Abstract
Cluster headache (CH) is a primary headache disorder where the aetiological and pathophysiological mechanisms still are largely unknown. An increased risk of CH in first- and second-degree relatives suggests the importance of genetic factors. Mutations of the P/Q type calcium channel alpha 1 subunit (CACNA1A) gene on chromosome 19p13 have been shown to cause several neurological disorders with a wide clinical spectrum, mainly episodic diseases. Missence mutations of the gene cause familial hemiplegic migraine (FHM) and it is also likely to be involved in the more common forms of migraine. The CACNA1A gene is thus a promising candidate gene for CH. In this study we performed an association analysis of an intragenic polymorphic (CA)n-repeat with marker D19S1150 and a (CAG)n-repeat in the 3′UTR region, in 75 patients with CH according to IHS criteria and 108 matched controls. Genotypes and allele frequencies were similarly distributed in patients and controls. Linkage disequilibrium between the two markers was similar in patients and controls. We conclude that an importance of the CACNA1A gene in sporadic CH is unlikely.
Keywords
Introduction
Cluster headache (CH) is characterized by recurrent, unilateral attacks of severe headache. The attacks most commonly appear in clusters, so called cluster periods. CH is considered a neurovascular headache disorder with the trigeminovascular system and the cranial parasympathetic nervous system being involved (1–3), but the pathophysiology is still largely unknown. The genetic background to migraine and other headaches has attracted an increased interest in recent years (4, 5). Heredity has previously been regarded as being of minor importance for the aetiology of CH, but in recent years its importance has been increasingly recognized. Several studies have reported a positive family history of CH (6–14). Russell showed in a Danish genetic epidemiological survey a 14.1-fold significant increased risk of CH in first-degree relatives and a 2.3-fold increased risk in second-degree relatives (10, 12). Kudrow and Kudrow (7) showed in an American survey an 45.6-fold increased risk in first-degree relatives, but the familial occurrence in this study might have been overestimated
The brain-specific P/Q type calcium channel alpha 1A subunit gene CACNA1A on chromosome 19p13 has been shown to be involved in a few mainly episodic neurological disorders. Different types of mutations in this gene are described in three disorders: familial hemiplegic migraine (FHM), episodic ataxia type 2 (EA2) and spinocerebellar ataxia type 6 (SCA6). Four different missence mutations have been described in five unrelated FHM families (19–24). Two mutations disrupting the reading frame cause EA2 (20, 23, 25). In SCA6 there is an expansion of a polymorphic CAG-repeat within the open reading frame (26). Affected sib-pair analysis with highly informative microsatellite markers in the FHM locus on 19p13 in migraine with and without aura has shown significantly increased sharing of alleles, indicating involvement of the CACNA1A gene even in the more common forms of migraine (27, 28, 30). Another study has shown linkage to chromosome 19p13 in one of four families with familial typical migraine (29).
Calcium acts as an intracellular second messenger by regulating several events in the cell; both biochemical and neuronal events such as neurotransmitter release (31–33). P-type neuronal calcium channels mediate serotonin (5-HT) release (34, 35). Most models of migraine and CH suggest that serotonin has a central role in the pathophysiology. Verapamil, an L-calcium channel blocker, is effective as prophylactic treatment in CH although high doses are often needed. The mechanism of action is not yet known but it is likely that verapamil may act by non-L-channel mechanisms, perhaps by interacting with one or more of the other voltage-dependent calcium channels (36).
Mutations of the CACNA1A gene result in a wide clinical spectrum varying from mild and fully reversible symptoms to severely progressive symptoms. The CACNA1A gene is thus an interesting candidate for conferring susceptibility to CH.
Materials and methods
Patients and control samples
Blood samples from 75 Caucasian outpatients with sporadic CH were collected, 56 (74.7%) males and 19 (25.3%) females. The mean age was 49.4 years (range 24–74 years). Six patients had chronic and 69 patients had episodic cluster headache. They had all been seen by us and clearly fulfilled the International Headache Society (IHS) criteria for CH (37). The control group consisted of 108 Caucasian persons, 74 (68.5%) males and 34 (31.5%) females. The mean age of the controls was 48.9 years (range 25–72 years). The control group, drawn from a larger group of controls to match for age and gender, consisted of blood donors, healthy volunteers and spouses of patients.
The study was approved by the local ethics committee at Huddinge University Hospital.
DNA isolation and genotyping
DNA was extracted from peripheral blood using a modified salting out method (38). For this association study we chose two highly polymorphic intragenic markers, the (CA)n-repeat marker D19S1150 (forward 5′-GGAGAAGCATAGAAAAGCCA-3′ and reverse 5′-CCTGTTGAAAACTCCTGACC-3′) and the (CAG)n triplet repeat sequence in the 3-prime-UTR (exon 47) with forward primer sequence S-5-F1 (5′-CACGTGTCCTATTCCCCTGTGATCC-3′) and reversed primer sequence S-5-R1(5′-TGGGTACCTCCGAGGGCCGCTGGTG-3′) as described by Zhuchenko et al. (26). For marker D19S1150, PCR was performed in a final volume of 7 μl consisting of 50 m
Electrophoresis of the amplified DNA fragments was performed on polyacrylamide gels on an ABI 377 DNA sequencer. Fragment sizes were determined by comparison with internal lane size standards. The results were analysed using the GENESCAN/GENOTYPER software (version 1.1; PE Biosystems).
Statistical analysis
Allele and phenotype frequencies for patients and controls were compared using Fisher's exact test. The level of significance was chosen as P < 0.05. The large number of comparisons made in the study should be considered in evaluating deviations observed. We also performed a Monte Carlo simulation test to assess the likelihood of our data as described by Sham & Curtis (39). Linkage disequilibrium (LD) was assessed by Fisher's exact test and by chi-square analysis between allele pairs of the two markers. The sample size allows 80% power (with a two-sided CI of 95%) to detect a RR of 2.5 of an allele with a frequency of 13% in controls.
Results
In our dataset there were 11 alleles of the dinucleotide repeat polymorphisms (D19S1150), ranging in size from 144 to 168 bp. Phenotype and allele frequencies (defined as carriership of the different alleles) of this marker are shown in Tables 1 and 2. The 158 bp allele was somewhat more frequent in CH patients and the 156 bp allele was somewhat more frequent in controls, but none of the comparisons were statistically significant.
Phenotype frequencies of CACNA1A dinucleotide repeat (marker D19S1150) alleles in cluster headache patients and controls

Allele frequencies of CACNA1A dinucleotide repeat (marker D19S1150) in cluster headache patients and controls

Refers to numbers of alleles.
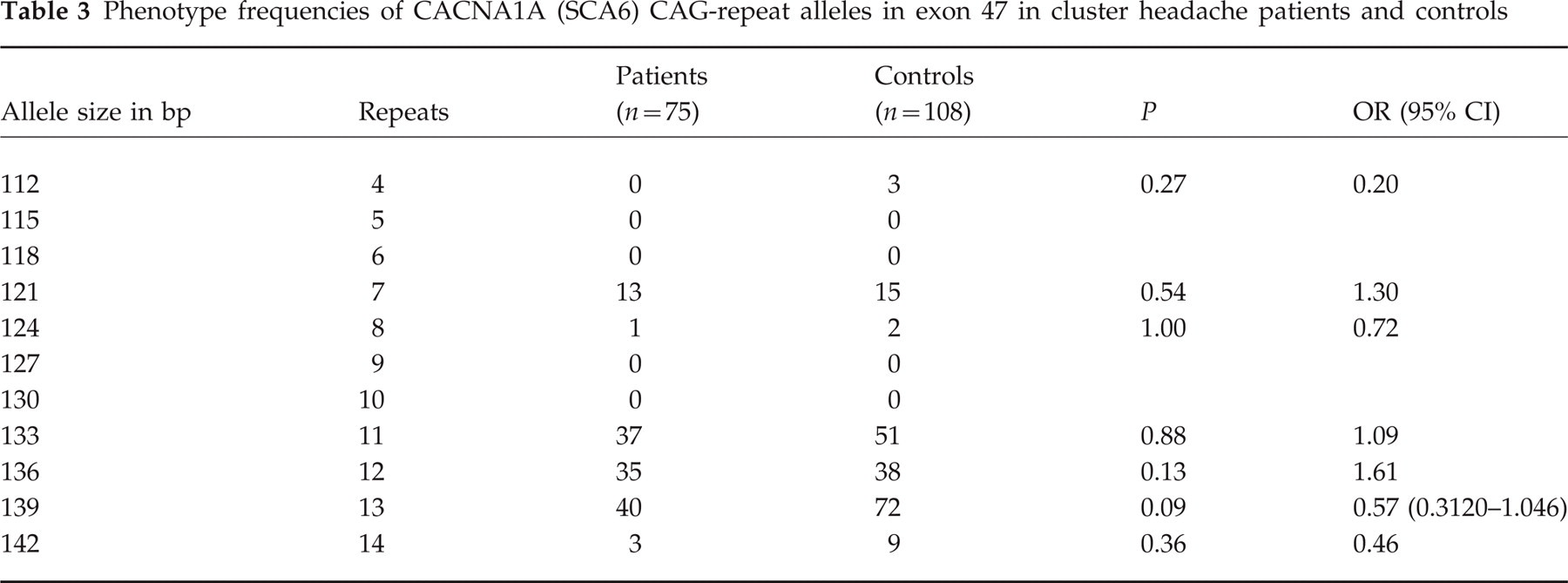
For the CAG-repeat in the 3′ coding region we detected alleles ranging in size from 112 to 142, with estimated numbers of repeats ranging from 4 to 14. The reported repeat lengths in control populations in other studies vary from 4 to 20 repeats (20, 26, 40, 41). We observed amplified fragments varying from 112 to 142 base pairs, corresponding to 4–14 repeats (Genebank accession number Z 80155). Phenotype and allele frequencies are shown in Tables 3 and 4. The 14 repeat allele was somewhat more frequent in the control group but differences were not significant.
Phenotype frequencies of CACNA1A (SCA6) CAG-repeat alleles in exon 47 in cluster headache patients and controls
Allele frequencies of CACNA1A (SCA6) CAG-repeat in exon 47 in cluster headache patients and controls

Refers to numbers of alleles.
The genotypes for the two polymorphisms in the chronic CH cases did not differ remarkably from the rest of the CH patients.
Chi-square comparisons of phenotype frequencies with Monte Carlo simulation confirmed the lack of significance (P = 0.398 for D19S1150 marker and P = 0.365 for the CAG-repeat).
We analysed the extent of LD between specific alleles of the two markers. The distribution of genotypes was similar in patients and controls, with a minor increase of the combined alleles, 162/139, in patients (23% vs. 13% in controls, OR = 1.8, P = 0.16, data not shown). The 154 allele occurred more often together with the 136 allele in both patients and controls (OR = 2.39, P < 0.01). This indicates a slight LD between the two markers that appeared to be similar in patients and controls.
Discussion
This study, to our knowledge the first of its kind in CH, offers no support for an association between CH and two polymorphic markers of the CACNA1A gene, the (CA)n repeat marker D19S1150 and the coding (CAG)n poly glu SCA6 expansion polymorphism. The observed slight differences in phenotype and allele frequencies fall well inside what would be expected by chance. Based on the power estimations performed, we conclude that it is unlikely that genetic variation within this gene contributes greatly to the susceptibility to CH.
CACNA1A gene mutations are described in a few neurological disorders, calcium acts as an important messenger in the regulation of many neuronal events and verapamil, an L-channel-blocker, is an effective prophylactic treatment in CH as mentioned in the introduction. Given these findings, the CACNA1A gene becomes a relevant candidate in conferring CH susceptibility, especially as it is preferentially expressed within the central nervous system. Therefore, even if no evidence for an association was found with CH, other subunits of the same or other calcium channels may be selected in subsequent studies.
It is likely that a number of individuals in the control group would suffer from common or classical migraine. As the CACNA1A gene could just as well be considered a candidate gene for migraine as for CH, the hypothetical presence of an association with this gene in migraine would somewhat diminish the statistical power of the present study. However, this negative effect is not likely to be large, and there is not yet enough data available to evaluate the importance of the CACNA1A gene in the more common forms of migraine.
We chose to study two highly polymorphic multi-allelic markers of the CACNA1A gene, assuming that either they should be of functional importance for CH themselves or that they should be in linkage disequilibrium (LD) with other hypothetical disease-causing mutations. Indeed, the significant LD observed between pairs of the more common alleles of the two markers suggests that LD is quite extensive within this gene, at least between relatively recent polymorphisms. On the other hand, functionally important single nucleotide polymorphisms could be of an older date, which would have allowed LD to weaken. In that situation, only an analysis of the polymorphisms themselves would give the final answer to whether an association exists. So far, no such polymorphisms have been reported within this gene and we did not consider the effort of sequencing long sequences of this gene in large numbers of individuals as relevant at this stage. Thus, lacking other direct indications of an importance of this gene in CH, we consider the present data sufficient for the time being. Clearly a minor contribution to sporadic CH or a major contribution in a subgroup, for example familial cases of CH, cannot be ruled out. The 19p13 chromosomal region remains of great interest in the genetics of neurological disorders.
Footnotes
Acknowledgements
This study was supported by grants from the Karolinska Institute, Astra Zeneca and Glaxo Wellcome.
