3 (F (1,28)=4.87, P=0.04) in the group with than without aura.
Discussion
Whereas the movement-related potential of healthy children was lateralized and positive, that of migraine children was centralized and exhibited a negative shift. The negative shift was even more pronounced and had an earlier onset in migraine with aura than without. The migraine with aura group also showed distinct lateralization of negativity. The migraine with aura group thus showed the most ‘mature’ potentials, i.e. the ones that were most similar to movement-related potentials in adults. A left lateralized early negative component was followed, albeit at the central lead, by a late negative component. Children without aura only exhibited the latter without, however, the early positive component evident in healthy children. It is conceivable that the differences between the three groups mirror the development of the movement-related potential. Accordingly, after a phase of lateralized positivity, a late negative component emerges centrally at first, after which the early component becomes steadily more negative and the potential lateralizes again to the left. It is as yet unclear why migraine children show more ‘mature’ motor-related potentials than healthy children. The results are, however, broadly in keeping with CNV findings. Adult migraine patients have shown an increased CNV amplitude (4, 7, 28) as have children suffering from migraine (8). CNV amplitude has been related to alterations in neurotransmitter function (29).
Tecce (30) reported a positive correlation between dopamine activity and CNV amplitude in healthy adult subjects. Abnormal activity of the dopaminergic system has also been found in migraine subjects, suggestive of increased dopamine receptor sensitivity (31–33).
Serotonergic activity has been directly related to migraine. An increased level of its metabolite 5-HIAA was found in urine during migraine attacks (34), indicative of an elevated metabolic rate. Platelet serotonine level was also increased in the intervals between attacks (35), supporting a model of cyclic activity. A threshold model has been proposed according to which susceptibility to attacks increases during intervals, to be reset during attacks (36, 37). It remains to be explained in which way the cyclic serotonergic activity is related to elevated motor-related potentials in migraine and, in particular, in aura patients.
The elevated CNV amplitude in migraineurs has been interpreted in terms of high arousal (38–40). Schoenen et al. (2) found a significant correlation with the clinical response to beta blockers. The theory of cortical hyperexcitability has been assessed recently by means of transcranial magnetic stimulation of visual cortex (41, 42) and motor cortex (43–45) in migraine with aura. Results from these studies are inconsistent and fail to support the model of general cortical hyperexcitability.
Kropp & Gerber (7) attributed the elevated early CNV amplitude to its slow rate of habituation in migraine patients. Retarded habituation was thought to be a cyclic process and more pronounced during the days before the onset of migraine attacks. This was confirmed in adults (10, 12) and in children suffering from migraine (10). However, another group of researchers failed to replicate these results in adult subjects with migraine with aura (6). Habituation refers to response attenuation due to repeated presentation of a stimulus, i.e. changes in information processing rather than motor-related processes. It could, however, be speculated that the elevated motor-related potential of migraine children may be due either to increased motor programming or to persistent attention to action. Studies using neuro-imaging methods have shown that new motor tasks are associated with pre-frontal and pre-motor activation that diminishes with repeated execution of the movement, presumably after they have been programmed (46). Activity of the lateral pre-motor areas and the anterior cingulate has also been associated with attention to action (47). Both may contribute to elevated negativity over fronto-central leads (48). Our result of an elevated negativity over frontal and central areas in migraine children is consistent with both explanations. Neuro-imaging studies will be needed to investigate the cortical areas that contribute to the elevated motor-related potential in migraine.
Footnotes
Acknowledgements
This study was supported by the German Federal Minister of Science and Technology (BMFT grant no. 0701515). We are grateful to Raymiand Pothmann and My Khan Truong for their help in carrying out this study.
