Abstract
The 5-HT1B/1D receptor agonist sumatriptan is effective in aborting acute attacks of migraine and is known to cause constriction of cranial arteries as well as some peripheral blood vessels. The present study set out to investigate whether 5-HT1B and/or 5-HT1D receptors mediate contractions of the human isolated middle meningeal and temporal arteries (models for anti-migraine efficacy) and coronary artery and saphenous vein (models for side-effect potential). Concentration-response curves were made with sumatriptan (1 n
Keywords
Introduction
Sumatriptan, the first of the 5-HT1B/1D receptor agonists, is highly effective in aborting migraine headaches (1, 2). It is believed that migraine headache results from dilation of extracerebral cranial blood vessels and that sumatriptan, as well as other triptans, constricts these dilated vessels (2, 3). Although the triptans are known to be cranioselective, they all have the propensity to constrict the coronary artery (4–7). Constriction of the coronary artery may lead to cardiovascular adverse events, including myocardial ischaemia and infarction in predisposed individuals (8).
The triptans are high affinity agonists at 5-HT1B and 5-HT1D receptors, but their vasoconstrictor effect seems to be mediated via the 5-HT1B receptor (2). Using RT-PCR techniques, it has also been shown that the 5-HT1B receptor mRNA is predominant over 5-HT1D receptor mRNA in the human middle cerebral (9, 10), middle meningeal (11), temporal (12) and coronary (13, 14) arteries. The 5-HT1B receptor protein has also been localized in the smooth muscle layer as well as endothelium of the human middle cerebral (15), middle meningeal (4, 16) and coronary (4, 13, 16) arteries, where the 5-HT1D receptor protein is not, or poorly, expressed. Functional pharmacological in vitro studies suggest that sumatriptan behaves as a full agonist in blood vessels (17–21), which do not contract in response to selective 5-HT1D receptor agonists (14, 22).
A more direct evidence for a 5-HT1B receptor-mediated vasoconstriction to sumatriptan can be obtained from antagonist studies. Until now, studies in the human isolated middle meningeal (7, 18, 23) and coronary (7, 17, 24, 25) arteries and saphenous vein (21) have used non-selective 5-HT1B/1D receptor antagonists (GR127935, GR55562 and GR125743). Recently, SB224289 and BRL15572 have been introduced as selective 5-HT1B and 5-HT1D receptor antagonists, respectively (26, 27). The use of these compounds revealed that sumatriptan-induced contractions in human isolated temporal (12) and small pulmonary (28) arteries as well as canine (29) and porcine (30) carotid vascular beds are mediated via the 5-HT1B receptor. Using both functional in vitro and in situ hybridization techniques, we investigated the role of 5-HT1B and 5-HT1D receptors in mediating contractions of the human isolated middle meningeal and temporal arteries (models for therapeutic efficacy in migraine) and coronary artery and saphenous vein (models for peripheral side-effect potential) (6, 7, 31).
Material and methods
Tissue collection
The middle meningeal (four male, five female; age 30–72 years) and temporal (three female; age 45–59 years) arteries and saphenous vein (eight male, one female; age 45–78 years) were obtained postoperatively from patients undergoing craniotomy (middle meningeal and temporal artery: eight aneurysms; four meningiomas) or coronary artery bypass grafting (saphenous vein) at the Erasmus University Medical Centre, Rotterdam, The Netherlands. The blood vessels were placed in a propylene tube filled with ice-cold (0–4°C) physiological saline, transported immediately to the laboratory and used within 2 h of surgery.
The right epicardial coronary artery was obtained from nine heart-beating organ donors (three male, six female; 37–64 years) who died of non-cardiac disorders (six cerebrovascular accident, two cerebral infarction, one head trauma). The Rotterdam Heart Valve Bank, Rotterdam, The Netherlands provided the hearts, after donor mediation by Bio Implant Services Foundation/Eurostransplant Foundation, Leiden, The Netherlands. The vessel was stored overnight in a modified Krebs bicarbonate solution (see below) and used the next day.
Organ bath experiments
Measurement of vascular contractions
The methods used were similar to those described in detail earlier (6, 7). Briefly, approximately 4-mm segments, obtained from pieces of the middle meningeal (n=6), temporal (n=3) and coronary (n=6) arteries and saphenous vein (n=6), were mounted on metal prongs in organ baths, containing a modified Krebs bicarbonate solution (pH 7.4; 37°C), aerated with 95% O2 and 5% CO2. The composition (mM) of the Krebs bicarbonate solution was NaCl 119, KCl 4.7, CaCl2 1.25 (or 2.5 for coronary artery and saphenous vein), MgSO4 1.2, KH2PO4 1.2, NaHCO3 25 and glucose 11.1 (or 8.3 for coronary artery and saphenous vein). The cyclo-oxygenase inhibitor indomethacin (0.1 µ
Changes in isometric tension were registered on recording set-ups from either EMKA Technology (Paris, France) for the middle meningeal and temporal arteries or Harvard Apparatus (South Natick, Massachusetts, USA) for the coronary artery and saphenous vein. The segments were allowed to equilibrate for at least 30 min, stretched to a passive tension of 4 mN (middle meningeal and temporal arteries), 15 mN (coronary artery) or 10 mN (saphenous vein). Since sumatriptan is metabolized by monoamine oxidase (32), we then treated the segments with pargyline (100 µ
After washing, the segments were allowed to equilibrate for 60 min, with replacement of the Krebs solution every 15 min. The segments were then studied in a paired parallel experimental set-up (6, 7), where a single concentration response curve to sumatriptan (1 n
Data presentation
Contractile responses were expressed as percentage of the contractile response to 1 µ
Statistical analysis
Differences between the (apparent) Emax of sumatriptan in the absence or presence of antagonists were analysed with a paired t-test, using Graphpad Prism 3.0 (Graphpad Software Inc., San Diego, California, USA). In case the Emax values in the absence of antagonists were similar to those in the presence of antagonist, the differences in Hill slopes and pEC50 values of mean concentration response curves between vehicle and antagonist groups were analysed with a one-way analysis of variance (
Molecular biological experiments
In situ hybridization
5-HT1B and 5-HT1D receptor mRNAs were localized employing non-radioactive in situ hybridization on the human middle meningeal artery, coronary artery and saphenous vein (n=3 each). After cleaning the surrounding tissue the blood vessels were fixed for 24 h in 4% paraformaldehyde dissolved in phosphate buffered saline. Chinese hamster ovary (CHO) cells expressing either the human recombinant 5-HT1B or 5-HT1D receptor served as positive controls. After dehydration with increasing percentage of ethanol in phosphate buffered saline, the blood vessels were embedded in paraffin and 5-µm thick sections were cut with a microtome (model HM325, Microm GmbH, Walldorf, Germany). The sections were mounted on superfrost plus® glass slides (Menzel-Glaser, Braunschweig, Germany) and dried at 37°C for 48 h.
Recombinant plasmid DNAs encoding the human 5-HT1B (Genbank accession number D10995, nucleotide 40–390, length 350 bp) or 5-HT1D (Genbank accession number M81589; nucleotide 1–400, length 400 bp) receptor were employed for the preparation of non-radioactive cRNA probes. Linearized DNA templates were transcribed to synthesize the sense and antisense cRNA probes, using T7 or SP6 RNA polymerase as per protocol described for the DIG-RNA labelling kit. DIG-labelled cRNA probes were quantified by dot blotting and using serial dilutions of standard DIG-labelled control RNA supplied in the kit. Treatment of tissue sections and cells and subsequent hybridization was performed as described earlier (33). The tissues were hybridized with 25 ng of cRNA probe per slide for 16 h at 55°C and the DIG-labelled hybrids were detected by incubation with antidigoxigenin antibody (1 : 2000 dilution) conjugated to alkaline phosphatase for 2.5 h at room temperature. The immunodetection of DIG-labelled hybrids was done using 4-nitroblue tetrazolium chloride (NBT) as chromogen and 5-bromo-4-chloro-3-indolyl-phosphate (BCIP) as coupling agent. Slides were counter-stained with nuclear red solution, dehydrated with ethanol gradients and mounted with Euparal (Chroma-Gesellschaft, Schmid GmbH, Köngen, Germany). Cells and tissue sections were visualized under a light microscope (model Leica DM RBE, Leica NL, BV, Rijswijk, The Netherlands) and photographed using a CCD video camera (Sony DXC-950, Sony Corporation, Tokyo, Japan). The sense riboprobes were included as negative controls, which showed no or little staining compared with antisense riboprobes.
Endothelial staining
Serial sections of 5-µm thickness were processed for immunohistochemical localization of endothelial cell marker, CD31. Sections were deparaffinized, rehydrated and incubated with pronase (1 mg/ml) at 37°C for 10 min prior to incubation with specific purified mouse monoclonal antibodies raised against human CD31 (Neomarkers, Union City, California, USA). To block non-specific binding, sections were incubated with 10% normal goat serum diluted in 5% bovine serum albumin in phosphate buffered saline (pH=7.4). Subsequently, sections were incubated overnight at 4°C with primary antibodies, CD31 (1 : 75 v/v). Incubation for 30 min with secondary biotinylated anti-immunoglobulins (Multilink®, 1 : 75 v/v, Biogenex, San Ramon, USA) and tertiary complex of streptavidin conjugated to alkaline phosphatase (Label® 1 : 50 v/v, Biogenex) were used to enhance the detection sensitivity. Colour was developed using New Fuchsine as chromogen, while endogenous alkaline phosphatase activity was inhibited by 0.01
Ethical approval
The Medical Ethics Committee of the Erasmus University Medical Centre Rotterdam, dealing with the use of human material for scientific experiments, approved the protocols for this investigation.
Compounds and kits
For pharmacological study, the following compounds were used: atropine sulphate, bradykinin acetate, corticosterone, 5-hydroxytryptamine creatinine sulphate (serotonin; 5-HT), imipramine hydrochloride, indomethacin hydrochloride, mepyramine maleate, pargyline hydrochloride, prostaglandin F
2α tris salt (PGF
2α
) and substance P acetate (all purchased from Sigma Chemical Co.), mesulergine hydrochloride (gift: Novartis AG, Basel, Switzerland), prazosin hydrochloride (gift: Pfizer, Sandwich, UK), sumatriptan succinate, SB224289 (2,3,6,7-tetrahydro-1′-methyl-5-[2′-methyl-4′(5-methyl-1,2,4-oxadiazo l-3-yl) biphenyl-4-carbonyl] furo [2,3f]indole-3-spiro-4′-piperidine hydrochloride) and BRL15572 (1-3-chlorophenyl-4-[3,3-diphenyl(2-(S,R) hydroxypropanyl) piperazine] hydrochloride), both from GlaxoSmithKline, Harlow, Essex, UK, courtesy of Dr A. A. Parsons. Stock solutions of corticosterone (100 m
For molecular biology experiments, the materials used were: acetic anhydride, diethyl pyrocarbonate, levamisol, maleic acid, triethanolamine and xylene (Sigma Chemical Co.), antidigoxigenin-AP Fab fragments, blocking reagent for nucleic hybridization, DIG-RNA labelling kit, glycine and RNase T1 (Boehringer Mannheim, Almere, The Netherlands), bovine serum albumin, dextran sulphate, ethylene diaminetetraacetic acid, ficoll, hering sperm DNA, phenol and Tris-HCl (Amersham Pharmacia Biotech Benelux, Roosendaal, The Netherlands), BCIP, formamide, NBT, proteinase K, sodium citrate and yeast tRNA (Life Technologies, Breda, The Netherlands), DNase, RNase inhibitors and Triton-X-100 (Promega Benelux, Leiden, The Netherlands) and RNase away solution (Molecular Bio-products, San Diego, CA, USA).
Results
Relaxation responses to substance P and bradykinin
The relaxation to substance P (10 n
Effects of sumatriptan on cranial arteries
Concentration response curves to sumatriptan in the middle meningeal and temporal arteries are depicted in Fig. 1. In both blood vessels, sumatriptan elicited a concentration-dependent contraction. The Emax values (efficacy) of sumatriptan in the middle meningeal and temporal arteries were 83 ± 15% and 68 ± 28% of the contraction to 1 µ
Apparent Emax and pEC50 values of sumatriptan in contracting human isolated blood vessels in the absence or presence of antagonists

Data are means± SEM (n = 3–6). Apparent Emax is presented as percentage of contraction elicited by either 1 µ
∗Significantly different from sumatriptan control (P < 0.05).

Cumulative concentration response curves to sumatriptan in the human isolated middle meningeal (n = 5–6) and temporal (n = 3) arteries in the absence (control; ○) or presence of SB224289 (1 µ
The responses to sumatriptan in both cranial arteries were antagonized by the 5-HT1B receptor antagonist SB224289 (1 µ
Due to the insurmountable antagonism of sumatriptan by SB224289, pKB values could not be estimated. Alternatively, we calculated the negative logarithm of sumatriptan concentration eliciting a response equivalent to 25% of sumatriptan control Emax (pEC25%) in the absence or presence of the antagonists in the middle meningeal artery; the temporal artery data was not further processed as SB224289 virtually abolished sumatriptan-induced contractions (Table 2). While BRL15572 was ineffective, the pEC25% of sumatriptan was significantly decreased by SB224289. No additional antagonism was observed with the combination of the two antagonists (Table 2).
pEC25% of sumatriptan in contracting the middle meningeal artery and saphenous vein in the absence or presence of antagonists

Data are mean± SEΜ (n = 4–6). pEC25% represents the negative logarithm of the concentrations of sumatriptan eliciting a contraction equivalent to 25% of individual control Emax (middle meningeal artery, 20.8 ± 3.8% of the response to 1 µ
∗Significantly different from sumatriptan control (P < 0.05).
Effect of sumatriptan on peripheral vessels
Sumatriptan also contracted the human coronary artery and saphenous vein in a concentration-dependent manner. The pEC50 and (apparent) Emax of sumatriptan were, respectively, 5.7 ± 0.1 and 13 ± 2% of the response to 100 m
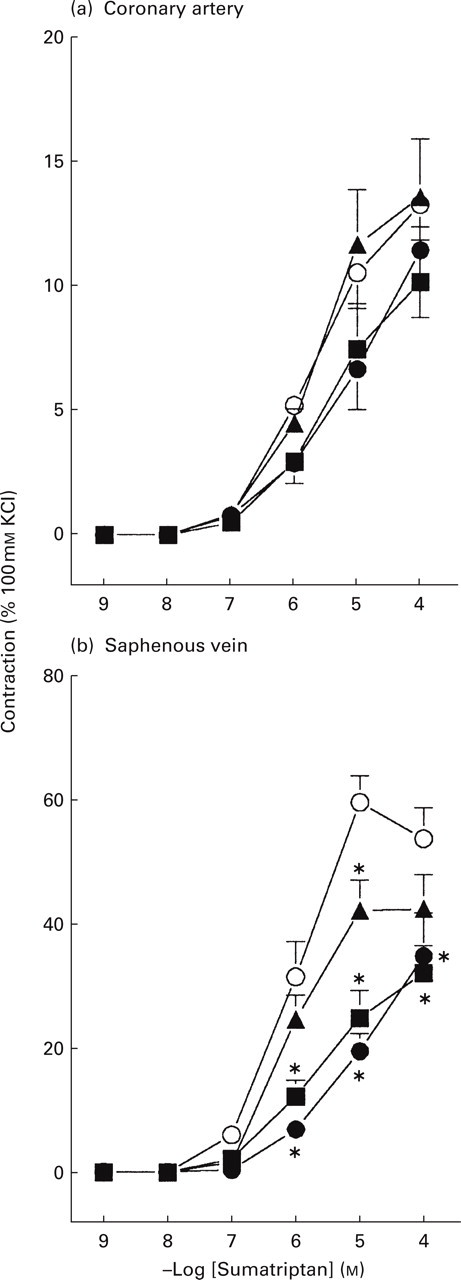
Cumulative concentration response curves to sumatriptan in the human isolated coronary artery (n = 5–6) and saphenous vein (n = 4–6) in the absence (control; ○) or presence of SB224289 (1 µ
In the coronary artery, SB224289 caused a small parallel rightward shift in the concentration response curve to sumatriptan, yielding a significant decrease in the pEC50 (5.0 ± 0.1) with no change in the apparent Emax (11 ± 3% of the response to 100 m
In the saphenous vein, the response to sumatriptan was antagonized by SB224289 (1 µ
In situ hybridization
The specificity of mRNA signals and standardization of the in situ hybridization conditions were first established by the use of specific sense and antisense probes in CHO cells expressing either the human 5-HT1B or 5-HT1D receptor. Strong cytoplasmic and nuclear mRNA expression as a dark purple/blue colour for respective antisense riboprobes was observed in these cells, whereas sense riboprobes depicted no staining. Furthermore, the sense riboprobes did not show any specific mRNA expression assessed as purple/blue staining in the tissue samples included in this study. If any staining with sense riboprobes was detected, it was always far less than the staining with antisense probes (data not shown).
Cellular localization of mRNAs for the 5-HT1B and 5-HT1D receptors in the human middle meningeal and coronary arteries and saphenous vein is depicted in Fig. 3. In the middle meningeal artery, hybridization with antisense riboprobe showed 5-HT1B receptor mRNA signals in the medial smooth muscle layer as well as in the endothelial cells, whereas adventitial cells showed only a faint staining. Although not so intense, the coronary artery and saphenous vein also expressed the 5-HT1B receptor mRNA in the vascular smooth muscle cells. Specific expression for the 5-HT1B receptor was seen in the luminal endothelial cells in the case of coronary artery. In order to verify the in situ hybridization data for the endothelial cell expression of 5-HT1B receptor mRNA, CD31 immunohistochemistry was performed and it confirmed a cytoplasmic endothelial cell staining (Fig. 3). In contrast to the 5-HT1B receptor mRNA, no signals were detected for the 5-HT1D receptor mRNA in both smooth muscle and endothelial cells in the middle meningeal artery and saphenous vein. However, a weak staining for the 5-HT1D receptor mRNA was noticed in some endothelial cells in the coronary artery and in adventitial cells in the case of middle meningeal as well as coronary arteries (Fig. 3).
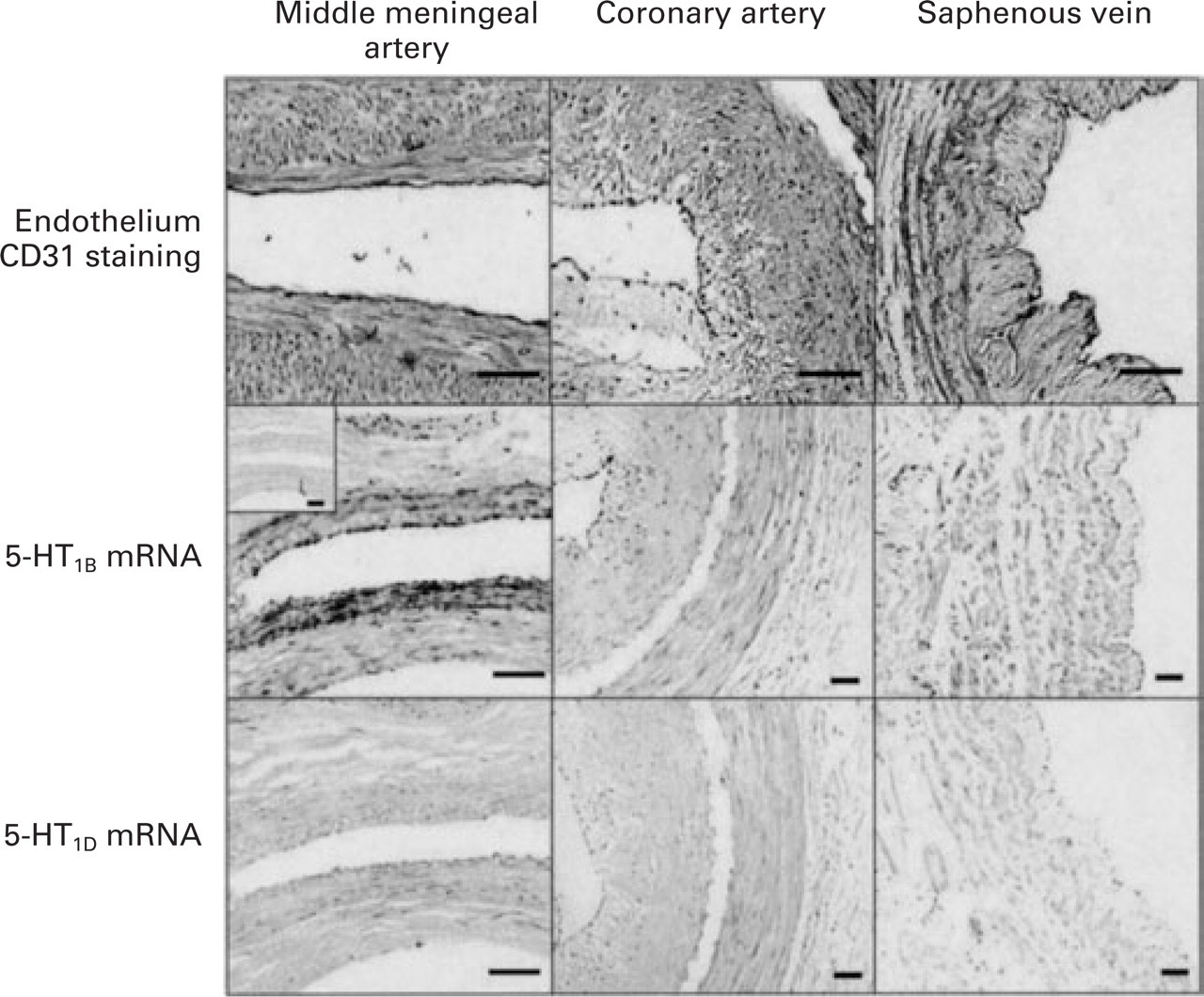
Photographs of immunohistochemical staining with CD31 endothelial marker (top panels) and in situ hybridization with DIG-labelled cRNA probes for h5-HT1B (middle panels) and h5-HT1D (lower panels) receptor mRNAs in the human middle meningeal and coronary arteries and saphenous vein. The reddish colour in the top panels denotes endothelial cells, while the purple precipitates in the middle panels correspond to 5-HT1B receptor mRNA signals. The 5-HT1D receptor mRNA (lower panels) as well as the sense riboprobes, as exemplified by the left middle panel inset, did not show signals in the three blood vessels. Scale bar = 50 µ
Discussion
Craniovascular selectivity of sumatriptan
Sumatriptan contracted in a concentration-dependent manner both cranial and peripheral blood vessels, used as models for anti-migraine activity (middle meningeal and temporal artery) and possible peripheral side-effect potential (coronary artery and saphenous vein), respectively. The potency of sumatriptan was higher at the middle meningeal and temporal arteries (pEC50: 6.7 each) than at the coronary artery (pEC50: 5.7) and saphenous vein (pEC50: 6.1). Compared with the other vessels, the efficacy (Emax) of sumatriptan in the coronary artery was clearly lower (see Table 1). These data, which confirm the cranioselectivity of sumatriptan, are in accordance with other studies dealing with middle meningeal (4, 6, 18, 23), temporal (12, 18) and coronary (6, 7, 13, 17, 31, 34) arteries and the saphenous vein (6, 7, 21).
Receptors mediating sumatriptan-induced contractions
The data obtained in in vitro functional studies show that the 5-HT1B receptor antagonist SB224289 clearly attenuated sumatriptan-induced contractions, although the 5-HT1D receptor antagonist BRL15572 had little effect on the concentration response curves to sumatriptan; the combination of SB224289 and BRL15572 did not reveal any additional antagonism. These results establish that the 5-HT1B receptor mediates the contractile effects of sumatriptan in both cranial and peripheral blood vessels, while the 5-HT1D receptor does not seem to play any role. The latter conclusion is also supported by the fact that selective 5-HT1D receptor agonists (PNU-109291 and L775 606) did not contract isolated blood vessels (14, 22, 35) and, interestingly, one such compound (PNU-142633) was also found ineffective in migraine (36). Based on the high affinity of sumatriptan for the 5-HT1F receptor (pKi: 7.9) (37), it has been argued that this receptor might play a role in the therapeutic action of sumatriptan (38). Although we cannot rule this out, the 5-HT1F receptor is not involved in vasoconstriction, as selective 5-HT1F receptor agonists (LY344864 and LY334370) show no vasoconstrictor effect (14, 39, 40). In addition, Verheggen et al. (12) have suggested that in the presence of SB224289 high concentrations of sumatriptan can elicit contractions of the human isolated temporal artery via the 5-HT2A receptor. This was not the case in the present studies, probably because we included the mixed 5-HT2/7 receptor antagonist mesulergine (pKB: 9.1 and 8.2, respectively) (41), in the Krebs solution. However, the 5-HT2 receptor antagonist ketanserin was unable to block sumatriptan-induced contractions in the human middle meningeal (18) and coronary (17, 34) arteries as well as the saphenous vein (21).
It may be noted that SB224289 antagonized the responses to sumatriptan in an insurmountable manner in the middle meningeal artery, whereas in the temporal artery the contractions were virtually abolished. In the coronary artery, there was a weak competitive (surmountable) antagonism, whilst in the saphenous vein an intermediate antagonistic response was observed. The nature of the difference in the antagonistic behaviour of SB224289 in these blood vessels is not clear. A possible explanation (42) could be that SB224289, which acts as an inverse agonist in cells expressing recombinant 5-HT1B receptors (43), may also do so at constitutive 5-HT1B receptors. Another explanation for the observed differences could be the influence of receptor density and receptor reserve, but, in view of considerably higher 5-HT1B receptor density in the meningeal artery as compared with the coronary artery (16), we should have observed a surmountable antagonism in the middle meningeal artery and insurmountable antagonism in the coronary artery. Finally, in view of the localization of the 5-HT1B receptor on the endothelium (see below), the effect of SB224289 may be influenced to a different degree by endothelial factors (either contractile or relaxing) released upon activation of the 5-HT1B receptor. Whatever the mechanism, differences in the nature of antagonism have also been observed between sumatriptan and the non-selective 5-HT1B/1D receptor antagonist GR127935 (14, 23).
It is surprising that SB224289 proved to be a weak antagonist in our experiments as its pKB (6.4 ± 0.2) against sumatriptan in the coronary artery resembled more its pKi (6.2) at the 5-HT1D receptor and was far less than its pKi at the 5-HT1B receptor (8.2, 26, 27). However, if 5-HT1D receptors were involved, we would have found BRL15572 to be an even more potent antagonist than SB224289; this was obviously not the case. The involvement of 5-HT1B receptor in the sumatriptan-induced coronary artery contraction is also supported by previous investigations using non-selective 5-HT1B/1D receptor antagonists (6, 7, 13, 24, 34). However, the weaker antagonism by SB224289 than these non-selective antagonists suggests that an additional mechanism is partly responsible for the coronary contraction induced by sumatriptan. The nature of this additional mechanism is not known, but it cannot be related to 5-HT2 receptors in view of the presence of mesulergine in the organ bath. Certainly, this additional mechanism is of interest with regard to the sumatriptan-related cardiac side-effects and may prove relevant in future anti-migraine drug development.
Vascular localization of 5-HT1B and 5-HT1D receptor mRNA
Although RT-PCR studies have shown the presence of 5-HT1B receptor mRNA in the human middle meningeal (11) and coronary (13, 14) arteries, the cellular localization of 5-HT receptor subtypes at mRNA level has not been demonstrated. Our results of in situ hybridization showed the mRNA expression of 5-HT1B receptor in both smooth muscle and endothelium, whereas the expression of 5-HT1D receptor mRNA was weak, if any, in the three human blood vessels investigated (Fig. 3). These results are in agreement with the 5-HT1B and 5-HT1D receptor protein localization in coronary and middle meningeal arteries (4, 13, 16). The localization of 5-HT1B receptor mRNA within the smooth muscle cells of these blood vessels, and the lack of 5-HT1D receptor mRNA, strongly suggest that contraction to sumatriptan is mediated via the 5-HT1B receptor rather than the 5-HT1D receptor. Although the exact role of the endothelial 5-HT1B receptor in these vessels is not well understood, we cannot rule out the release of relaxing and/or contractile substances upon stimulation.
Conclusion
In conclusion, our data show that sumatriptan contracts the human middle meningeal, temporal and coronary arteries and saphenous vein via the 5-HT1B receptor, but not 5-HT1D receptor. In the human coronary artery, the contraction may also be mediated by an unknown, SB224289-resistant, mechanism.
Footnotes
Acknowledgements
The authors wish to express their gratitude to the staff of the Neurosurgery (Head: Professor C.J.J. Avezaat) and Cardiothoracic Surgery (Head: Professor A.J.J.C. Bogers) Units, Erasmus University Medical Centre, Rotterdam, as well as the Rotterdam Heart Valve Bank (Dr A.P.A. Stegmann) for providing human blood vessels. We thank Dr Mike Mosley (Pfizer Ltd, Sandwich, UK) for kindly providing us with recombinant plasmid DNAs encoding human 5-HT1B and 5-HT1D receptors and transfected CHO cells. Dr H. Motulsky (Graphpad Software Inc., San Diego, California, USA) and Dr P.G.H. Mulder (Department of Epidemiology and Biostatistics, Erasmus University Medical Centre, Rotterdam) helped us with statistical analyses of the data.
