Abstract
The mechanistically novel benzopyran derivative SB-220453, which is undergoing clinical evaluation in migraine, exhibits a high affinity for a selective, but not yet characterized, binding site in the human brain. It inhibits nitric oxide release and cerebral vasodilatation following cortical spreading depression as well as carotid vasodilatation induced by trigeminal nerve stimulation in the cat. The aim of our study was to investigate the contractile properties of SB-220453 on a number of human isolated blood vessels (coronary artery, saphenous vein and middle meningeal artery) as well as atrial and ventricular cardiac trabeculae. While sumatriptan induced marked contractions in three blood vessels investigated, SB-220453 was devoid of any effect. In atrial and ventricular cardiac trabeculae, neither SB-220453 nor sumatriptan displayed a positive or negative inotropic effect. Since SB-220453 did not contract the middle meningeal artery, we conclude that potential anti-migraine effects are not mediated via a direct cerebral vasoconstriction. The lack of activity of SB-220453 in coronary artery, saphenous vein and cardiac trabeculae demonstrates that the compound is unlikely to cause adverse cardiac side-effects.
Keywords
Introduction
Sumatriptan, an indole sulphonamide with agonist activity at 5-HT1B/1D receptors, is highly effective in aborting attacks of migraine and cluster headache. The drug is generally well tolerated, but up to 15% of patients consistently report chest symptoms, including chest pressure, tightness and pain, often mimicking angina (1–3). Although extracardiac mechanisms have been invoked (4), chest symptoms may well be caused by coronary vasoconstriction, which has been observed after sumatriptan both in vivo (5) and in vitro (6–9). In some cases, the use of sumatriptan, like that of ergotamine (10, 11), was even associated with myocardial infarction (12, 13) and cardiac arrest (14). ‘Second generation’ sumatriptan-like anti-migraine drugs are aimed at, in addition to achieving high efficacy and long duration of action, avoiding coronary vasoconstrictor activity (15). However, these drugs also contract human isolated coronary artery and seem to have a similar coronary side-effect potential to sumatriptan (9, 16, 17).
Due to concerns about cardiac side-effects, it would be highly desirable to develop anti-migraine drugs that act via a mechanism not involving 5-HT1B/1D receptors. Indeed, the mechanistically novel benzopyran SB-220453 (18) has no significant affinity at 5-HT1B/1D receptors, nor does it show any activity in a large number of receptor, ion channel and enzyme assays (18). SB-220453 exhibits a high affinity for a selective, but structurally unknown binding site in the human brain (19) and may be active in the treatment of migraine via blockade of excessive cortical excitability. SB-220453 inhibits neurogenic inflammation in rat brain meninges (18), nitric oxide release associated with cortical spreading depression as well as carotid vasodilatation induced by trigeminal nerve stimulation in the cat (19, 20). In the present study, we investigated the effects of SB-220453 on a number of human isolated blood vessels (coronary artery, saphenous vein and middle meningeal artery) as well as human isolated atrial and ventricular cardiac trabeculae. Sumatriptan was used for comparison.
Methods
Preparation of tissue
Human isolated coronary artery
Right epicardial coronary arteries were obtained from six heart beating organ donors (two male, four female; age 37–63 years), who died of non-cardiac disorders (five of cerebrovascular accident, one head trauma) less than 24 h before the tissue was taken to the laboratory. Hearts, provided by the Rotterdam Heart Valve Bank (Bio Implant Services/Eurotransplant Foundation) after removal of the aortic and pulmonary valves for transplantation purposes, were stored at 0–4°C in a sterile organ-protecting solution immediately after circulatory arrest. After arrival at the laboratory, the right coronary artery was removed and placed in a cold, oxygenated (95% O2/5% CO2) Krebs buffer solution of the following composition (m
On the following day, the artery was cut into segments of 3–4 mm length, excluding distinct, macroscopically visible atherosclerotic lesions. The vessel segments were mounted in 15-ml organ baths filled with oxygenated Krebs buffer solution at 37°C. After equilibration for at least 30 min and a wash every 15 min, the vessel segments were stretched to a stable tension of about 15 mN (9). Changes in tissue tension were measured using an isometric transducer (Harvard, South Nattick, MA) and recorded on a flatbed recorder (Servogor 124; Goerz, Neudorf, Austria).
Human isolated saphenous vein
Human saphenous veins were obtained post-operatively from four patients (two male, two female; 68–79 years) undergoing coronary bypass surgery. The tissue was immediately placed in cold saline and was brought to the laboratory within 15 min. Subsequently, the vein was cleaned of connective tissue and placed in a cold, oxygenated Krebs buffer solution (for composition, see above). After overnight storage, the vein was cut into segments of 3–4 mm length. The vessel segments were mounted in 15-ml organ baths filled with oxygenated Krebs buffer solution at 37°C. After equilibration for at least 30 min and a wash every 15 min, the vessel segments were stretched to a stable tension of about 10 mN, which we previously determined to be optimal for contraction measurements (21). Contractions were measured with an isometric transducer (Harvard) and recorded on a flatbed recorder (Servogor 124; Goerz).
Human isolated middle meningeal artery
Human middle meningeal arteries were obtained from 10 patients (three male, seven female; 30–71 years) undergoing craniotomy during neurosurgical procedures. In such patients, a part of the skull is temporarily removed to gain access to the brain and a small redundant portion of a branch of the middle meningeal artery is usually found attached to the dural sheath covering the removed piece of the skull. After careful removal from the dura mater, this arterial piece was placed in cold saline and brought to the laboratory immediately. Upon arrival at the laboratory, the artery was cleaned of connective tissue and was placed in cold oxygenated Krebs buffer solution of the following composition (m
Because endogenous prostaglandin synthesis frequently caused spontaneous increases in baseline tension in pilot experiments, the cyclo-oxygenase inhibitor indomethacin (0.1 μ
Human atrial and ventricular cardiac trabeculae
As described above (coronary artery section), hearts were obtained from five heart beating organ donors (two male, three female; age 36–57 years), who died of non-cardiac disorders (all cerebrovascular accident). Immediately after arrival at the laboratory, right atrial and left ventricular trabeculae of approximately 1 mm thickness were carefully dissected and mounted in a 15-ml organ bath in the same Krebs buffer solution as used for the coronary artery and saphenous vein. The trabeculae were paced at 1 Hz using electrical field stimulation (5 ms, 15–20 V) delivered by a Grass S6 Square Wave Stimulator (Quincy, MA). Resting tension was set to 0.75 mN and 2.0 mN for atrial and ventricular tissue, respectively. Changes in contraction were recorded with a Harvard force transducer on a flatbed recorder (Servogor 124; Goerz). The preparation was allowed to stabilize during 1 h with a wash every 15 min (24).
Experimental protocol
Human isolated coronary artery and saphenous vein
Segments were exposed to K+ (30 m
Human isolated middle meningeal artery
Since the addition of K+ frequently increased basal tone (unpublished observations), vessel segments were exposed to prostaglandin F
2α (0.1 μ
Atrial and ventricular cardiac trabeculae
A concentration response curve to noradrenaline (10 n
Compounds
Sumatriptan succinate was a kind gift from GlaxoWellcome (Dr H. E. Connor, Ware, UK). SB-220453 ((-)-cis-6-acetyl-4S-(3-chloro-4-fluoro-benzoylamino)3,4-dihydro-2,2-dimethyl-2H-benzo[b]pyran-3S-ol) was kindly provided by SmithKline Beecham (Dr A. A. Parsons, Harlow, UK). U46619 (9,11-dideoxy-11α,9α-epoxy, methanoprostaglandin F 2α), prostaglandin F 2α (Tris salt), bradykinin acetate, substance P acetate and DMSO were purchased from Sigma Chemical Co. (St Louis, MO). Indomethacin was obtained from the pharmacy of Erasmus University Medical Centre Rotterdam (Rotterdam, The Netherlands). The chemicals used for the Krebs buffer solutions were purchased from Merck (Darmstadt, Germany).
Sumatriptan was dissolved in distilled water or, where indicated, in DMSO and further diluted in distilled water. Indomethacin and SB-220453 were dissolved in DMSO and further diluted in distilled water. The other compounds were dissolved in distilled water.
Statistical analysis
Concentration response curves were analysed using GraphPad software (GraphPad Software Inc., San Diego, CA) to determine pEC50 values (negative logarithm of the concentration eliciting 50% of the maximal contractile response, Emax). When a plateau in the concentration response curve was not reached, the response observed with the highest concentration used (100 μ
At 100 μ
All data in the text and illustrations are presented as mean ±
Ethical approval
The Ethical Committee of the Erasmus University Medical Centre Rotterdam approved this study.
Results
Human isolated coronary artery
All coronary artery segments relaxed after substance P (1 n
Sumatriptan induced a concentration-dependent contraction, which was independent of the solvent (distilled water and DMSO) used; Emax, 12 ± 3% and 7 ± 1% of K+-induced contraction, respectively; pEC50, 6.0 ± 0.2% and 6.4 ± 0.2%, respectively. The Emax of sumatriptan did not correlate to the relaxant response to substance P (Pearson's r = 0.117). SB-220453 induced no response (Fig. 1, left panel). After precontraction with U46619 (17 ± 3% of K+-induced contraction), the Emax to sumatriptan was substantially augmented in two out of the four experiments. However, the mean Emax (39 ± 24% for sumatriptan dissolved in distilled water and 38 ± 17% for sumatriptan dissolved in DMSO) was, as reported previously (25), not significantly different from values in these four experiments in the absence of U46619 (16 ± 3% for sumatriptan dissolved in distilled water and 9 ± 1% for sumatriptan dissolved in DMSO). The pEC50 of sumatriptan in the presence of U46619 (6.7 ± 0.3 and 6.6 ± 0.3 for sumatriptan dissolved in distilled water and DMSO, respectively) was also not significantly increased, when compared with those in the absence of U46619 (6.2 ± 0.3% for sumatriptan dissolved in distilled water and 6.5 ± 0.2% for sumatriptan dissolved in DMSO). Similar to the experiments performed on vessel segments in the absence of U46619, SB-220453 failed to either contract or relax the coronary artery in the presence of U46619 (Fig. 1, right panel).
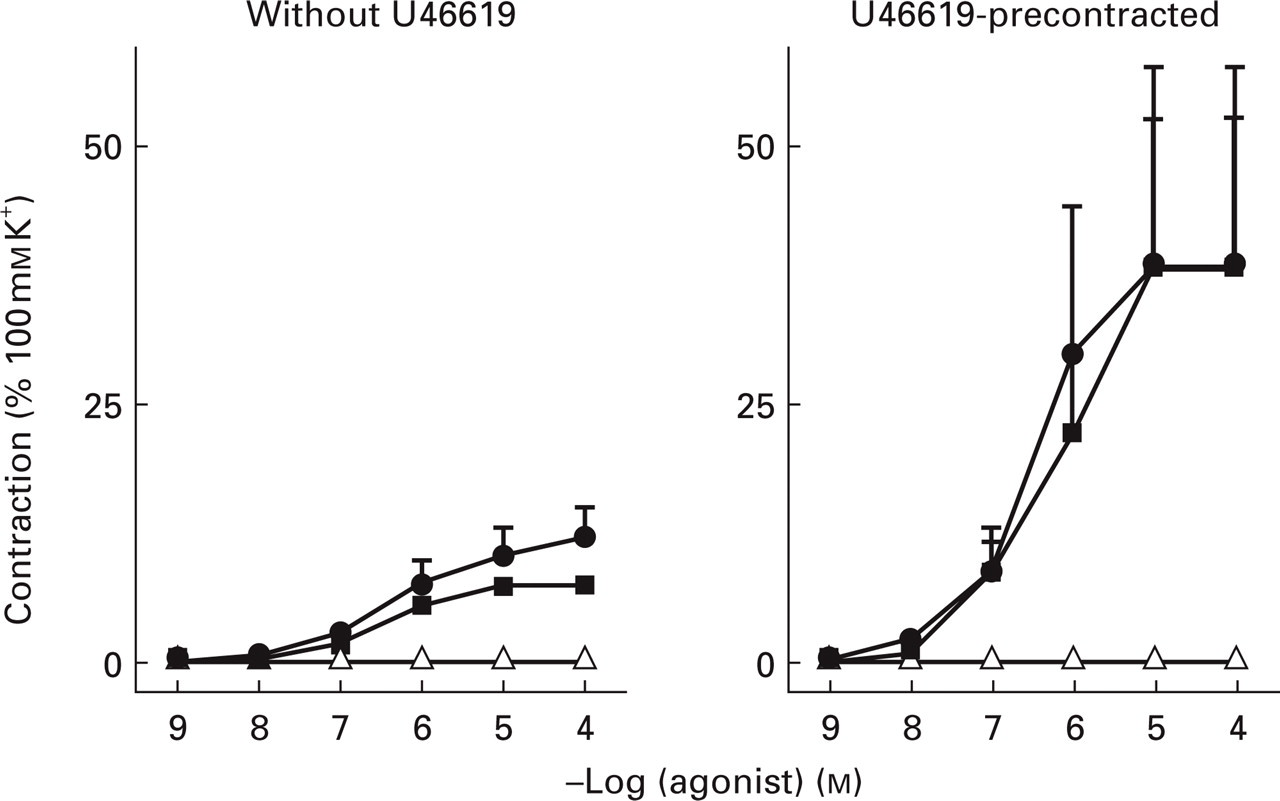
Concentration response curves in human isolated coronary arteries to sumatriptan (dissolved in distilled water (•) or dimethylsulfoxide (DMSO; ▪)) and SB-220453 (▵, dissolved in DMSO). Experiments were performed in the absence of U46619 (left panel, n = 6) or after precontraction with U46619 (3–10 n
Human isolated saphenous vein
Saphenous vein segments relaxed after bradykinin (1 μ
In all experiments, sumatriptan (dissolved in DMSO) induced a concentration-dependent contraction (Emax 52 ± 5%, pEC50 6.3 ± 0.1), which was unrelated to the relaxant response to bradykinin (Pearson's r =−0.081). SB-220453 did not induce a contraction in any of the concentrations used (Fig. 2). The concentration response curves to sumatriptan that were constructed after the concentration response curve to SB-220453 did not differ from those constructed before SB-220453 (Emax 59 ± 10%; pEC50 6.2 ± 0.2). Also after the concentration response curve to sumatriptan, SB-220453 failed to contract the saphenous vein.

Concentration response curves in human isolated saphenous vein (n = 4) to sumatriptan (▪) and SB-220453 (▵), both dissolved in dimethylsulfoxide. Whereas sumatriptan induced a concentration-dependent contraction in all experiments, SB-220453 had no effect.
Human isolated middle meningeal artery
Middle meningeal artery segments relaxed to substance P (10 n
Sumatriptan (dissolved in distilled water) induced a concentration-dependent contraction in all experiments (Emax 105 ± 18%, pEC50 6.9 ± 0.2), which was not related to the relaxant response to substance P (Pearson's r =− 0.155). SB-220453 did not induce any contraction (Fig. 3). After construction of the concentration response curves to SB-220453, the contraction to sumatriptan (dissolved in DMSO) did not differ significantly from that to sumatriptan dissolved in distilled water (Emax 101 ± 2%; pEC50 6.8 ± 0.4).
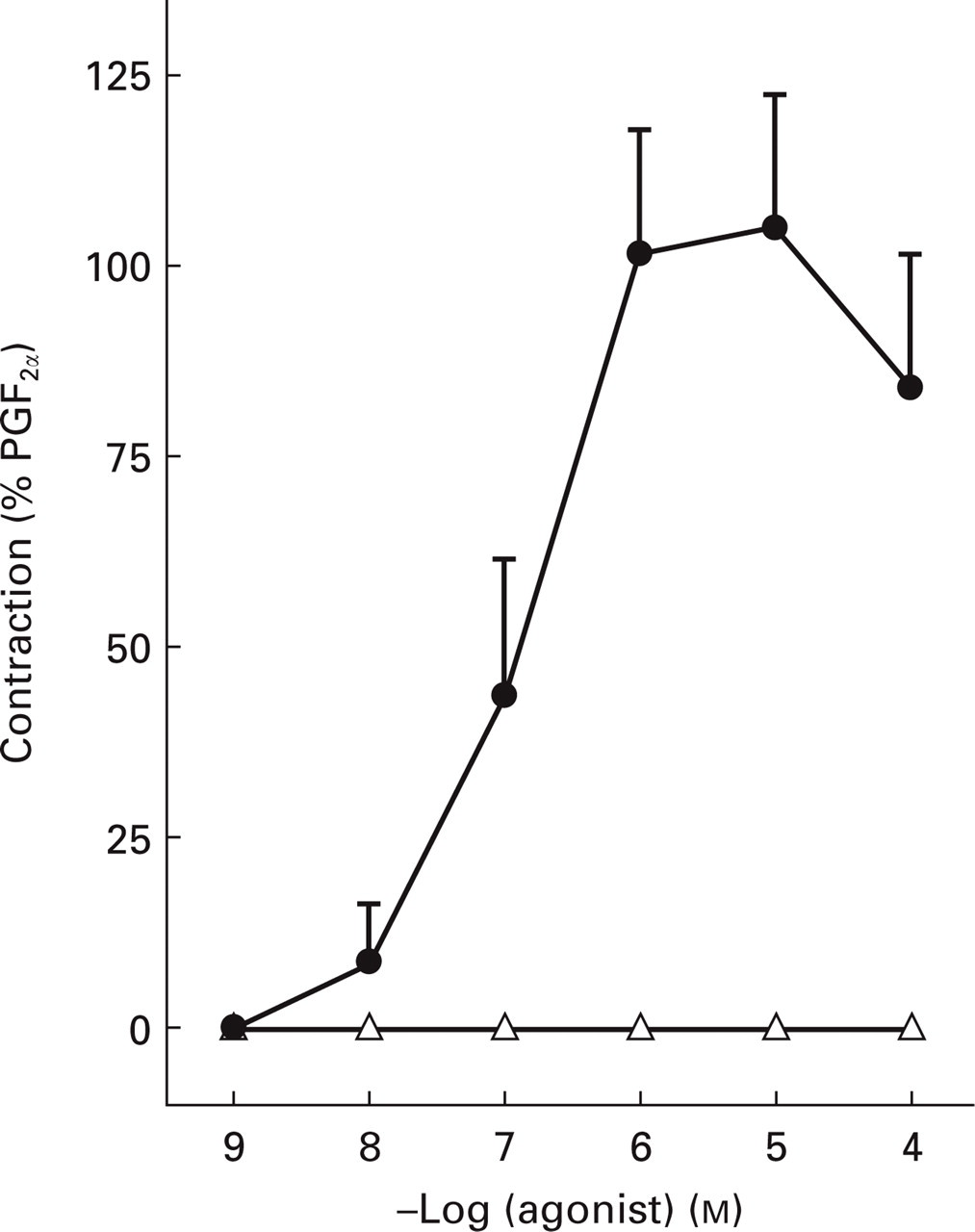
Concentration response curves in human isolated middle meningeal artery to sumatriptan (dissolved in distilled water, •; n = 5) and SB-220453 (▵, dissolved in dimethylsulfoxide; n = 4).
Human atrial and ventricular trabeculae
As we reported earlier (24), baseline contractile force was significantly lower in the atrial (0.64 ± 0.13 mN, n = 17 trabeculae) than in ventricular (3.04 ± 0.45 mN, n = 20 trabeculae) tissue. In both tissues, noradrenaline (10 n
Neither SB-220453 nor sumatriptan displayed a positive inotropic effect on atrial and ventricular trabeculae (Fig. 4). At concentrations ≥10 μ

Concentration response curves in human isolated atrial (left panel, n = 3–5) and ventricular (right panel, n = 5) trabeculae to sumatriptan (dissolved in distilled water, •, or dimethylsulfoxide (DMSO; ▪) and SB-220453 (▵, dissolved in DMSO). NA, Noradrenaline.
Discussion
Human isolated blood vessels
SB-220453 did not induce any significant contraction of the human isolated coronary artery, saphenous vein or middle meningeal artery. In contrast, sumatriptan, investigated in parallel, produced marked contractions. Since contractions to some agonists can be ‘unmasked’ or augmented in the presence of increased tension (7, 25), we also investigated the coronary artery contraction in the presence of the thromboxane A2 analogue U46619. Indeed, the contraction to sumatriptan was augmented in two of the coronary arteries investigated (in accordance with our previous findings (25)), but even in these precontracted segments, SB-220453 failed to elicit any contraction.
In all blood vessels, a slight relaxation was observed at the highest concentration of SB-220453 and sumatriptan (both dissolved in DMSO), but not with sumatriptan dissolved in distilled water. Therefore, this relaxation may be assigned to the solvent DMSO. However, despite this relaxant response, SB-220453 did not affect the concentration response curve to sumatriptan (see Figs 2 and 3).
It is known that the presence or absence of functional endothelium can influence contractile responses in blood vessels (26, 27). In our study, the endothelial quality of the blood vessels varied, as is illustrated by the relaxation to substance P (coronary artery 9–107%, middle meningeal artery 19–76%) or bradykinin (saphenous vein 10–107%). The contraction to sumatriptan was not related to the endothelial quality, though in a large post hoc study we have previously demonstrated that coronary artery contraction to sumatriptan slightly increases with increasing functional integrity of the endothelium (26). Because in none of the blood vessels did SB-220453 induce any contraction, our results suggest that the lack of response to these compounds is not dependent on the quality of the endothelium.
It is conceivable that overnight storage of the coronary artery and saphenous vein segments may have affected responses to SB-220453. However, several arguments suggest that this was probably not the case. First, the endothelial function in the present study was similar to that reported in coronary arteries obtained directly after explantation of the heart (28, 29). Second, porcine coronary arteries display similar basic contractile as well as relaxant properties, whether investigated immediately or after 24 h storage (30). Lastly, as in coronary artery and saphenous vein, SB-220453 also failed to contract middle meningeal artery, which was studied within 2 h of surgical removal.
Atrial and ventricular cardiac trabeculae
Apart from a negative inotropic effect associated with the solvent DMSO, no inotropic response was observed with SB-220453 or sumatriptan in either atrial or ventricular cardiac trabeculae. Sumatriptan did not display any effect on cardiac trabecular contractility despite expression of both 5-HT1B and 5-HT1D mRNA in human atrium and ventricle (31). However, the present results are consistent with the observed lack of haemodynamic effects of sumatriptan in patients, where no negative or positive inotropic effects were demonstrated (32). These studies are also in accordance with in vitro studies performed on guinea pig (33, 34) cardiac tissue. In fact, the role of 5-HT1B and 5-HT1D receptors on the human heart is still poorly understood (36), although the 5-HT1D receptor has been reported to mediate inhibition of noradrenaline release in human atrium (37).
In conclusion, our results do not provide any evidence of human blood vessel contraction or altered myocardial contractility in response to SB-220453. Since SB-220453 did not contract the middle meningeal artery, we conclude that potential therapeutic efficacy is independent of cerebral vasoconstriction. SB-220453 did not contract coronary artery, saphenous vein or cardiac trabeculae, and is therefore likely to be devoid of adverse cardiovascular side-effects observed with serotonergic agonists.
